Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (32): 5243-5248.doi: 10.12307/2024.495
High tibial osteotomy promotes cartilage regeneration in the treatment of knee osteoarthritis
Fu Qiangchang1, 2, 3, 4, Zheng Liming1, 2, 3, 4, Jiang Lifeng1, 2, 3, 4
- 1Department of Orthopedic Surgery, the Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou 310052, Zhejiang Province, China; 2Orthopedics Research Institute of Zhejiang University, Hangzhou 310052, Zhejiang Province, China; 3Key Laboratory of Motor System Disease Research and Precision Therapy of Zhejiang Province, Hangzhou 310052, Zhejiang Province, China; 4Clinical Research Center of Motor System Disease of Zhejiang Province, Hangzhou 310052, Zhejiang Province, China
-
Received:2023-08-02Accepted:2023-09-25Online:2024-11-18Published:2023-12-29 -
Contact:Jiang Lifeng, MD, Associate chief physician, Department of Orthopedic Surgery, the Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou 310052, Zhejiang Province, China; Orthopedics Research Institute of Zhejiang University, Hangzhou 310052, Zhejiang Province, China; Key Laboratory of Motor System Disease Research and Precision Therapy of Zhejiang Province, Hangzhou 310052, Zhejiang Province, China; Clinical Research Center of Motor System Disease of Zhejiang Province, Hangzhou 310052, Zhejiang Province, China Zheng Liming, MD, Researher, Department of Orthopedic Surgery, the Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou 310052, Zhejiang Province, China; Orthopedics Research Institute of Zhejiang University, Hangzhou 310052, Zhejiang Province, China; Key Laboratory of Motor System Disease Research and Precision Therapy of Zhejiang Province, Hangzhou 310052, Zhejiang Province, China; Clinical Research Center of Motor System Disease of Zhejiang Province, Hangzhou 310052, Zhejiang Province, China -
About author:Fu Qiangchang, Master candidate, Department of Orthopedic Surgery, the Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou 310052, Zhejiang Province, China; Orthopedics Research Institute of Zhejiang University, Hangzhou 310052, Zhejiang Province, China; Key Laboratory of Motor System Disease Research and Precision Therapy of Zhejiang Province, Hangzhou 310052, Zhejiang Province, China; Clinical Research Center of Motor System Disease of Zhejiang Province, Hangzhou 310052, Zhejiang Province, China -
Supported by:National Natural Science Foundation of China (Youth Project), No. 81801371 (to JLF); China Postdoctoral Science Foundation (General Project), No. 2022M722769 (to ZLM)
CLC Number:
Cite this article
Fu Qiangchang, Zheng Liming, Jiang Lifeng. High tibial osteotomy promotes cartilage regeneration in the treatment of knee osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5243-5248.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
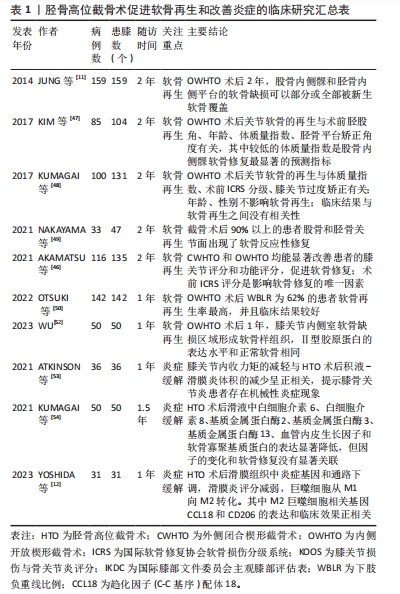
正常膝关节的内侧间室承受了60%-80%的压缩负荷,而下肢力线的微小改变会使应力负荷再分布,从而导致局部异常应力微环境,通过机械信号转导激活细胞内一系列级联生化反应,导致软骨基质的分解和炎症反应的加剧。在一项针对Kellgren-Lawrence分级1/2级的膝骨关节炎患者为期24个月随访中发现,胫骨内侧近端角每降低1°,内侧间室狭窄进展的概率增加了21%[13]。同时,下肢力线异常可导致软骨下骨发生骨质病变,包括微骨折伴骨碎片、坏死、纤维化,进而通过软骨下骨-软骨轴加速软骨的退行性改变[14]。总的来说,下肢力线的异常是导致软骨磨损、退变的重要原因,深入理解软骨细胞对异常力学微环境的响应对治疗膝骨关节炎具有重要的科学价值。 2.1 软骨细胞外基质与力学微环境的研究进展 关节软骨处在复杂的力学微环境中,受到多种动态力学刺激,主要包括压缩、剪切以及旋转应力。合适的应力水平可通过调节软骨基质合成-分解代谢、局部炎症水平以维持关节软骨稳态。过度的应力负荷打破软骨基质代谢和促炎-抗炎平衡,导致膝骨关节炎的发生和进展[15]。膝关节活动时软骨不同层面的机械应力各不相同,阐明软骨的组织结构有助于深入了解软骨细胞所处的力学微环境。 关节软骨的细胞外基质结构和组成并不是均匀的,而是通过高度差异化的结构以实现降低摩擦和承载负荷的功能。软骨从关节软骨表面向软骨下骨可以分为浅表区、中间区、深层区3个部分。在浅表区,小而扁平的软骨细胞和横向分布的Ⅱ型胶原纤维分散关节活动中的剪切力,并分泌润滑素实现润滑;中间区同时承受压缩力和剪切力,因此Ⅱ型胶原纤维通过随机排列以抵抗来自多个方向的力;在深层区,软骨细胞更大更圆,分泌数量更多的细胞外基质,Ⅱ型胶原纤维更厚且垂直于关节表面排列以抵抗压缩负荷,蛋白多糖浓度更高以保留水分和发挥减震作用[16]。除了力的大小和频率,细胞外基质的完整性在负荷是否启动软骨细胞的细胞外基质分解代谢中发挥重要作用。 从微观角度,软骨主要由细胞外基质和软骨细胞构成,占组织体积5%的软骨细胞分散在高度结构化的细胞外基质中。细胞外基质由Ⅱ型胶原蛋白网络和充填其中的蛋白聚糖(如聚集聚糖、润滑素、串珠素)和糖胺聚糖(如透明质酸)组成[17]。近年研究表明,靠近软骨细胞周围2-4 μm区域中存在一类特殊的细胞外基质,其特征是富含Ⅵ型胶原蛋白且不含Ⅱ型胶原蛋白,同时含有基底膜蛋白多糖、聚集蛋白聚糖、透明质酸、双糖链蛋白多糖、Ⅸ型胶原蛋白、层粘连蛋白和纤连蛋白等成分,被称为细胞周基质。细胞周基质与软骨细胞形成了相互联系的结构、功能和代谢单元,称为“软骨单元”。研究表明细胞周基质的变性是膝骨关节炎中最早发生的事件之一,其破坏可以作为膝骨关节炎启动或进展的关键事件[18]。由于其独特的组成和结构,细胞周基质比邻近的细胞外基质硬度低一个数量级左右,在应力载荷下往往优先压缩[19]。细胞周基质作为一种桥梁,在应力从细胞外基质到软骨细胞的传递中起着关键调节作用。研究表明,细胞周基质一方面在高组织应力的软骨浅表区中降低驻留的软骨细胞应力,一方面在低组织应力的软骨深层区中放大驻留的软骨细胞应力[20]。细胞周基质通过自身的应力调节作用使得细胞水平的应力在整个软骨组织中更加均匀。此外,细胞周基质的形变会改变软骨细胞周围微环境中的流体流动和渗透应力,从而激活软骨细胞的机械传感器,调节软骨细胞的生理活动。除了调节软骨细胞的力学微环境外,细胞周基质还作为软骨细胞所需的生长因子和调节因子的存储库,存储成纤维细胞生长因子2、转化生长因子β、结缔组织生长因子等。当生理水平的应力作用于细胞周基质时,其响应性释放存储的细胞因子作用于软骨细胞,促进损伤软骨的修复[21]。因此,给予软骨细胞合适的力学微环境,有助于延缓甚至逆转膝骨关节炎软骨损伤的进展。 2.2 软骨细胞对力学微环境的响应机制 如上所述,软骨细胞处于一个动态的应力环境中,为了识别和响应应力刺激,细胞外应力需要通过机械传感器转换为细胞内的生化信号或电信号以诱导细胞反应。机械传感器是力学信号转换为生化信号或电信号的关键组成部分,目前参与软骨细胞机械信号转导的主要传感器包括初级纤毛、整合素、细胞骨架和核骨架、机械敏感离子通道和线粒体[22-24]。由于机械应力负荷必须通过机械传感器才能诱导细胞功能的改变,因此作者根据机械传感器的种类分别总结其在应力调控软骨细胞中的作用,关注机械传感器在骨关节炎发生、发展中的研究现状。 2.2.1 初级纤毛-整合素系统 初级纤毛是一种细胞骨架细胞器,从细胞表面发出后伸入周围的细胞周基质中,在机械应力作用下会发生弯曲、形变,具有机械敏感性[22]。当软骨细胞遭受过度的周期性压缩应力时,初级纤毛的数量和长度呈可逆性减少,这种变化在解除应力后能得到逆转[25]。细胞水平研究发现,纤毛的存在是应力刺激调控软骨细胞的细胞外基质合成-分解代谢的关键[26]。THOMPSON等[27]也发现,只有在完整的纤毛存在时,10%循环拉伸应变才能诱导血小板反应蛋白解整合素金属肽酶5(a disintegrin and metalloproteinase with thrombospondin motifs 5,ADAMTS-5)的表达。当应变幅度达到20%,软骨细胞过度拉伸将引起初级纤毛拆卸,从而导致应变无法诱导ADAMTS-5的上调。在转基因小鼠中特异性敲除软骨细胞中的初级纤毛,软骨组织厚度增加但硬度降低,且膝骨关节炎的生物学标记物(如X型胶原蛋白、Runt相关转录因子2、基质金属蛋白酶13和ADAMTS-5)显著性上升[28]。 整合素存在于软骨细胞的初级纤毛上,将细胞内细胞骨架连接到细胞外的细胞周基质上,由此参与机械刺激转导。研究表明,机械压力可通过整合素依赖性途径激活FAK/ERK/PI3K等多条信号通路,保护软骨细胞免于凋亡[29]。另外,机械刺激可诱导软骨细胞通过整合素途径提高白细胞介素4的分泌,从而抑制白细胞介素1β介导的软骨基质降解[30]。而过高的机械应力则可通过刺激骨架重组从而触发αV整合素介导的转化生长因子β活化,导致转化生长因子β在机械应力集中的软骨区域表达高活性,促进局部的软骨损伤[31]。 2.2.2 细胞骨架与核骨架 软骨细胞周围的机械负荷会直接引起细胞形变,导致由细胞微管、微丝和中间纤维组成的细胞骨架和核骨架结构的变化,通过间接影响细胞器和直接作用于细胞骨架相关分子2种方式调节细胞的活动。 关于机械应力通过细胞骨架间接影响的细胞器主要有细胞核和线粒体2种。一方面,细胞核受细胞骨架结构的动态变化影响,通过核骨架介导的信号传导进一步影响软骨细胞DNA浓度和基因表达谱,调节软骨基质的代谢。例如,应力压迫导致软骨细胞变形、细胞核体积变小,改变软骨基质的代谢平衡[32]。最近REYNOLDS等[33]发现将软骨细胞中细胞骨架的收缩力增加到与成纤维细胞相关的水平,软骨细胞会出现部分成纤维细胞的表型,从而导致细胞外基质的异常矿化。另一方面,线粒体也可响应机械应力的刺激以激活一系列下游信号通路。机械负荷既可通过细胞骨架引起线粒体变形,又可产生高动能导致急性线粒体功能障碍和细胞呼吸改变,诱导软骨细胞损伤。JIANG等[24]发现生理负荷刺激软骨细胞线粒体产生少量活性氧,参与细胞内信号传导。然而,过度的机械负荷会损害呼吸链复合功能,导致线粒体释放大量活性氧并下调超氧化物歧化酶2表达,破坏氧化还原平衡,导致软骨细胞凋亡和软骨变性,进而导致软骨损伤。 除了间接影响重要细胞器的功能外,机械应力还可以直接影响细胞骨架相关的途径和分子,调控软骨细胞的功能。例如软骨细胞的循环压缩应变会导致肌动蛋白解聚,而压缩应变或静水压力都会诱导肌动蛋白细胞骨架可逆性重塑,进而导致Ⅱ型胶原分泌的增加[34]。过载应力(细胞收缩力> 40 pN)则被证明可激活肌动蛋白介导的转化生长因子β过表达,诱导骨关节炎软骨变性[31]。总的来说,机械应力可以通过影响细胞骨架/核骨架可逆性调控膝骨关节炎的进展。 2.2.3 机械门控型钙离子通道 机械门控型钙离子通道作为软骨细胞中的经典机械传感器,在机械刺激下发生构象变化,导致作为第二信使的Ca2+内流,参与细胞行为的调控[35],常见的有TRPV4和Piezo通道2种。TRPV4主要调节细胞内Ca2+来转导软骨细胞感受到的机械负荷和渗透压刺激[36-37]。适度机械负荷通过激活TRPV4增强软骨细胞合成代谢并抑制分解代谢基因表达,促进基质的积累和机械性能的增强,同时抑制白细胞介素1β介导的一氧化氮和前列腺素E2释放来调节骨关节炎的炎症反应,从而起到治疗膝骨关节炎的作用[36,38]。过度的机械应力则可以通过TRPV4诱导软骨细胞凋亡[39]。与主要通过响应生理负荷诱导软骨细胞合成代谢反应的TRPV4不同,Piezo机械敏感离子通道与有害机械刺激的机械转导有关[40]。Piezo通道中的Piezo1和Piezo2都存在于软骨细胞中,它们通过协同作用在机械应力超载的情况下导软骨降解和炎症[41]。机械应力升高会激活Piezo1和Piezo2,导致细胞内钙离子达到毒性浓度,过度激活下游途径,诱导细胞凋亡和软骨细胞降解[40,42]。过度的机械拉伸通过上调Piezo1表达导致软骨细胞周期停滞并最终抑制软骨细胞增殖[43]。 2.3 胫骨高位截骨术通过纠正下肢力线促进软骨再生和改善炎症的研究现状 2.3.1 单纯胫骨高位截骨术即可促进膝骨关节炎患者软骨再生和炎症改善 胫骨高位截骨术主要用于治疗以内侧间室退变为主的膝骨关节炎。该手术通过针对胫骨的截骨矫形,纠正下肢力线,在内外侧关节间室重新分配应力负荷,从而起到治疗膝骨关节炎的效果。手术的主要目的都是恢复下肢冠状面上的理想机械轴,使下肢力线从髋关节中心穿过膝关节到达踝关节中心[44]。相比较全膝关节置换术和单髁膝关节置换术,胫骨高位截骨术保留了膝关节的结构,改善了膝关节的活动范围[45]。目前多项研究已证明,在矫正下肢力线后的患者中,异常应力负荷的去除改善了力学微环境,在改善患者症状的同时观察到了受损软骨的再生和炎症的缓解,见表1[11-12,46-54]。据JUNG等[11]报道,在利用胫骨高位截骨术充分矫正内翻畸形后2年,无需额外的软骨再生策略,原磨损区域可被再生纤维软骨覆盖。后续的多项研究也证明了仅行胫骨高位截骨术改变内侧关节的异常负荷就会激活软骨的反应性修复,在软骨缺损处产生纤维软骨[46-52]。外侧闭合楔形截骨术和内侧开放楔形截骨术在促进软骨再生上都能取得良好的效果[46]。影响术后关节软骨再生的因素包括术前胫股角、术前ICRS分级、体质量指数、年龄和术后力线矫正程度[11,47-48,50]。除了促进软骨再生,胫骨高位截骨术还可以改善膝关节的炎症反应[54]。ATKINSON等[53]通过检测行走时膝关节内收力矩以观察膝关节内侧负荷,同时利用核磁共振检测膝关节髌上积液-滑膜炎体积表示关节炎症水平,证明了膝关节内侧负荷的减少与关节腔内炎症显著相关。近期研究表明,这种变化可能与巨噬细胞M1/M2的极化相关[12]。 "
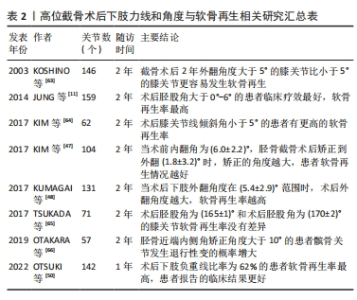
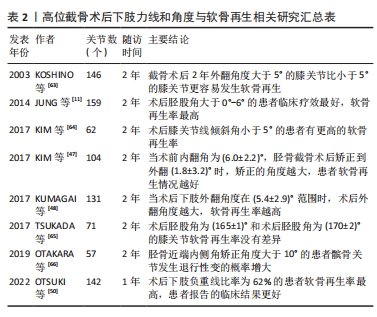
2.3.2 准确的力线纠正是实现软骨再生的必要条件 前人研究证明,下肢力线的矫正准确度与患者症状改善、术后软骨再生具有显著的相关性。术后胫股角大于0°且小于6°,术后下肢负重线比率在62%更有利于患者软骨再生[11,48,50,55]。目前认为术后3°-6°外翻能达到最好的治疗效果,过度矫正会导致功能结局恶化和外侧间室退化,矫正不足则不能解决内侧间室的问题。正常的胫骨平台关节面有平均10°的后倾,其中内侧后倾约12°,外侧后倾约7°。胫骨平台后倾角会影响膝关节活动和矢状面的稳定性。因此术中应尽量不改变其角度,保留胫骨平台生理性倾斜。正常的膝关节线为0°,手术导致关节线倾斜角过大会增加关节面的剪切力,降低关节面的软骨再生率[56]。影响力线纠正的准确度主要取决于术前规划、手术方式和截骨精度3个方面。术前规划截骨的最佳角度目前尚缺乏成熟的方法,基于人工智能的术前规划是未来可能的方向。胫骨高位截骨术最常见的手术方式包括外侧闭合楔形截骨术和内侧开放楔形截骨术,两种手术方式都能有效纠正下肢力线,同时也会改变膝关节各组成部分的空间关系。在力线纠正精度上,通常认为外侧闭合楔形截骨术力线纠正仅局限于单个平面,矫正精度较低,而内侧开放楔形截骨术矫正精度高,而且有机会进行双平面下肢力线纠正[57]。无论使用哪种手术方式,精准截骨对于术后力线纠正的准确度都是必不可少的,应用计算机辅助技术、定制截骨导板、手术机器人系统可提高手术精确性,提高力线矫正的准确性,是未来的发展趋势[58-62],相关研究总结见表2[11,47-48,50,63-66]。"
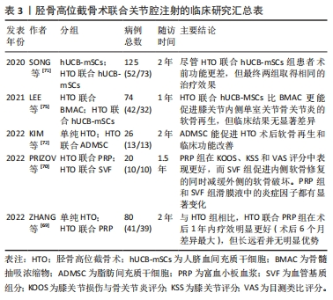
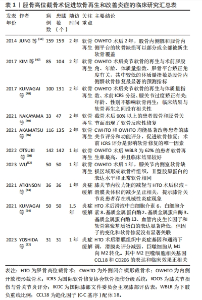
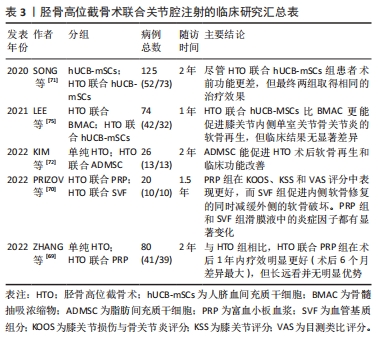
2.3.3 胫骨高位截骨术联合关节腔内注射/关节镜技术促进软骨再生的研究进展 尽管单独应用胫骨高位截骨术即可促进软骨再生,但再生的纤维软骨的生物力学特性和功能远不如正常透明软骨[11]。而且软骨再生耗费时间较长、覆盖面积小,受限于患者年龄、体质量指数、术前关节损坏程度,严重影响了胫骨高位截骨术的适用范围[46-48]。如前所述,通过改善力学微环境,胫骨高位截骨术可为软骨再生和修复创造良好的理化环境,以实现结构和功能恢复。因此,胫骨高位截骨术联合软骨再生策略具有重要价值。研究表明,胫骨高位截骨术联合关节镜技术可有效缩短患者的住院时间、缓解疼痛、促进软骨再生,同时降低血清和滑膜液炎症因子白细胞介素1β、白细胞介素6和白细胞介素17水平[52,67-68]。 除了联合关节镜技术外,胫骨高位截骨术联合关节腔内注射透明质酸、富血小板血浆、血管基质组分、干细胞等亦受到了广泛重视。胫骨高位截骨术联合关节内注射富血小板血浆、血管基质组分能提高术后内侧膝关节间隙,缓解疼痛和改善关节活动范围[69-70]。与单纯应用胫骨高位截骨术相比,联合应用同种异体人脐血间充质干细胞、脂肪间充质干细胞、骨髓间充质干细胞都提升了患者的治疗效果[71-72]。有报道称,胫骨高位截骨术联合关节内注射自体外周血干细胞后,获得的再生软骨的质量与天然关节透明软骨非常相似[73]。除了促进关节软骨的再生与修复,关节腔内注射也可以缓解关节炎症[70,71,74]。PRIZOV等[70]发现富血小板血浆和血管基质组分联合胫骨高位截骨术治疗能显著改变滑膜液的细胞因子浓度。随着新的材料、药物科学的进步,促软骨再生、调控局部炎症的药物不断涌现,联合药物治疗的胫骨高位截骨术有望成为全新的抗膝骨关节炎治疗策略,见表3[69-72,75]。"
| [1] LOESER RF, COLLINS JA, DIEKMAN BO. Ageing and the pathogenesis of osteoarthritis. Nat Rev Rheumatol. 2016;12(7):412-420. [2] SANCHEZ-LOPEZ E, CORAS R, TORRES A, et al. Synovial inflammation in osteoarthritis progression. Nat Rev Rheumatol. 2022;18(5):258-275. [3] SAKELLARIOU G, CONAGHAN PG, ZHANG W, et al. EULAR recommendations for the use of imaging in the clinical management of peripheral joint osteoarthritis. Ann Rheum Dis. 2017;76(9):1484-1494. [4] HUNTER DJ, BIERMA-ZEINSTRA S. Osteoarthritis. Lancet. 2019;393(10182):1745-1759. [5] KATZ JN, ARANT KR, LOESER RF. Diagnosis and Treatment of Hip and Knee Osteoarthritis: A Review. JAMA. 2021;325(6):568-578. [6] CAO Y, LUO J, HAN S,et al. A model-based quantitative analysis of efficacy and associated factors of platelet rich plasma treatment for osteoarthritis. Int J Surg. 2023;109(6):1742-1752. [7] TSCHOPP M, PFIRRMANN CWA, FUCENTESE SF, et al. A Randomized Trial of Intra-articular Injection Therapy for Knee Osteoarthritis. Invest Radiol. 2023;58(5):355-362. [8] BENNELL KL, PATERSON KL, METCALF BR, et al. Effect of Intra-articular Platelet-Rich Plasma vs Placebo Injection on Pain and Medial Tibial Cartilage Volume in Patients With Knee Osteoarthritis: The RESTORE Randomized Clinical Trial. JAMA. 2021;326(20):2021-2030. [9] HUANG Y, LOBENHOFFER P, JIANG XY. Development of knee-preserving osteotomy in China. Sci Bull (Beijing). 2023;68(2):125-128. [10] WU ZX, REN WX, WANG ZQ. Proximal fibular osteotomy versus high tibial osteotomy for treating knee osteoarthritis: a systematic review and meta-analysis. J Orthop Surg Res. 2022;17(1):470. [11] JUNG WH, TAKEUCHI R, CHUN CW, et al. Second-look arthroscopic assessment of cartilage regeneration after medial opening-wedge high tibial osteotomy. Arthroscopy. 2014;30(1):72-79. [12] YOSHIDA S, NISHITANI K, YOSHITOMI H, et al. Knee Alignment Correction by High Tibial Osteotomy Reduces Symptoms and Synovial Inflammation in Knee Osteoarthritis Accompanied by Macrophage Phenotypic Change From M1 to M2. Arthritis Rheumatol. 2023;75(6):950-960. [13] PALMER JS, JONES LD, MONK AP, et al. Varus alignment of the proximal tibia is associated with structural progression in early to moderate varus osteoarthritis of the knee. Knee Surg Sports Traumatol Arthrosc. 2020;28(10):3279-3286. [14] HAYASHI D, ENGLUND M, ROEMER FW, et al. Knee malalignment is associated with an increased risk for incident and enlarging bone marrow lesions in the more loaded compartments: the MOST study. Osteoarthritis Cartilage. 2012;20(11): 1227-1233. [15] CHANG SH, MORI D, KOBAYASHI H, et al. Excessive mechanical loading promotes osteoarthritis through the gremlin-1-NF-κB pathway. Nat Commun. 2019;10(1):1442. [16] POOLE AR, KOJIMA T, YASUDA T, et al. Composition and structure of articular cartilage: a template for tissue repair. Clin Orthop Relat Res. 2001;(391 Suppl):S26-33. [17] HODGKINSON T, KELLY DC, CURTIN CM, et al. Mechanosignalling in cartilage: an emerging target for the treatment of osteoarthritis. Nat Rev Rheumatol. 2022; 18(2):67-84. [18] CHERY DR, HAN B, LI Q, et al. Early changes in cartilage pericellular matrix micromechanobiology portend the onset of post-traumatic osteoarthritis. Acta Biomater. 2020;111:267-278. [19] GUILAK F, ALEXOPOULOS LG, UPTON ML, et al. The pericellular matrix as a transducer of biomechanical and biochemical signals in articular cartilage. Ann N Y Acad Sci. 2006;1068:498-512. [20] CHOI JB, YOUN I, CAO L, et al. Zonal changes in the three-dimensional morphology of the chondron under compression: the relationship among cellular, pericellular, and extracellular deformation in articular cartilage. J Biomech. 2007;40(12):2596-2603. [21] TANG X, MUHAMMAD H, MCLEAN C, et al. Connective tissue growth factor contributes to joint homeostasis and osteoarthritis severity by controlling the matrix sequestration and activation of latent TGFβ. Ann Rheum Dis. 2018;77(9):1372-1380. [22] TAO F, JIANG T, TAO H, et al. Primary cilia: Versatile regulator in cartilage development. Cell Prolif. 2020;53(3):e12765. [23] MOBASHERI A, MATTA C, UZIELIENÈ I, et al. The chondrocyte channelome: A narrative review. Joint Bone Spine. 2019;86(1):29-35. [24] JIANG W, LIU H, WAN R, et al. Mechanisms linking mitochondrial mechanotransduction and chondrocyte biology in the pathogenesis of osteoarthritis. Ageing Res Rev. 2021;67:101315. [25] MCGLASHAN SR, KNIGHT MM, CHOWDHURY TT, et al. Mechanical loading modulates chondrocyte primary cilia incidence and length. Cell Biol Int. 2010; 34(5):441-446. [26] WANN AK, ZUO N, HAYCRAFT CJ, et al. Primary cilia mediate mechanotransduction through control of ATP-induced Ca2+ signaling in compressed chondrocytes. FASEB J. 2012;26(4):1663-1671. [27] THOMPSON CL, CHAPPLE JP, KNIGHT MM. Primary cilia disassembly down-regulates mechanosensitive hedgehog signalling: a feedback mechanism controlling ADAMTS-5 expression in chondrocytes. Osteoarthritis Cartilage. 2014;22(3):490-498. [28] IRIANTO J, RAMASWAMY G, SERRA R, et al. Depletion of chondrocyte primary cilia reduces the compressive modulus of articular cartilage. J Biomech. 2014; 47(2):579-582. [29] MA D, KOU X, JIN J, et al. Hydrostatic Compress Force Enhances the Viability and Decreases the Apoptosis of Condylar Chondrocytes through Integrin-FAK-ERK/PI3K Pathway. Int J Mol Sci. 2016;17(11):1847. [30] MILLWARD-SADLER SJ, WRIGHT MO, Lee H, et al. Integrin-regulated secretion of interleukin 4: A novel pathway of mechanotransduction in human articular chondrocytes. J Cell Biol. 1999;145(1):183-189. [31] ZHEN G, GUO Q, LI Y, et al. Mechanical stress determines the configuration of TGFβ activation in articular cartilage. Nat Commun. 2021;12(1):1706. [32] LEE DA, KNIGHT MM, BOLTON JF, et al. Chondrocyte deformation within compressed agarose constructs at the cellular and sub-cellular levels. J Biomech. 2000;33(1):81-95. [33] REYNOLDS N, MCEVOY E, GHOSH S, et al. Image-derived modeling of nucleus strain amplification associated with chromatin heterogeneity. Biophys J. 2021; 120(8):1323-1332. [34] LAUER JC, SELIG M, HART ML, et al. Articular Chondrocyte Phenotype Regulation through the Cytoskeleton and the Signaling Processes That Originate from or Converge on the Cytoskeleton: Towards a Novel Understanding of the Intersection between Actin Dynamics and Chondrogenic Function. Int J Mol Sci. 2021;22(6):3279. [35] DELCO ML, BONASSAR LJ. Targeting calcium-related mechanotransduction in early OA. Nat Rev Rheumatol. 2021;17(8):445-446. [36] O’CONOR CJ, LEDDY HA, BENEFIELD HC, et al. TRPV4-mediated mechanotransduction regulates the metabolic response of chondrocytes to dynamic loading. Proc Natl Acad Sci U S A. 2014;111(4):1316-1321. [37] CLARK AL, VOTTA BJ, KUMAR S, et al. Chondroprotective role of the osmotically sensitive ion channel transient receptor potential vanilloid 4: age- and sex-dependent progression of osteoarthritis in Trpv4-deficient mice. Arthritis Rheum. 2010;62(10):2973-2983. [38] FU S, MENG H, INAMDAR S, et al. Activation of TRPV4 by mechanical, osmotic or pharmaceutical stimulation is anti-inflammatory blocking IL-1β mediated articular cartilage matrix destruction. Osteoarthritis Cartilage. 2021;29(1):89-99. [39] XU B, XING R, HUANG Z, et al. Excessive mechanical stress induces chondrocyte apoptosis through TRPV4 in an anterior cruciate ligament-transected rat osteoarthritis model. Life Sci. 2019;228:158-166. [40] DU G, LI L, ZHANG X, et al. Roles of TRPV4 and piezo channels in stretch-evoked Ca2+ response in chondrocytes. Exp Biol Med (Maywood). 2020;245(3):180-189. [41] LEE W, LEDDY HA, CHEN Y, et al. Synergy between Piezo1 and Piezo2 channels confers high-strain mechanosensitivity to articular cartilage. Proc Natl Acad Sci U S A. 2014;111(47):E5114-E5122. [42] LEE W, GUILAK F, LIEDTKE W. Role of Piezo Channels in Joint Health and Injury. Curr Top Membr. 2017;79:263-273. [43] SUN Y, LENG P, LI D, et al. Mechanism of Abnormal Chondrocyte Proliferation Induced by Piezo1-siRNA Exposed to Mechanical Stretch. Biomed Res Int. 2020; 2020:8538463. [44] AMIS AA. Biomechanics of high tibial osteotomy. Knee Surg Sports Traumatol Arthrosc. 2013;21(1):197-205. [45] CAO Z, MAI X, WANG J, et al. Unicompartmental Knee Arthroplasty vs High Tibial Osteotomy for Knee Osteoarthritis: A Systematic Review and Meta-Analysis. J Arthroplasty. 2018;33(3):952-959. [46] AKAMATSU T, KUMAGAI K, YAMADA S, et al. No differences in clinical outcomes and cartilage repair between opening wedge and closed wedge high tibial osteotomies at short-term follow-up: A retrospective case series analysis. J Orthop Surg (Hong Kong). 2021;29(2):23094990211020366. [47] KIM KI, SEO MC, SONG SJ, et al. Change of Chondral Lesions and Predictive Factors After Medial Open-Wedge High Tibial Osteotomy With a Locked Plate System. Am J Sports Med. 2017;45(7):1615-1621. [48] KUMAGAI K, AKAMATSU Y, KOBAYASHI H, et al. Factors affecting cartilage repair after medial opening-wedge high tibial osteotomy. Knee Surg Sports Traumatol Arthrosc. 2017;25(3):779-784. [49] NAKAYAMA H, KANTO R, ONISHI S, et al. Cartilage repair examined by second-look arthroscopy following double-level osteotomy performed for osteoarthritic knees with severe varus deformity. Knee. 2021;29:411-417. [50] OTSUKI S, IKEDA K, ISHITANI T, et al. Impact of the Weightbearing Line on Cartilage Regeneration of the Medial Knee Compartment after Open-Wedge High Tibial Osteotomy, Based on Second-Look Arthroscopy. Cartilage. 2022;13(4):87-93. [51] SPAHN G, KLINGER HM, HARTH P, et al. Knorpelregeneration nach valgisierender Tibiakopfosteotomie. Ergebnisse einer arthroskopischen Studie [Cartilage regeneration after high tibial osteotomy. Results of an arthroscopic study]. Z Orthop Unfall. 2012;150(3):272-279. [52] WU J, ZHAO B, LUO W, et al. Arthroscopy combined with high tibial osteotomy promotes cartilage regeneration in osteoarthritis. J Orthop Surg (Hong Kong). 2023;31(1):10225536231165357. [53] ATKINSON HF, BIRMINGHAM TB, PRIMEAU CA, et al. Association between changes in knee load and effusion-synovitis: evidence of mechano-inflammation in knee osteoarthritis using high tibial osteotomy as a model. Osteoarthritis Cartilage. 2021;29(2):222-229. [54] KUMAGAI K, FUJIMAKI H, YAMADA S, et al. Changes of synovial fluid biomarker levels after opening wedge high tibial osteotomy in patients with knee osteoarthritis. Osteoarthritis Cartilage. 2021;29(7):1020-1028. [55] 张润杰,王浩浩,芦升升,等.胫骨高位截骨术后软骨再生影响因素的研究进展[J].实用骨科杂志,2021,27(2):153-156. [56] NAKAYAMA H, SCHRÖTER S, YAMAMOTO C, et al. Large correction in opening wedge high tibial osteotomy with resultant joint-line obliquity induces excessive shear stress on the articular cartilage. Knee Surg Sports Traumatol Arthrosc. 2018;26(6):1873-1878. [57] DENG Y, YUAN Y, REN Y, et al. Differences between open-wedge versus closed-wedge high tibial osteotomy on clinical and radiological outcomes and adverse events. Int J Clin Exp Med. 2018;11(5):4371-4388. [58] JACQUET C, SHARMA A, FABRE M, et al. Patient-specific high-tibial osteotomy’s ‘cutting-guides’ decrease operating time and the number of fluoroscopic images taken after a Brief Learning Curve. Knee Surg Sports Traumatol Arthrosc. 2020;28(9):2854-2862. [59] PREDESCU V, GROSU AM, GHERMAN I, et al. Early experience using patient-specific instrumentation in opening wedge high tibial osteotomy. Int Orthop. 2021;45(6):1509-1515. [60] CERCIELLO S, OLLIVIER M, CORONA K, et al. CAS and PSI increase coronal alignment accuracy and reduce outliers when compared to traditional technique of medial open wedge high tibial osteotomy: a meta-analysis. Knee Surg Sports Traumatol Arthrosc. 2022;30(2):555-566. [61] 范少勇,龚礼,周明,等.3D打印截骨导板辅助开放楔形胫骨高位截骨治疗膝骨性关节炎[J].江西中医药,2023,54(8):43-45. [62] 李军,梁帅,王健.模拟手术结合3D打印个性化导航模板在胫骨高位截骨术中的应用[J].中国临床解剖学杂志,2023,41(3):354-359. [63] KOSHINO T, WADA S, ARA Y, et al. Regeneration of degenerated articular cartilage after high tibial valgus osteotomy for medial compartmental osteoarthritis of the knee. Knee. 2003;10(3):229-236. [64] KIM CW, SEO SS, LEE CR, et al. Factors affecting articular cartilage repair after open-wedge high tibial osteotomy. Knee. 2017;24(5):1099-1107. [65] TSUKADA S, WAKUI M. Is overcorrection preferable for repair of degenerated articular cartilage after open-wedge high tibial osteotomy? Knee Surg Sports Traumatol Arthrosc. 2017;25(3):785-792. [66] OTAKARA E, NAKAGAWA S, ARAI Y, et al. Large deformity correction in medial open-wedge high tibial osteotomy may cause degeneration of patellofemoral cartilage: A retrospective study. Medicine (Baltimore). 2019;98(5):e14299. [67] ZHAO B, XIAO Q, LIU B. Effects of High Tibial Osteotomy Combined with Arthroscopy on Pain and Inflammation Markers in Patients with Medial Knee Osteoarthritis. J Invest Surg. 2022;35(4):891-897. [68] LI Q, WANG H, WANG D. Effects of high tibial osteotomy combined with arthroscopy on local inflammation degree and gait activity index in patients with medial knee osteoarthritis. Pak J Med Sci. 2023;39(1):161-165. [69] ZHANG Q, XU W, WU K, et al. Intra-articular Pure Platelet-Rich Plasma Combined With Open-Wedge High Tibial Osteotomy Improves Clinical Outcomes and Minimal Joint Space Width Compared With High Tibial Osteotomy Alone in Knee Osteoarthritis: A Prospective Study. Arthroscopy. 2022;38(2):476-485. [70] PRIZOV A, TCHETINA E, EREMIN I, et al. Differences in Synovial Cytokine Profile Associated with Long-Term Clinical Outcomes in Patients with Knee Osteoarthritis Undergoing Corrective Osteotomy with Platelet-Rich Plasma or Stromal Vascular Fraction Post-Treatments. Int J Mol Sci. 2022;23(21):12835. [71] SONG JS, HONG KT, KONG CG, et al. High tibial osteotomy with human umbilical cord blood-derived mesenchymal stem cells implantation for knee cartilage regeneration. World J Stem Cells. 2020;12(6):514-526. [72] KIM JH, KIM KI, YOON WK, et al. Intra-articular Injection of Mesenchymal Stem Cells After High Tibial Osteotomy in Osteoarthritic Knee: Two-Year Follow-up of Randomized Control Trial. Stem Cells Transl Med. 2022;11(6):572-585. [73] SAW KY, ANZ A, JEE CS, et al. High Tibial Osteotomy in Combination With Chondrogenesis After Stem Cell Therapy: A Histologic Report of 8 Cases. Arthroscopy. 2015;31(10):1909-1920. [74] WONG KL, LEE KB, TAI BC, et al. Injectable cultured bone marrow-derived mesenchymal stem cells in varus knees with cartilage defects undergoing high tibial osteotomy: a prospective, randomized controlled clinical trial with 2 years’ follow-up. Arthroscopy. 2013;29(12):2020-2028. [75] LEE NH, NA SM, AHN HW, et al. Allogenic Human Umbilical Cord Blood-Derived Mesenchymal Stem Cells Are More Effective Than Bone Marrow Aspiration Concentrate for Cartilage Regeneration After High Tibial Osteotomy in Medial Unicompartmental Osteoarthritis of Knee. Arthroscopy. 2021;37(8):2521-2530. |
| [1] | Yang Yifeng, Ye Nan, Wang Lin, Guo Shuaicheng, Huang Jian. Signaling pathway of dexmedetomidine against ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1464-1469. |
| [2] | Li Zhifei, Yang Yin, Chen Hualong, Liang Qinqiu, Zhong Yuanming, Zhang Yisheng. Finite element analysis of the correlation between tilt angle of titanium cage and postoperative subsidence of titanium cage after anterior subtotal cervical corpectomy, decompression and fusion [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1313-1319. |
| [3] | Chen Mengmeng, Bao Li, Chen Hao, Jia Pu, Feng Fei, Shi Guan, Tang Hai. Biomechanical characteristics of a novel interspinous distraction fusion device BacFuse for the repair of lumbar degenerative disease [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1325-1329. |
| [4] | Liang Cheng, Zhang Linqi, Wang Guan, Li Wen, Duan Ke, Li Zhong, Lu Xiaobo, Zhuo Naiqiang. Finite element and biomechanical analysis of different implants in repair for unilateral unstable pelvic posterior ring injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1336-1341. |
| [5] | Yang Junliang, Lu Tan, Xu Biao, Jiang Yaqiong, Wang Fucheng. Three-dimensional finite element analysis of effects of partial anterior cruciate ligament rupture on knee joint stress [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1347-1353. |
| [6] | Li Yongjie, Fu Shenyu, Xia Yuan, Zhang Dakuan, Liu Hongju. Correlation of knee extensor muscle strength and spatiotemporal gait parameters with peak knee flexion/adduction moment in female patients with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1354-1358. |
| [7] | Du Changling, Shi Hui, Zhang Shoutao, Meng Tao, Liu Dong, Li Jian, Cao Heng, Xu Chuang. Efficacy and safety of different applications of tranexamic acid in high tibial osteotomy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1409-1413. |
| [8] | Weng Rui, Lin Dongxin, Guo Haiwei, Zhang Wensheng, Song Yuke, Lin Hongheng, Li Wenchao, Ye Linqiang. Abnormal types of intervertebral disc structure and related mechanical loading with biomechanical factors [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1436-1442. |
| [9] | Wang Weiqing, Zhou Yue. Chronic inflammation regulates adipose tissue fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1307-1312. |
| [10] | Huang Xiarong, Hu Lizhi, Sun Guanghua, Peng Xinke, Liao Ying, Liao Yuan, Liu Jing, Yin Linwei, Zhong Peirui, Peng Ting, Zhou Jun, Qu Mengjian. Effect of electroacupuncture on the expression of P53 and P21 in articular cartilage and subchondral bone of aged rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1174-1179. |
| [11] | Wang Ji, Zhang Min, Li Wenbo, Yang Zhongya, Zhang Long. Effect of aerobic exercise on glycolipid metabolism, skeletal muscle inflammation and autophagy in type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1200-1205. |
| [12] | Han Bing, Liu Hongbin, Wang Hehong, Zhao Hanqing, Zhao Riguang, Sun Yiyan, Zhang Yu. Correlation between lower limb alignment and risk factors of patellofemoral pain syndrome in young men [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1211-1216. |
| [13] | Mu Bingtao, Yu Jingwen, Liu Chunyun, Guo Minfang, Meng Tao, Yang Pengwei, Wei Wenyue, Song Lijuan, Yu Jiezhong, Ma Cungen. Immunomodulatory effect of astragaloside IV on T cells of experimental autoimmune encephalomyelitis mice [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1057-1062. |
| [14] | Wang Qiang, Li Shiyun, Xiong Ying, Li Tiantian. Biomechanical changes of the cervical spine in internal fixation with different anterior cervical interbody fusion systems [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 821-826. |
| [15] | Wei Yuanbiao, Lin Zhan, Chen Yanmei, Yang Tenghui, Zhao Xiao, Chen Yangsheng, Zhou Yanhui, Yang Minchao, Huang Feiqi. Finite element analysis of effects of sagittal cervical manipulation on intervertebral disc and facet joints [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 827-832. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||