Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (15): 2338-2345.doi: 10.12307/2024.250
Previous Articles Next Articles
Mechanism of black phosphorus regulating oxidative stress-inflammation cascade in retarding intervertebral disc degeneration
Kou Yu, Gu Yong, Chen Liang
- First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China
-
Received:2022-12-13Accepted:2023-02-14Online:2024-05-28Published:2023-09-19 -
Contact:Chen Liang, Professor, Doctoral supervisor, Chief physician, First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China -
About author:Kou Yu, Master candidate, First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China -
Supported by:National Natural Science Foundation of China, No. 81972078, No. 82072438 (to CL)
CLC Number:
Cite this article
Kou Yu, Gu Yong, Chen Liang. Mechanism of black phosphorus regulating oxidative stress-inflammation cascade in retarding intervertebral disc degeneration[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(15): 2338-2345.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
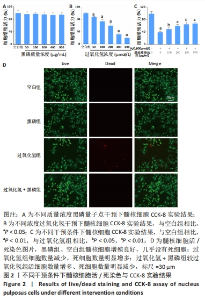
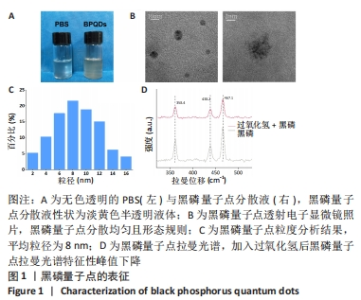
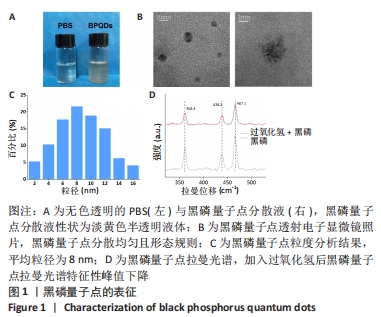
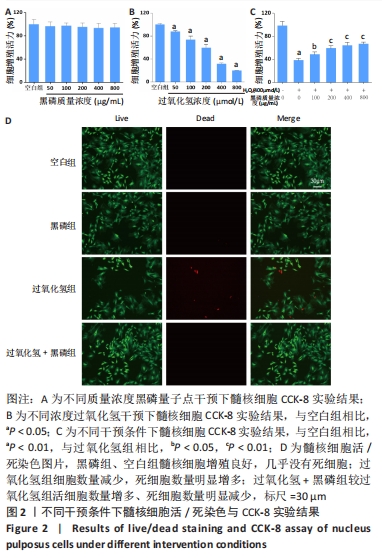
2.2 黑磷量子点体外细胞实验结果 2.2.1 黑磷量子点生物相容性及保护髓核细胞增殖活性能力 CCK-8实验结果显示,黑磷量子点质量浓度在100,200,400,800 μg/mL时,髓核细胞的增殖活性与空白组无明显差异(P > 0.05),见图2A,进一步验证了黑磷量子点良好的生物相容性。过氧化氢浓度在50,100,200,400,800 μmol/L时,髓核细胞的增殖活性受到抑制,且这一作用随过氧化氢浓度的增高更加明显,见图2B。使用黑磷量子点与过氧化氢共同干预髓核细胞,相较空白组,过氧化氢的干预显著降低了髓核细胞的增殖活性,当黑磷量子点质量浓度在100,200,400,800 μg/mL时髓核细胞增殖活性有所恢复,黑磷量子点质量浓度在100,200 μg/mL时细胞增殖活性升高(P < 0.05,P < 0.01);黑磷量子点质量浓度在200,400,800 μg/mL时3组细胞增殖活性无明显差异(P > 0.05),见图2C。活/死染色结果显示,黑磷组、空白组髓核细胞增殖良好,几乎没有死细胞,说明黑磷量子点生物相容性良好,对细胞无毒性作用;过氧化氢组细胞数量减少,死细胞数量明显增多;黑磷+过氧化氢组较过氧化氢干预组活细胞数量增多、死细胞数量明显减少,见图2D,说明黑磷量子点在氧化应激环境下对髓核细胞具有保护作用。"
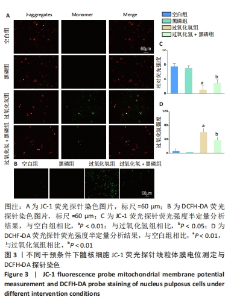
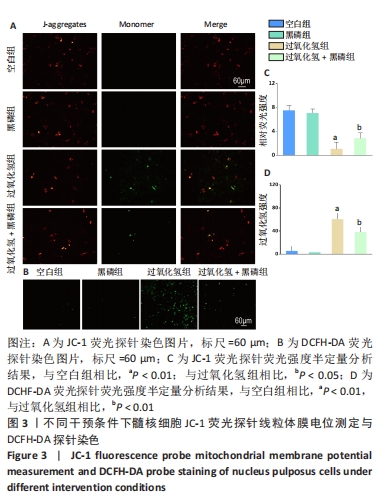
2.2.2 黑磷量子点调控氧化应激导致的髓核细胞线粒体膜电位降低 线粒体膜电位下降标志着细胞凋亡的早期。通过JC-1荧光探针检测髓核细胞线粒体膜电位,结果显示,过氧化氢组线粒体膜电位较空白组降低(P < 0.01),黑磷+过氧化氢组线粒体膜电位较过氧化氢组升高(P < 0.05),而空白组及黑磷组线粒体膜电位无差异(P > 0.05),见图3A、C。 2.2.3 黑磷量子点对细胞内活性氧清除效果 DCHF-DA荧光染料可被细胞内活性氧氧化,形成具有绿色荧光的2’,7’二氯荧光素。根据荧光强度定量分析结果,发现过氧化氢组绿色荧光强度最高,黑磷+过氧化氢组绿色荧光强度显著降低,而空白组与黑磷组绿色荧光强度极低,见图3B、D。这些结果表明,黑磷量子点可以保护细胞免受过氧化氢诱导的细胞内氧化应激。"
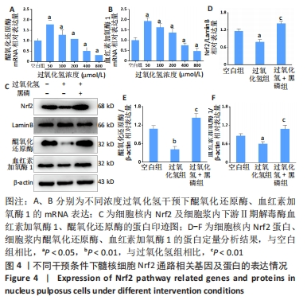
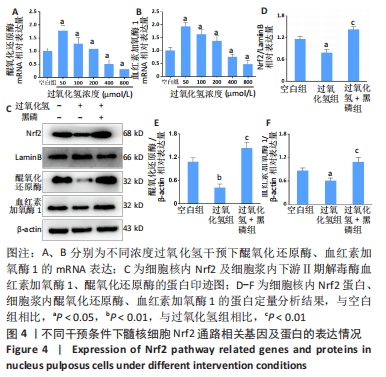
2.2.4 黑磷量子点促进Nrf2核转移及其下游酶谱 RT-qPCR检测结果显示,与空白组比较,50,100,200 μmol/L的过氧化氢增加了髓核细胞内血红素加氧酶1和醌氧化还原酶的mRNA水平(P < 0.05),当过氧化氢浓度达到400 μmol/L以上时,髓核细胞内血红素加氧酶1和醌氧化还原酶的mRNA水平显著下降(P < 0.05),见图4A、B,说明在该浓度过氧化氢下髓核细胞内Nrf2通路活性下降,其下游的Ⅱ期解毒酶基因表达下降。Nrf2的核转位是Nrf2/ARE信号通路激活的标志[34]。实验提取了细胞核蛋白以检测Nrf2的激活,如图4C-F所示,与空白组相比,细胞毒性浓度的过氧化氢干预显著抑制了Nrf2蛋白的核转位,血红素加氧酶1和醌氧化还原酶的蛋白表达也被过氧化氢处理显著抑制;然而,与单独过氧化氢处理相比,黑磷量子点与过氧化氢共同处理后细胞中的Nrf2蛋白核转位显著增加,血红素加氧酶1和醌氧化还原酶的蛋白表达也显著增加。这些结果表明,黑磷量子点有助于Nrf2核转位,提高Ⅱ期解毒酶的表达。"
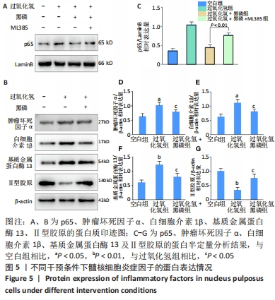
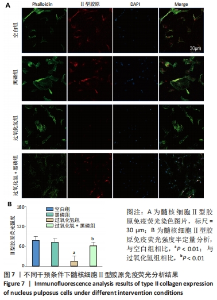
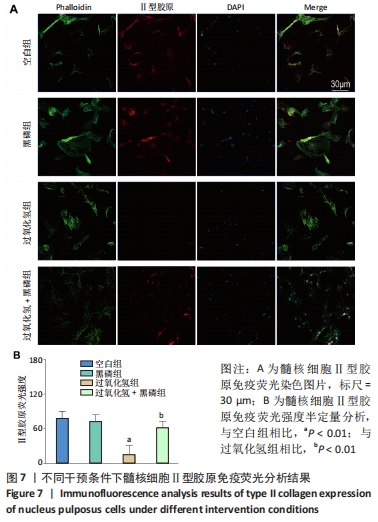
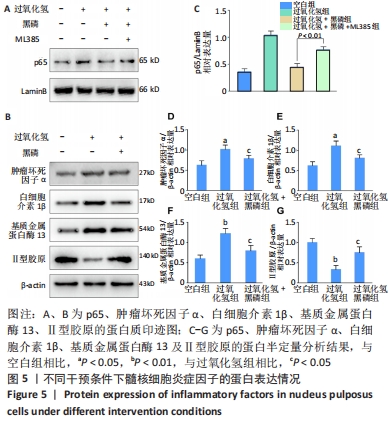
2.2.5 黑磷量子点通过激活Nrf2通路抑制核因子κB炎症通路 核因子κB通路是椎间盘退变中被激活的经典炎症通路之一[21],有文献报道,Nrf2通路对核因子κB通路存在抑制作用[12,19-20]。 为了证实Nrf2对核因子κB的抑制作用,检测了空白组、过氧化氢组、黑磷+过氧化氢组及黑磷+过氧化氢+ML385组中p65的核转位水平,结果表明,过氧化氢组p65核转位水平高于空白组(P < 0.01),但与过氧化氢组相比,黑磷+过氧化氢干预后p65核转位水平降低(P < 0.01);然而,Nrf2特异性抑制剂ML385阻断了黑磷量子点对核因子κB的抑制作用(P < 0.01),见图5A、C。 2.2.6 黑磷量子点影响髓核细胞炎症因子及髓核退变相关分子表达 Western Blot检测结果表明,与空白组比较,过氧化氢组相白细胞介素1β、肿瘤坏死因子α等炎症细胞因子的蛋白表达显著增加(P < 0.05),基质金属蛋白酶13的蛋白表达也显著增加(P < 0.01),而细胞外基质成分Ⅱ型胶原的蛋白表达显著减少(P < 0.01),黑磷量子点的加入逆转了上述趋势,见图5B及图5D-G。"
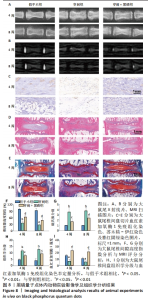
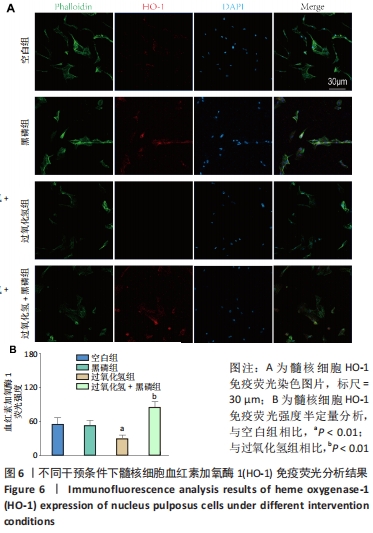
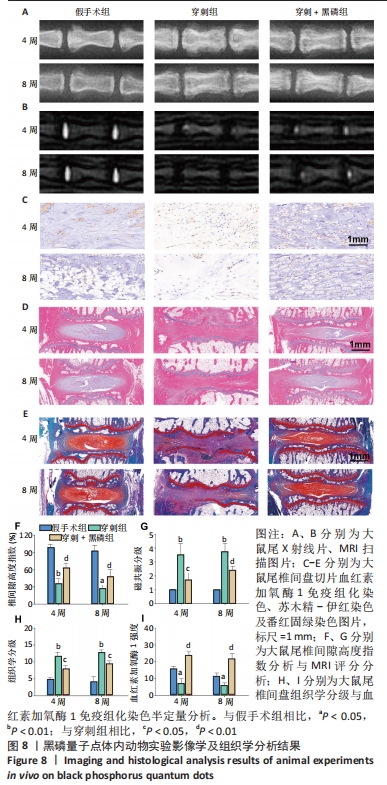
2.3 黑磷量子点动物体内实验结果 2.3.1 实验动物数量分析 30只大鼠全部进入结果分析。 2.3.2 影像学评估结果 如图8A、F所示,穿刺组术后4,8周的椎间盘高度明显低于假手术组(P < 0.01),黑磷+穿刺组后4,8周的椎间盘高度明显高于穿刺组(P < 0.01)。MRI也能可靠地反映椎间盘的再生,T2加权信号较高,表明髓核含水量较高。根据改进的汤姆逊分类法,MRI图像根据T2加权信号强度被分类为Ⅰ-Ⅳ级[35]。术后4,8周,穿刺组T2加权信号显著低于假手术组(P < 0.01),黑磷+穿刺组髓核T2加权信号均高于穿刺组(P < 0.05,P < 0.01),见图8B、G。说明黑磷量子点的加入起到了延缓椎间盘退变的效果。 2.3.3 组织学评价结果 苏木精-伊红和番红固绿染色结果见图8D、E、H所示,不论是在术后4周还是8周,相较于假手术组,穿刺组椎间盘髓核组织胶原含量减少,髓核和纤维环之间的边界模糊;与穿刺组相比,黑磷+穿刺组椎间盘组织的变性和结构损伤均有所改善,髓核和纤维环之间有明确的边界,髓核组织的胶原含量有所提高;术后4周和8周,黑磷+穿刺组的组织学评分显著高于穿刺组(P < 0.05),但低于假手术组(P < 0.05)。 免疫组化染色结果显示,术后4,8周,穿刺组大鼠椎间盘组织内血红素加氧酶1表达量较假手术组降低,黑磷+穿刺组大鼠椎间盘组织内血红素加氧酶1表达量高于穿刺组,见图8C、8I。证明黑磷量子点在体内促进了Nrf2通路的激活,起到了抗氧化应激作用。"
| [1] CIEZA A, CAUSEY K, KAMENOV K, et al. Global estimates of the need for rehabilitation based on the Global Burden of Disease study 2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2021;396(10267):2006-2017. [2] ADAMS MA, ROUGHLEY PJ. What is intervertebral disc degeneration, and what causes it? Spine (Phila Pa 1976). 2006;31(18):2151-2161. [3] 解志锋,刘清,刘冰,等.腰椎间盘疲劳损伤的生物力学特性[J].中国组织工程研究, 2021,25(3):339-343. [4] LE MAITRE CL, FREEMONT AJ, HOYLAND JA. Accelerated cellular senescence in degenerate intervertebral discs: a possible role in the pathogenesis of intervertebral disc degeneration. Arthritis Res Ther. 2007;9(3):R45. [5] DING F, SHAO ZW, XIONG LM. Cell death in intervertebral disc degeneration. Apoptosis. 2013; 18(7):777-785. [6] DIMOZI A, MAVROGONATOU E, SKLIROU A, et al. Oxidative stress inhibits the proliferation, induces premature senescence and promotes a catabolic phenotype in human nucleus pulposus intervertebral disc cells. Eur Cell Mater. 2015;30:89-102; discussion 103. [7] SONG D, GE J, WANG Y, et al. Tea Polyphenol Attenuates Oxidative Stress-Induced Degeneration of Intervertebral Discs by Regulating the Keap1/Nrf2/ARE Pathway. Oxid Med Cell Longev. 2021; 2021:6684147. [8] LU Y, ZHOU L, HE S, et al. Lycopene alleviates disc degeneration under oxidative stress through the Nrf2 signaling pathway. Mol Cell Probes. 2020;51:101559. [9] VO N, NIEDERNHOFER LJ, NASTO LA, et al. An overview of underlying causes and animal models for the study of age-related degenerative disorders of the spine and synovial joints. J Orthop Res. 2013;31(6):831-837. [10] NAVARRO-YEPES J, BURNS M, ANANDHAN A, et al. Oxidative stress, redox signaling, and autophagy: cell death versus survival. Antioxid Redox Signal. 2014;21(1):66-85. [11] FENG C, YANG M, LAN M, et al. ROS: Crucial Intermediators in the Pathogenesis of Intervertebral Disc Degeneration. Oxid Med Cell Longev. 2017;2017:5601593. [12] MA Q. Role of nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol. 2013;53:401-426. [13] ESPINOSA-DIEZ C, MIGUEL V, MENNERICH D, et al. Antioxidant responses and cellular adjustments to oxidative stress. Redox Biol. 2015;6:183-197. [14] MOTOHASHI H, KATSUOKA F, ENGEL JD, et al. Small Maf proteins serve as transcriptional cofactors for keratinocyte differentiation in the Keap1-Nrf2 regulatory pathway. Proc Natl Acad Sci U S A. 2004;101(17):6379-6384. [15] ZHANG X, LIANG S, GAO X, et al. Protective Effect of Chitosan Oligosaccharide against Hydrogen Peroxide-Mediated Oxidative Damage and Cell Apoptosis via Activating Nrf2/ARE Signaling Pathway. Neurotox Res. 2021;39(6):1708-1720. [16] TAO W, SUN W, LIU L, et al. Chitosan Oligosaccharide Attenuates Nonalcoholic Fatty Liver Disease Induced by High Fat Diet through Reducing Lipid Accumulation, Inflammation and Oxidative Stress in C57BL/6 Mice. Mar Drugs. 2019;17(11):645. [17] SALOMONE F, GODOS J, ZELBER-SAGI S. Natural antioxidants for non-alcoholic fatty liver disease: molecular targets and clinical perspectives. Liver Int. 2016;36(1):5-20. [18] ZHANG Y, AHMAD KA, KHAN FU, et al. Chitosan oligosaccharides prevent doxorubicin-induced oxidative stress and cardiac apoptosis through activating p38 and JNK MAPK mediated Nrf2/ARE pathway. Chem Biol Interact. 2019;305:54-65. [19] LI W, KHOR TO, XU C, et al. Activation of Nrf2-antioxidant signaling attenuates NFkappaB-inflammatory response and elicits apoptosis. Biochem Pharmacol. 2008;76(11):1485-1489. [20] MA Q, KINNEER K, YE J, et al. Inhibition of nuclear factor kappaB by phenolic antioxidants: interplay between antioxidant signaling and inflammatory cytokine expression. Mol Pharmacol. 2003;64(2):211-219. [21] ZHANG GZ, LIU MQ, CHEN HW, et al. NF-κB signalling pathways in nucleus pulposus cell function and intervertebral disc degeneration. Cell Prolif. 2021;54(7):e13057. [22] FU HD, LI ZB, XIE HH, et al. Different-sized black phosphorus nanosheets with good cytocompatibility and high photothermal performance. Rsc Adv. 2017;7(24):14618-14624. [23] BRENT JR, GANGULI AK, KUMAR V, et al. On the stability of surfactant-stabilised few-layer black phosphorus in aqueous media. Rsc Adv. 2016;6(90):86955-86958. [24] HOU J, WANG H, GE Z, et al. Treating Acute Kidney Injury with Antioxidative Black Phosphorus Nanosheets. Nano Lett. 2020;20(2):1447-1454. [25] CHENG L, CHEN Z, CAI Z, et al. Bioinspired Functional Black Phosphorus Electrospun Fibers Achieving Recruitment and Biomineralization for Staged Bone Regeneration. Small. 2020;16(50):e2005433. [26] RAUCCI MG, FASOLINO I, CAPORALI M, et al. Exfoliated Black Phosphorus Promotes in Vitro Bone Regeneration and Suppresses Osteosarcoma Progression through Cancer-Related Inflammation Inhibition. ACS Appl Mater Interfaces. 2019;11(9):9333-9342. [27] WANG Z, LIU Z, SU C, et al. Biodegradable Black Phosphorus-based Nanomaterials in Biomedicine: Theranostic Applications. Curr Med Chem. 2019;26(10):1788-1805. [28] BIAN J, CAI F, CHEN H, et al. Modulation of Local Overactive Inflammation via Injectable Hydrogel Microspheres. Nano Lett. 2021;21(6): 2690-2698. [29] XU YC, GU Y, CAI F, et al. Metabolism Balance Regulation via Antagonist-Functionalized Injectable Microsphere for Nucleus Pulposus Regeneration. Adv Funct Mater. 2020;30(52): 14. [30] 叶旭文,顾勇,陈亮.负载姜黄素可注射微球延缓椎间盘的退变[J].中国组织工程研究, 2023,27(12):1884-1891. [31] MASUDA K, AOTA Y, MUEHLEMAN C, et al. A novel rabbit model of mild, reproducible disc degeneration by an anulus needle puncture: correlation between the degree of disc injury and radiological and histological appearances of disc degeneration. Spine (Phila Pa 1976). 2005;30(1):5-14. [32] CHEN H, LIU Z, WEI B, et al. Redox responsive nanoparticle encapsulating black phosphorus quantum dots for cancer theranostics. Bioact Mater. 2021;6(3):655-665. [33] GÓMEZ-PÉREZ J, BARNA B, TÓTH IY, et al. Quantitative Tracking of the Oxidation of Black Phosphorus in the Few-Layer Regime. ACS Omega. 2018;3(10):12482-12488. [34] BELLEZZA I, GIAMBANCO I, MINELLI A, et al. Nrf2-Keap1 signaling in oxidative and reductive stress. Biochim Biophys Acta Mol Cell Res. 2018;1865(5):721-733. [35] HAN B, ZHU K, LI FC, et al. A simple disc degeneration model induced by percutaneous needle puncture in the rat tail. Spine (Phila Pa 1976). 2008;33(18):1925-1934. [36] HUA W, LI S, LUO R, et al. Icariin protects human nucleus pulposus cells from hydrogen peroxide-induced mitochondria-mediated apoptosis by activating nuclear factor erythroid 2-related factor 2. Biochim Biophys Acta Mol Basis Dis. 2020;1866(1):165575. [37] Tang Z, Hu B, Zang F, et al. Nrf2 drives oxidative stress-induced autophagy in nucleus pulposus cells via a Keap1/Nrf2/p62 feedback loop to protect intervertebral disc from degeneration. Cell Death Dis. 2019;10(7):510. [38] HE R, CUI M, LIN H, et al. Melatonin resists oxidative stress-induced apoptosis in nucleus pulposus cells. Life Sci. 2018;199:122-130. [39] KIM KW, CHUNG HN, HA KY, et al. Senescence mechanisms of nucleus pulposus chondrocytes in human intervertebral discs. Spine J. 2009;9(8):658-666. [40] FINKEL T. Signal transduction by reactive oxygen species. J Cell Biol. 2011;194(1):7-15. [41] THURAKKAL S, ZHANG X. Recent Advances in Chemical Functionalization of 2D Black Phosphorous Nanosheets. Adv Sci (Weinh). 2020;7(2):1902359. [42] WANG H, YANG X, SHAO W, et al. Ultrathin Black Phosphorus Nanosheets for Efficient Singlet Oxygen Generation. J Am Chem Soc. 2015;137(35):11376-11382. [43] DING F, SHAO ZW, YANG SH, et al. Role of mitochondrial pathway in compression-induced apoptosis of nucleus pulposus cells. Apoptosis. 2012;17(6):579-590. [44] NASTO LA, ROBINSON AR, NGO K, et al. Mitochondrial-derived reactive oxygen species (ROS) play a causal role in aging-related intervertebral disc degeneration. J Orthop Res. 2013;31(7):1150-1157. [45] BUENDIA I, MICHALSKA P, NAVARRO E, et al. Nrf2-ARE pathway: An emerging target against oxidative stress and neuroinflammation in neurodegenerative diseases. Pharmacol Ther. 2016;157: 84-104. [46] WANG P, ZHANG S, LIU W, et al. Selenium Attenuates TBHP-Induced Apoptosis of Nucleus Pulposus Cells by Suppressing Mitochondrial Fission through Activating Nuclear Factor Erythroid 2-Related Factor 2. Oxid Med Cell Longev. 2022;2022:7531788. [47] LI Y, CHEN L, GAO Y, et al. Oxidative Stress and Intervertebral Disc Degeneration: Pathophysiology, Signaling Pathway, and Therapy. Oxid Med Cell Longev. 2022;2022:1984742. [48] FENG C, ZHANG Y, YANG M, et al. Oxygen-Sensing Nox4 Generates Genotoxic ROS to Induce Premature Senescence of Nucleus Pulposus Cells through MAPK and NF-κB Pathways. Oxid Med Cell Longev. 2017;2017:7426458. [49] 杨林,石军,郭中华,等.基于高分子材料的椎间盘组织工程:研究重点和热点[J].中国组织工程研究,2021,25(16):2589-2596. |
| [1] | Yang Yifeng, Ye Nan, Wang Lin, Guo Shuaicheng, Huang Jian. Signaling pathway of dexmedetomidine against ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1464-1469. |
| [2] | Wang Weiqing, Zhou Yue. Chronic inflammation regulates adipose tissue fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1307-1312. |
| [3] | Wang Ji, Zhang Min, Li Wenbo, Yang Zhongya, Zhang Long. Effect of aerobic exercise on glycolipid metabolism, skeletal muscle inflammation and autophagy in type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1200-1205. |
| [4] | Liu Xin, Hu Man, Zhao Wenjie, Zhang Yu, Meng Bo, Yang Sheng, Peng Qing, Zhang Liang, Wang Jingcheng. Cadmium promotes senescence of annulus fibrosus cells via activation of PI3K/Akt signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1217-1222. |
| [5] | Mu Bingtao, Yu Jingwen, Liu Chunyun, Guo Minfang, Meng Tao, Yang Pengwei, Wei Wenyue, Song Lijuan, Yu Jiezhong, Ma Cungen. Immunomodulatory effect of astragaloside IV on T cells of experimental autoimmune encephalomyelitis mice [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1057-1062. |
| [6] | Zhang Kefan, Shi Hui. Research status and application prospect of cytokine therapy for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 961-967. |
| [7] | Zhang Ya, Mu Qiuju, Wang Zilin, Liu Hongjie, Zhu Lili. Hydrogel loaded with platelet-rich plasma promotes wound healing in diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 690-696. |
| [8] | Fu Qiangchang, Zheng Liming, Jiang Lifeng. High tibial osteotomy promotes cartilage regeneration in the treatment of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5243-5248. |
| [9] | Li Yang, Ma Fei, Leng Yebo, Xu Shicai, He Baoqiang, Zhou Jiajun, Liao Yehui, Tang Qiang, Tang Chao, Wang Qing, Zhong Dejun. Correlation between intervertebral disc degeneration and hyperuricemia [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5091-5096. |
| [10] | Zhang Yunxin, Zhang Cunxin, Wang Qian, Xu Xinliang, Lyu Chaoliang, Ni Yong. Syringin inhibits intervertebral disc degeneration in rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5104-5109. |
| [11] | Ren Weiliang, Jiao Yongwei, Zhang Jian, Yang Liying, Yang Qi. Modulatory effect of resveratrol on oxidative stress and inflammatory factors in the joint fluid of rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5154-5158. |
| [12] | Hou Zengtao, Dong Zhiwei, Zhang Jinfeng, Yang Xiaohui, Fan Xiao. Platelet-rich fibrin regulates apoptosis to promote cartilage repair in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5167-5171. |
| [13] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Effect of gelatin methacryloyl hydrogel loaded with salvianolic acid B on intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 380-386. |
| [14] | Zhang Tiandong, Peng Qingping, Liu Huan, Feng Jianguo, Yi Qian, Huang Wenhua. Semen cuscutae in the treatment of osteoarthritis: network pharmacology analysis and experimental validation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(28): 4516-4521. |
| [15] | Guan Jinqi, Sun Pingping, Bian Jing, Yan Xue, Zhang Weimin. Gastrodin intervention attenuates inflammatory injury in ischemic stroke rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(28): 4535-4540. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||