Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (19): 2960-2967.doi: 10.12307/2023.606
Previous Articles Next Articles
Effects of iRoot BP Plus combined with Yunnan Baiyao on mineralization of dental pulp stem cells under inflammatory conditions
Zhao Ying1, Liang Guangzhi2, Zhou Yuqi2, Wang Tianqi2, Liu Yunxia2, Liu Xiaoying3, Zhang Juanjuan2, Sun Yan2
- 1Affiliated Hospital of Weifang Medical University, Weifang 261035, Shandong Province, China; 2School of Stomatology, Weifang Medical University, Weifang 261000, Shandong Province, China; 3School of Life Science and Technology, Weifang Medical University, Weifang 261000, Shandong Province, China
-
Received:2022-03-11Accepted:2022-07-09Online:2023-07-08Published:2022-11-28 -
Contact:Sun Yan, Master, Associate professor, School of Stomatology, Weifang Medical University, Weifang 261000, Shandong Province, China -
About author:Zhao Ying, Master, Affiliated Hospital of Weifang Medical University, Weifang 261035, Shandong Province, China
CLC Number:
Cite this article
Zhao Ying, Liang Guangzhi, Zhou Yuqi, Wang Tianqi, Liu Yunxia, Liu Xiaoying, Zhang Juanjuan, Sun Yan. Effects of iRoot BP Plus combined with Yunnan Baiyao on mineralization of dental pulp stem cells under inflammatory conditions[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(19): 2960-2967.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
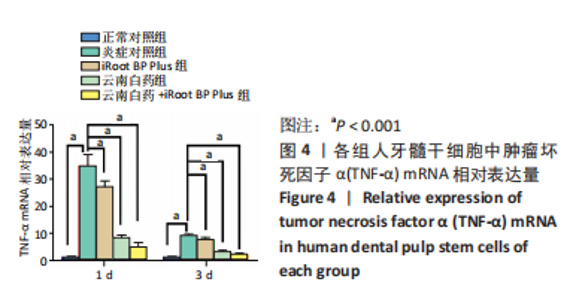
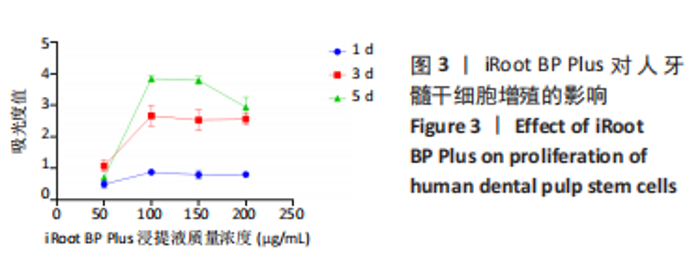
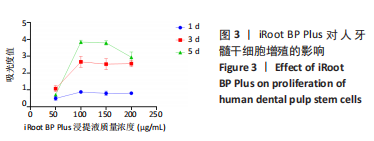
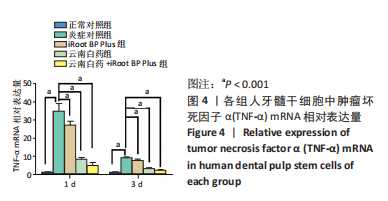
随着时间的增加,不同质量浓度云南白药组的牙髓干细胞相对数量均增加。第1天,当云南白药质量浓度≤75 μg/mL时,随着云南白药质量浓度的升高细胞数量无明显变化,当云南白药质量浓度为100 μg/mL时,细胞数量有所升高;第3天,当云南白药质量浓度≤75 μg/mL时,随着云南白药质量浓度的升高细胞数量无明显变化,当云南白药质量浓度为100 μg/mL时,细胞数量出现下降;第5天,当云南白药质量浓度≤50 μg/mL时,细胞数量略有降低,而云南白药质量浓度为50-75 μg/mL时,细胞数量随着云南白药质量浓度的增加而升高,而在云南白药质量浓度为100 μg/mL时,细胞数量出现明显下降,即当云南白药浸提液质量浓度为75 μg/mL时,药物对牙髓干细胞增殖的影响最小,细胞毒性小,因而在后续实验中,云南白药浸提液选用75 μg/mL质量浓度。 随着时间增加,不同质量浓度iRoot BP Plus浸提液组细胞数量同样整体呈增加趋势。第1天和第3天,当iRoot BP Plus浸提液质量浓度≤100 μg/mL时,细胞数量随iRoot BP Plus质量浓度升高而增加,当iRoot BP Plus质量浓度≥100 μg/mL时,细胞数量无明显变化;在第5天,在质量浓度≤100 μg/mL时细胞数量随着iRoot BP Plus质量浓度升高而增加,当质量浓度为150 μg/mL时,细胞数量未见继续增加,且当质量浓度升至200 μg/mL时,细胞数量反而较之前下降,通过上述实验结果,选择质量浓度为100 μg/mL的iRoot BP Plus浸提液应用于后续实验。 2.3 云南白药及iRoot BP Plus浸提液对脂多糖诱导下牙髓干细胞炎症的影响 炎症对照组肿瘤坏死因子α mRNA相对表达量均高于正常对照组(P < 0.001),表明经脂多糖处理后细胞进入炎症状态,细胞炎症模型建立成功。经云南白药、iRoot BP Plus及其联合应用处理1,3 d后,肿瘤坏死因子α mRNA相对表达量均低于炎症对照组(P < 0.001),表明上述2种药物对炎症相关因子肿瘤坏死因子α mRNA均具有抑制作用,在上述2个时间点云南白药组对肿瘤坏死因子α mRNA相对表达量的抑制作用明显强于iRoot BP Plus组,云南白药+iRoot BP Plus组对肿瘤坏死因子α mRNA相对表达量的抑制作用强于2个单独处理组。初步认定云南白药对牙髓干细胞炎症状态的抑制作用较iRoot BP Plus具有明显优势,且2种药物具有协同作用,见图4。"
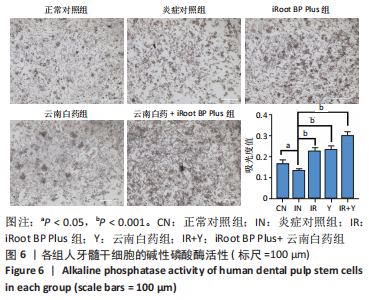
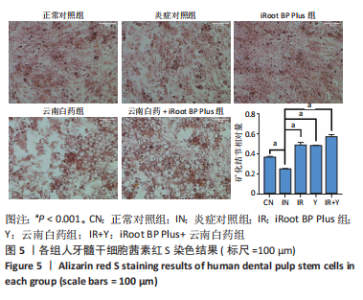
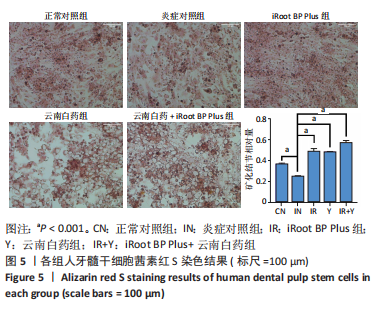
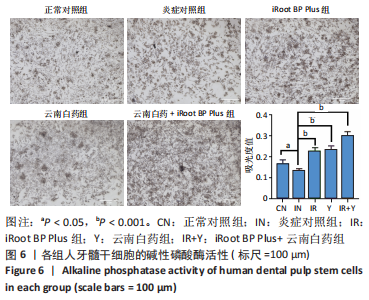
2.5 云南白药及iRoot BP Plus浸提液对脂多糖诱导下牙髓干细胞碱性磷酸酶活性的影响 炎症对照组碱性磷酸酶活性低于正常对照组,表明脂多糖刺激牙髓干细胞后碱性磷酸酶的表达降低(P < 0.05)。云南白药组、iRoot BP Plus组及iRoot BP Plus+云南白药组的碱性磷酸酶活性均高于炎症对照组(P < 0.001)。云南白药组与iRoot BP Plus组碱性磷酸酶活性比较无统计学意义,但2种药物联合应用组碱性磷酸酶活性高于药物单独刺激组(P < 0.001),见图6。表明云南白药及iRoot BP Plus可提高牙髓干细胞的碱性磷酸酶活性,且两者联合应用对于碱性磷酸酶活性的促进具有正向作用。"
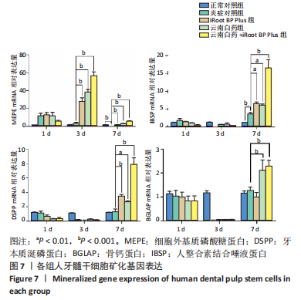
2.6 云南白药及iRoot BP Plus浸提液对脂多糖诱导下牙髓干细胞矿化相关基因表达的影响 用实时荧光定量PCR法检测第1,3,7天时矿化相关基因的表达,见图7,使用2-ΔΔCT公式计算,通过观察统计结果得出,经药物刺激后,MEPE mRNA的表达量大幅度增加发生在第3天,刺激3 d后,正常对照组与炎症对照组相比无统计学意义(P > 0.05),与炎症对照组相比,云南白药组、iRoot BP Plus组和云南白药+ iRoot BP Plus组MEPE mRNA表达均明显升高(P < 0.001),且云南白药+iRoot BP Plus组明显高于云南白药组和iRoot BP Plus组(P < 0.001);刺激7 d后,正常对照组MEPE mRNA表达高于炎症对照组(P < 0.001),云南白药组、iRoot BP Plus组和云南白药+iRoot BP Plus组MEPE mRNA表达均高于炎症对照组(P < 0.001),云南白药+iRoot BP Plus组MEPE mRNA表达高于云南白药组和iRoot BP Plus组(P < 0.001)。"
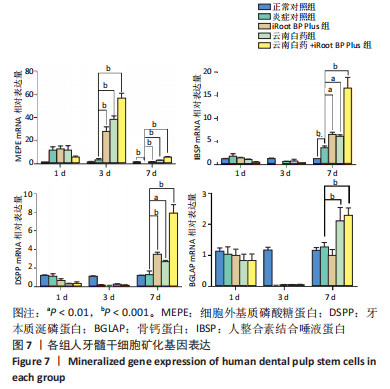
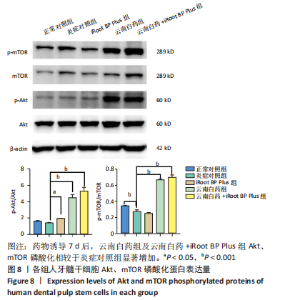
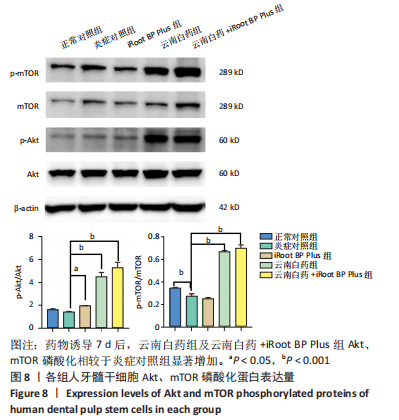
IBSP mRNA的相对表达量在药物刺激7 d后出现明显差异,此时正常对照组低于炎症对照组(P < 0.01),云南白药组和iRoot BP Plus组高于炎症对照组(P < 0.01),云南白药+iRoot BP Plus组高于炎症对照组(P < 0.001),云南白药+iRoot BP Plus组高于云南白药组和iRoot BP Plus组(P < 0.001)。 DSPP mRNA的相对表达量在药物刺激7 d后出现明显差异,正常对照组与炎症对照组相比无统计学意义(P > 0.05),iRoot BP Plus组和云南白药+iRoot BP Plus组高于炎症对照组(P < 0.001),云南白药组高于炎症对照组(P < 0.01),云南白药+iRoot BP Plus组高于云南白药组和iRoot BP Plus组(P < 0.001)。 在刺激7 d后,云南白药组和云南白药+iRoot BP Plus组的BGLAP mRNA相对表达量高于炎症对照组(P < 0.001),而iRoot BP Plus组与炎症对照组相比均无统计学差异(P > 0.05)。 2.7 云南白药及iRoot BP Plus浸提液与脂多糖诱导下牙髓干细胞Akt/mTOR通路的关系 Akt磷酸化水平在正常对照组、炎症对照组之间无显著差异(P > 0.05);iRoot BP Plus组(P < 0.05)、云南白药组(P < 0.001)、iRoot BP Plus+云南白药组(P < 0.001)Akt磷酸化水平明显高于炎症对照组。mTOR磷酸化水平在炎症对照组与iRoot BP Plus组无明显差异,云南白药组及iRoot BP Plus+云南白药组与炎症对照组比较,磷酸化水平均明显增加(P < 0.001),见图8。"
| [1] LINAS N, DECERLE N, MUNOZ-SANCHEZ ML, et al. Long-term Outcomes of Full Pulpotomy in Permanent Molars for Patients Treated in a Single, Short Session under Special Conditions. J Endod. 2020;46(11):1597-1604. [2] TAN SY, YU VSH, LIM KC, et al. Long-term Pulpal and Restorative Outcomes of Pulpotomy in Mature Permanent Teeth. J Endod. 2020;46(3):383-390. [3] ZANINI M, HENNEQUIN M, COUSSON PY. A Review of Criteria for the Evaluation of Pulpotomy Outcomes in Mature Permanent Teeth. J Endod. 2016;42(8):1167-1174. [4] CUSHLEY S, DUNCAN HF, LAPPIN MJ, et al. Pulpotomy for mature carious teeth with symptoms of irreversible pulpitis: A systematic review. J Dent. 2019;88:103158. [5] LUIZ DE OLIVEIRA DA ROSA W, MACHADO DA SILVA T, FERNANDO DEMARCO F, et al. Could the application of bioactive molecules improve vital pulp therapy success? A systematic review. J Biomed Mater Res A. 2017;105(3):941-956. [6] PEZELJ-RIBARIC S, ANIC I, BREKALO I, et al. Detection of tumor necrosis factor alpha in normal and inflamed human dental pulps. Arch Med Res. 2002;33(5):482-484. [7] SILVA AC, FARIA MR, FONTES A, et al. Interleukin-1 beta and interleukin-8 in healthy and inflamed dental pulps. J Appl Oral Sci. 2009;17(5):527-532. [8] RAO Q, KUANG J, MAO C, et al. Comparison of iRoot BP Plus and Calcium Hydroxide as Pulpotomy Materials in Permanent Incisors with Complicated Crown Fractures: A Retrospective Study. J Endod. 2020;46(3):352-357. [9] LU J, LI Z, WU X, et al. iRoot BP Plus promotes osteo/odontogenic differentiation of bone marrow mesenchymal stem cells via MAPK pathways and autophagy. Stem Cell Res Ther. 2019;10(1):222. [10] CHEN X, ZHANG H, ZHONG J, et al. Comparison of indirect pulp treatment and iRoot BP Plus pulpotomy in primary teeth with extremely deep caries: a prospective randomized trial. Clin Oral Investig. 2021;25(5):3067-3076. [11] ABOU ELREASH A, HAMAMA H, GRAWISH M, et al. A laboratory study to test the responses of human dental pulp stem cells to extracts from three dental pulp capping biomaterials. Int Endod J. 2021;54(7):1118-1128. [12] LI R, ALEX P, YE M, et al. An old herbal medicine with a potentially new therapeutic application in inflammatory bowel disease. Int J Clin Exp Med. 2011;4(4):309-319. [13] TANG ZL, WANG X, YI B, et al. Effects of the preoperative administration of Yunnan Baiyao capsules on intraoperative blood loss in bimaxillary orthognathic surgery: a prospective, randomized, double-blind, placebo-controlled study. Int J Oral Maxillofac Surg. 2009;38(3):261-266. [14] 黎慧,于浩滢,杨小佳,等.云南白药提取物对大鼠实验性牙周炎的治疗作用[J].华西药学杂志,2018,33(6):472-474. [15] WANG S, JIAO J, DONG X, et al. Effect of total knee replacement combined with Yunnan Baiyao on the expression of serum IL-6, TNF-α, VCAM-1 and MMP-9 in patients with knee osteoarthritis. Panminerva Med. 2020 Aug 5. doi: 10.23736/S0031-0808.20.04016-1. Online ahead of print. [16] GRONTHOS S, MANKANI M, BRAHIM J, et al. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci U S A. 2000;97(25): 13625-13630. [17] MIURA M, GRONTHOS S, ZHAO M, et al. SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci U S A. 2003;100(10): 5807-5812. [18] ABD-ELMEGUID A, YU DC, KLINE LW, et al. Dentin matrix protein-1 activates dental pulp fibroblasts. J Endod. 2012;38(1):75-80. [19] SILLER AF, WHYTE MP. Alkaline Phosphatase: Discovery and Naming of Our Favorite Enzyme. J Bone Miner Res. 2018;33(2):362-364. [20] HALLING LINDER C, EK-RYLANDER B, KRUMPEL M, et al. Bone Alkaline Phosphatase and Tartrate-Resistant Acid Phosphatase: Potential Co-regulators of Bone Mineralization. Calcif Tissue Int. 2017;101(1):92-101. [21] CHAUDHARY SC, KUZYNSKI M, BOTTINI M, et al. Phosphate induces formation of matrix vesicles during odontoblast-initiated mineralization in vitro. Matrix Biol. 2016;52-54:284-300. [22] SIMÃO AM, BOLEAN M, HOYLAERTS MF, et al. Effects of pH on the production of phosphate and pyrophosphate by matrix vesicles’ biomimetics. Calcif Tissue Int. 2013;93(3):222-232. [23] MALAVAL L, AUBIN JE, VICO L. Role of the small integrin-binding ligand N-linked glycoprotein (SIBLING), bone sialoprotein (BSP) in bone development and remodeling. Osteoporos Int. 2009;20(6):1077-1080. [24] ZHANG YL, YIN JH, DING H, et al. Protective effect of VK2 on glucocorticoid-treated MC3T3-E1 cells. Int J Mol Med. 2017;39(1):160-166. [25] ZOCH ML, CLEMENS TL, RIDDLE RC. New insights into the biology of osteocalcin. Bone. 2016;82:42-49. [26] AN Y, SONG Y, WANG Z, et al. Effect of low-intensity pulsed ultrasound on the biological behaviors of bone marrow mesenchymal stem cells on titanium with different surface topographies. Am J Transl Res. 2018;10(1):67-76. [27] RAHMAN SU, OH JH, CHO YD, et al. Fibrous Topography-Potentiated Canonical Wnt Signaling Directs the Odontoblastic Differentiation of Dental Pulp-Derived Stem Cells. ACS Appl Mater Interfaces. 2018;10(21):17526-17541. [28] BERTACCHINI J, HEIDARI N, MEDIANI L, et al. Targeting PI3K/AKT/mTOR network for treatment of leukemia. Cell Mol Life Sci. 2015;72(12):2337-2347. [29] MANFREDI GI, DICITORE A, GAUDENZI G, et al. PI3K/Akt/mTOR signaling in medullary thyroid cancer: a promising molecular target for cancer therapy. Endocrine. 2015;48(2):363-370. [30] FERRERO H. Commentary on regulation of leiomyoma stem cell proliferation by WNT4 via AKT-dependent β-Catenin activation. Fertil Steril. 2020;114(6): 1192-1193. [31] JIANG Y, LIU F, ZOU F, et al. PBX homeobox 1 enhances hair follicle mesenchymal stem cell proliferation and reprogramming through activation of the AKT/glycogen synthase kinase signaling pathway and suppression of apoptosis. Stem Cell Res Ther. 2019;10(1):268. [32] LEI X, JIAO J. UTX Affects Neural Stem Cell Proliferation and Differentiation through PTEN Signaling. Stem Cell Reports. 2018;10(4):1193-1207. [33] RUSSELL RC, FANG C, GUAN KL. An emerging role for TOR signaling in mammalian tissue and stem cell physiology. Development. 2011;138(16): 3343-3356. [34] ZHOU R, MA Y, QIU S, et al. Metformin promotes cell proliferation and osteogenesis under high glucose condition by regulating the ROS‑AKT‑mTOR axis. Mol Med Rep. 2020;22(4):3387-3395. [35] ALSALLEEH F, CHUNG N, STEPHENSON L. Antifungal activity of endosequence root repair material and mineral trioxide aggregate. J Endod. 2014;40(11):1815-1819. |
| [1] | Fang Xingyan, Tian Zhenli, Zhao Zheyi, Wen Ping, Xie Tingting. Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Guo Shuhui, Yang Ye, Jiang Yangyang, Xu Jianwen. Screening and validation of neurogenic bladder miRNA-mRNA regulatory network [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-8. |
| [3] | Tang Liang, Li Xiheng, Niu Ruijuan, Li Xinyue, Zou Xinying, Mao Tianjiao, Li Jiang. Naringin regulates the function of RAW264.7 macrophages to affect the osteogenic differentiation of MC-3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1205-1210. |
| [4] | Liang Jiaqi, Liu Hengxu, Yang Jinxin, Yang Yi, Deng Xuhui, Tan Mingjian, Luo Jiong. Health benefit relationship between exercise and intestinal bacteria [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1292-1299. |
| [5] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [6] | Zhang Tingting, Liu Juan, Zhang Xu. Bioactivity of phase-transition lysozyme for surface modification of zirconia all-ceramic implant material mediating hydroxyapatite coating [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1043-1049. |
| [7] | Wang Min, Yin Xiushan, Wang Yingxi, Zhang Yan, Zhao Long, Xia Shuyue. Inhalation of bone marrow mesenchymal stem cells-derived exosomes alleviates inflammatory injury in chronic obstructive pulmonary disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 827-834. |
| [8] | Qiao Luhui, Ma Ziyu, Guo Haoyu, Hou Yudong. Comparison of puerarin and icariin on the biological properties of mouse preosteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 872-877. |
| [9] | Li Zhichao, Tan Guoqing, Su Hui, Xu Zhanwang, Xue Haipeng. Regulatory role of non-coding RNAs as potential therapeutic targets in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 758-764. |
| [10] | Zhang Lichen, Chen Liang, Gu Yong. Inorganic ion bionic periosteum regulates immune microenvironment to promote bone repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 346-353. |
| [11] | Zong Mingrui, Liu Haiyan, Li Bing, Wu Xiuping. Application of carboxymethyl chitosan in tissue engineering of stomatology [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 447-452. |
| [12] | Liu Huan, Li Han, Ma Yunhao, Zhong Weijian, Ma Guowu. Osteogenic capacity of partially demineralized dentin particles in the maxillary sinus lift [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 354-359. |
| [13] | Wang Ziyang, Zuo Enjun. Application of biological materials in vital pulp therapy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 427-433. |
| [14] | Chen Bohao, He Qi, Yang Junzheng, Pan Zhaofeng, Xiao Jiacong, Li Miao, Li Shaocong, Zeng Jiaxu, Wang Haibin, Zheng Jia, Zhang Meng. Significance of Piezo1 protein in the pathogenesis of osteonecrosis of femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(27): 4414-4420. |
| [15] | Sun Fangyuan, Meng Jialei, Ma Yuhui, Geng Huan, Zhang Tao. Effects of ligustrazine on inflammation and oxidative stress in rat cardiomyocytes induced by lipopolysaccharide [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3253-3258. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||