Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (3): 478-485.doi: 10.12307/2023.063
Previous Articles Next Articles
Application of functional peptides for biomedical diagnosis
Wu Tong1, Yin Caiyun1, Zhao Mingzhe2, Zhu Yishen1
- 1Nanjing Tech University, Nanjing 211816, Jiangsu Province, China; 2Nanjing University of Chinese Medicine, Nanjing 210023, Jiangsu Province, China
-
Received:2021-03-26Accepted:2021-05-16Online:2023-01-28Published:2022-06-01 -
Contact:Zhu Yishen, Professor, Nanjing Tech University, Nanjing 211816, Jiangsu Province, China -
About author:Wu Tong, Master candidate, Nanjing Tech University, Nanjing 211816, Jiangsu Province, China -
Supported by:the Key Project of Social Development in Jiangsu Province, No. BE2019736 (to ZYS)
CLC Number:
Cite this article
Wu Tong, Yin Caiyun, Zhao Mingzhe, Zhu Yishen. Application of functional peptides for biomedical diagnosis[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 478-485.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
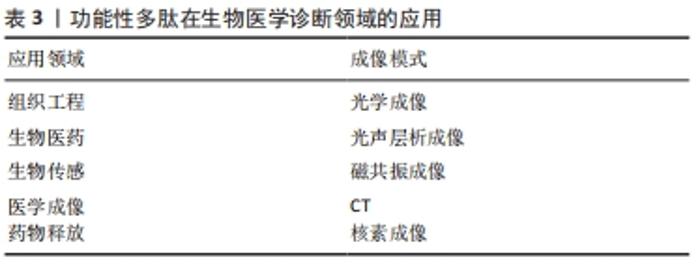
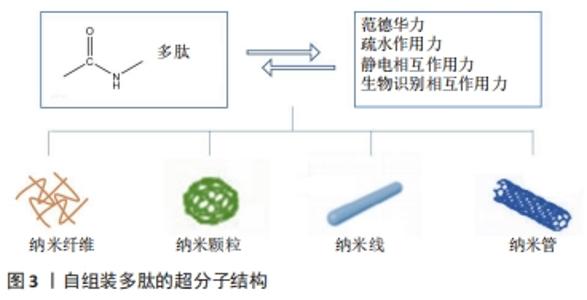
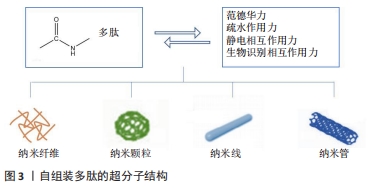
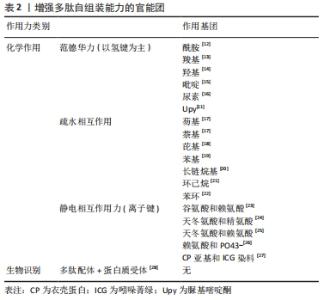
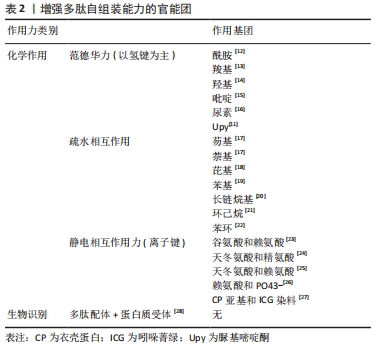
此外,通过脲基嘧啶酮(Urea Pyrimidone,UPy)获得四重氢键体系,能进一步提高多肽的自组装作用[11]。 2.2.3 多肽自组装的另一个主要驱动力是多肽及其衍生物之间的π-π相互作用 CARNY等[29]对苯丙氨酰-苯丙氨酸(FF)二肽进行了研究,发现侧链苯环之间的π-π堆积作用可以促进多肽自组装形成纳米管状结构。此外,含有芴基、萘基的芳香族多肽衍生物芳环间也能形成π-π相互作用[17]。 当多肽衍生物含有疏水基团时,不仅存在疏水相互作用,还与氢键产生协同作用。例如,将环己烷三羰基三氯化物通过酰胺键偶联到具有MH-OMe序列的多肽,获得了一类良好自组装能力的水凝胶,疏水性侧基将酰胺与水屏蔽,形成分子内氢键,疏水内核的疏水相互作用也为自组装提供了作用力[21,30]。静电相互作用力对多肽自组装也有重要作用。RADA4(RADARADARADARADA)是含有带正电荷的精氨酸残基(Arg)和带负电荷的天冬氨酸残基(Asp)的离子互补型自组装多肽,通过正负离子相互作用以及丙氨酸侧链甲基的n-n作用形成自组装多肽水凝胶[24]。雀麦花叶病毒衣壳蛋白亚基对吲哚菁绿(Indocyanine Green,ICG)的包封也是一种自组装过程,主要受带负电的ICG分子和带正电的衣壳蛋白亚基之间的静电作用力控制[27]。 多肽自组装的另一驱动力是蛋白质与多肽配体之间的相互作用。ZHANG等[28]设计了融合蛋白ULD-TIP-1 (ULD:泛素样结构域,Ubiquitin-like Domain;TIP-1:Tax相互作用蛋白1,Tax-interacting Protein-1),形成4个结合位点的紧密四聚体,ULD辅助四聚体的形成,TIP-1提供多肽的结合位点,该融合蛋白能够增强自组装纳米纤维之间的相互作用形成自组装多肽水凝胶。 多肽分子在水溶液中溶解后通过自组装作用力自发或触发地由一级结构向二级结构转变,形成α-螺旋、β折叠和β发夹等常见的二级结构,导致多肽发生自组装行为。 2.3 功能性多肽与生物医学诊断领域的应用 功能性多肽生物相容性好、无免疫原性和无毒副反应,体内降解产物为人体可吸收的氨基酸,通过分子间相互作用(范德华力、疏水相互作用和静电相互作用等)将功能性多肽结构单元构建形成高效稳定的生物材料,用于生物组织工程、生物医药及生物传感器等领域,进行医学成像、控制药物释放等。功能性多肽用于医学成像具有高度特异性,在医学诊断和治疗中发挥重要的作用。目前通过整合各种成像组分(放射性同位素和荧光分子等),开发了高度特异性的多肽类成像探针,用于各种不同的成像模式 [31],见表3。 "

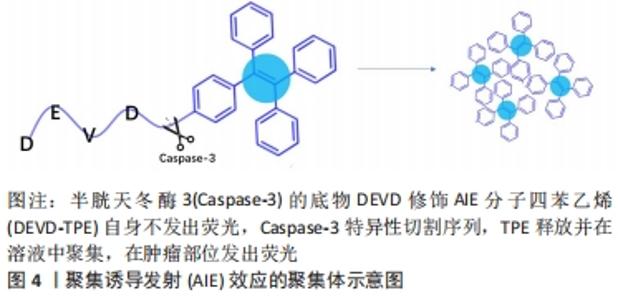
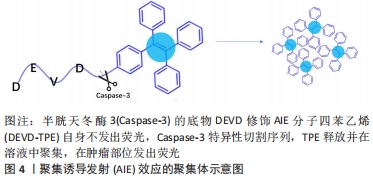
2.3.1 用于光学成像的功能性多肽 光学成像是指光在生物组织传播过程中吸收和散射,通过不同的采样技术实现成像。光学成像技术无损伤、非接触,具有高灵敏度、高分辨率和成本低等特点,是医学诊断领域普遍应用的一种成像技术。通过原位或异位构建多肽,可以形成以荧光增强或猝灭为检测目标的多肽类荧光分子探针[32]。一些应用于光学成像的功能性多肽序列,见表4。有研究开发了聚集诱导发射(aggregation induced emission,AIE)效应的聚集体用于荧光生物成像[43]。AIE分子单体几乎不发荧光,但聚集状态下显示明亮荧光。通过组合功能性多肽,能够有效地在活细胞中原位构建AIE聚集体,从而实现荧光增强,成像活细胞的特定生物过程或测定酶活性。比如监测细胞凋亡过程,用半胱天冬酶3(Caspase-3)的底物DEVD修饰AIE分子四苯乙烯(tetraphenylethylene,TPE),构建水溶性AIE前体,见图4。"


在凋亡细胞模型中,Caspase-3切割DEVD-TPE释放TPE,游离的TPE聚集后发出荧光[44]。在细胞毒性试验中与对照组无显著差异,小鼠器官切片未见明显的病理改变,说明该材料几乎没有全身毒性,且在肿瘤部位注射24 h后仍可观察到,有较好的肿瘤特异性和滞留特性。 荧光分子成像可以方便安全地提供较高的空间和时间分辨率。多肽能够与靶点特异性结合、被酶特异性切割,具有靶向性和成像功能。使用多肽荧光探针进行荧光成像不仅作为一种临床诊断方法,而且作为一种分析生物现象的方法,引起广泛关注。合理利用这些多肽化合物的光学特性,可以研制出用于多种靶点光学成像的多肽分子探针。 2.3.2 用于光声层析成像的功能性多肽 光声效应(photo acoustic effect,PA)于1880年首次发现[45],是指物质吸收短脉冲激光束,将能量转换为声音(通过热量,引起局部压力变化)的现象[45-46]。20世纪90年代中期,光声效应用于生物医学成像,结合了光学吸收特性和超声探测优势,在临床医学和基础生物学领域迅速发展。功能性多肽作为光声层析成像造影剂,不仅具有高通透性和滞留效应,还具有高灵敏度和高特异性。表5列举了一些能够用于光声层析成像的多肽纳米材料。 "

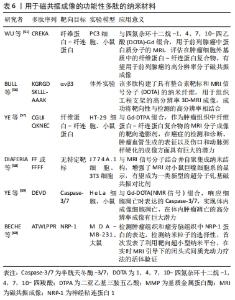
有研究将RGD结合到单壁碳纳米管(single walled carbon nanotubes,SWCNT)用于整合素αvβ3阳性U87肿瘤模型的体内光声层析成像[53]。与SWCNTs相比,这种对比剂在活体组织中的光声对比度显著提高300倍。同时发现通过堆积作用将ICG染料附着到纳米管表面,增强了SWNT-RGD的光声信号,从而提高成像灵敏度,可以检测到先前报道1/20的癌细胞。 WANG等[54]制备了一种可激活Caspase-3的PA探针1-RGD用于体内化学诱导的肿瘤细胞凋亡分子成像。该探针由2-氰基-6-羟基喹啉、D-半胱氨酸残基、Caspase-3裂解序列DEVD、还原性谷胱甘肽二硫键、近红外染料ICG组成,通过Caspase-3触发肽底物的切割,环化并自组装成纳米颗粒,MTT细胞毒性检测显示100 mmol/L内各个浓度的1-RGD对细胞增殖几乎没有影响,与阴性对照组相比,阿霉素处理的肿瘤细胞中PA信号明显升高。鉴于光声层析成像空间分辨率高、穿透深,1-RGD可用于分析凋亡信号在整个肿瘤组织中的分布,有助于早期和实时评估体内肿瘤的治疗效果。 光声成像能够有效进行生物组织结构和功能成像,为研究生物组织形态结构,生理特征,病理特征,代谢功能等提供了重要手段,通过超分子策略,使功能性多肽包含PA材料,这些PA材料可以协同增强光声层析成像性能[27]。 2.3.3 用于磁共振成像的功能性多肽 MRI利用细胞中不同原子核的自旋特性进行成像,是一种强大的功能性成像模式,可以得到软组织(如肌肉,关节和大脑)的高分辨率图像。多肽通过折叠或非共价作用力组装后,可作为磁共振成像潜在造影剂,受到广泛的关注。顺磁性化合物如超顺磁性氧化铁纳米颗粒通过与多肽复合,能够增加生物相容性,以应对来自机体环境的变化,改善其聚集和降解,在体内成像性能高、能够与组织和组织液相互作用,用于靶向到特定器官或肿瘤等特定区域成像进行临床诊疗[55]。用于磁共振成像的功能性多肽纳米材料,见表6。 "


基于与Gd(III)(HPN3DO3A)标记相连的氨基酸序列V3A3(VVVAAA),有研究开发了一系列两亲性多肽(Amphiphilic Peptides,PA)分子[61],V3A3在pH值大于7.0的溶液中β-折叠,形成高纵横比的纳米纤维,Gd(III)可以提高配合物的稳定性,相对于典型的单体造影剂,弛豫度显著提高。用Gd(III)-PA配合物在小鼠体内模型中对PA凝胶进行监测,显示其在4 d内都有保留。这些Gd(III)-PA自组装形成的超分子结构为监测生物材料植入体内后随时间推移而产生的变化提供了一个有效途径。 有研究报道,由pH值响应性二嵌段共聚物(polyethylene glycol-block-poly diisopropanol amino ethyl methacrylate cohydroxyl methacrylate,PDPA)、光敏剂二氢卟吩e6(Chlorin e6,Ce6)、阿霉素聚合物前药,组成多功能胶束,胶束在血液中不表现荧光、MRI和光动力(Photodynamic Therapy,PDT)活性[62]。 功率密度≤1.5 W/cm2时,胶束导致的细胞死亡可以忽略不计,但在2.0 W/cm2的功率密度下,胶束处理的MCF-7/ADR细胞中超过40 %的细胞被杀死,在使用巴氏霉素A1(bafilomycin A1,Baf-A1)预孵育后,光毒性明显降低。当该胶束被癌细胞摄取在细胞内解离时,在内吞囊泡中转变为活跃状态,诱导荧光成像的荧光信号增加7.5%。在近红外激光照射下,胶束将近红外光转换为局部热,以增强抗癌药物对肿瘤的穿透、肿瘤特异性光热治疗和PA成像。此外,胶束可以在酸性微环境中放大磁共振信号进行MRI。该胶束在荧光、MRI、光声层析成像三模式成像和联合治疗显示出良好的应用前景。磁共振技术在医学成像领域有许多优势,磁共振检查没有放射性,能够清晰地显示人体内部组织结构,具有较高的图像分辨率,在先天性疾病、创伤和肿瘤疾病的诊断上效果显著。通过鉴别细胞表面肽配体细微差异,功能性多肽能够靶向到特定细胞,一般对正常细胞不会产生毒性作用。与市面上的MRI对比剂(如Magnevist和Resovist)相比,功能性多肽作为探针显示出更高的r2/r1的比值,并且已经应用到肝脏病变成像,如肝硬化和肿瘤等领域[55]。 2.3.4 用于CT检查的功能性多肽 CT是一种基于X射线吸收差异的强大诊断工具,能够提供高分辨率3D断层图像[63]。CT成像通常使用高X射线衰减对比剂增强对比度,如碘、钡、铋等。目前临床获批的碘对比剂具有肾脏毒性,对患者治疗过程有一定影响;金纳米粒对正常细胞活动和代谢有一定影响,且受环境影响较大;金属对比剂使用剂量较高,不良反应大,使用不便。功能性多肽则易于制备和保存,且性质稳定,相对分子量小,体内代谢清除快,是CT分子影像探针的理想化合物,目前用于CT检查的功能性多肽纳米材料,见表7。 "

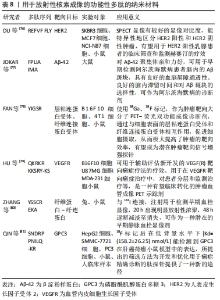
有研究者开发了一种新型人表皮生长因子受体2(human epidermal growth factor receptor 2,HER2)靶向探针[71],将H10F(KLRLEWNR)与SBz-HYNIC偶联,99Tcm标记多肽,得到99Tcm-HYNIC-H10F。建立SK-BR3和MDA-MB-361两种HER2阳性乳腺癌模型,采用SPECT/CT监测和量化曲妥珠单抗的治疗反应。初步临床研究显示,2位女性乳腺癌患者使用该探针后1周内均未发生严重不良事件或生命体征明显改变,SPECT结果显示患者1的99Tcm-HYNIC-H10F的T/NT比值明显低于患者2,肿瘤HER2 FISH试验和IHC染色显示患者1 HER2阴性,患者2 HER2阳性,与该探针显像结果一致。数据显示,该探针可以清晰反映乳腺肿瘤中HER2表达的动态变化,给药4 d后放射性显著降低,且在治疗过程中不与曲妥珠单抗竞争性结合,能反映肿瘤对曲妥珠单抗治疗的早期反应,有望进一步开展乳腺癌临床研究,帮助制定个性化治疗策略。 ZAHID等[72]合成了99Tcm标记的CTP肽(APWHL SSQYS RT)靶向心脏组织,放射化学实验表明,使用微克级(7-15 μg)的CTP,就可以向心脏提供足够量的辐射,与目前临床使用的99Tcm-甲氧基异丁基异腈(99Tcm-Sestamibi)剂量相似,给药后最快5 min心肌达到摄取峰值,主要通过肾脏排泄,缩短体内滞留时间,尽可能地降低造影剂辐射造成的影响。此外,对人IPSC的实验表明CTP不受物种限制,不仅限于其对小鼠心脏的转导能力,而且它具有人类应用的潜力,可作为一种新型心脏靶向载体,应用于诊断成像和靶向治疗。 有研究设计了一种99Tcm标记的多肽P5+14(GGGYS KAQKA QAKQA KQAQK AQKAQ AKQAK QAQKA QKAQA KQAKQ A),该多肽制备重复性好,产率高,纯化后能达到90%以上[73],能够保持较高的生物活性,能对体内全身脏器淀粉样蛋白进行成像,在注射后4 h内保持稳定的放射性标记,除肾脏外,所有含有淀粉样蛋白的器官如肝、胰腺、脾、胃、肠和心脏比无淀粉样蛋白的器官保留了更多的标记肽,游离探针大部分通过肾脏和膀胱排泄,这些数据与用碘同位素放射性标记的这种肽的生物分布一致,可替代放射性碘化肽进行全身诊断和成像。 在近几十年中,CT作为一种无创成像技术,在临床诊断、基础生物学等领域广泛使用,随着科技的进步,多肽纳米材料用于CT对比剂领域飞速发展。不断完善的多肽纳米对比剂将有望实现对肿瘤部位的靶向成像,提高疾病诊断、治疗效率,减轻患者的痛苦,具有巨大的应用潜力。 2.3.5 用于放射性核素成像的功能性多肽 RI主要包括正电子发射断层扫描(positron pmission computed tomography,PET)和单光子发射计算机断层扫描(single photon emission computed tomography,SPECT)[74],该方法将放射性核素标记在适当的化合物上,引入体内以形成放射性,在受检部位按一定浓度规律进行分布,然后根据放出射线的特性,在体外用探测器进行跟踪探测。通过光点记录、闪烁焦像或断层扫描等方法获得反映放射性核素在脏器或组织中的浓度分布图像。 放射性核素标记的多肽受体显像是核医学中最新颖最具活力的分支领域,用于放射性核素成像的功能性多肽纳米材料,见表8。 "

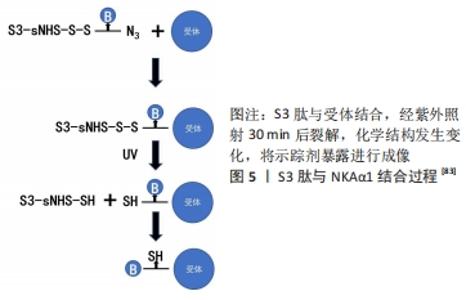
多肽类放射性药物具有下列优点:①化学合成配体多肽,克服了放射免疫显像的抗体异源性问题;②制备技术可控,以保证其理化和生物性能;③多肽配体和受体具有高度亲和性,保证了放射性配体有效聚集;④多肽分子量低,可以通过生理屏障,更主要的是血液清除快,降低了放射性本底,提高了T/NT比值(肿瘤组织与非肿瘤组织放射性计数比值)[75]。 CHEN等[82]设计了一种新型探针18F-Al-NODA-Bn-p-SCN-GGGRDN-IF7,IF7多肽(IFLLWQR)可以靶向肿瘤细胞中过度表达的膜联蛋白1(annexin A1,Anxa1),RDN增加亲水性,GGG降低空间位阻,可以增加肿瘤摄取,降低本底信号,使用双功能螯合剂NODA-Bn-p-SCN及Al18F标记法,提高了标记效率。注射后30 min,肿瘤组织摄取值为(5.74±1.13)%ID/g,肿瘤的摄取显著高于血液和心脏、肠道等器官的摄取。肝、肾中放射性水平较高,表明放射性代谢产物通过肝、肾排泄。除肝脏和肾脏外,其他主要器官的摄取值均明显低于肿瘤的摄取值,没有发现明显的骨摄取,这表明示踪剂在体内是稳定的。研究结果显示该探针在肿瘤中有显著的放射性积聚,肿瘤靶向性好,血药清除快,可作为监测表达Anxa1的表皮样癌PET示踪剂。 以Na+/K+泵ATP酶1(NKAα1)为靶点,有研究发现了一种肿瘤特异性靶向的S3(CSISS LTHC)肽[83],并制备了一种基于S3肽的放射性示踪剂18F-ALF-NOTA-S3,用于乳腺癌和其他NKAα1过表达癌症的PET成像,见图5。 "
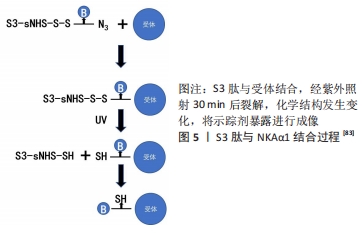

临床研究表明,S3与RGD有相同的代谢特征,分子质量低能够迅速从血液和正常组织中清除,成像方式类似RGD,MDAMB-231肿瘤细胞对18F-S3的摄取达到(5.14±0.26)%ID/g。动态示踪18F-AlF-NOTA-S3时,肿瘤/背景比高,且在注射后50 min保持不变。该示踪剂首次将NKA家族亚单位作为潜在的临床诊断生物标记物用于癌症分子成像,具有高灵敏度、稳定性和低毒性,为特征性肿瘤的表型显像提供了一种新途径。SPECT缺乏精确的解剖学影像学信息,人们逐渐用SPECT/CT代替单一SPECT成像。研究显示以SPECT扫描成像融合CT成像可提高诊断准确性。多肽分子探针容易标记及纯化,可以用化学方法来修饰探针,没有毒性和免疫原性,使用放射性核素标记肿瘤靶向性多肽序列,将其制成多肽探针用于肿瘤成像,在正常组织器官中清除快,肿瘤靶向性良好,具有广阔的应用前景。 "

| [1] AL-HALIFA S, BABYCH M, ZOTTIG X, et al. Amyloid self-assembling peptides: potential applications in nanovaccine engineering and biosensing. Peptide Sci. 2019;111(1):2475-8817. [2] CHEN J, ZOU X. Self-assemble peptide biomaterials and their biomedical applications. Bioact Mater. 2019;4:120-131. [3] ESKANDARI S, GUERIN T, TOTH I. Recent advances in self-assembled peptides: lmplications for targeted drug delivery and vaccine engineering. Adv Drug Deliv Rev. 2017;110:169-187. [4] ZHOU J, LI J, DU XW, et al. Supramolecular biofunctional materials. Biomaterials. 2017;129:1-27. [5] MODLIN I M, KIDD M, FARHADI J. Bayliss and Starling and the nascence of endocrinology. Regul Pept. 2000;93(1):109-123. [6] SOUTHARD GL, BROOKE GS, PETTEE JM. Solid phase peptide synthesis. I. A mild method of solid phase peptide synthesis employing an enamine nitrogen protecting group and a benzhydryl resin as a solid support. Tetrahedron Lett. 1969;10(40):3505-3508. [7] BORNSCHEIN W, GOLDMANN FL, DRESSLER J. Diagnostik der exokrinen Pankreasinsuffizienz mit einem synthetischen chymotrypsinspezifischen Peptid. J Mol Med. 1978;56(4):197-205. [8] RUOSLAHTI E, PIERSCHBACHER M. New perspectives in cell adhesion: RGD and integrins. Science. 1987;238(4826):491-497. [9] ESTROFF LA, HAMILTON AD. Water gelation by small organic molecules. Chem Rev. 2004;104(3):1201-1217. [10] STEVENS MM, ALLEN S, SAKATA JK, et al. pH-Dependent behavior of surface-immobilized artificial leucine zipper proteins. Langmuir. 2004;20(18):7747-7752. [11] WANG H, HEILSHORN SC. Adaptable Hydrogel Networks with Reversible Linkages for Tissue Engineering. Adv Mater. 2015;27(25):3717-3736. [12] JAYAWARNA V, RICHARDSON S, HIRST A. Introducing chemical functionality in Fmoc-peptide gels for cell culture. Acta Biomater. 2009;5(3):934-943. [13] DU XQ, LI P, LU SQ, et al. Highly directional co-assembly of 2,6-pyridinedicarboxylic acid and 4-hydroxypyridine based on low molecular weight gelators. J Mol Liq. 2013;180(4):129-134. [14] DOU XQ, ZHANG D, FENG CL. Wettability of supramolecular nanofibers for controlled cell adhesion and proliferation. Langmuir. 2013;29(49):15359-15366. [15] LIU GF, JI W, WANG WL, et al. Multiresponsive hydrogel coassembled from phenylalanine and azobenzene derivatives as 3D scaffolds for photoguiding cell adhesion and release. ACS Appl Mater Interfaces. 2015;7(1):301-307. [16] YAMANAKA M. Development of C3-symmetric Tris-urea low-molecular-weight gelators. Cheminform. 2016;16(2):768-782. [17] WEIBIN B, LIFANG C. Solvent-induced controllable self-assembly of poly(9,9-dihexylfluorene). Chem Lett. 2014;43(3):331-333. [18] Lo PY, Lee GY, Zheng JH, et al. Intercalating pyrene with peptide as a novel self-assembly nano-carrier for colon cancer suppression in vitro and in vivo. Mater Sci Eng C. 2020;109:0928-4931. [19] HANEIN D, GEIGER B, ADDADI L. Differential adhesion of cells to enantiomorphous crystal surfaces. Science. 1994;263(5152):1413-1416. [20] HARTGERINK JD, BENIASH E, STUPP SI. Self-assembly and mineralization of peptide-amphiphile nanofibers. Science. 2001;294(5547):1684-1688. [21] LIAO G, WANG L, YU W. Application of novel targeted molecular imaging probes in the earlydiagnosis of upper urinary tract epithelial carcinoma. Oncol Lett. 2018;16(5):6349-6354. [22] Feng CL, Dou X, Zhang D, et al. A highly efficient self‐assembly of responsive c2‐cyclohexane‐derived gelators. Macromol Rapid Commun. 2012;33(18):1535-1541. [23] HONG Y, PRITZKER MD, LEGGE RL, et al. Effect of NaCl and peptide concentration on the self-assembly of an ionic-complementary peptide EAK16-II. Colloids Surf B. 2005;46(3):152-161. [24] KABIRI M, BUSHNAK I, MCDERMOT MT, et al. Toward a mechanistic understanding of ionic self-complementary peptide self-assembly:role of water molecules and ions. Biomacromolecules. 2013;14(11):3943-3950. [25] MUJEEB A, MILLER AF, SAIANI A, et al. Self-assembled octapeptide scaffolds for in vitro chondrocyte culture. Acta Biomater. 2013;9(1):4609-4617. [26] KUMAR VA, TAYLOR NL, SHI S, et al. Self-assembling multidomain peptides tailor biological responses through biphasic release. Biomaterials. 2015; 52(1):71-78. [27] LEI W, PEIPEI Y, XIAOXIAO Z, et al. Self-assembled nanomaterials for photoacoustic imaging. Nanoscale. 2016;8(5):2488-2509. [28] ZHANG X, CHU X, WANG L, et al. Rational design of a tetrameric protein to enhance interactions between self-assembled fibers gives molecular hydrogels. Angew Chem Int Ed. 2012;51(18):4388-4392. [29] CARNY O, SHALEV DE, GAZIT E. Fabrication of coaxial metal nanocables using a self-assembled peptide nanotube scaffold. Nano Lett. 2006;6(8):1594-1597. [30] VAN BOMMEL KJC, VAN DER POL C, MUIZEBELT I, et al. Responsive cyclohexane-based low-molecular-weight hydrogelators with modular architecture. Angew Chem Int Ed. 2004;43(13):1663-1667. [31] WEISSLEDER R, PITTET MJ. Imaging in the era of molecular oncology. Nature. 2008;452(7187):580-589. [32] LEE S, XIE J, CHEN X. Peptides and peptide hormones for molecular imaging and disease diagnosis. Chem Rev. 2010;110(5):3087-3111. [33] FAN Z, SUN L M, HUANG Y J, et al. Bioinspired fluorescent dipeptide nanoparticles for targeted cancer cell imaging and real-time monitoring of drug release. Nat. Nanotechnol. 2016;11(4):388-394. [34] WU J, ZAWISTOWSKI A, EHRMANN M, et al. Peptide functionalized polydiacetylene liposomes act as a fluorescent turn-on sensor for bacterial lipopolysaccharide. J Am Chem Soc. 2011;133(25):9720-9723. [35] YANG PP, ZHAO XX, XU AP, et al. Reorganization of self-assembled supramolecular materials controlled by hydrogen bonding and hydrophilic-lipophilic balance. J Mater Chem B. 2016;4(15):2662-2668. [36] PARK Y, HONG HY, MOON HJ, et al. A new atherosclerotic lesion probe based on hydrophobically modified chitosan nanoparticles functionalized by the atherosclerotic plaque targeted peptides. J Control Release. 2008;128(3):217-223. [37] He M, Li J, Tan S, et al. Photodegradable Supramolecular Hydrogels with Fluorescence Turn-On Reporter for Photomodulation of Cellular Microenvironments. J Am Chem Soc. 2013;135(50):18718-18721. [38] TANG Y, WU Z, ZHANG CH, et al. Enzymatic activatable self-assembled peptide nanowire for targeted therapy and fluorescence imaging of tumors. Chem Commun. 2016;52(18):3631-3634. [39] SUN HY, LIU Y, ZHANG CT, et al. Peptidic β-sheets induce Congo red-derived fluorescence to improve the sensitivity of HIV-1 p24 detection. Anal Methods. 2017;9(7):1185-1189. [40] WANG L, LI W, LU J, et al. Supramolecular nano-aggregates based on bis(pyrene) derivatives for lysosome-targeted cell imaging. J Phys Chem C. 2013;117(50): 26811-26820. [41] LIANG G, REN H, RAO J. A biocompatible condensation reaction for controlled assembly of nanostructures in living cells. Nat Chem. 2010;2(1):54-60. [42] CHIEN MP, THOMPSON MP, BARBACK CV, et al. Enzyme‐Directed Assembly of a Nanoparticle Probe in Tumor Tissue. Adv Mater. 2013;25(26):3599-3604. [43] WANG D, SU HF, KWOK RTK, et al. Rational design of a water-soluble NIR AIEgen, and its application in ultrafast wash-free cellular imaging and photodynamic cancer cell ablation. Chem Sci. 2018;9(15):3685-3693. [44] YI H, LEILEI S, YUE S, et al. A fluorescent light-up aggregation-induced emission probe for screening gefitinib-sensitive non-small cell lung carcinoma. Biomater Sci. 2017;5(4):792-799. [45] DIMA A, BURTON NC, NTZIACHRISTOS V. Multispectral optoacoustic tomography at 64, 128, and 256 channels. J Biomed Opt. 2014;19(3):36021. [46] MINGHUA X, LV W. Photoacoustic imaging in biomedicine. Rev Sci Instrum. 2006; 77(4):041101. [47] YANG K, ZHU L, NIE L, et al. Visualization of protease activity in vivo using an activatable photo-acoustic imaging probe based on CuS nanoparticles. Theranostics. 2014;4(2):134-141. [48] LEVI J, SATHIRACHINDA A, GAMBHIR SS. A high affinity, high stability photoacoustic agent for imaging gastrin releasing peptide receptor in prostate cancer. Clin Cancer Res. 2014;20(14):3721-3729. [49] ZHANG T, CUI H, FANG CY, et al. Targeted nanodiamonds as phenotype-specific photoacoustic contrast agents for breast cancer. Nanomedicine. 2015;10(4):573-587. [50] LEVI J, KOTHAPALLI SR, MA TJ, et al. Design, synthesis, and imaging of an activatable photoacoustic probe. J Am Chem Soc. 2010;132(32):11264-11269. [51] WU X, YU G, LINDNER D, et al. Peptide targeted high-resolution molecular imaging of prostate cancer with MRI. Ame J Nucl Med Mol I. 2014;4(6):525. [52] ZHANG D, QI GB, ZHAO YX, et al. Photoacoustic imaging: in situ formation of nanofibers from purpurin18-peptide conjugates and the assembly induced retention effect in tumor sites. Adv Mater. 2015;27(40):6125-6130. [53] DE LA ZERDA A, LIU ZA, BODAPATI S, et al. Ultrahigh sensitivity carbon nanotube agents for photoacoustic molecular imaging in living mice. Nano Lett. 2010;10(6):2168-2172. [54] WANG Y, HU X, WENG J, et al. A photoacoustic probe for the imaging of tumor apoptosis by caspase‐mediated macrocyclization and self‐assembly. Angew Chem Int Ed. 2019;58(15):4886-4890. [55] CHEE HL, GAN CRR, NG M, et al. Biocompatible peptide-coated ultrasmall superparamagnetic iron oxide nanoparticles for in vivo contrast-enhanced magnetic resonance imaging. ACS Nano. 2018;12(7):6480-6491. [56] BULL SR, GULER MO, BRAS RE, et al. Self-assembled peptide amphiphile nanofibers conjugated to MRI contrast agents. Nano Lett. 2005;5(1):1-4. [57] YE F, JEONG E, JIA Z, et al. A peptide targeted contrast agent specific to fibrin-fibronectin complexes for cancer molecular imaging with MRI. Bioconjug Chem. 2008;19(12):2300-2303. [58] DIAFERIA C, GIANOLIO E, PALLADINO P, et al. Peptide materials obtained by aggregation of polyphenylalanine conjugates as gadolinium-based magnetic resonance imaging contrast agents. Adv Funct Mater. 2015;25(45):7003-7016. [59] YE D, SHUHENDLER AJ, PANDIT P, et al. Caspase-responsive smart gadolinium-based contrast agent for magnetic resonance imaging of drug-induced apoptosis. Chem Sci. 2014;5(10):3845-3852. [60] BECHET D, AUGER F, COULEAUD P, et al. Multifunctional ultrasmall nanoplatforms for vascular-targeted interstitial photodynamic therapy of brain tumors guided by real-time MRI. Nanomedicine. 2015;11(3):657-670. [61] PRESLAR AT, PARIGI G, MCCLENDON MT, et al. Gd(III) labeled peptide nanofibers for reporting on biomaterial localization in vivo. ACS Nano. 2014;8(7):7325-7332. [62] WANG T, WANG D, YU H, et al. Intracellularly acid-switchable multifunctional micelles for combinational photo/chemotherapy of the drug-resistant tumor. ACS Nano. 2016;10(3):3496-3508. [63] DEKRAFFT KE, XIE Z, CAO G, et al. Iodinated nanoscale coordination polymers as potential contrast agents for computed tomography. Angew Chem Int Edit. 2010;48(52):9901-9904. [64] KHODADUST F, AHMADPOUR S, ALIGHOLIKHAMSEH N, et al. An improved 99mTc-HYNIC-(Ser)3-LTVSPWY peptide with EDDA/tricine as co-ligands for targeting and imaging of HER2 overexpression tumor. Eur J Med Chem. 2018;144:767-773. [65] RANA S, NISSEN F, MARR A, et al. Optimization of a novel peptide ligand targeting human carbonic anhydrase IX. Plos One. 2012;7(5):1-11. [66] FLECHSIG P, LINDNER T, LOKTEV A, et al. PET/CT imaging of NSCLC with a αvβ6 integrin-targeting peptide. Mol Imaging Biol. 2019;21(5):973-983. [67] TORIIHARA A, DUAN H, THOMPSON HM, et al. 18F-FPPRGD2 PET/CT in patients with metastatic renal cell cancer. Eur J Nucl Med Mol I. 2019;46(7):1518-1523. [68] LI L, WU Y, WANG Z, et al. SPECT/CT imaging of the novel HER2-targeted peptide probe 99mTc-HYNIC-H6F in breast cancer mouse models. J Nucl Med. 2017;58(5):821-826. [69] ZHENG L, DING X, LIU K, et al. Molecular imaging of fibrosis using a novel collagen-binding peptide labelled with 99mTc on SPECT/CT. Amino Acids. 2017;49(1):89-101. [70] ZHOU J, DU X, WANG J, et al. Enzyme-instructed self-assembly of peptides containing phosphoserine to form supramolecular hydrogels as potential soft biomaterials. Front Chem Sci Eng. 2017;11(4):509-515. [71] WU Y, LI L, WANG Z, et al. Imaging and monitoring HER2 expression in breast cancer during trastuzumab therapy with a peptide probe 99mTc-HYNIC-H10F. Eur J Nucl Med Mol I. 2020;47(11):2613-2623. [72] ZAHID M, FELDMAN KS, GARCIA-BORRERO G, et al. Cardiac targeting peptide, a novel cardiac vector:studies in bio-distribution, imaging application, and mechanism of transduction. Biomolecules. 2018;8(4):147. [73] KENNEL SJ, STUCKEY A, MCWILLIAMS-KOEPPEN HP, et al. Tc-99m radiolabeled peptide p5 + 14 is an effective probe for spect imaging of systemic amyloidosis. Mol Imaging Biol. 2016;18(4):483-489. [74] HAYNIE TP. Radionuclide imaging techniques in the detection of cancer. Cancer. 1967;20(5):607-613. [75] YAN S, FENG W, LELE Z, et al. The experimental study on anti-tumor effect of “1”3”1I-Tyr-octreotide in nude mice bearing human non-small cell lung cancer. Chinese J Nucl Med. 2009;29(1):34-38. [76] DU S, LUO C, YANG G, et al. Developing PEGylated reversed d-peptide as a novel her2-targeted SPECT imaging probe for breast cancer detection. Bioconjug Chem. 2020;31(8):1971-1980. [77] JOKAR S, BEHNAMMANESH H, ERFANI M, et al. Synthesis, biological evaluation and preclinical study of a novel 99mTc-peptide: a targeting probe of amyloid-β plaques as a possible diagnostic agent for Alzheimer’s disease. Bioorg Chem. 2020;99:103857. [78] FAN L, JIARUI Y, SI C, et al. Peptide-rhodamine B probes containing laminin/fibronectin receptor-targeting sequence (YIGSR/RGD) for fluorescent imaging in cancers. Talanta. 2020;212:0039-9140. [79] HU K, SHANG J, XIE L, et al. PET imaging of VEGFR with a novel 64Cu-labeled peptide. ACS Omega. 2020;5(15):8508-8514. [80] ZHANG CL, LIU M, WU R, et al. Radiolabeled YSSCREKA peptide for targeting early thrombosis. J Nucl Med. 2018;59:1085. [81] QIN Y, CHENG S, LI Y, et al. The development of a Glypican-3-specific binding peptide using in vivo and in vitro two-step phage display screening for the PET imaging of hepatocellular carcinoma. Biomater Sci. 2020;8(20):5656-5665. [82] CHEN F, XIAO Y, SHAO K, et al. PET Imaging of a novel anxal-targeted peptide 18F-Al-NODA-Bn-p-SCN-GGGRDN-IF7 in A431 cancer mouse models. J Labelled Compd Rad. 2020;63(12):494-501. [83] WANG Q, LI SB, ZHAO YY, et al. Identification of a sodium pump Na+/K+ ATPase α1-targeted peptide for PET imaging of breast cancer. J Control Release. 2018; 281:178-188. |
| [1] | Song Yiwei, Lin Xiangxiang, Zhang Jianan, Chen Jue, Lu Haiping. Consistency of gingival thickness measurement based on cone-beam CT imaging and cone-beam CT superimposed intraoral scan imaging [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 569-573. |
| [2] | Gao Wenping, Zhang Zhihua, Han Fei, Hao Weiguo. Near-infrared fluorescence imaging for intraoperative neuroimaging: current applications and future development [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5225-5230. |
| [3] | Li Kun, Zhou Zheyuan, Wang Jian, Zhang Yan, Zhao Yan, He Xuetong, Li Ke, Chen Simin, Wu Xingyu, Wang Xing, Zhang Shaojie. Clinical significance of digital measurement of occipital condyle and foramen magnum in children [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(18): 2830-2834. |
| [4] | Hao Shuai, Ma Xun, Zhang Yannan, Zhao Haoliang, Liu Qingqing. Morphological classification of CT reconstruction of the narrowest part of pediculoisthmic component [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(18): 2876-2880. |
| [5] | Wang Wenli, Luo Lankun, Fu Yating, Chu Cheng, Ma Xiao, Liu Hua. Measurement and analysis of gingival biotype in maxillary anterior teeth using cone-beam computed tomography [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(23): 3616-3620. |
| [6] | Sun Lianlian, Liu Yongchao, Wang Zhixing. Repair effects of two kinds of bone repair materials on rabbit femoral defects compared using synchrotron-radiation-based micro-computed tomography [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3343-3348. |
| [7] | Wang Jian, Wang Xing, Li Kun, Gao Shang, Wang Chaoqun, Zhang Shaojie, Li Zhijun. Digital morphology of foramen magnum and occipital condyle: an observation based on three-dimensional reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3117-3122. |
| [8] | Zhan Ying, Wu Xiaodan, Hao Shanhu. Establishment of an animal model of radioactive 131I-induced hypothyroidism in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2631-2636. |
| [9] | Gan Tian, Wang Wenyuan, Yan Shujin, Hao Lan, Ran Haitao, Wang Zhigang, Xia Jizhu. Near infrared photoresponsive nanoparticles loaded with LXR agonists for photothermal immunotherapy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1863-1869. |
| [10] | Deng Zhaoyang, Yang Zhaohui. Three-dimensional model analysis of tibial plateau fracture lines [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 334-339. |
| [11] | Yang Ruijuan, Li Yangyang, Cai Ruiyan, Liu Huibin, Guo Chun. Interleukin-1 alpha induces osteoclast activation and bone loss [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(23): 3691-3699. |
| [12] | Li Yufan, Lin Mingyue, Wang Chenxin, Zhang Rui, Li Yubao, Chen Li, Zou Qin. Application of micro-CT in rabbit tibial osteomyelitis modeling [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(18): 2874-2880. |
| [13] | Li Jinglian, Zhang Hongfei, Yan Jiapeng, Zhou Jiabin, Yu Hao. Lag screw path for fixation of sacroiliac joint dislocation through S1 pedicle [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(15): 2337-2341. |
| [14] | Hu Weiran, Song Yuepeng, Shi Xinge, Ma Haohao, Wang Hongqiang, Zhang Kai, Gao Kun, Shao Jia, Gao Yanzheng. Sensitivity and specificity of coronal computed tomography reconstruction and traditional methods for determining screw loosening in spinal internal fixation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(15): 2372-2376. |
| [15] | Du Xueting, Yang Yang, Huang Wenhua, Chen Wubiao. Clinical application and breakthrough of three-dimensional printing based on medical imaging technology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(18): 2887-2894. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||