Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (20): 3259-3265.doi: 10.12307/2023.482
Previous Articles Next Articles
Role and application of bone morphogenetic protein 2 in the repair of osteochondral defects
Wang Buyu, Zhang Yong, Li Feifei, Dong Xiaoyu, Deng Jiang, Ruan Shiqiang
- Third Affiliated Hospital of Zunyi Medical University (First People’s Hospital of Zunyi City), Zunyi 563000, Guizhou Province, China
-
Received:2022-07-14Accepted:2022-08-24Online:2023-07-18Published:2022-11-21 -
Contact:Ruan Shiqiang, MD, Chief physician, Master’s supervisor, Third Affiliated Hospital of Zunyi Medical University (First People’s Hospital of Zunyi City), Zunyi 563000, Guizhou Province, China -
About author:Wang Buyu, Physician, Third Affiliated Hospital of Zunyi Medical University (First People’s Hospital of Zunyi City), Zunyi 563000, Guizhou Province, China -
Supported by:Joint Science and Technology Research and Development Fund Project of Zunyi First People’s Hospital & Zunyi Science and Technology Bureau, No. HZ(2019)186 (to RSQ); Guizhou Provincial Science and Technology Program Projects, Nos. ZK[2021] 393 and LC[2022]029 (to RSQ); Science and Technology Fund Project of Guizhou Provincial Health and Health Commission, No. gzwkj2021-259 (to RSQ)
CLC Number:
Cite this article
Wang Buyu, Zhang Yong, Li Feifei, Dong Xiaoyu, Deng Jiang, Ruan Shiqiang. Role and application of bone morphogenetic protein 2 in the repair of osteochondral defects[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3259-3265.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

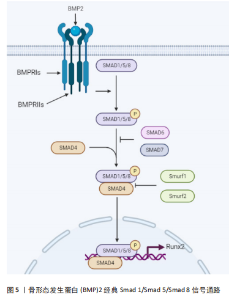
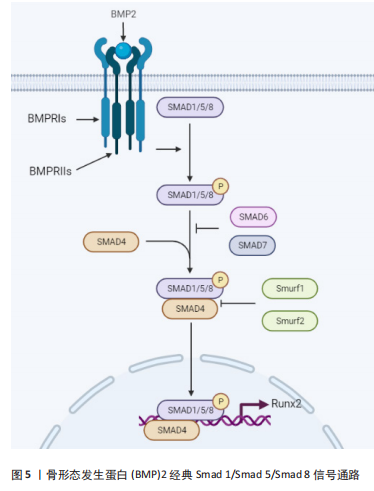
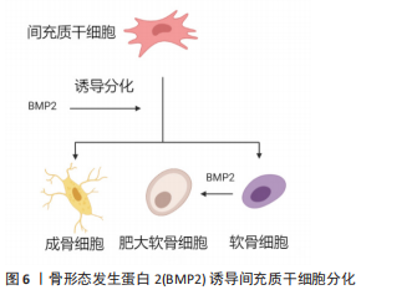
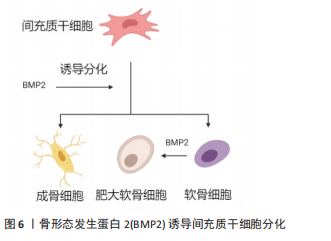
目前关于BMP2对骨骼发育和诱导干细胞成骨诱导的研究是骨科领域的一大热点。BMP2信号通过丝氨酸(Ser)/苏氨酸(Thr)蛋白激酶受体转导,即Ⅰ型受体(BMPRIs)和Ⅱ型受体(BMPRIIs)。这两种受体类型结合成一个功能复合物,把BMP2信号传导入细胞内。BMP2配体先激活BMPRⅡ,然后,BMPRⅡ将BMPRⅠ磷酸化。被激活的BMPRI招募并磷酸化Smad依赖性信号通路,然后将信号转入细胞核并控制成骨基因的表达。激活的BMPRI磷酸化Smad依赖性信号途径,也就是经典Smad 1/5/8途径,与Smad 4结合形成混合复合物,被输送到细胞核并调控靶基因转录。在胞浆中Smad 6或7通过阻止受体激活的Smad(R-Smad)磷酸化来负向调节Smad信号,抑制成骨相关基因的表达,阻断成骨细胞的分化。泛素连接酶1和2通过直接和Smad 1/5/8/4复合体结合,诱导复合体泛素化来抑制成骨相关基因的表达。进入细胞核后,BMP2信号可以刺激主要的成骨转录因子Runt相关转录因子2(Runt-related transcription factor 2,Runx2),Runx2在诱导干细胞成骨方面起着关键作用[10-11]。Runx2属于Runx家族,该家族有DNA结合域Runt,由Runx1、Runx2和Runx3组成,Runx2在成骨细胞和软骨细胞中表达。在软骨细胞中,Runx2在静止的软骨细胞中微弱表达,在肥大前的软骨细胞中表达上调,这种上调的表达一直保持到分化为肥大软骨细胞。Runx2在未分化的间质细胞中微弱表达,其表达在前骨细胞中上调,在未成熟的成骨细胞中达到最高水平,在成熟的成骨细胞中下调[12]。Runx2缺陷的小鼠缺乏成骨细胞和骨形成,软骨细胞的成熟受到明显抑制[13]。 2.2 BMP2对软骨的作用 2.2.1 BMP2诱导软骨分化和维持软骨细胞表型 目前关于BMP2如何诱导软骨分化的机制尚不清楚。但有学者通过对软骨表达基因-Sox9的研究发现,Sox9的过度表达导致BMP2诱导的间充质干细胞中Smad7的表达减少;Sox9抑制了BMP2诱导的骨细胞蛋白的表达,同时增强了间充质干细胞中软骨标志物Col2a1的表达,Sox9在间充质干细胞中的表达促进了BMP2诱导的软骨生成并抑制了BMP2诱导的软骨内层骨化;小鼠肢体移植实验显示,Sox9扩大了BMP2刺激的软骨细胞增殖区,而Smad7促进了BMP2诱导的生长板肥大区[14]。综上所述,该实验表明,Sox9可能通过下调Smad7促进BMP2诱导的软骨生成,这有希望被用于软骨组织工程。有学者通过深入研究BMP2的受体,发现BMP2的Ⅰ型受体有3种,分别是ALK-3 (BMPRIA)、ALK-6 (BMPRIB)和ALK-2,而Ⅱ型受体也有3种,不过是BMP家族通用的受体,BMP2受体分类见表1。与BMPR1A信号相比,更强的BMPR1B信号可防止软骨细胞肥大[15]。但具体如何使BMP2信号更好地刺激BMPR1B,目前还没有具体的研究。"


2.2.2 BMP2通过调节软骨细胞的糖代谢来控制软骨发育 正常情况下,人体内的大部分软骨细胞处于低氧的环境中,尤其是经常容易受到损伤的关节软骨,它是一种无血管组织,通过滑膜液和软骨下骨的扩散获得营养和氧气供应,因此关节软骨一生都维持在低氧环境中[18],故而糖酵解是软骨细胞中最重要的能量产生途径。而软骨损伤的时候,软骨细胞所处的内环境遭到破坏,软骨细胞的代谢可能会发生改变,不利于受损软骨的修复。最近的研究表明,在软骨发育过程中,BMP2严格控制葡萄糖的无氧酵解,来维持软骨细胞的正常代谢[19]。MONACO等[20]发现,在含有BMP2培养基的情况下,在2%的氧气浓度下,软骨生成明显减少了碱性磷酸酶的表达,减少了Ⅰ型胶原的沉积,产生了更稳定和更少的肥大软骨表型,2%的氧气浓度在蛋白质水平上没有抑制成骨分化,但减少了碱性磷酸酶和骨钙素的表达;而在20%的氧气浓度下,成骨过程中碱性磷酸酶和骨钙素发生上调。这项研究表明,在体外使用相同的供体细胞群时,氧气浓度的调节,结合组织特异性生长因子和培养基成分,可以促进软骨或软骨内分化,但其具体的机制还不清楚,作者推测软骨细胞在低氧浓度下,糖酵解产生的能量较少,用来维持自身表型;而在高氧环境中,软骨细胞的糖代谢产生更高的能量,促使自身转变成肥大软骨细胞最后成骨。 2.3 BMP2在各类骨缺损修复中的应用 2.3.1 BMP2在颅骨缺损修复中的作用 颅骨的自我修复能力极差,相关研究表明缺损直径在8 mm以上的颅骨缺损在12个月后仍难以自然愈合,缺损部位会被致密的纤维组织填充,但应用BMP2之后能够大幅缩短颅骨的愈合时间[21]。一项研究表明,BMP2能够吸收胶原海绵,并在4周内促使缺损8 mm的大鼠颅骨完全再生,在8周内即可恢复到正常骨密度,而且无论是大肠埃希菌源性的BMP2和哺乳动物源性的BMP2都能做到这一点[22]。REEVES等[23]以30∶70的比例用羟基磷灰石/磷酸三钙(HA/TCP)的双相生物材料修复了兔颅骨缺损,在对照实验中应用BMP2的一组成骨量显著增加,而且应用BMP2的一组缺损处的骨再生具有更高的一致性,而未应用BMP2的骨再生主要存在于缺损的边缘处,有向中央发展的趋向性。AHMED等[24]用腺病毒介导的BMP2体外转染骨髓间充质干细胞后进行猪颅骨缺损的修复,直径4 cm的缺损在6个月内被修复,苏木精-伊红染色三维CT观察下,可见新生的颅骨为双层皮质骨,无论是硬度还是厚度都要优于生物多聚物组的修复效果。有学者研究在BMP2介导下的不同载体修复犬颅骨缺损,结果显示无论是胶原海绵、羟基磷灰石/磷酸三钙、胶原海绵+皮松质骨片、牛Ⅰ型胶原+羟基磷灰石/磷酸三钙都能诱导新骨形成,在修复6个月后各组的X射线阻射率都能够达到99.9%,但不同材料都有异位骨形成[25]。 2.3.2 BMP2在颌面骨缺损修复中的作用 口腔颌面骨具有支撑面型的作用,如果发生颌面骨缺损不但会影响面部器官的功能性,还会影响患者的容貌,给患者造成心理负担[26]。颌面骨具有微妙的三维结构特性,在颌面骨的缺损修复上也具有一定的难度[27]。BMP2介导的生物工程骨能够满足各种类型缺损修复的需求,2007年已被FDA批准用于牙槽嵴增高术和鼻窦提升术[28]。 SUN等[29]的研究中,将BMP2转染的骨髓间充质干细胞与天然珊瑚相结合,用于修复12 mm直径的兔眶下缘骨缺损,在16周内基本完成修复,三维CT观察显示缺损部位愈合良好,无论是成骨量还是骨密度都要优于未用BMP2介导的组织工程骨。GUIHARD等[30]的报道中显示,重组人BMP2复合胶原用于修复下颌骨缺损,在18个月的随访中,所有患者在4个月内完成缺损处的新骨形成,并且在随访期间内所有患者均达到骨性愈合。在一项动物实验中,用BMP2介导自体骨髓间充质干细胞结合天然珊瑚做生物支架修复大鼠下颌升支骨缺损,4周内有新骨形成,8周内有成熟新骨形成,而未用BMP2介导的对照组在8周时仅有少量脂肪组织和极少量的新骨形成[31]。ZHANG等[32]应用负载BMP2的羟基磷灰石/磷酸三钙材料修复鼠下颌骨缺损,2周内可观察到有骨小梁形成,4周内可见再血管化和大量的矿化沉积,8周时影像学检查可见缺损部位再生度达到80%以上,且组织学分析结果为成熟骨组织。 2.3.3 BMP2在四肢长骨缺损修复中的作用 长骨缺损是长期困扰临床的一种治疗难题,长骨开放性骨折后的骨不连、骨缺损严重影响患者的预后[33]。2004年起FDA允许重组人BMP2用于开放型胫骨骨折的临床治疗,近年来BMP2在各类长骨缺损修复上也有了较好的应用。HE等[34-35]将BMP2转染的胶原海绵用于兔桡骨15 mm临界缺损修复,12周内支架材料完全吸收,基本完成骨重建和骨髓腔再通。同样是一项兔桡骨缺损修复实验中,用BMP2活化的纳米骨浆进行15 mm桡骨临界缺损修复,4周时可见骨痂形成,且修复边缘的界限模糊,8周内基本完成修复工作,12周时纳米骨浆被完全吸收,完成髓腔再通,骨皮质连接良好,且BMP2活化组的新骨抗压强度要明显高于单纯纳米骨浆组[36]。PENG等[37]将负载BMP2的聚己内酯支架用于修复兔尺骨骨干缺损,组织学评估提示骨修复速度显著高于单纯应用聚己内酯组和空白对照组,并且在8周内完成修复。LIN等[38]将BMP2转染的脂肪组织和肌肉组织用于修复大鼠股骨中段缺损,放射学观察在修复10 d后形成骨桥,8周内缺损处已经达到正常骨强度,相比之下脂肪组织的修复稳定性和修复速度都不如肌肉组织。LOU等[39]将BMP2介导的生物组织材料用于修复慢性胫骨骨髓炎导致的骨缺损,并与自体骨移植效果进行了对比,结果显示研究组的愈合时间显著低于对照组,愈合率显著高于对照组。李侠等[40]采用磷酸钙骨水泥联合BMP2作为植骨材料,研究了该方案在治疗老年肱骨近端骨折中的疗效,结果显示与单纯的磷酸钙骨水泥填充相比,结合BMP2的实验组骨性愈合时间更短,且术后1年内实验组的关节功能评分显著高于对照组。NOMURA等[41]用BMP2结合胶原海绵进行成人胫骨缺损修复,缺损平均4 cm,结果与自体骨移植的效果和安全性相近。PENG等[42]通过组织学和放射学观察证实,BMP2结合胶原海绵能够加速大鼠胫骨节段性骨缺损的修复。SU等[43]的研究中用聚己内酯修复大鼠胫骨缺损,观察到局部注射BMP2能够加速骨痂形成,且有效缩短了成骨时间。ZHANG等[44]则将BMP2转染的人成纤维细胞进行大鼠腓骨缺损修复,虽然在6周内成功完成缺损修复,但观察到额外的骨修复,考虑原因可能是由于修复对象为非负重骨所致。 2.3.4 BMP2在软骨缺损修复中的作用 软骨的生物力学特性相对特殊,且具有高度的耐用性,在没有神经支配和血液供应的情况下,自身的修复能力有限,寻求一种高效的修复方法十分必要[45]。DRUMMOND等[46]利用大鼠创伤愈合模型,评估了BMP2浸泡缝线对软骨修复的治疗效果,在进行动物实验之前,将Vicryl可吸收缝合线预浸泡在BMP2溶液中,大鼠剑突(一种无血管的透明软骨结构)通过手术断裂,然后用普通缝线或预先浸泡在BMP2溶液中的缝线进行修复。体外评估表明,用 10 μg/mL的BMP2预浸Vicryl可吸收缝线,可使生长因子持续释放7 d;组织学分析表明,与未浸泡的对照缝合线相比,在大鼠剑突模型上应用这种BMP-2浸泡的缝合线明显改善了无血管软骨的愈合。TATARA等[47]采用33%的胶原凝胶进行兔膝骨软骨的全层缺损修复,在持续应用BMP2局部注射1周后进行组织学观察,8周内新生软骨的厚度达到正常水平。ZHU等[48]构建了大鼠股骨远端关节截骨模型,并将BMP2转染的BMSCs盐溶液注射截骨部位,观察到在3周内完成了软骨缺损的修复。GE等[49]采用BMP2介导的胶原海绵进行了兔膝骨软骨缺损修复,组织学分析显示12周内缺损面恢复正常,支架材料全部吸收且软骨表面光滑,与周围组织结合良好,新生软骨基本达到正常厚度,单纯胶原海绵修复的兔膝关节缺损修复不完全,表面凹陷有断裂现象,成纤维细胞排列混乱。 2.3.5 BMP2在脊柱融合中的作用 2002年FDA批注重组人BMP2可被用于前路腰椎融合,这是BMP2首次进入临床脊柱融合。SAMY[50]将重组人BMP2联合髂嵴骨移植物用于后外侧腰椎融合术中,在术后2年的随访中,联合BMP2的总融合率达到97%,而单纯髂嵴骨移植物组患者的融合率仅为77%,两组均未出现硬脊膜骨化、软组织骨化或板状骨再生的情况,提示BMP2在脊柱融合中的疗效及安全性较好。SUWANMANEE等[51] 将BMP2复合同种异体骨环用于前路腰椎椎间融合术的患者,在术后2年的随访中,BMP2组患者的融合率达到100%,而单纯骨环组患者仅为84%。WANG等[52]的研究中,观察了88例前路腰椎椎间融合术患者应用BMP2的效果,同样将BMP2和单纯骨融合组进行了对比,在术后6,12,24个月时 BMP2组患者的骨融合情况均优于未应用BMP2组。KANG等[53]的报道中对比了后外侧腰椎融合术患者应用自体骨和重组人BMP2融合聚己内酯支架的愈合率,结果显示两种方案在愈合率对比上无统计学差异,提示重组人BMP2可用于诱导成骨,作用与自体骨相当。王雪峰等[54]则对60岁以上人群的腰椎融合情况进行了观察,发现重组人BMP2与自体骨相比,术中的手术并发症少、效价比更高。NGUYEN等[55]则应用重组人BMP2复合胶原海绵成功治疗了脊柱融合失败的7例儿童病例,肯定了BMP2在儿童脊柱融合中的应用效果。此外还有研究对BMP2结合不同异体移植物的效果进行了观察,发现各种移植物融合时间存在差异,但应用BMP2的愈合率均与单纯应用同种移植物存在差异,提示BMP2在脊柱融合方面具有较高的应用价值[56-57]。 BMP2在各类骨缺损修复中的应用见表2。"

| [1] Nguyen V, Meyers CA, Yan N, et al. BMP2-induced bone formation and neural inflammation. J Orthop. 2017;14(2):252-256. [2] Park SY, Kim KH, Kim S, et al. BMP2 Gene Delivery-Based Bone Regeneration in Dentistry. Pharmaceutics. 2019;11(8):393. [3] Salazar VS, Gamer LW, Rosen V. BMP signalling in skeletal development, disease and repair. Nat Rev Endocrinol. 2016;12(4):203-221. [4] Nguyen V, Meyers CA, Yan N, et al. BMP2-induced bone formation and neural inflammation. J Orthop. 2017;14(2):252-256. [5] Katagiri T, Watabe T. Bone Morphogenetic Proteins. Cold Spring Harb Perspect Biol. 2016;8(6):a021899. [6] 曹一汀. FGF2调控软骨下骨活性促进关节软骨损伤修复的研究[D].北京:北京协和医学院,2017. [7] 鉴晓东. 羟基丁酸-羟基辛酸共聚体/胶原一体化骨软骨支架复合骨髓间充质干细胞修复兔关节骨软骨缺损的实验研究[D].太原:山西医科大学,2017. [8] Yang J, Zhang YS, Yue K, et al. Cell-laden hydrogels for osteochondral and cartilage tissue engineering. Acta Biomater. 2017;57(15):1-25 [9] Boushell MK, Hung CT, Hunziker EB, et al. Current strategies for integrative cartilage repair. Connect Tissue Res. 2017;58(5):393-406. [10] Katagiri T, Watabe T. Bone Morphogenetic Proteins. Cold Spring Harb Perspect Biol. 2016;8(6):a021899. [11] Lowery JW, Rosen V. The BMP Pathway and Its Inhibitors in the Skeleton. Physiol Rev. 2018;98(4):2431-2452. [12] Komori T. Runx2, an inducer of osteoblast and chondrocyte differentiation. Histochem Cell Biol. 2018;149(4):313-323. [13] Komori T. Regulation of Proliferation, Differentiation and Functions of Osteoblasts by Runx2. Int J Mol Sci. 2019;20(7):1694. [14] Zhao C, Jiang W, Zhou N, et al. Sox9 augments BMP2-induced chondrogenic differentiation by downregulating Smad7 in mesenchymal stem cells (MSCs). Genes Dis. 2017;4(4):229-239. [15] Mang T, Kleinschmidt-Doerr K, Ploeger F, et al. BMPR1A is necessary for chondrogenesis and osteogenesis, whereas BMPR1B prevents hypertrophic differentiation. J Cell Sci. 2020;133(16): jcs246934. [16] NAKAMURA S, ITO T, OKAMOTO K, et al. Acceleration of bone regeneration of horizontal bone defect in rats using collagen-binding basic fibroblast growth factor combined with collagen scaffolds. J Periodontol. 2019;90(9):1043-1052. [17] SHARMA U, PAL D, PRASAD R, et al. Alkaline phosphatase:an overview. Indian J Clin Biochem. 2014;29(3):269-278. [18] Lepetsos P, Papavassiliou KA, Papavassiliou AG, et al. Redox and NF-κB signaling in osteoarthritis. Free Radic Biol Med. 2019;132:90-100. [19] Hollander JM, Zeng L. The Emerging Role of Glucose Metabolism in Cartilage Development. Curr Osteoporos Rep. 2019;17(2):59-69. [20] Monaco G, Ladner YD, El Haj AJ, et al. Mesenchymal Stromal Cell Differentiation for Generating Cartilage and Bone-Like Tissues In Vitro. Cells. 2021;10(8):2165. [21] Zhang W, Zhao F, Huang D, et al. Strontium-substituted submicrometer bioactive glasses modulate macrophage responses for improved bone regeneration. ACS Appl Mater Interfaces. 2016;8(45): 30747-30758. [22] Zhang Q, Hubenak J, Iyyanki T, et al. Engineering vascularized soft tissue flaps in an animal model using human adipose-derived stem cells and VEGF+PLGA/PEG microspheres on a collagen-chitosan scaffold with a flow-through vascular pedicle. Biomaterials. 2015;73:198-213. [23] Reeves AR, Spiller KL, Freytes DO, et al. Controlled release of cytokines using silk-biomaterials for macrophage polarization. Biomaterials. 2015;73:272-283. [24] Ahmed S, Ikram S. Chitosan & its derivatives: a review in recent Innovations. IJPSR. 2015;6(1):14-30. [25] Ruan J, Wang XS, Yu Z, et al. Enhanced physiochemical and mechanical performance of chitosan-grafted graphene oxide for superior osteoinductivity. Adv Funct Mater. 2016;26(7):1085-1097. [26] Mori G, D’Amelio P, Faccio R, et al. The interplay between the bone and the immune system. Clin Dev Immunol. 2013;2013:720504. [27] Zheng ZW, Chen YH, Wu DY, et al. Development of an accurate and proactive immunomodulatory strategy to improve bone substitute material-mediated osteogenesis and angiogenesis. Theranostics. 2018; 8(19):5482-5500. [28] Madl CM, Mehta M, Duda GN, et al. Presentation of BMP2 mimicking peptides in 3D hydrogels directs cell fate commitment in osteoblasts and mesenchymal stem cells. Biomacromolecules. 2014; 15(2):445-455. [29] Sun J, Zhang Y, Li B, et al. Controlled release of BMP2 from a collagen-mimeticpeptide-modifiedsilkfibroi nnanohydroxyapatite scaffold for bone regeneration. J Mater Chem B. 2017;5(44):8770-8779. [30] Guihard P, Danger Y, Brounais B, et al. Induction of osteogenesis in mesenchymal stem cells by activated monocytes/macrophages depends on oncostatin M signaling. Stem Cells. 2012;30(4):762-772. [31] Gibon E, Lu L, Goodman SB. Aging, inflammation, stem cells, and bone healing. Stem Cell Res Ther. 2016;7:44. [32] Zhang Y, Böse T, Unger RE, et al. Macrophage type modulates osteogenic differentiation of adipose tissue MSCs. Cell Tissue Res. 2017;369(2):273-286. [33] Lin T, Pajarinen J, Nabeshima A, et al. Establishment of NF-Κb sensing and interleukin-4 secreting mesenchymal stromal cells as an “on-demand” drug delivery system to modulate inflammation. Cytotherapy. 2017;19(9):1025-1034. [34] He XT, Li X, Yin Y, et al. The effects of conditioned media generated by polarized macrophages on the cellular behaviours of bone marrow mesenchymal stem cells. J Cell Mol Med. 2018;22(2):1302-1315. [35] He XT, Li X, Xia Y, et al. Building capacity for macrophage modulation and stem cell recruitment in high-stiffness hydrogels for complex periodontal regeneration: experimental studies in vitro and in rats. Acta Biomater. 2019;88:162-180. [36] Shu Y, Yu Y, Zhang S, et al. The immunomodulatory role of sulfated chitosan in BMP2-mediated bone regeneration. Biomater Sci. 2018; 6(9):2496-2507. [37] PENG WX, WANG L. Adenovirus-mediated expression of BMP2 and BFGF in bone marrow mesenchymal stem cells combined with demineralized bone matrix for repair of femoral head osteonecrosis in beagle dogs. Cell Physiol Biochem. 2017;43(4):1648-1662. [38] Lin D, Zhang J, Bai F, et al. Fabrication and clinical application of easy-to-operate pre-cured CPC/rhBMP2 micro-scaffolds for bone regeneration. Am J Transl Res. 2016;8(3):1379-1396. [39] Lou TF, Wen G, Wang CY, et al. L-shaped corticotomy with bone flap sliding in the management of chronic tibial osteomyelitis: surgical technique and clinical results. J Orthop Surg Res. 2019;14(1):47. [40] 李侠,陆晴友,潘玉涛,等.可注射磷酸钙骨水泥联合骨形态发生蛋白-2治疗老年肱骨近端骨折疗效分析[J].实用骨科杂志,2015, 21(10):883-887. [41] NOMURA K, KIMIRA Y, OSAWA Y, et al. Stimulation of the Runx2 P1 promoter by collagen-derived dipeptide prolyl-hydroxyproline bound to Foxg1 and Foxo1 in osteoblasts. Biosci Rep. 2021:34779485. [42] PENG W, ZHANG J, ZHANG H, et al. Effects of lentiviral transfection containing bFGF gene on the biological characteristics of rabbit BMSCs. J Cell Biochem. 2018;119(10):8389-8397. [43] SU Z, HE L, SHANG H, et al. Overexpression of bone morphogenetic protein-1 promotes osteogenesis of bone marrow mesenchymal stem cells in vitro. Med Sci Monit. 2020;26:e920122. [44] Zhang C, Meng C, Guan D, et al. BMP2 and VEGF165 transfection to bone marrow stromal stem cells regulate osteogenic potential in vitro. Medicine(Baltimore). 2018;97(5):e9787. [45] YAMAGUCHI H, TERAJIMA M, KITAMI M, et al. IFT20 is critical for collagen biosynthesis in craniofacial bone formation. Biochem Biophys Res Commun. 2020;533(4):39-744. [46] Drummond N, W Bruner B, Heggeness MH, et al. Effect of BMP-2 Adherent to Resorbable Sutures on Cartilage Repair: A Rat Model of Xyphoid Process. Materials (Basel). 2020;13(17):3764. [47] Tatara AM, Mikos AG. Tissue engineering in orthopaedics. J Bone Joint Surg Am. 2016;98(13):1132-1139. [48] ZHU S, LIU Z, YUAN C, et al. Bidirectional ephrinB2-EphB4 signaling regulates the osteogenic differentiation of canine periodontal ligament stem cells. Int J Mol Med. 2020;45(3):897-909. [49] GE YW, LIU ZQ, SUN ZY, et al. Titanium particle-mediated osteoclastogenesis may be attenuated via bidirectional ephrin-B2/eph-B4 signaling in vitro. Int J Mol Med. 2018;42(4):2031-2041. [50] Samy AM. Management of osteonecrosis of the femoral head: a novel technique. Indian J Orthop. 2016;50(4):359-365. [51] Suwanmanee T, Hu G, Gui T, et al. Integration-deficient lentiviral vectors expressing codon-optimized R338L human FIX restore normal hemostasis in hemophilia B mice. Mol Ther. 2014;22(3):567-574. [52] Wang CL, Xiao F, Wang CD, et al. Gremlin2 suppression increases the hBMP2-induced osteogenesis of human bone-marrow-derived mesenchymal stem cells via the hBMP2 /Smad /Runx2 signaling pathway. J Cell Biochem. 2016;118(2):286-297. [53] KANG JW, SUH DH, PARK JH, et al. Repair of large segmental bone defect using vascularized small corticocancellous bone in rabbit femur. J Reconstr Microsurg. 2017;33(9):649-659. [54] 王雪峰,尚希福.三种填充材料修复胸腰椎骨质疏松性骨折的疗效对比[J].中国组织工程研究,2019,23(6):863-869. [55] Nguyen V, Meyers CA, Yan N, et al. BMP2-induced bone formation and neural inflammation. J Orthop. 2017;14(2):252-256. [56] Bae SJ, Kim HJ, Won HY, et al. Acceleration of osteoblast differentiation by a novel osteogenic compound, DMP-PYT, through activation of both the BMP and Wnt pathways. Sci Rep. 2017;7(1): 8455. [57] 钱光,余月明,董有海,等.磷酸钙复合BMP2 增强骨质疏松椎体椎弓根螺钉内固定[J].中国矫形外科杂志,2019,27(8):735-739. [58] De la Vega RE, Atasoy-Zeybek A, Panos JA, et al. Gene therapy for bone healing: lessons learned and new approaches. Transl Res. 2021;236:1-16. [59] Mesfin A, BUCHOWSKI M, ZEBALA LP, et al. High-Dose rhBMP2 for Adults: Major and Minor Complications-A Study of 502 Spine Cases. J Bone Joint Surg Am. 2013;95:1546-1553. [60] Bae HW, Strenge KB, Ashraf N, et al. Transient soft-tissue edema associated with implantation of increasing doses of rhBMP2 on an absorbable collagen sponge in an ectopic rat model. J Bone Joint Surg Am. 2012;94:1845-1852. [61] Fu JN, Wang X, Yang M, et al. Scaffold-Based Tissue Engineering Strategies for Osteochondral Repair. Front Bioeng Biotechnol. 2022;9: 812383. |
| [1] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [3] | Wen Xinghua, Ding Huanwen, Cheng Kai, Yan Xiaonan, Peng Yuanhao, Wang Yuning, Liu Kang, Zhang Huiwu. Three-dimensional finite element model analysis of intramedullary nailing fixation design for large femoral defects in Beagle dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1371-1376. |
| [4] | Yang Zhishan, Tang Zhenglong. YAP/TAZ, a core factor of the Hippo signaling pathway, is involved in bone formation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1264-1271. |
| [5] | Zhao Lu, Zhao Yifei, Gao Da, Liu Yanfang, Fu Tingting, Xu Jiangyan. Expression of suppressor of Zeste 12 in kidney tissues of rats with diabetic nephropathy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1179-1186. |
| [6] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [7] | Yang Yitian, Wang Lu, Yao Wei, Zhao Bin. Application of the interaction between biological scaffolds and macrophages in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1071-1079. |
| [8] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [9] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [10] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [11] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [12] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [13] | Liu Wentao, Feng Xingchao, Yang Yi, Bai Shengbin. Effect of M2 macrophage-derived exosomes on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 840-845. |
| [14] | Long Yanming, Xie Mengsheng, Huang Jiajie, Xue Wenli, Rong Hui, Li Xiaojie. Casein kinase 2-interaction protein-1 regulates the osteogenic ability of bone marrow mesenchymal stem cells in osteoporosis rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 878-882. |
| [15] | Li Xinyue, Li Xiheng, Mao Tianjiao, Tang Liang, Li Jiang. Three-dimensional culture affects morphology, activity and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 846-852. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||