Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (35): 5733-5740.doi: 10.12307/2022.919
Telomeres and telomerase: mechanism of exercise retarding aging telomeres
Yang Ling1, Huang Sen2
- 1School of Physical Education, Shaoguan University, Shaoguan 512000, Guangdong Province, China; 2Hunan Institute of Sports Science, Changsha 410008, Hunan Province, China
-
Received:2021-11-29Accepted:2022-01-19Online:2022-12-18Published:2022-05-18 -
Contact:Huang Sen, PhD, Associate researcher, Hunan Institute of Sports Science, Changsha 410008, Hunan Province, China -
About author:Yang Ling, PhD candidate, School of Physical Education, Shaoguan University, Shaoguan 512000, Guangdong Province, China -
Supported by:2021 Humanities and Social Sciences Project of the Ministry of Education of China, No. 21YJAZH049 (to YL [project participant])
CLC Number:
Cite this article
Yang Ling, Huang Sen. Telomeres and telomerase: mechanism of exercise retarding aging telomeres[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5733-5740.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

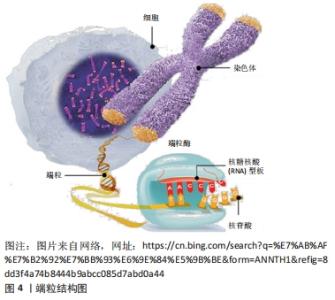
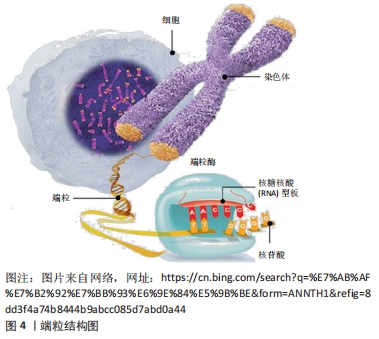
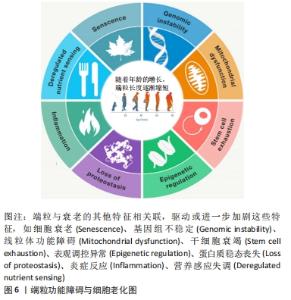
2.1.2 端粒酶的结构和功能 端粒酶是一种自身携带RNA模板的末端反转录酶, 是由端粒酶反转录酶、端粒酶RNA模板及端粒酶相关蛋白构成的核糖核蛋白复合体[20]。端粒酶可以通过自身携带的RNA为模板,合成端粒末端重复序列,弥补在细胞分裂过程中减少的端粒长度[21]。正常体细胞中端粒酶活性很低[22],只有在干细胞[23]、肿瘤细胞和生殖细胞中端粒酶才具有高活性[24-25],因此体细胞在进行有限次数的分裂后使端粒长度到极短,引起细胞凋亡,导致衰老;端粒酶活性受端粒酶反转录酶的影响,此外,端粒末端的结合蛋白复合体也会对端粒酶的活性有调节作用。由此可见,衰老的调节依赖于端粒酶对端粒长度的调控,以及端粒和端粒酶的联合作用[26-27]。 2.2 端粒与衰老 衰老是一种进行性退行性状态,伴有组织干细胞耗竭、组织炎症、基质改变、细胞衰老和代谢功能障碍。这些细胞和组织的变化反映了线粒体和蛋白稳态、细胞间通讯、营养感应、表观遗传学和DNA修复等潜在的分子畸变,导致基因组不稳定和破坏,包括端粒功能障碍。"

2.2.2 端粒与老年疾病 当端粒长度缩短到某一数值时,细胞分裂停止,产生凋亡,因此,端粒长度可作为年龄老化的生物标志物。缩短的端粒限制了细胞增殖的速度,与疾病风险和死亡率密切有关[28]。最近研究发现白细胞和骨骼肌细胞的端粒长度可能与年龄相关疾病及其并发症之间有关,但其机制目前尚不清楚。 (1)端粒系统与心血管疾病:研究显示,心血管疾病患者比健康人的白细胞端粒长度短300 bp [29],这个差异相当于8.6年的年龄差[30]。动脉粥样硬化与心脑血管疾病的发生密切相关,端粒长度的缩短也是引起动脉粥样硬化的诱因,端粒结合蛋白因子2表达的降低或缺失可导致血管平滑肌细胞衰老,进一步促进动脉粥样硬化,增加斑块程度[31-32];另外,端粒缩短会增加急性心肌梗死的风险[33]。虽然目前的文献表明端粒缩短可作为预测心血管疾病的生物标志物,但端粒长度对心血管疾病的预测准确性尚不清楚[34]。运动可以增加心血管疾病患者白细胞端粒长度[35],但具体机制还不清楚。 (2)端粒系统与肿瘤:在衰老期间,机体免疫系统受损,无法清除所有衰老细胞,致使留在组织中的非功能衰老细胞危及组织结构和功能,使细胞再生能力受到限制,促使组织癌变[36]。大多数体细胞端粒酶的活性较低,少数分裂旺盛的细胞,如干细胞、肿瘤细胞等出现端粒酶活性高表达[37-38]。端粒酶反转录酶不仅维持端粒长度,使细胞永生,也在肿瘤发展过程中诱导癌细胞的转移、抗凋亡和抗分化[39]。在肝癌中,端粒酶反转录酶变化与端粒缩短呈正相关,端粒酶活性和端粒长度可能成为肝癌的生物标志物[40]。 (3)端粒系统与神经退行性疾病:神经退化性疾病是影响衰老的最常见的疾病之一,如阿尔茨海默病、帕金森病、亨廷顿症、癫痫等。由于端粒长度与衰老相关,端粒在一定程度上参与了神经退化性疾病的发生发展。越来越多的研究证实阿尔茨海默病的痴呆系数与端粒长度有关[41],端粒长度可预测阿尔茨海默病的进展[42]。大多数研究显示帕金森病患者的端粒长度与健康对照组之间没有统计学差异,但有少数研究显示帕金森病患者的白细胞端粒长度比健康人的短[43]。越来越多的证据表明,端粒长度与脑卒中的死亡率和认知能力呈正相关[44-45]。由此可见,端粒长度有望成为预测神经退行性疾病发生发展的因子。 (4)端粒系统与肥胖:大多数研究显示白细胞端粒长度与肥胖相关表型(如体质量指数、体质量和腰围)密切相关[40];研究显示,肥胖受试者体质量、体质量指数和腰围指数降低,伴随着白细胞端粒长度的增加[46];但也有一些研究指出,白细胞端粒长度缩短与单纯性的肥胖表型不相关[47]。另有研究显示,肥胖相关表型对白细胞端粒长度的影响具有性别特异性,其中女性不良影响高于男性[48]。以上研究结果不一致的原因可能是研究对象的种族、年龄、肥胖程度导致的。在探寻端粒系统与肥胖的机制研究中显示,端粒结合蛋白因子1是肥胖机体端粒缩短的主要原因[49]。定期参加体育运动可降低肥胖受试者体质量,延长白细胞端粒长度[50],其具体机制是否与运动调控端粒结合蛋白因子1有关,需要进一步研究。 (5)端粒系统与糖尿病:糖尿病是一种以各种器官功能障碍为特征的疾病。最近的研究表明,糖尿病发生发展和白细胞端粒长度之间密切相关[51],糖尿病患者胰腺β细胞中的端粒长度缩短,导致胰岛素分泌能力受损,加速细胞死亡;过度的氧化应激也会使端粒长度缩短,损害胰腺功能[52]。糖尿病患者的白细胞端粒长度比健康人短[53],伴有糖尿病并发症(如视网膜病变、早期肾病和心血管疾病)的糖尿病患者比单纯糖尿病患者的白细胞端粒长度更短[40]。在端粒维持基因中,端粒酶反转录酶、端粒RNA模板、端锚聚合酶、端粒酶活性相关蛋白1、端粒结合因子1和端粒结合因子2中的单核苷酸多态性与2型糖尿病患者β细胞的端粒长度消耗及其病理生理学有很强的联系[54]。一项长达10年的研究发现,白细胞端粒长度的缩短与体质量指数、胰岛素和葡萄糖增加有关[53]。端粒长度有望作为预测糖尿病发生发展的生物标志物。 2.3 运动延缓衰老的端粒机制 衰老的发生依赖于端粒酶对端粒长度的调整,以及端粒与端粒酶的联合作用。运动对端粒的影响主要有运动对端粒长度和端粒活性的改变。 "
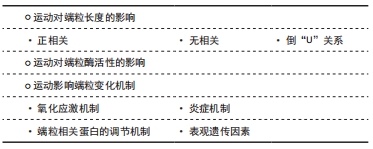
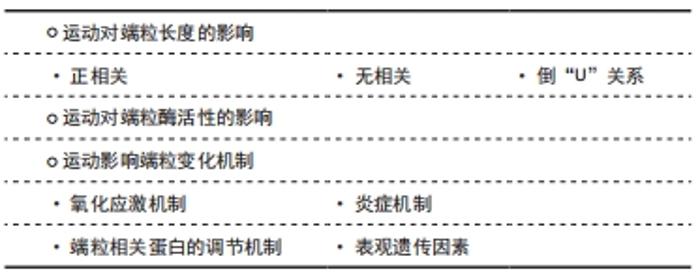
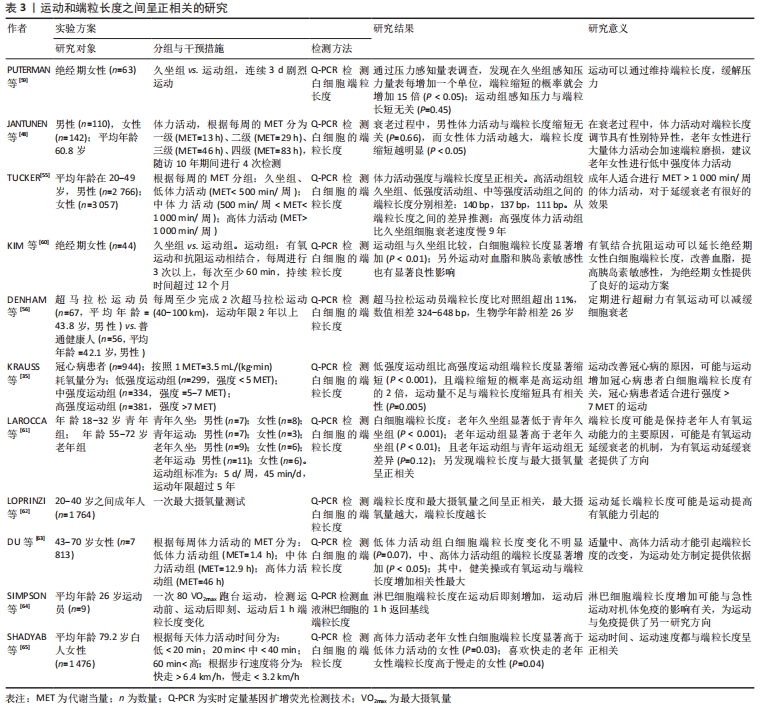
2.3.1 运动对端粒长度的影响 现有研究中运动与端粒长度之间的关系尚有较多的争议,有3种不同的研究结果,即运动与端粒长度的正相关关系、无相关关系和倒U型关系。 (1)正相关:科学运动可以提高人体各项功能,增强免疫力,改善身体健康,延缓衰老,其中原因与运动可以延长端粒长度有关,见表3[35,48,55-56,59-65]。运动员与经常参加体育锻炼者比久坐人群拥有更长的端粒[55]。DENHAM 等[56]对超级马拉松运动员的一项研究证明,与静坐少动的人比较,超级马拉松运动员的端粒更长,两者差异约等于16年的生物年龄,表明体育活动在预防年龄引起的端粒长度磨损中发挥了重要作用。在健康个体和双胞胎志愿者中,运动与白细胞端粒长度呈正相关[57],而久坐行为与端粒长度呈负相关。TUCKER[55]研究也发现了经常参加运动的成年人白细胞端粒长度比久坐不动人的端粒长度多了140个碱基对,两者差异约等于7年的生物年龄。研究显示,只有5年以上的长期运动才能使端粒长度增加,急性运动或者短期运动不能改变端粒长度[58]。但有研究发现,短期运动也可以延长端粒长度[59-60]。运动延长端粒长度受运动时间长短的影响。 "
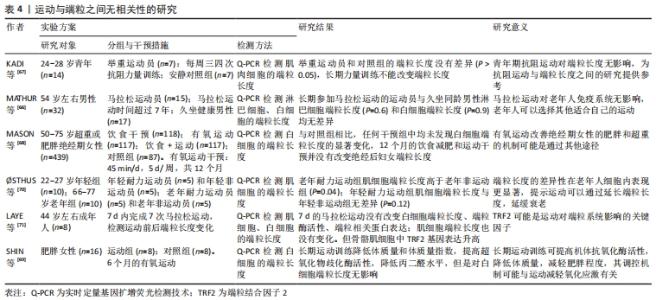
(2)无相关:有研究表明,运动不能延长端粒长度,他们之间没有相关性,见表4[66-71]。MATHUR等[66]研究发现,长期参加马拉松运动的运动员与久坐同龄男性白细胞端粒长度无差异,长时间耐力运动不能增加端粒长度。KADI等[67]探讨了抗阻运动与端粒长度的关系,将7名举重运动员与同龄普通人比较,发现举重运动员和普通人的端粒长度没有差异,长期力量训练并不能改变端粒长度。MASON等[68]将 439 名肥胖或超重的老年女性随机分为饮食减肥组、有氧运动组、饮食+运动组、对照组,运动组每天45 min的中高强度的有氧运动,每周5 d,运动干预12个月;结果显示,干预组的白细胞端粒长度与对照组相比无显著差异。SHIN等[69]研究发现6个月的有氧运动降低了肥胖女性的体质量,增加了血液中超氧化物歧化酶活性,但是对白细胞端粒长度没有影响。以上研究结果的原因,可能是研究对象自身健康差异性导致运动与端粒长度关联性较低,此外,横断面自我报告的体力活动调查的误差性,也可能是运动与端粒长度无关联性的原因。 "
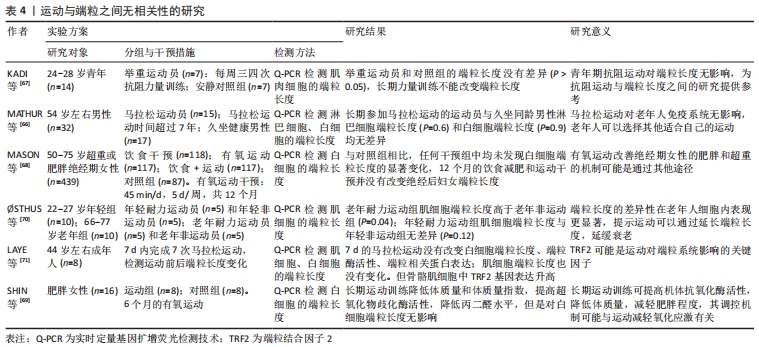

(3)倒U关系:一些学者认为运动与端粒长度之间关系不是正相关和无相关,而是倒U关系,见表5[58,72-73]。一项针对老年女性运动的横断面研究表明,每周运动时间≥17 h的女性受试者比每周运动时间< 1.25 h的受试者,其端粒长度较长,更利于身体健康[65]。LUDLOW等[58]将年龄在50-70岁研究对象分为久坐组、低体力活动组、中体力活动组和高体力活动组,分别检测各组受试者的外周血单核细胞的端粒长度,结果显示,与久坐组端粒长度比较,中体力活动组的端粒长度明显增加,而高体力活动组、低体力活动组无差异。由此可见,体力活动与端粒长度之间存在倒U型的关系。?STHUS等[70]研究表明,端粒长度与运动之间倒U型关系在年龄较大的耐力运动受试者中较为明显,年轻耐力受试者中不明显,推测运动与端粒长度的倒U型现象可能与年龄有关。 "
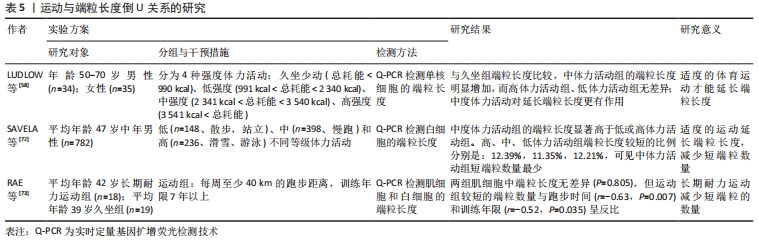

综上所述,以上多个横向研究描述了身体体力活动或运动训练与端粒长度的关系,报道了3种不同的关系:正相关、无相关、倒U型关系;研究中样本量的大小、运动和身体体力活动数据采集的变化、端粒长度测定的方法、用于提取DNA的细胞类型、受试个体的年龄,都可能是导致结果不一致的原因。但大多数研究学者认为体育锻炼和端粒长度之间存在剂量相关关系,适宜的运动可以延缓端粒长度的缩短。此外,应在特定年龄进行不同形式的体育活动,以获得对端粒长度维持的积极作用。 2.3.2 运动对端粒酶活性的影响 与久坐不动的老年人比较,长期参加运动的老年运动员的循环白细胞端粒酶活性升高、端粒稳定蛋白增加、衰老标志物的表达下降;且长期参加运动的老年运动员和年轻运动员白细胞的端粒酶活性无差异[74],这可能是长期运动提高机体适应、增加端粒酶活性、延缓衰老的原因。相对于长期运动,急性运动中也发现端粒酶活性的改变,研究显示,急性耐力运动可以提高健康青年男性外周血单核细胞端粒酶的活性[75]。但也有结论相反的研究,CLUCKEY等[76]对比年轻受试者[(22±1)岁]和老年受试者[(60±2)岁]在一次高强度间歇运动后端粒酶活性和端粒相关蛋白表达变化,结果显示,运动后即刻,年轻组端粒酶活性显著升高,老年组端粒酶活性变化不明显;运动后1 h,老年组端粒结合因子2表达显著增加,年轻组无变化。PUTERMAN等[77]对老年久坐人员进行24周有氧运动干预,每周3-5次,每次40 min,受试者外周血单核细胞端粒酶活性在运动后没有变化。运动对端粒酶活性的影响与年龄有一定的关系,不同年龄之间存在偏倚。 2.3.3 运动影响端粒变化机制 (1)氧化应激机制:氧化应激是指细胞内活性氧水平升高,会对细胞造成损害。机体在氧化代谢中会产生一些活性氧基团或分子,如活性氧。活性氧能破坏细胞中的脂质、蛋白质和DNA,造成细胞结构和功能的改变。端粒中的5-GGG片段容易遭受活性氧破坏,出现双链断裂、长度缩短,因此,氧化抗氧化失衡是影响端粒长度重要的因素之一。OIKAWA 等[78]研究发现,氧化应激会对端粒G序列上面特定位点的DNA造成损伤,持续的氧化应激会破坏端粒DNA的修复,加速端粒长度的缩短速度。轻度氧化应激会使精子细胞端粒长度延长,过度氧化应激则导致端粒长度相应缩短[79]。虽然,有研究表明端粒长度缩短可作为慢性氧化应激的潜在标志物[80],运动可能是通过降低体内氧化应激水平,提高端粒酶活性,增加端粒长度,但具体调控途径还需要进一步验证。 (2)炎症机制:机体的慢性炎症可以提高白细胞的周转率,激活细胞分裂,提高细胞复制,导致端粒长度缩短,功能失调,引起细胞衰老。研究发现,细胞衰老与循环巨噬细胞中转录因子NF-κB的过度活跃和炎症因子如肿瘤坏死因子α、白细胞介素6和γ干扰素的过度表达有关[81],运动训练可以降低机体炎症标志物的水平[82]。有研究表明,白细胞端粒长度缩短与白细胞介素6、肿瘤坏死因子α水平升高有关[83],同时慢性炎症也可提高白细胞的周转率,从而增加端粒磨损。而运动训练可以引起白细胞介素6、肿瘤坏死因子α水平下降,因此推测,运动可能通过减轻炎症来维持端粒长度。 (3)端粒相关蛋白的调节机制:端粒结合蛋白复合体作为重要的调控因子来调控端粒长度和端粒酶活性。端粒结合因子1、端粒结合因子2 和保护端粒1作为复合体的关键因子,保护端粒1-肽酶1可以与端粒DNA结合,增强端粒酶的活性[84],端粒结合因子2可以将端粒末端进行折叠,维持端粒结构稳定。研究显示,21 d有氧运动可以上调小鼠心肌细胞端粒结合因子2的表达,提高2倍端粒酶活性,减少凋亡蛋白p53和p16的表达[85],提示端粒酶活性的上调与端粒结合因子2的表达有关。有氧运动对不同的端粒调控蛋白具有不同的调节效应:一方面可降低端粒结合因子1的表达水平,从而减弱对端粒酶的负性调控作用;另一方面通过显著增强端粒结合因子2和保护端粒1的表达,更有利于端粒酶的提升,同时加强端粒酶的结合能力,两者综合作用达到端粒结构完整、端粒酶活性提高。综上所述,这可能是运动保护端粒的另一种原因。 (4)表观遗传因素:端粒长度和端粒酶活性也受表观遗传因素的影响,如DNA甲基化;端粒的长度与端粒反转录酶的表达密切相关,而端粒反转录酶的表达是由DNA甲基化调控的[86],同型半胱氨酸可以通过DNA甲基转移酶1促进甲基化敏感转录因子,降低人端粒酶反转录酶的表达和活性,从而降低内皮细胞端粒酶活性,促进细胞衰老[87]。研究显示,4周的有氧运动可以下调DNA甲基转移酶3的表达水平和改变同型半胱氨酸浓度[88],提示运动能通过下调DNA甲基转移酶介导的甲基化水平,减少同型半胱氨酸生成,提高端粒酶活性,延缓衰老。研究表明,抗阻运动对同型半胱氨酸水平无影响,运动对同型半胱氨酸水平的影响与运动量显著相关,不受运动强度的影响,这也可能是运动与端粒长度无相关变化的原因[89]。另外,micro RNA(micro RNA-138、micro RNA 491-5p、micro RNA 1266、micro RNA 1207-5p、micro RNA 1182等)都可通过结合于端粒反转录酶3'上的互补序列而干扰端粒反转录酶RNA的正常翻译,抑制端粒反转录酶水平表达,运动有可能通过调节micro RNA影响端粒。综上可见,表观遗传因素的改变可能是运动调节端粒的另一机制,但是无论何种机制,对于该机制的具体信号调节途径还需要实验性验证。 "

| [1] 高瑗,原新.中国老年人口健康转移与医疗支出[J].人口研究,2020, 44(2):60-72. [2] 乔玉成,王卫军. 规律运动干预人类衰老过程的表观遗传学机制研究进展[J].北京体育大学学报,2016,39(1):61-67,75. [3] MCCLINTOCK B. The Behavior in Successive Nuclear Divisions of a Chromosome Broken at Meiosis. Proc Natl Acad Sci U S A. 1939;25(8):405-416. [4] WADE N. Hayflick’s Tragedy: The Rise and Fall of a Human Cell Line. Science. 1976;192(4235):125-127. [5] WATSON JD. Origin of concatemeric T7 DNA. Nat New Biol. 1972;239(94): 197-201. [6] BLACKBURN EH, GALL JG. A tandemly repeated sequence at the termini of the extrachromosomal ribosomal RNA genes in Tetrahymena. J Mol Biol. 1978;120(1):33-53. [7] SZOSTAK JW, BLACKBURN EH. Cloning yeast telomeres on linear plasmid vectors. Cell. 1982;29(1):245-255. [8] GREIDER CW, BLACKBURN EH. Identification of a specific telomere terminal transferase activity in Tetrahymena extracts. Cell. 1985;43(2 Pt 1):405-413. [9] GREIDER CW, BLACKBURN EH. A telomeric sequence in the RNA of Tetrahymena telomerase required for telomere repeat synthesis. Nature. 1989;337(6205):331-337. [10] LUNDBLAD V, BLACKBURN EH. RNA-dependent polymerase motifs in EST1: tentative identification of a protein component of an essential yeast telomerase. Cell. 1990;60(4):529-530. [11] COHN M, BLACKBURN EH. Telomerase in yeast. Science. 1995;269(5222):396-400. [12] LINGNER J, HUGHES TR, SHEVCHENKO A, et al. Reverse transcriptase motifs in the catalytic subunit of telomerase. Science. 1997;276(5312):561-567. [13] COUNTER CM, MEYERSON M, EATON EN, et al. The catalytic subunit of yeast telomerase. Proc Natl Acad Sci U S A. 1997;94(17):9202-9207. [14] WEINRICH SL, PRUZAN R, MA L, et al. Reconstitution of human telomerase with the template RNA component hTR and the catalytic protein subunit hTRT. Nat Genet. 1997;17(4):498-502. [15] SADAIE M, NAITO T, ISHIKAWA F. Stable inheritance of telomere chromatin structure and function in the absence of telomeric repeats. Genes Dev. 2003;17(18):2271-2282. [16] BIESSMANN H, MASON JM. Telomerase-independent mechanisms of telomere elongation. Cell Mol Life Sci. 2003;60(11):2325-2333. [17] MOYZIS RK, BUCKINGHAM JM, CRAM LS, et al. A highly conserved repetitive DNA sequence, (TTAGGG)n, present at the telomeres of human chromosomes. Proc Natl Acad Sci U S A. 1988;85(18):6622-6626. [18] 刘惠芬,李峰,彭东旭,等.端粒、端粒酶及靶向抗衰老研究[J].现代预防医学,2017,44(3):557-560. [19] SMITH EM, PENDLEBURY DF, NANDAKUMAR J. Structural biology of telomeres and telomerase. Cell Mol Life Sci. 2020;77(1):61-79. [20] MUSGROVE C, JANSSON LI, STONE MD. New perspectives on telomerase RNA structure and function. Wiley Interdiscip Rev RNA. 2018;9(2):10. [21] JIANG J, WANG Y, SUŠAC L, et al. Structure of Telomerase with Telomeric DNA. Cell. 2018;173(5):1179-1190.e13. [22] BREY EM, GREISLER HP. Telomerase expression in somatic cells. Lancet. 2005;365(9477):2068-2069. [23] TORRES-MONTANER A. The telomere complex and the origin of the cancer stem cell. Biomark Res. 2021;9(1):81. [24] MEYERSON M, COUNTER CM, EATON EN, et al. hEST2, the putative human telomerase catalytic subunit gene, is up-regulated in tumor cells and during immortalization. Cell. 1997;90(4):785-795. [25] OZTURK S. Telomerase activity and telomere length in male germ cells. Biol Reprod. 2015;92(2):53. [26] 吕欣桐.端粒、端粒酶与抗衰老的研究进展[J].医学信息(上旬刊),2011, 24(5):2857-2858. [27] DER G, BATTY GD, BENZEVAL M, et al. Is telomere length a biomarker for aging: cross-sectional evidence from the west of Scotland? PLoS One. 2012; 7(9):e45166. [28] BERNADOTTE A, MIKHELSON VM, SPIVAK IM. Markers of cellular senescence. Telomere shortening as a marker of cellular senescence. Aging (Albany NY). 2016;8(1):3-11. [29] O’DONNELL CJ, DEMISSIE S, KIMURA M, et al. Leukocyte telomere length and carotid artery intimal medial thickness: the Framingham Heart Study. Arterioscler Thromb Vasc Biol. 2008;28(6):1165-1171. [30] SAMANI NJ, BOULTBY R, BUTLER R, et al. Telomere shortening in atherosclerosis. Lancet. 2001;358(9280):472-473. [31] NOWAK R, SIWICKI JK, CHECHLINSKA M, et al. Telomere shortening and atherosclerosis. Lancet. 2002;359(9310):976; author reply 976-977. [32] FERNÁNDEZ-ALVIRA JM, FUSTER V, DORADO B, et al. Short Telomere Load, Telomere Length, and Subclinical Atherosclerosis: The PESA Study. J Am Coll Cardiol. 2016;67(21):2467-2476. [33] OLIVIERI F, LORENZI M, ANTONICELLI R, et al. Leukocyte telomere shortening in elderly Type2DM patients with previous myocardial infarction. Atherosclerosis. 2009;206(2):588-593. [34] TELLECHEA M, GIANOTTI TF, ALVARIÑAS J, et al. Telomere length in the two extremes of abnormal fetal growth and the programming effect of maternal arterial hypertension. Sci Rep. 2015;5:7869. [35] KRAUSS J, FARZANEH-FAR R, PUTERMAN E, et al. Physical fitness and telomere length in patients with coronary heart disease: findings from the Heart and Soul Study. PLoS One. 2011;6(11):e26983. [36] KIM SH SH, KAMINKER P, CAMPISI J. Telomeres, aging and cancer: in search of a happy ending. Oncogene. 2002;21(4):503-511. [37] MENDER I, ZHANG A, REN Z, et al. Telomere Stress Potentiates STING-Dependent Anti-tumor Immunity. Cancer Cell. 2020;38(3):400-411.e6. [38] YUAN X, DAI M, XU D. Telomere-related Markers for Cancer. Curr Top Med Chem. 2020;20(6):410-432. [39] HUANG DS, WANG Z, HE XJ, et al. Recurrent TERT promoter mutations identified in a large-scale study of multiple tumour types are associated with increased TERT expression and telomerase activation. Eur J Cancer. 2015;51(8):969-976. [40] NAULT JC, NINGARHARI M, REBOUISSOU S, et al. The role of telomeres and telomerase in cirrhosis and liver cancer. Nat Rev Gastroenterol Hepatol. 2019;16(9):544-558. [41] JASKELIOFF M, MULLER FL, PAIK JH, et al. Telomerase reactivation reverses tissue degeneration in aged telomerase-deficient mice. Nature. 2011; 469(7328):102-106. [42] TEDONE E, AROSIO B, COLOMBO F, et al. Leukocyte Telomere Length in Alzheimer’s Disease Patients with a Different Rate of Progression. J Alzheimers Dis. 2015;46(3):761-769. [43] ZEE RY, CASTONGUAY AJ, BARTON NS, et al. Relative leukocyte telomere length and risk of incident ischemic stroke in men: a prospective, nested case-control approach. Rejuvenation Res. 2010;13(4):411-414. [44] MARTIN-RUIZ C, DICKINSON HO, KEYS B, et al. Telomere length predicts poststroke mortality, dementia, and cognitive decline. Ann Neurol. 2006; 60(2):174-180. [45] ROLYAN H, SCHEFFOLD A, HEINRICH A, et al. Telomere shortening reduces Alzheimer’s disease amyloid pathology in mice. Brain. 2011;134(Pt 7):2044-2056. [46] GARCÍA-CALZÓN S, GEA A, RAZQUIN C, et al. Longitudinal association of telomere length and obesity indices in an intervention study with a Mediterranean diet: the PREDIMED-NAVARRA trial. Int J Obes (Lond). 2014; 38(2):177-182. [47] IGLESIAS MOLLI AE, PANERO J, DOS SANTOS PC, et al. Metabolically healthy obese women have longer telomere length than obese women with metabolic syndrome. PLoS One. 2017;12(4):e0174945. [48] JANTUNEN H, WASENIUS NS, GUZZARDI MA, et al. Physical Activity and Telomeres in Old Age: A Longitudinal 10-Year Follow-Up Study. Gerontology. 2020;66(4):315-322. [49] GRUN LK, TEIXEIRA NDR JR, MENGDEN LV, et al. TRF1 as a major contributor for telomeres’ shortening in the context of obesity. Free Radic Biol Med. 2018;129:286-295. [50] SHIN YA. How Does Obesity and Physical Activity Affect Aging?: Focused on Telomere as a Biomarker of Aging. J Obes Metab Syndr. 2019;28(2):92-104. [51] WANG J, DONG X, CAO L, et al. Association between telomere length and diabetes mellitus: A meta-analysis. J Int Med Res. 2016;44(6):1156-1173. [52] TAMURA Y, TAKUBO K, AIDA J, et al. Telomere attrition and diabetes mellitus. Geriatr Gerontol Int. 2016;16 Suppl 1:66-74. [53] WILLEIT P, RASCHENBERGER J, HEYDON EE, et al. Leucocyte telomere length and risk of type 2 diabetes mellitus: new prospective cohort study and literature-based meta-analysis. PLoS One. 2014;9(11):e112483. [54] SETHI I, BHAT GR, SINGH V, et al. Role of telomeres and associated maintenance genes in Type 2 Diabetes Mellitus: A review. Diabetes Res Clin Pract. 2016;122:92-100. [55] TUCKER LA. Physical activity and telomere length in U.S. men and women: An NHANES investigation. Prev Med. 2017;100:145-151. [56] DENHAM J, NELSON CP, O’BRIEN BJ, et al. Longer leukocyte telomeres are associated with ultra-endurance exercise independent of cardiovascular risk factors. PLoS One. 2013;8(7):e69377. [57] BENDIX L, GADE MM, STAUN PW, et al. Leukocyte telomere length and physical ability among Danish twins age 70+. Mech Ageing Dev. 2011;132 (11-12):568-572. [58] LUDLOW AT, ZIMMERMAN JB, WITKOWSKI S, et al. Relationship between physical activity level, telomere length, and telomerase activity. Med Sci Sports Exerc. 2008;40(10):1764-1771. [59] PUTERMAN E, LIN J, BLACKBURN E, et al. The power of exercise: buffering the effect of chronic stress on telomere length. PLoS One. 2010;5(5):e10837. [60] KIM JH, KO JH, LEE DC, et al. Habitual physical exercise has beneficial effects on telomere length in postmenopausal women. Menopause. 2012; 19(10):1109-1115. [61] LAROCCA TJ, SEALS DR, PIERCE GL. Leukocyte telomere length is preserved with aging in endurance exercise-trained adults and related to maximal aerobic capacity. Mech Ageing Dev. 2010;131(2):165-167. [62] LOPRINZI PD. Cardiorespiratory Capacity and Leukocyte Telomere Length Among Adults in the United States. Am J Epidemiol. 2015;182(3):198-201. [63] DU M, PRESCOTT J, KRAFT P, et al. Physical activity, sedentary behavior, and leukocyte telomere length in women. Am J Epidemiol. 2012;175(5): 414-422. [64] SIMPSON RJ, COSGROVE C, CHEE MM, et al. Senescent phenotypes and telomere lengths of peripheral blood T-cells mobilized by acute exercise in humans. Exerc Immunol Rev. 2010;16:40-55. [65] SHADYAB AH, LAMONTE MJ, KOOPERBERG C, et al. Leisure-time physical activity and leukocyte telomere length among older women. Exp Gerontol. 2017;95:141-147. [66] MATHUR S, ARDESTANI A, PARKER B, et al. Telomere length and cardiorespiratory fitness in marathon runners. J Investig Med. 2013;61(3): 613-615. [67] KADI F, PONSOT E, PIEHL-AULIN K, et al. The effects of regular strength training on telomere length in human skeletal muscle. Med Sci Sports Exerc. 2008;40(1):82-87. [68] MASON C, RISQUES RA, XIAO L, et al. Independent and combined effects of dietary weight loss and exercise on leukocyte telomere length in postmenopausal women. Obesity (Silver Spring). 2013;21(12):E549-554. [69] SHIN YA, LEE JH, SONG W, et al. Exercise training improves the antioxidant enzyme activity with no changes of telomere length. Mech Ageing Dev. 2008;129(5):254-260. [70] ØSTHUS IB, SGURA A, BERARDINELLI F, et al. Telomere length and long-term endurance exercise: does exercise training affect biological age? A pilot study. PLoS One. 2012;7(12):e52769. [71] LAYE MJ, SOLOMON TP, KARSTOFT K, et al. Increased shelterin mRNA expression in peripheral blood mononuclear cells and skeletal muscle following an ultra-long-distance running event. J Appl Physiol (1985). 2012; 112(5):773-781. [72] SAVELA S, SAIJONMAA O, STRANDBERG TE, et al. Physical activity in midlife and telomere length measured in old age. Exp Gerontol. 2013;48(1):81-84. [73] RAE DE, VIGNAUD A, BUTLER-BROWNE GS, et al. Skeletal muscle telomere length in healthy, experienced, endurance runners. Eur J Appl Physiol. 2010; 109(2):323-330. [74] WERNER C, FÜRSTER T, WIDMANN T, et al. Physical exercise prevents cellular senescence in circulating leukocytes and in the vessel wall. Circulation. 2009;120(24):2438-2447. [75] ZIETZER A, BUSCHMANN EE, JANKE D, et al. Acute physical exercise and long-term individual shear rate therapy increase telomerase activity in human peripheral blood mononuclear cells. Acta Physiol (Oxf). 2017;220(2): 251-262. [76] CLUCKEY TG, NIETO NC, RODONI BM, et al. Preliminary evidence that age and sex affect exercise-induced hTERT expression. Exp Gerontol. 2017;96:7-11. [77] PUTERMAN E, WEISS J, LIN J, et al. Aerobic exercise lengthens telomeres and reduces stress in family caregivers: A randomized controlled trial - Curt Richter Award Paper 2018. Psychoneuroendocrinology. 2018;98:245-252. [78] OIKAWA S, KAWANISHI S. Site-specific DNA damage at GGG sequence by oxidative stress may accelerate telomere shortening. FEBS Lett. 1999;453(3): 365-368. [79] MISHRA S, KUMAR R, MALHOTRA N, et al. Mild oxidative stress is beneficial for sperm telomere length maintenance. World J Methodol. 2016;6(2):163-170. [80] BOONEKAMP JJ, BAUCH C, MULDER E, et al. Does oxidative stress shorten telomeres? Biol Lett. 2017;13(5):20170164. [81] ZHANG J, RANE G, DAI X, et al. Ageing and the telomere connection: An intimate relationship with inflammation. Ageing Res Rev. 2016;25:55-69. [82] SOLTANI N, MARANDI SM, KAZEMI M, et al. The Exercise Training Modulatory Effects on the Obesity-Induced Immunometabolic Dysfunctions. Diabetes Metab Syndr Obes. 2020;13:785-810. [83] LYU L, HE S, ZHANG H, et al. TNFα Mediates the Interaction of Telomeres and Mitochondria Induced by Hyperglycemia: A Rural Community-Based Cross-Sectional Study. Oxid Med Cell Longev. 2020;2020:8235873. [84] WANG F, PODELL ER, ZAUG AJ, et al. The POT1-TPP1 telomere complex is a telomerase processivity factor. Nature. 2007;445(7127):506-510. [85] WERNER C, HANHOUN M, WIDMANN T, et al. Effects of physical exercise on myocardial telomere-regulating proteins, survival pathways, and apoptosis. J Am Coll Cardiol. 2008;52(6):470-482. [86] SHARMA NK, REYES A, GREEN P, et al. Human telomerase acts as a hTR-independent reverse transcriptase in mitochondria. Nucleic Acids Res. 2012; 40(2):712-725. [87] ZHANG D, SUN X, LIU J, et al. Homocysteine accelerates senescence of endothelial cells via DNA hypomethylation of human telomerase reverse transcriptase. Arterioscler Thromb Vasc Biol. 2015;35(1):71-78. [88] 刘波,陈祥和,杨康,等.骨代谢紊乱的表观遗传重编程与运动调控[J].中国组织工程研究,2021,25(20):3210-3218. [89] DEMINICE R, RIBEIRO DF, FRAJACOMO FT. The Effects of Acute Exercise and Exercise Training on Plasma Homocysteine: A Meta-Analysis. PLoS One. 2016;11(3):e0151653. |
| [1] | Wang Jianping, Zhang Xiaohui, Yu Jinwei, Wei Shaoliang, Zhang Xinmin, Xu Xingxin, Qu Haijun. Application of knee joint motion analysis in machanism based on three-dimensional image registration and coordinate transformation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-5. |
| [2] | Tan Xinfang, Guo Yanxing, Qin Xiaofei, Zhang Binqing, Zhao Dongliang, Pan Kunkun, Li Yuzhuo, Chen Haoyu. Effect of uniaxial fatigue exercise on patellofemoral cartilage injury in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-6. |
| [3] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [4] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [5] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [6] | Gu Zhengqiu, Xu Fei, Wei Jia, Zou Yongdi, Wang Xiaolu, Li Yongming. Exploratory study on talk test as a measure of intensity in blood flow restriction training [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1154-1159. |
| [7] | Wang Qin, Shen Cheng, Liao Jing, Yang Ye. Dapagliflozin improves renal injury in diabetic nephropathy rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1216-1222. |
| [8] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [9] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [10] | Tang Wenjing, Wu Siyuan, Yang Chen, Tao Xi. Inflammatory responses in post-stroke depression [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1278-1285. |
| [11] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [12] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [13] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [14] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [15] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||