Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (32): 5227-5231.doi: 10.12307/2023.814
Previous Articles Next Articles
Mechanism of clock gene Bmal1 in temporomandibular joint osteoarthritis
Du Chendan, Du Bingli, Ning Xiao, Zong Mingrui, Li Bing, Wu Xiuping
- Shanxi Key Laboratory of Prevention and Treatment of Oral Diseases and New Materials, School and Hospital of Stomatology, Shanxi Medical University, Taiyuan 030000, Shanxi Province, China
-
Received:2022-10-11Accepted:2022-11-30Online:2023-11-18Published:2023-03-23 -
Contact:Wu Xiuping, MD, Chief physician, Doctoral supervisor, Shanxi Key Laboratory of Prevention and Treatment of Oral Diseases and New Materials, School and Hospital of Stomatology, Shanxi Medical University, Taiyuan 030000, Shanxi Province, China -
About author:Du Chendan, Master candidate, Shanxi Key Laboratory of Prevention and Treatment of Oral Diseases and New Materials, School and Hospital of Stomatology, Shanxi Medical University, Taiyuan 030000, Shanxi Province, China -
Supported by:the Scientific Research Project of Shanxi Province Returned Overseas Students, No. HGKY2019-055 (to LB); Central Government-Guided Local Funds for Scientific and Technological Development (Transfer of scientific and technological achievements), No. YDZJSX2021C01 (to WXP); the Science and Technology Innovation of Shanxi Provincial Education Department, No. 2020L0211 (to DBL)
CLC Number:
Cite this article
Du Chendan, Du Bingli, Ning Xiao, Zong Mingrui, Li Bing, Wu Xiuping. Mechanism of clock gene Bmal1 in temporomandibular joint osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(32): 5227-5231.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
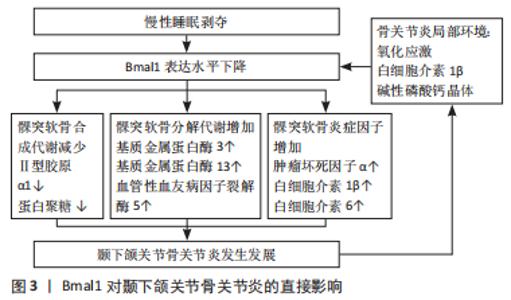
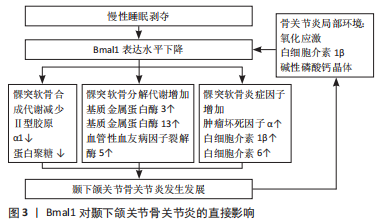
2.1 Bmal1对TMJOA的直接影响 正常的昼夜节律是维持髁突软骨代谢平衡的重要因素,许多研究证实了软骨代谢的昼夜节律变化。1962年SIMMONS[10]发现软骨细胞增殖和生长板软骨代谢的日变化。现已证实生长板软骨细胞在早晨增殖加速,中午达到高峰,导致基质合成增加及生长板扩展[11]。近来ALAGHA等[12]证实软骨形成标记基因遵循昼夜振荡模式,将时钟同步可以显著增加软骨基质的生成,并提高Sox9、蛋白聚糖和Ⅱ型胶原α1基因的表达。Bmal1作为核心昼夜节律因子,在健康的颞下颌关节软骨中呈节律性表达,并参与许多与软骨相关因子的昼夜节律表达[13]。Bmal1缺失会降低磷酸化Smad2/3和Nfatc2及软骨基质相关因子Sox9、蛋白聚糖和Ⅱ型胶原α1的表达水平,但会增加磷酸化的Smad1/5的水平,这证明软骨基质的合成代谢和分解代谢均受时钟基因Bmal1的控制[9]。研究发现,Bmal1的节律性表达与机体的休息-活动周期相结合,猜测这可能是为了将关节的清理阶段(例如基质降解酶)和重建阶段(例如基质降解酶抑制剂)在时间上分开,使两个相反的代谢过程互不干扰,从而有益于关节活动后修复,这一假设在皮肤组织中得到证实[14]。GOSSAN等[15]在一项研究中报道,在软骨中有619个基因(占转录表达基因的3.9%)表现出昼夜节律的表达模式,不仅包括参与软骨稳态的基因,还包括在骨关节炎发病机制中具有潜在作用的基因。研究发现,在骨性关节炎小鼠模型的软骨退化早期数个时钟基因被破坏[15]。同样在TMJOA小鼠模型中Bmal1表达水平降低,提示TMJOA的发生发展与时钟基因Bmal1密切相关[16]。 动物实验证实,慢性睡眠剥夺可以导致大鼠颞下颌关节发生骨关节炎样病理改变,表现为睡眠剥夺后髁突软骨表面粗糙、胶原纤维排列紊乱、软骨细胞增殖减少、颞下颌关节滑膜和髁突软骨中基质降解酶表达增加,炎症因子如肿瘤坏死因子α、白细胞介素1β、白细胞介素6和诱导型一氧化氮合酶等表达增加[17-18]。同时发现,睡眠剥夺后大鼠颞下颌关节中钟基因Bmal1显著下降,这提示钟基因Bmal1的异常表达可能是TMJOA发生的诱因之一[19]。DUDEK等[9]通过软骨特异性敲除Bmal1证实,当小鼠软骨组织中缺失Bmal1会引起软骨组织昼夜节律的丧失,同时伴有软骨基质中软骨合成因子Ⅱ型胶原α1、蛋白聚糖等下调,基质金属蛋白酶3、基质金属蛋白酶13、血管内皮生长因子和Runt相关转录因子2的表达升高,软骨内稳态向分解代谢状态转变,引起骨关节炎。缺乏 Bmal1 的软骨细胞会产生更多的炎症因子,加速成年小鼠肢体关节的炎症[20]。相反,骨关节炎环境中的因素也可以引起软骨细胞昼夜节律时钟的变化,如氧化应激、白细胞介素1β和碱性磷酸钙晶体等均可降低Bmal1表达,需要注意的是肿瘤坏死因子α对软骨组织的昼夜节律及时钟基因的表达几乎没有影响[21]。CHEN等[22]的研究发现,下调Bmal1的表达可以激活MAPK/ERK信号通路从而诱发TMJOA,而Bmal1质粒治疗可缓解小鼠的关节炎症,并降低颞下颌关节中的P-ERK及基质降解酶含量。此外,Bmal1 还可以通过转化生长因子β、Wnt/β及缺氧诱导因子1α-血管内皮生长因子信号通路等来调控软骨的合成代谢与分解代谢,进而影响关节疾病的发生发展[23]。尽管睡眠剥夺是动物模型中建立颞下颌关节损伤的一种有效方法,但睡眠剥夺除了可以中断中央时钟,还可以引发一系列的代谢疾病,如肥胖、糖尿病、心血管疾病等[24]。SANCHEZ等[25]做了一份关于TMJOA与睡眠障碍的相关性研究,Meta分析中低质量的证据显示TMJOA与睡眠质量有关,但极低质量的证明显示TMJOA与睡眠障碍无关。所以慢性睡眠剥夺引起TMJOA是时钟紊乱因素还是代谢紊乱因素,或者是二者共同作用的结果尚不清楚,因此中央时钟中断本身是否对软骨内稳态有不利影响还有待确定[16]。Bmal1对TMJOA的直接影响见图3。"
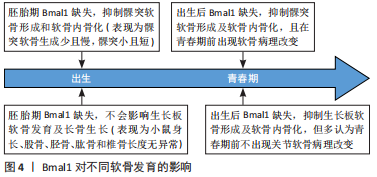
2.2 Bmal1对TMJOA的间接影响 2.2.1 Bmal1与下颌骨发育 短小髁突是TMJOA易发的解剖因素,短小的髁突使颞下颌关节活动空间加大,髁突更易超越关节结节;髁突表面受力增加,使它存在容易受伤的解剖基础,同时短小的髁突可以引起咬合异常,咬合异常同样是TMJOA发病的主要原因[26]。髁突的发育源自髁突软骨,与其他四肢关节的关节软骨不同,髁突软骨被认为是继发性软骨,其生长发育可分为两个阶段,即成熟和矿化[27]。髁突软骨的生长发育是一个复杂的生物学过程,该过程不仅受到各种局部自分泌/旁分泌因子的严格调节,还受到昼夜节律的控制[28]。研究表明,Bmal1 能通过影响髁突软骨的软骨形成和软骨内骨化调控小鼠髁突及下颌骨的发育。在全基因敲除Bmal1小鼠的胚胎和出生后发育过程中,髁突软骨中的软骨形成因子表达水平降低,出现下颌骨发育不良、髁突短小畸形[29]。Micro-CT显示Bmal1缺乏后髁突的骨量、骨矿物质密度显著下降;组织学检测显示Bmal1缺乏会导致髁突软骨的增殖层和肥厚层变薄,软骨细胞的增殖减少、凋亡增加[27]。在临床中也发现,骨性下颌发育不全的青少年下颌骨组织中Bmal1的表达水平低[30]。 目前关于Bmal1调控髁突发育的研究较少,已证实Bmal1通过调控印度刺猬蛋白、骨保护素、基质金属蛋白酶3等信号通路在下颌骨的生长发育中发挥作用,这为下颌骨发育不良的发病机制提供了新见解。此外,ZHOU等[29]发现,在小鼠生命周期的早期,给予激活剂SAG、骨保护素可部分挽救因Bmal1缺乏引起的髁突短小表型,为治疗下颌骨发育不良提出了新策略。在正畸临床中,应用髁突生长改建具有昼夜节律性的特性,在髁突增生改建昼夜节律的峰值期戴用矫治器,有利于提高矫治效率[31]。需要注意的是,Bmal1影响胚胎软骨发育的结论可能只适用于髁突软骨。一项研究发现,在胚胎期消融Bmal1基因未观察到Bmal1 KO组有明显的形态学异常,身体、股骨、胫骨、肱骨和脊椎(L1-L5)的长度方面均未发现显著差异,这表明胚胎发生期消融Bmal1不影响长骨生长,但在出生后消融Bmal1会影响软骨内骨发育[32]。两种完全相反的结论可能是因为髁突软骨和四肢关节软骨的软骨内成骨阶段不同,然而,需要更多的研究来支持这一假设[33]。Bmal1在不同软骨发育中的作用见图4。"

2.2.2 Bmal1与衰老 颞下颌关节因承担发音和咀嚼功能,髁突软骨会发生进行性退变,随着年龄的增长,常发生骨关节炎[34]。通常认为衰老导致TMJOA是因为:随着年龄的增长软骨基质中糖基化终末产物的富集使软骨变脆;衰老的软骨细胞对细胞因子敏感,细胞因子诱导的基质金属蛋白酶增加,软骨基质趋于代谢分解状态;随着年龄增长而发展的慢性低度炎症会进一步损害关节组织的稳态[14]。目前认为,昼夜节律失调也是衰老引起骨关节炎的机制之一。研究发现,敲除Bmal1基因的小鼠表现出各种早衰症状,包括体质量减轻、皮下脂肪减少、白内障、寿命缩短和骨关节炎等[35]。敲除Bmal1导致早衰症状可能与其调控的氧化应激和炎症有关[36]。因此,BROWN等[37]认为高龄与膝骨关节炎易感的关系或许能用昼夜节律失调解释。这可能是因为Bmal1在老年关节软骨中表达量减少导致软骨细胞衰老,衰老的软骨细胞可能通过氧化应激反应产生白细胞介素 6、白细胞介素1等炎症因子和基质金属蛋白酶13、基质金属蛋白酶14等基质金属蛋白酶导致膝骨关节炎的发生[5]。相反,衰老也是影响昼夜节律稳定性的内在因素。随着年龄的增长,髁突软骨的昼夜节律变钝,时钟基因Bmal1表达下降[38]。已被证明,在老年小鼠软骨中昼夜节律的振幅降低了约40%[39]。这种变化与随着年龄的增长颞下颌关节髁突软骨的退行性病变是一致的,这凸显了钟基因Bmal1在年龄相关的颞下颌关节退行性变中的潜在作用[38]。该研究还发现,老年颞下颌关节髁突会出现软骨退化、骨重塑不耦合和昼夜节律消失的表现[38]。尽管衰老破坏软骨时钟的具体机制未知,但目前研究发现可能与Sirt1有关[40]。Sirt1是一种参与衰老、应激反应和能量代谢的蛋白去乙酰化酶,因衰老而下降,Sirt1表达下降也被认为是小鼠中央时钟昼夜节律紊乱的原因。此外,正常情况下中央时钟和关节组织中的外周时钟在时间上协调使局部生理功能适应日常节律环境(明暗、进食-禁食、体温调节和机械负荷循环[41]。而衰老过程中昼夜节律的精确度丧失可能使身体活动周期(由中央时钟控制)与关节昼夜节律(由局部时钟控制)错位,导致关节组织对机械刺激做出不恰当的反应,这种脱节将增加关节损伤风险[14]。值得注意的是,研究发现老年小鼠经过地塞米松治疗后,软骨组织的节律性可以恢复到与年轻成年小鼠相似程度,这暗示老年小鼠软骨组织中的固有起搏机制可能仍然是完整[15]。因此,老化的软骨时钟可能会因加强中央时钟控制的昼夜节律信号而重新调整,这可能会对骨关节炎未来的治疗方法产生重大影响。 2.2.3 Bmal1与肥胖 近来发现昼夜节律时钟与肥胖存在密切关系,昼夜节律时钟紊乱会增加肥胖的风险[42]。研究认为敲除Bmal1的小鼠更容易诱发肥胖,可能是Bmal1缺失导致Elovl6和Scd1的表达减少,而后者是脂肪组织中参与合成脂肪酸生物的时钟控制基因[43-44]。已知肥胖是TMJOA的危险因素[45],研究发现,高脂饮食诱发肥胖的小鼠其髁突软骨出现骨关节炎样改变,表现为软骨形态改变、炎症因子和软骨基质降解酶表达上调、瘦素表达过度[46]。有趣的是,KC等[47]将昼夜节律紊乱与肥胖结合起来研究二者对骨关节炎的影响,发现昼夜节律的破坏加上高脂肪饮食会加剧小鼠膝关节骨关节炎样病变。因此DU等[46]认为可以利用降脂药来缓解TMJOA的炎症,这为TMJOA的治疗提供了新的思路。 2.2.4 Bmal1与机械负荷 机械负荷是调节颞下颌关节软骨基质稳态的重要因素。研究发现机械刺激调节软骨时钟蛋白的表达,这一观点被 Bmal1/Clock复合物中的Clock基因所支持。大规模DNA芯片分析表明,Clock可能是一个机械敏感的时钟基因,因为施加机械应力后Clock显著上调[48]。Clock基因可促进软骨年再生、延缓衰老相关的关节退行性变,或许每天有规律的温和锻炼可以有效保护关节软骨[49]。需要注意,在软骨特异性Bmal1敲除的研究发现,膝关节软骨完整性丧失,但髋关节软骨未受影响,猜测可能是Bmal1水平降低使软骨细胞对机械负荷敏感,增加了患病风险。因此了解Bmal1敲除后髋关节软骨被保留的机制,有助于理解不同关节发生骨性关节炎的危险因素不同[16]。Bmal1对TMJOA的间接影响见表1。"

| [1] HU Z, XIAO M, CAI H, et al. Glycyrrhizin regulates rat TMJOA progression by inhibiting the HMGB1-RAGE/TLR4-NF-κB/AKT pathway. J Cell Mol Med. 2022;26: 925-936. [2] LI B, GUAN G, MEI L, et al. Pathological mechanism of chondrocytes and the surrounding environment during osteoarthritis of temporomandibular joint. Cell Mol Med. 2021;25:4902-4911. [3] HE D, AN Y, LI Y, et al. RNA sequencing reveals target genes of temporomandibular joint osteoarthritis in rats after the treatment of low-intensity pulsed ultrasound. Gene. 2018;672:126-136. [4] OUDET C, PETROVIC A. Growth rhythms of the cartilage of the mandibular condyle: effects of orthopaedic appliances. Int J Chronobiol. 1978;5:545-564. [5] 周晓红,李柏村,李佳,等. 从肝论治取穴针灸治疗膝骨关节炎模型大鼠关节功能与昼夜节律的关系[J]. 中国组织工程研究,2021,25(14):2192-2198. [6] ALBRECHT U. Timing to perfection: the biology of central and peripheral circadian clocks. Neuron. 2012;74:246-260. [7] CHEN G, TANG Q, YU S, et al. The biological function of BMAL1 in skeleton development and disorders. Life Sci. 2020;253:117636. [8] ZHU Q, BELDEN WJ. Molecular Regulation of Circadian Chromatin. J Mol Biol. 2020;432:3466-3482. [9] DUDEK M, GOSSAN N, YANG N, et al. The chondrocyte clock gene Bmal1 controls cartilage homeostasis and integrity. J Clin Invest. 2016;126:365-376. [10] SIMMONS DJ. Diurnal periodicity in epiphyseal growth cartilage. Nature. 1962; 195:82-83. [11] STEVENSON S, HUNZIKER EB, HERRMANN W, et al. Is longitudinal bone growth influenced by diurnal variation in the mitotic activity of chondrocytes of the growth plate?. J Orthop Res. 1990;8:132-135. [12] ALAGHA MA, VAGO J, KATONA É, et al. A Synchronized Circadian Clock Enhances Early Chondrogenesis. Cartilage. 2021;13:53S-67S. [13] TAKARADA T, KODAMA A, HOTTA S, et al. Clock genes influence gene expression in growth plate and endochondral ossification in mice. J Biol Chem. 2012;287:36081-36095. [14] ERENBAUM F, MENG QJ. The brain-joint axis in osteoarthritis: nerves, circadian clocks and beyond. Nat Rev Rheumatol. 2016;12:508-516. [15] GOSSAN N, ZEEF L, HENSMAN J, et al. The circadian clock in murine chondrocytes regulates genes controlling key aspects of cartilage homeostasis. Arthritis Rheum. 2013;65:2334-2345. [16] POULSEN RC, HEARN JI, DALBETH N. The circadian clock: a central mediator of cartilage maintenance and osteoarthritis development?. Rheumatology (Oxford). 2021;60:3048-3057. [17] LIANG C, YANG T, WU G, et al. Therapeutic effect of low-intensity pulsed ultrasound on temporomandibular joint injury induced by chronic sleep deprivation in rats. Am J Transl Res. 2019;11:3328-3340. [18] DONG Y, WU G, ZHU T, et al. VEGF promotes cartilage angiogenesis by phospho-ERK1/2 activation of Dll4 signaling in temporomandibular joint osteoarthritis caused by chronic sleep disturbance in Wistar rats. Oncotarget. 2017;8:17849-17861. [19] LIANG C, YANG T, WU G,et al. The Optimal Regimen for the Treatment of Temporomandibular Joint Injury Using Low-Intensity Pulsed Ultrasound in Rats with Chronic Sleep Deprivation. Biomed Res Int. 2020;2020:5468173. [20] URSINI F, DE GIORGI A, D’ONGHIA M, et al. Chronobiology and Chronotherapy in Inflammatory Joint Diseases. Pharmaceutics. 2021;13:1832. [21] RONG J, ZHU M, MUNRO J, et al. Altered expression of the core circadian clock component PERIOD2 contributes to osteoarthritis-like changes in chondrocyte activity. Chronobiol Int. 2019;36:319-331. [22] CHEN G, ZHAO H, MA S, et al. Circadian Rhythm Protein Bmal1 Modulates Cartilage Gene Expression in Temporomandibular Joint Osteoarthritis via the MAPK/ERK Pathway. Front Pharmacol. 2022;11:527744. [23] AKAGI R, AKATSU Y, FISCH KM, et al. Dysregulated circadian rhythm pathway in human osteoarthritis: NR1D1 and BMAL1 suppression alters TGF-β signaling in chondrocytes. Osteoarthritis Cartilage. 2017;25:943-951. [24] ZHAO Y, LU X, WAN F, et al. Disruption of Circadian Rhythms by Shift Work Exacerbates Reperfusion Injury in Myocardial Infarction. J Am Coll Cardiol. 2022; 79:2097-2115. [25] SANCHEZ EA, MARTINEA O, GARCIA M, et al. Association between Sleep Disorders and Sleep Quality in Patients with Temporomandibular Joint Osteoarthritis: A Systematic Review. Biomedicines. 2022;10:2143. [26] NOJIMA K, NAGATA M, OOTAKE T, et al. Surgical Orthodontic Treatment Involving Mandibular Premolar Extraction in Patient with Mandibular Retrusion Associated with Temporomandibular Joint Osteoarthritis. Bull Tokyo Dent Coll. 2019;60:139-149. [27] YU S, TANG Q, XIE M, et al. Circadian BMAL1 regulates mandibular condyle development by hedgehog pathway. Cell Prolif. 2020;53:e12727. [28] KOSHI R, MATSUMOTO K, IMANISHI Y, et al. Morphological characteristics of interalveolar septum and mandible in BMAL1 gene knockout mice. J Oral Sci. 2020;63:83-86. [29] ZHOU X, YU R, LONG Y, et al. BMAL1 deficiency promotes skeletal mandibular hypoplasia via OPG downregulation. Cell Prolif. 2018;51:e12470. [30] ZHAO J, ZHOU X, TANG Q, et al. BMAL1 Deficiency Contributes to Mandibular Dysplasia by Upregulating MMP3. Stem Cell Reports. 2018;10:180-195. [31] 熊国平,陈扬熙,罗颂椒. 功能矫形与大鼠髁突内源性甲状旁腺素昼夜节律变化的研究[J]. 中华口腔医学杂志,2000,35(5):394. [32] MA Z, JIN X, QIAN Z, et al. Deletion of clock gene Bmal1 impaired the chondrocyte function due to disruption of the HIF1α-VEGF signaling pathway. Cell Cycle. 2019;18:1473-1489. [33] SONG X, BAI H, MENG X, et al. Drivers of phenotypic variation in cartilage: Circadian clock genes. J Cell Mol Med. 2021;25:7593-7601. [34] ROBERTS WE, STOCUM DL. Part II: Temporomandibular Joint (TMJ)-Regeneration, Degeneration, and Adaptation. Curr Osteoporos Rep. 2018;16:369-379. [35] KONDRATOV RV, KONDRATOVA AA, GORBACHEVA VY, et al. Early aging and age-related pathologies in mice deficient in BMAL1, the core componentof the circadian clock. Genes Dev. 2006;20:1868-1873. [36] FAN R, PENG X, XIE L, et al. Importance of Bmal1 in Alzheimer’s disease and associated aging-related diseases: Mechanisms and interventions. Aging Cell. 2022;21:e13704. [37] BROWN SA, PAGANI L, CAJOCHEN C, et al. Systemic and cellular reflections on ageing and the circadian oscillator: a mini-review. Gerontology. 2011;57(5):427-434. [38] CHA S, LEE SM, WANG J, et al. Enhanced Circadian Clock in MSCs-Based Cytotherapy Ameliorates Age-Related Temporomandibular Joint Condyle Degeneration. Int J Mol Sci. 2021;22:10632. [39] YANG N, MENG QJ. Circadian Clocks in Articular Cartilage and Bone: A Compass in the Sea of Matrices. J Biol Rhythms. 2016;31:415-427. [40] YANG W, KANG X, LIU J, et al. Clock Gene Bmal1 Modulates Human Cartilage Gene Expression by Crosstalk With Sirt1. Endocrinology. 2016;157:3096-3107. [41] MORRIS H, GONCALVES CF, DUDEK M, et al. Tissue physiology revolving around the clock: circadian rhythms as exemplified by the intervertebral disc. Ann Rheum Dis. 2021;80:828-839. [42] HASAN N, NAGATA N, MORISHIGE JI, et al. Brown adipocyte-specific knockout of Bmal1 causes mild but significant thermogenesis impairment in mice. Mol Metab. 2021;49:101202. [43] SHIMBA S, ISHII N, OHTA Y, et al. Brain and muscle Arnt-like protein-1 (BMAL1), a component of the molecular clock, regulates adipogenesis. Proc Natl Acad Sci U S A. 2005;102:12071-12076. [44] JOUFFE C, WEGER BD, MARTIN E, et al. Disruption of the circadian clock component BMAL1 elicits an endocrine adaption impacting on insulin sensitivity and liver disease. Proc Natl Acad Sci U S A. 2022;119:e2200083119. [45] TAKAOKA R, KUYAMA K, YATANI H, et al. Involvement of an FTO gene polymorphism in the temporomandibular joint osteoarthritis. Clin Oral Investig. 2022;2:2965-2973. [46] DU J, JIANG Q, MEI L, et al. Effect of high fat diet and excessive compressive mechanical force on pathologic changes of temporomandibular joint. Sci Rep. 2020;10:17457. [47] KC R, LI X, FORSYTH CB, et al. Osteoarthritis-like pathologic changes in the knee joint induced by environmental disruption of circadian rhythms is potentiated by a high-fat diet. Sci Rep. 2015;5:16896. [48] KANBE K, INOUE K, XIANG C, et al. Identification of clock as a mechanosensitive gene by large-scale DNA microarray analysis: downregulation in osteoarthritic cartilage. Mod Rheumatol. 2006;16:131-136. [49] LIANG C, LIU Z, SONG M, et al. Stabilization of heterochromatin by CLOCK promotes stem cell rejuvenation and cartilage regeneration. Cell Res. 2021;31:187-205. [50] LU KH, LU PW, LU EW, et al. The potential remedy of melatonin on osteoarthritis. J Pineal Res. 2021;71:e12762. |
| [1] | Du Xueting, Zhang Xiaodong, Chen Yanjun, Wang Mei, Chen Wubiao, Huang Wenhua. Application of compressed sensing technology in two-dimensional magnetic resonance imaging of the ankle joint [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1396-1402. |
| [2] | Ruan Ling, Wang Guanghua, Wu Rongping, Jin Zhan, Lyu Zhenqing, Zhang Nan, Li Shoubang. Correlation between exercise intensity and lipid metabolism disorder and oxidative stress in a high-diet rat model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1149-1155. |
| [3] | Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244. |
| [4] | Xue Ting, Zhang Xinri, Kong Xiaomei. Mesenchymal stem cell therapy for pneumoconiosis using nanomaterials combined with multi-modal molecular imaging [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1133-1140. |
| [5] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [6] | Xiong Juan, Guan Yalin, Yang Yutong, Wang Fan, Liu Zhongshan. Application of stem cells to skin anti-aging [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 948-954. |
| [7] | Liu Guangluan, Guo Zonglei, Ge Jin, Huang Dong, Wang Yehua. Anatomic risk factors for medial meniscus posterior root tears combined with anterior cruciate ligament injuries [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 663-668. |
| [8] | Zhao Siqi, Du Juan, Qu Haifeng, Li Jianmin, Zhang Yuxin, Liu Junjie. Effects of enriched environment combined with melatonin on learning and memory function and brain neuron apoptosis in SAMP8 mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 701-706. |
| [9] | Gu Mingxi, Wang Bo, Tian Fengde, An Ning, Hao Ruihu, Wang Changcheng, Guo Lin. Comparison of early efficacy and safety of simultaneous and staged bilateral total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 565-571. |
| [10] | Guo Yingqi, Gong Xianxu, Zhang Yan, Xiao Han, Wang Ye, Gu Wenguang. Meniscus extrusion and patellofemoral joint cartilage injury and bone marrow lesions: MRI semi-quantitative score [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 600-605. |
| [11] | Liu Hao, Yang Hongsheng, Zeng Zhimou, Wang Liping, Yang Kunhai, Hu Yongrong, Qu Bo. Lumbar MRI vertebral bone quality score to evaluate the severity of osteoporosis in postmenopausal women [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 606-611. |
| [12] | Yuan Haibo, Li Dongya, Pan Bin, Guan Kai, Chen Feng, Yuan Feng, Wu Jibin. Sagittal related factors of upper lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(31): 4984-4989. |
| [13] | Li Xiaojuan, Zhang Yuanzhi, Yang Xiaoguang, Gao Yang, Wu Qiong. Quantitative evaluation of bone structure changes in femoroacetabular impingement syndrome by magnetic resonance imaging [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(31): 4965-4970. |
| [14] | Wu Tong, Yin Caiyun, Zhao Mingzhe, Zhu Yishen. Application of functional peptides for biomedical diagnosis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 478-485. |
| [15] | Chen Chao, Wang Xuenan, Zhan Enyu, Lyu Zhengpin, Zhang Fan. Mechanism underlying the mutual regulation between circadian rhythm and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(29): 4696-4703. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||