Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (14): 2272-2277.doi: 10.3969/j.issn.2095-4344.0772
Previous Articles Next Articles
Adipose tissue engineering construction using adipose-derived stem cells and scaffolds in breast reconstruction
Guo Xiao1, Feng Rui2
- 1Tianjin Medical University, Tianjin 300072, China; 2Department of Breast Surgery, Tianjin Central Hospital of Gynecology Obstetrics, Tianjin 300072, China
-
Received:2017-12-26Online:2018-05-18Published:2018-05-18 -
Contact:Feng Rui, Professor, Chief physician, Master’s supervisor, Department of Breast Surgery, Tianjin Central Hospital of Gynecology Obstetrics, Tianjin 300072, China -
About author:Guo Xiao, Master candidate, Tianjin Medical University, Tianjin 300072, China
CLC Number:
Cite this article
Guo Xiao, Feng Rui. Adipose tissue engineering construction using adipose-derived stem cells and scaffolds in breast reconstruction[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(14): 2272-2277.
share this article

2.1 支架材料 应用于脂肪组织工程的支架材料需具备良好的生物相容性、可降解性及三维多孔结构,可为种子细胞提供可进行黏附、生长和增殖的结构支持,以改善细胞浸润活力,利于细胞进行营养物质交换,促进血管生成并形成正常的细胞信号传导通路。此外支架材料还应具备一定的机械强度以保持原有形状。然而,目前的支架材料普遍缺乏可连接宿主血运循环的3D毛细血管网[2],血管再生缓慢,制备工艺复杂且可注射性差。近年来,由低免疫原性的脱细胞脂肪组织基质构成的支架材料因其在组织相容性、生物降解性和生物诱导性等方面具有的独特优势而备受关注,可用于支持临床多种人类组织或器官再生。支架材料优缺点及应用见表1。 2.1.1 人工合成高分子支架材料 人工合成高分子材料主要包括聚乳酸、聚乙醇酸、聚羟基乙酸、聚四氟乙烯等。Patrick等[1]首次将聚羟基乙酸联合前脂肪细胞进行短期体内实验并成功诱导脂肪组织生成,随后将聚羟基乙酸支架联合前脂肪细胞植入大鼠体内,观察到新生脂肪组织量在2个月内达到顶峰,随后出现减少趋势,并在5-12个月内观察到脂肪组织及聚羟基乙酸支架材料完全降解消失,认为聚羟基乙酸支架材料可作为软组织工程构建的支架材料。李春波等[4]利用新型聚羟基乙酸复合壳聚糖作为支架材料,研究其与兔脂肪干细胞的生物相容性,在加入成脂诱导培养基后第7天时可观察到脂滴形成,研究证实新型聚羟基乙酸/壳聚糖复合支架材料具有良好的生物相容性,可作为种子细胞实现增殖、黏附、分化的支架材料,但该支架材料如何在体内实现大体积脂肪组织构建仍需进一步的实验研究。最近,Rossi等[3]制备了一种交联精氨酸-甘氨酸-门冬氨酸3肽的聚酰氨基胺低分子大孔泡沫支架材料,分别利用上皮细胞、成纤维细胞和内皮细胞对其进行体外评估,结果证明聚酰氨基胺低分子大孔泡沫支架材料在体外可支持多种细胞类型的黏附和增殖,与此同时,体内研究证实其促进了前脂肪细胞成脂分化及体内血管组织生长。但人工合成高分子支架材料也存在若干限制,如聚羟基乙酸具有高度亲水性,不易加工成稳定结构,通常需要与其他材料联合或利用物理或化学方法进行交联,以提高支架材料的机械力学性能。此外人工合成类材料通常需添加昂贵的化学修饰或诱导液等[16-17],以提高合成材料的细胞黏附及定向分化能力。"

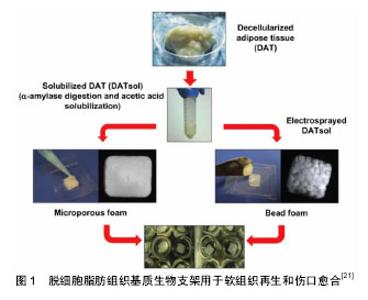
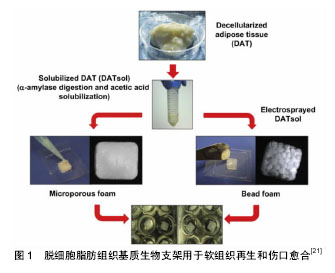
2.1.2 天然来源生物支架材料 天然支架材料主要分为天然多糖类和天然蛋白质类支架材料,分别包括壳聚糖、纤维素、甲壳素、透明质酸及胶原蛋白、纤维蛋白等。其中壳聚糖由于其良好的可降解性和生物相容性等生物学特性,已被广泛应用于组织工程中[6-7]。此外,天然生物支架材料常被加工为水凝胶形式,可作为药物或生长因子传递系统,也可作为注射材料用于修复或重建不规则形状的组织。Goisis等[18-19]发现透明质酸材料在微创移植中表现出良好的生物相容性,但在应用可注射透明质酸来源生物材料进行乳房重建时,部分患者可在治疗后30 d内观察到多个乳房肿块,肿块可干扰乳房X射线及超声检查,导致乳腺癌的误诊,因此,目前透明质酸来源生物材料应用于乳房重建时仍需进一步研究。作为脂肪酸的转运蛋白、白蛋白是许多亲脂性化合物的载体,能够通过促进脂质积累在脂肪细胞分化中起到重要作用,此外,卵清蛋白制备的支架材料相比其他天然支架材料更易获取且价格低廉,与人工合成材料相比具有更好的生物相容性和可降解性,但目前尚未有卵清蛋白支架材料应用于组织工程的相关报道。虽然天然生物来源支架材料具备促进种子细胞黏附、增殖、分化所需的天然微环境,但其局限性在于:其天然性质难以加工改性,降解速率不易控制,机械性能较低。 2.1.3 脱细胞脂肪组织基质 细胞外基质为细胞的迁移、增殖和分化提供了复杂的动态微环境,脱细胞化生物支架在去除引发免疫应答的细胞和抗原成分的同时,保留了关键细胞组分,如Ⅳ型胶原和层粘连蛋白等。同时,脱细胞化生物支架具备良好的组织特异性生物化学和力学性质,利于种子细胞进行生长、增殖、分化为组织或器官,从而对受损组织进行修复和再生。Flynn[15]和Turner等[14]的研究认为,脱细胞脂肪组织基质为脂肪组织生成提供了诱导微环境,可表达主要调节因子如脂蛋白脂肪酶、过氧化物酶体增殖物激活受体和增强子结合蛋白。相比于明胶等材料,脱细胞脂肪组织基质支架材料无需外源调节因子,即可用作软组织修复和重建的成脂生物支架(图1)[21]。值得一提的是,研究发现经不同脱细胞方法获得的脱细胞脂肪组织基质均基本保持了组织特异性,并能够诱导脂肪干细胞进行成脂分化[14-15,20]。Haddad等[13]利用取自腹部、乳房、心包和胸腺等部位的脱细胞脂肪组织基质材料,分别对乳房切除术后和乳房肿瘤切除术后患者进行乳房重建,在仰卧位和直立位下作可视化评估,并与正常乳房的对应变量模型进行比较,发现从腹部皮下脂肪组织和乳房衍生的脱细胞脂肪组织基质支架材料更适合用于乳房切除后乳房重建,而所有部位衍生的脱细胞脂肪组织基质支架材料在乳房肿瘤切除术后的乳房重建中均具有相似的性能。但如何快速、有效去除细胞内成分,并完整保留天然细胞外基质结构,仍是组织工程中脱细胞脂肪组织支架材料研究的关键。 2.2 种子细胞 间充质干细胞是存在于体内多种组织中,具有自我更新能力和多向分化潜能的干细胞,拥有易分离扩增、增殖能力强和低免疫原性等特性,对组织和各个器官系统的发育和再生起着重要的作用,成为了近年来组织工程和再生医学领域理想的种子细胞。 2.2.1 骨髓间充质干细胞 骨髓间充质干细胞具有易分离培养和免疫排斥反应小等优点,同时还具备强大的增殖能力及多向分化潜能,在体外适宜条件下能分化为成骨细胞、软骨细胞和脂肪细胞等,因此是组织工程理想的种子细胞。汪涛等[22]通过体外培养骨髓间充质干细胞,在成脂诱导液的作用下,第21天观察到其可成功定向分化为脂肪细胞。此外,大鼠和人类骨髓间充质干细胞已被证明在聚乳酸-羟基乙酸复合明胶支架材料上离体培养时,在纤维母细胞生长因子2的作用下能够向脂肪组织进行定向分化,这一结果为体内构建脂肪组织提供了依据[23-24]。但骨髓间充质干细胞由于潜在的并发症、需有创操作及体外增殖和分化能力受供者年龄影响等制约因素,作为组织工程理想的种子细胞还有待进一步改善。 2.2.2 乳腺干细胞 乳腺干细胞的增殖分化为乳房各个时期的分化发育提供了重要细胞来源。Shackleton 等[25-26]从活体中提取乳腺基底细胞,将其移植到免疫缺陷小鼠体内,形成了形态正常、功能完整的新生乳腺组织,证实了基底细胞中乳腺干细胞的存在,并可实现自我增殖,分化为成熟组织。Wang等[27]通过谱序追踪利用Procr标记的干细胞发现,乳腺干细胞在发育过程中能够分化为各种乳腺上皮细胞,并首次证实了乳腺干细胞具有多潜能性和高度再生能力,结束了乳腺中是否存在“多潜能”干细胞这一争议。但与此同时,提示了乳腺干细胞可能对乳腺癌的产生有重要影响。虽然该研究证实了乳腺干细胞实现乳房组织再生的可行性,但临床应用中依旧存在取材困难、定向分化成功率较低、易破坏健康乳腺组织等制约因素。此外,乳腺干细胞作为种子细胞的性质及发挥作用的分子机制仍需进一步研究探索。 2.2.3 人脂肪来源多能干细胞 2001年Zuk等[28]首次收集了抽脂术废弃的脂肪组织,利用胶原酶消化处理后,从中分离出脂肪来源干细胞,体外培养过程中发现该细胞群具有稳定的自我复制能力和低衰老性,与骨髓干细胞所特有的多向分化潜能等基本特征一致,可代替其作为备选干细胞来源。Heo等[29]对不同组织来源的干细胞在免疫表型、增殖潜力、多源分化和免疫调节能力等方面进行比较后得出结论:脂肪间充质干细胞与骨髓间充质干细胞不仅具有相同的体外三谱系(成骨、成脂、成软骨细胞)分化潜力,而且具有相似的表面抗原表达、旁分泌功能和基因表达谱。与骨髓间充质干细胞不同的是,脂肪组织来源干细胞具有取材容易、来源广泛,体内储备量大、对机体损伤小,能够在体外稳定增殖且衰亡率低。有研究表明1 mL骨髓抽吸物产生6×106个有核细胞,其中仅0.001%-0.01%是骨髓来源干细胞[30]。而1 g脂肪组织产生约2×106个有核细胞,其中多达10%为脂肪干细胞[31-32]。此外,与骨髓间充质干细胞相比,100 g脂肪组织可获得多达相同含量骨髓抽吸物300倍的间充质干细胞[33-34]。故少量脂肪组织即可获取大量干细胞,适宜大规模培养。Flynn等[15]将脂肪干细胞种植于脱细胞脂肪组织支架材料后体外培养14 d,观察到磷酸甘油脱氢酶活性和成脂相关基因的表达,如过氧化物酶体增殖物激活受体γ、脂蛋白脂肪酶等表达显著增高,同时可见细胞内脂滴形成,证实了脂肪干细胞联合脱细胞脂肪组织基质的复合物可促进脂肪干细胞的成脂分化。Mojallal[35]研究证实,脂肪干细胞的产量和增殖能力与供者年龄等无关,脂肪组织中所含干细胞的数量不会随着患者年龄的增加而减少。基于脂肪组织血管成分细胞中天然存在的脂肪干细胞的多向分化潜力及免疫调节作用,其可作为组织工程和再生医学最佳的干细胞来源,是脂肪组织工程理想的种子细胞[36-37]。 2.3 工程化脂肪组织构建的关键 局部微环境对诱导干细胞为成熟脂肪组织有很重要的作用,良好的血液供应是工程化移植物产生脂肪组织质量的关键因素,此外,良好的血运是关乎脂肪组织是否能够再生及长期稳定存活的基础,也是组织工程构建物植入体内后研究的难点。Lu等[38]认为脂肪干细胞具有促进血管重建功能,这可能是由于脂肪干细胞可分化成内皮细胞及脂肪干细胞可释放血管生长因子的间接作用导致。此外,有学者认为微血管内皮细胞释放的细胞外基质组分,具有直接刺激前脂肪细胞分化的能力,在体内其释放增殖和分化因子,共同促进脂肪组织扩增,在脂肪组织的发育和生长中发挥重要作用。Walton等[39]早前利用大鼠模型进行实验性研究,在纤维血管支架中添加成纤维细胞生长因子及前脂肪细胞来诱导脂质生成,通过硅胶外壳将构建体植入宿主相关部位,在第4-16周可观察到脂肪组织稳定生长,研究认为血管植入可维持较大体积脂肪的需血量,增强了脂肪移植物的耐久性,为大体积乳房缺损的构建提供了可能。此外,研究人员提出了不同的实验方案以促进早期组织血管化:局部加入血管生长因子、成纤维生长因子、干细胞生长因子等;病毒载体转染促血管生成基因;或者利用不同种子细胞进行共培养等,以使早期构建物尽快血管化,但目前为止都未能明显改善最终构建物的体积。 2.4 3D打印技术 3D打印技术旨在协调构建具有适当结构和材料构成的仿生支架,与传统方法相比,3D打印技术可使用各种各样的材料候选物,并且能够精确地定位生物材料、种子细胞及生长和分化因子,使之按照不同患者的需求分化成特定的结构。Pati等[40-41]通过开发脱细胞脂肪组织基质生物油墨来证明了该方法的实用性,其可与脂肪干细胞一起被精确打印成3D结构,通过这种技术,脂肪干细胞显示出较高的细胞活力和脂肪形成基因的表达。此外,该脱细胞脂肪组织生物油墨表现出较少慢性炎症的阳性组织浸润,并且在小鼠皮下植入时,能够促进成熟的脂肪组织形成。此外,研究表明,3D印刷水凝胶支架的结构和机械性对脂肪组织的形成同样具有重要作用[42]。尽管3D印刷支架的微观结构有益于种子细胞进行分化,但是如何将其转化为较大的、可供临床应用的宏观结构目前仍具有挑战性。 2.5 脂肪组织工程方法在乳房再造领域的应用前景 组织工程方法在人体浅表器官再造特别是乳房再造领域中的应用,正越来越多的引起临床外科医生和基础研究学者的重视,如何将组织工程充分血管化、获取可调节性质的高级支架材料,以及在如何阻止植入物免疫反应等方面已取得了重大进展[43-45]。组织工程方法修复或重建软组织的目的在于维持所需部位三维体积,并且促进患者自身健康组织的再生能力。但目前仍然存在有待进一步研究解决的问题:脂肪源性干细胞特异性表达蛋白及基因尚不明确,此外,来源于不同个体、不同部位的脂肪源性干细胞分化能力有明显差别,但其具体原因尚需进一步研究,与此同时,支架材料的优劣对组织工程的成败起着决定性作用。虽然对支架材料的研究已获得了重大进步,但大部分支架材料还有各种各样的缺陷。最后,如何构建足够体积的可供移植的成熟脂肪组织,仍是目前脂肪组织工程研究的重点及难点。Haddad等[13]认为,脱细胞脂肪组织支架材料的机械性能与正常乳腺脂肪组织的机械性质相似,使用脱细胞脂肪组织支架材料重建的乳房在生理负荷条件下可显示出规则的轮廓,与正常乳房组织的变形相似。此外,Chhaya等[46]将捕获的乳房切除术后乳房区域图像处理成3D模型,利用生物打印技术制造出高度多孔的可生物降解聚丙交酯聚合物支架材料,将人脐静脉血内皮细胞接种于该拥有乳房形状的构建体中,在体内模型中观察到持续再生的脂肪组织长达24周,该研究利用3D打印技术定做特定乳房形状的支架材料,为脂肪组织工程应用于乳房重建提供了新的思路。此外,Jotzu等[47]研究发现,脂肪间充质干细胞是肿瘤生长、侵袭和转移过程中发挥重要作用癌相关成纤维细胞的来源,可自发动员并影响癌症进展。Eterno等[48]为了证实脂肪干细胞是否在癌症复发中具有潜在致癌作用,将自体脂肪干细胞与来自人类供体原发性乳腺癌细胞相结合,建立模型,评估了其在体外和体内的相互作用后得出结论:脂肪干细胞本身不具有致癌性,并不诱导正常乳腺细胞向肿瘤转化,但是脂肪干细胞可增强c-Met基因表达的乳腺癌细胞致癌行为,产生支持癌细胞和血管生长的炎性微环境。"
| [1] Patrick CJ,Chauvin PB,Hobley J,et al.Preadipocyte seeded PLGA scaffolds for adipose tissue engineering.Tissue Eng.1999;5(2): 139-151.[2] Salgarello M,Visconti G,Rusciani A.Breast fat grafting with platelet-rich plasma: a comparative clinical study and current state of the art.Plast Reconstr Surg.2011;127(6):2176-2185.[3] Rossi E,Gerges I,Tocchio A,et al.Biologically and mechanically driven design of an RGD-mimetic macroporous foam for adipose tissue engineering applications.Biomaterials.2016;104:65-77.[4] 李春波,王红,陈增淦,等.兔脂肪干细胞(ADSCs)与聚羟基乙酸/壳聚糖(PLGA/CS)支架材料生物相容性研究[J].复旦学报(医学版), 2014,41(5): 610-616.[5] Norouzi M,Boroujeni SM,Omidvarkordshouli N,et al.Advances in skin regeneration: application of electrospun scaffolds.Adv Healthc Mater. 2015;4(8):1114-1133.[6] Sionkowska A,Kaczmarek B,Lewandowska K,et al.3D composites based on the blends of chitosan and collagen with the addition of hyaluronic acid.Int J Biol Macromol.2016;89:442-448.[7] Wang J,Wang L,Yu H,et al. Recent progress on synthesis, property and application of modified chitosan: An overview.Int J Biol Macromol. 2016;88:333-344.[8] 孔易,徐瑞,邢梦秋,等.壳聚糖/藻酸盐纳米膜在促进小鼠全层损伤皮肤创面愈合中的作用[J].第三军医大学学报,2015,37(21):2109-2114.[9] 张翠荣,谢华飞,贾振宇,等.壳聚糖基敏感性凝胶的研究进展[J].材料导报, 2012,26(3):92-95.[10] Isik S,Taskapilioglu MO,Atalay FO,et al.Effects of cross-linked high-molecular-weight hyaluronic acid on epidural fibrosis: experimental study.J Neurosurg Spine.2015;22(1):94-100.[11] Lam J,Truong NF,Segura T.Design of cell-matrix interactions in hyaluronic acid hydrogel scaffolds.Acta Biomater.2014;10(4):1571-1580.[12] Tan QW,Zhang Y,Luo JC,et al.Hydrogel derived from decellularized porcine adipose tissue as a promising biomaterial for soft tissue augmentation.J Biomed Mater Res A.2017;105(6):1756-1764.[13] Haddad SM,Omidi E,Flynn LE,et al.Comparative biomechanical study of using decellularized human adipose tissues for post-mastectomy and post-lumpectomy breast reconstruction.J Mech Behav Biomed Mater.2016;57:235-245.[14] Turner AE,Yu C,Bianco J,et al.The performance of decellularized adipose tissue microcarriers as an inductive substrate for human adipose-derived stem cells.Biomaterials.2012;33(18):4490-4499.[15] Flynn LE.The use of decellularized adipose tissue to provide an inductive microenvironment for the adipogenic differentiation of human adipose-derived stem cells.Biomaterials.2010;31(17):4715-4724.[16] Blit PH,Shen YH,Ernsting MJ,et al.Bioactivation of porous polyurethane scaffolds using fluorinated RGD surface modifiers.J Biomed Mater Res A.2010;94(4):1226-1235.[17] Guarnieri D,De Capua A,Ventre M,et al.Covalently immobilized RGD gradient on PEG hydrogel scaffold influences cell migration parameters. Acta Biomater.2010;6(7):2532-2539.[18] Goisis M,Yoshimura K,Heden P.Breast augmentation after Macrolane filler injections.Aesthetic Plast Surg.2011;35(4):684-686.[19] Goisis M,Savoldi A,Guareschi M.Is hyaluronic acid gel a good option for breast augmentation? Aesthetic Plast Surg.2011;35(1):134-136,137.[20] Brown BN,Freund JM,Han L,et al.Comparison of three methods for the derivation of a biologic scaffold composed of adipose tissue extracellular matrix.Tissue Eng Part C Methods.2011;17(4):411-421.[21] Yu C,Bianco J,Brown C,et al. Porous decellularized adipose tissue foams for soft tissue regeneration.Biomaterials.2013;34(13):3290-3302.[22] 汪涛,颜瑞巧,曹俊,等.miRNA-140-5p在人骨髓间充质干细胞成脂分化中的表达及其靶基因的预测[J].南方医科大学学报,2017,37(2):199-203.[23] Neubauer M,Hacker M,Bauer-Kreisel P,et al.Adipose tissue engineering based on mesenchymal stem cells and basic fibroblast growth factor in vitro.Tissue Eng.2005;11(11-12):1840-1851.[24] Hong L,Peptan I,Clark P,et al.Ex vivo adipose tissue engineering by human marrow stromal cell seeded gelatin sponge.Ann Biomed Eng. 2005;33(4):511-517.[25] Shackleton M,Vaillant F,Simpson KJ,et al.Generation of a functional mammary gland from a single stem cell.Nature.2006;439(7072):84-88.[26] Stingl J,Eirew P,Ricketson I,et al.Purification and unique properties of mammary epithelial stem cells.Nature.2006;439(7079):993-997.[27] Wang D,Cai C,Dong X,et al.Identification of multipotent mammary stem cells by protein C receptor expression.Nature. 2015;517(7532): 81-84.[28] Zuk PA,Zhu M,Mizuno H,et al.Multilineage cells from human adipose tissue: implications for cell-based therapies.Tissue Eng. 2001;7(2): 211-228.[29] Heo JS,Choi Y,Kim HS,et al.Comparison of molecular profiles of human mesenchymal stem cells derived from bone marrow, umbilical cord blood, placenta and adipose tissue.Int J Mol Med. 2016;37(1): 115-125.[30] Pittenger MF,Mackay AM,Beck SC,et al.Multilineage potential of adult human mesenchymal stem cells.Science.1999;284(5411):143-147.[31] Yoshimura K,Suga H,Eto H.Adipose-derived stem/progenitor cells: roles in adipose tissue remodeling and potential use for soft tissue augmentation.Regen Med.2009;4(2):265-273.[32] [32]Zhu Y,Liu T,Song K,et al.Adipose-derived stem cell: a better stem cell than BMSC.Cell Biochem Funct.2008;26(6):664-675.[33] Oedayrajsingh-Varma MJ,van Ham SM,Knippenberg M,et al.Adipose tissue-derived mesenchymal stem cell yield and growth characteristics are affected by the tissue-harvesting procedure.Cytotherapy. 2006; 8(2):166-177.[34] Aust L,Devlin B,Foster SJ,et al.Yield of human adipose-derived adult stem cells from liposuction aspirates.Cytotherapy.2004;6(1):7-14.[35] Mojallal A,Lequeux C,Shipkov C,et al.Influence of age and body mass index on the yield and proliferation capacity of adipose-derived stem cells.Aesthetic Plast Surg.2011;35(6):1097-1105.[36] Zielins ER,Paik K,Ransom RC,et al.Enrichment of Adipose-Derived Stromal Cells for BMPR1A Facilitates Enhanced Adipogenesis.Tissue Eng Part A.2016,22(3-4):214-221.[37] Lin G,Garcia M,Ning H,et al. Defining stem and progenitor cells within adipose tissue.Stem Cells Dev.2008;17(6):1053-1063.[38] Lu F,Mizuno H,Uysal CA,et al.Improved viability of random pattern skin flaps through the use of adipose-derived stem cells.Plast Reconstr Surg.2008;121(1):50-58.[39] Walton RL,Beahm EK,Wu L.De novo adipose formation in a vascularized engineered construct. Microsurgery.2004;24(5):378-384.[40] Pati F,Ha DH,Jang J,et al.Biomimetic 3D tissue printing for soft tissue regeneration.Biomaterials. 2015;62:164-175.[41] Pati F,Jang J,Ha DH,et al.Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink.Nat Commun. 2014;5: 3935.[42] Das S,Pati F,Choi YJ,et al.Bioprintable, cell-laden silk fibroin-gelatin hydrogel supporting multilineage differentiation of stem cells for fabrication of three-dimensional tissue constructs. Acta Biomater. 2015;11:233-246.[43] Kolesky DB,Homan KA,Skylar-Scott MA,et al.Three-dimensional bioprinting of thick vascularized tissues.Proc Natl Acad Sci U S A. 2016;113(12):3179-3184.[44] Kang HW,Lee SJ,Ko IK,et al.A 3D bioprinting system to produce human-scale tissue constructs with structural integrity. Nat Biotechnol. 2016;34(3):312-319.[45] Murphy SV,Atala A.3D bioprinting of tissues and organs. Nat Biotechnol. 2014;32(8):773-785.[46] Chhaya MP,Melchels FP,Holzapfel BM,et al.Sustained regeneration of high-volume adipose tissue for breast reconstruction using computer aided design and biomanufacturing.Biomaterials.2015;52:551-560.[47] Jotzu C,Alt E,Welte G,et al.Adipose tissue derived stem cells differentiate into carcinoma-associated fibroblast-like cells under the influence of tumor derived factors. Cell Oncol (Dordr). 2011;34(1): 55-67.[48] Eterno V,Zambelli A,Pavesi L,et al.Adipose-derived Mesenchymal Stem Cells (ASCs) may favour breast cancer recurrence via HGF/c-Met signaling.Oncotarget.2014;5(3):613-633.[49] Maijers MC,Niessen FB.Prevalence of rupture in poly implant Prothese silicone breast implants, recalled from the European market in 2010.Plast Reconstr Surg.2012;129(6):1372-1378.[50] Kolle SF,Fischer-Nielsen A,Mathiasen AB,et al. Enrichment of autologous fat grafts with ex-vivo expanded adipose tissue-derived stem cells for graft survival: a randomised placebo-controlled trial. Lancet.2013;382(9898):1113-1120.[51] Harrison BL,Malafa M,Davis K,et al.The discordant histology of grafted fat: a systematic review of the literature.Plast Reconstr Surg. 2015; 135(3):542e-555e.[1] Patrick CJ,Chauvin PB,Hobley J,et al.Preadipocyte seeded PLGA scaffolds for adipose tissue engineering.Tissue Eng.1999;5(2): 139-151.[2] Salgarello M,Visconti G,Rusciani A.Breast fat grafting with platelet-rich plasma: a comparative clinical study and current state of the art.Plast Reconstr Surg.2011;127(6):2176-2185.[3] Rossi E,Gerges I,Tocchio A,et al.Biologically and mechanically driven design of an RGD-mimetic macroporous foam for adipose tissue engineering applications.Biomaterials.2016;104:65-77.[4] 李春波,王红,陈增淦,等.兔脂肪干细胞(ADSCs)与聚羟基乙酸/壳聚糖(PLGA/CS)支架材料生物相容性研究[J].复旦学报(医学版), 2014,41(5): 610-616.[5] Norouzi M,Boroujeni SM,Omidvarkordshouli N,et al.Advances in skin regeneration: application of electrospun scaffolds.Adv Healthc Mater. 2015;4(8):1114-1133.[6] Sionkowska A,Kaczmarek B,Lewandowska K,et al.3D composites based on the blends of chitosan and collagen with the addition of hyaluronic acid.Int J Biol Macromol.2016;89:442-448.[7] Wang J,Wang L,Yu H,et al. Recent progress on synthesis, property and application of modified chitosan: An overview.Int J Biol Macromol. 2016;88:333-344.[8] 孔易,徐瑞,邢梦秋,等.壳聚糖/藻酸盐纳米膜在促进小鼠全层损伤皮肤创面愈合中的作用[J].第三军医大学学报,2015,37(21):2109-2114.[9] 张翠荣,谢华飞,贾振宇,等.壳聚糖基敏感性凝胶的研究进展[J].材料导报, 2012,26(3):92-95.[10] Isik S,Taskapilioglu MO,Atalay FO,et al.Effects of cross-linked high-molecular-weight hyaluronic acid on epidural fibrosis: experimental study.J Neurosurg Spine.2015;22(1):94-100.[11] Lam J,Truong NF,Segura T.Design of cell-matrix interactions in hyaluronic acid hydrogel scaffolds.Acta Biomater.2014;10(4):1571-1580.[12] Tan QW,Zhang Y,Luo JC,et al.Hydrogel derived from decellularized porcine adipose tissue as a promising biomaterial for soft tissue augmentation.J Biomed Mater Res A.2017;105(6):1756-1764.[13] Haddad SM,Omidi E,Flynn LE,et al.Comparative biomechanical study of using decellularized human adipose tissues for post-mastectomy and post-lumpectomy breast reconstruction.J Mech Behav Biomed Mater.2016;57:235-245.[14] Turner AE,Yu C,Bianco J,et al.The performance of decellularized adipose tissue microcarriers as an inductive substrate for human adipose-derived stem cells.Biomaterials.2012;33(18):4490-4499.[15] Flynn LE.The use of decellularized adipose tissue to provide an inductive microenvironment for the adipogenic differentiation of human adipose-derived stem cells.Biomaterials.2010;31(17):4715-4724.[16] Blit PH,Shen YH,Ernsting MJ,et al.Bioactivation of porous polyurethane scaffolds using fluorinated RGD surface modifiers.J Biomed Mater Res A.2010;94(4):1226-1235.[17] Guarnieri D,De Capua A,Ventre M,et al.Covalently immobilized RGD gradient on PEG hydrogel scaffold influences cell migration parameters. Acta Biomater.2010;6(7):2532-2539.[18] Goisis M,Yoshimura K,Heden P.Breast augmentation after Macrolane filler injections.Aesthetic Plast Surg.2011;35(4):684-686.[19] Goisis M,Savoldi A,Guareschi M.Is hyaluronic acid gel a good option for breast augmentation? Aesthetic Plast Surg.2011;35(1):134-136,137.[20] Brown BN,Freund JM,Han L,et al.Comparison of three methods for the derivation of a biologic scaffold composed of adipose tissue extracellular matrix.Tissue Eng Part C Methods.2011;17(4):411-421.[21] Yu C,Bianco J,Brown C,et al. Porous decellularized adipose tissue foams for soft tissue regeneration.Biomaterials.2013;34(13):3290-3302.[22] 汪涛,颜瑞巧,曹俊,等.miRNA-140-5p在人骨髓间充质干细胞成脂分化中的表达及其靶基因的预测[J].南方医科大学学报,2017,37(2):199-203.[23] Neubauer M,Hacker M,Bauer-Kreisel P,et al.Adipose tissue engineering based on mesenchymal stem cells and basic fibroblast growth factor in vitro.Tissue Eng.2005;11(11-12):1840-1851.[24] Hong L,Peptan I,Clark P,et al.Ex vivo adipose tissue engineering by human marrow stromal cell seeded gelatin sponge.Ann Biomed Eng. 2005;33(4):511-517.[25] Shackleton M,Vaillant F,Simpson KJ,et al.Generation of a functional mammary gland from a single stem cell.Nature.2006;439(7072):84-88.[26] Stingl J,Eirew P,Ricketson I,et al.Purification and unique properties of mammary epithelial stem cells.Nature.2006;439(7079):993-997.[27] Wang D,Cai C,Dong X,et al.Identification of multipotent mammary stem cells by protein C receptor expression.Nature. 2015;517(7532): 81-84.[28] Zuk PA,Zhu M,Mizuno H,et al.Multilineage cells from human adipose tissue: implications for cell-based therapies.Tissue Eng. 2001;7(2): 211-228.[29] Heo JS,Choi Y,Kim HS,et al.Comparison of molecular profiles of human mesenchymal stem cells derived from bone marrow, umbilical cord blood, placenta and adipose tissue.Int J Mol Med. 2016;37(1): 115-125.[30] Pittenger MF,Mackay AM,Beck SC,et al.Multilineage potential of adult human mesenchymal stem cells.Science.1999;284(5411):143-147.[31] Yoshimura K,Suga H,Eto H.Adipose-derived stem/progenitor cells: roles in adipose tissue remodeling and potential use for soft tissue augmentation.Regen Med.2009;4(2):265-273.[32] [32]Zhu Y,Liu T,Song K,et al.Adipose-derived stem cell: a better stem cell than BMSC.Cell Biochem Funct.2008;26(6):664-675.[33] Oedayrajsingh-Varma MJ,van Ham SM,Knippenberg M,et al.Adipose tissue-derived mesenchymal stem cell yield and growth characteristics are affected by the tissue-harvesting procedure.Cytotherapy. 2006; 8(2):166-177.[34] Aust L,Devlin B,Foster SJ,et al.Yield of human adipose-derived adult stem cells from liposuction aspirates.Cytotherapy.2004;6(1):7-14.[35] Mojallal A,Lequeux C,Shipkov C,et al.Influence of age and body mass index on the yield and proliferation capacity of adipose-derived stem cells.Aesthetic Plast Surg.2011;35(6):1097-1105.[36] Zielins ER,Paik K,Ransom RC,et al.Enrichment of Adipose-Derived Stromal Cells for BMPR1A Facilitates Enhanced Adipogenesis.Tissue Eng Part A.2016,22(3-4):214-221.[37] Lin G,Garcia M,Ning H,et al. Defining stem and progenitor cells within adipose tissue.Stem Cells Dev.2008;17(6):1053-1063.[38] Lu F,Mizuno H,Uysal CA,et al.Improved viability of random pattern skin flaps through the use of adipose-derived stem cells.Plast Reconstr Surg.2008;121(1):50-58.[39] Walton RL,Beahm EK,Wu L.De novo adipose formation in a vascularized engineered construct. Microsurgery.2004;24(5):378-384.[40] Pati F,Ha DH,Jang J,et al.Biomimetic 3D tissue printing for soft tissue regeneration.Biomaterials. 2015;62:164-175.[41] Pati F,Jang J,Ha DH,et al.Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink.Nat Commun. 2014;5: 3935.[42] Das S,Pati F,Choi YJ,et al.Bioprintable, cell-laden silk fibroin-gelatin hydrogel supporting multilineage differentiation of stem cells for fabrication of three-dimensional tissue constructs. Acta Biomater. 2015;11:233-246.[43] Kolesky DB,Homan KA,Skylar-Scott MA,et al.Three-dimensional bioprinting of thick vascularized tissues.Proc Natl Acad Sci U S A. 2016;113(12):3179-3184.[44] Kang HW,Lee SJ,Ko IK,et al.A 3D bioprinting system to produce human-scale tissue constructs with structural integrity. Nat Biotechnol. 2016;34(3):312-319.[45] Murphy SV,Atala A.3D bioprinting of tissues and organs. Nat Biotechnol. 2014;32(8):773-785.[46] Chhaya MP,Melchels FP,Holzapfel BM,et al.Sustained regeneration of high-volume adipose tissue for breast reconstruction using computer aided design and biomanufacturing.Biomaterials.2015;52:551-560.[47] Jotzu C,Alt E,Welte G,et al.Adipose tissue derived stem cells differentiate into carcinoma-associated fibroblast-like cells under the influence of tumor derived factors. Cell Oncol (Dordr). 2011;34(1): 55-67.[48] Eterno V,Zambelli A,Pavesi L,et al.Adipose-derived Mesenchymal Stem Cells (ASCs) may favour breast cancer recurrence via HGF/c-Met signaling.Oncotarget.2014;5(3):613-633.[49] Maijers MC,Niessen FB.Prevalence of rupture in poly implant Prothese silicone breast implants, recalled from the European market in 2010.Plast Reconstr Surg.2012;129(6):1372-1378.[50] Kolle SF,Fischer-Nielsen A,Mathiasen AB,et al. Enrichment of autologous fat grafts with ex-vivo expanded adipose tissue-derived stem cells for graft survival: a randomised placebo-controlled trial. Lancet.2013;382(9898):1113-1120.[51] Harrison BL,Malafa M,Davis K,et al.The discordant histology of grafted fat: a systematic review of the literature.Plast Reconstr Surg. 2015; 135(3):542e-555e. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||