Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (8): 1247-1252.doi: 10.3969/j.issn.2095-4344.0144
Previous Articles Next Articles
Effect of curcumin against capillarization of hepatic sinusoids and its mechanism
Duan Xue-lin, Peng Yue, Zhao Tie-jian, Wei Yan-fei, Li Gui-yu
- Department of Medical Physiology and Biochemistry, School of Basic Medical Science, Guangxi University of Chinese Medicine, Nanning 530200, Guangxi Zhuang Autonomous Region, China
-
Online:2018-03-18Published:2018-03-18 -
Contact:赵铁建。广西中医药大学基础医学院人体机能系,广西壮族自治区南宁市 530200 -
About author:Duan Xue-lin, Master, Associate professor, Department of Medical Physiology and Biochemistry, School of Basic Medical Science, Guangxi University of Chinese Medicine, Nanning 530200, Guangxi Zhuang Autonomous Region, China Peng Yue, M.D., Associate professor, Department of Medical Physiology and Biochemistry, School of Basic Medical Science, Guangxi University of Chinese Medicine, Nanning 530200, Guangxi Zhuang Autonomous Region, China Duan Xue-lin and Peng Yue contributed equally to this work. -
Supported by:the National Natural Science Foundation of China, No. 81403189, 81460628, 81660705 and 81560690; Scientific Research Project of Universities of Education Department of Guangxi Zhuang Autonomous Region, No. YB2014182
CLC Number:
Cite this article
Duan Xue-lin, Peng Yue, Zhao Tie-jian, Wei Yan-fei, Li Gui-yu. Effect of curcumin against capillarization of hepatic sinusoids and its mechanism[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(8): 1247-1252.
share this article
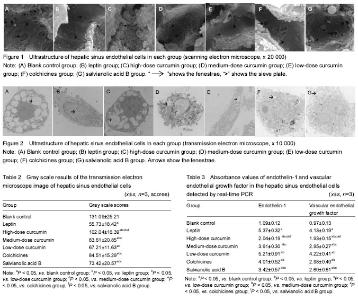
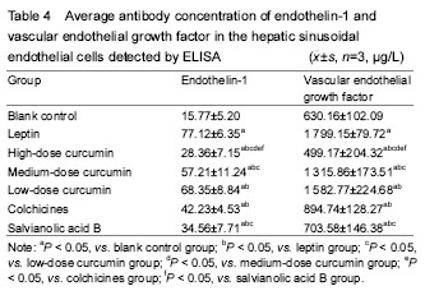
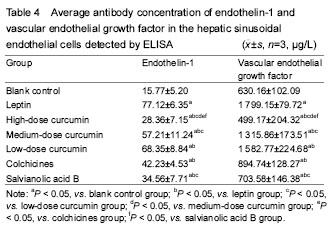
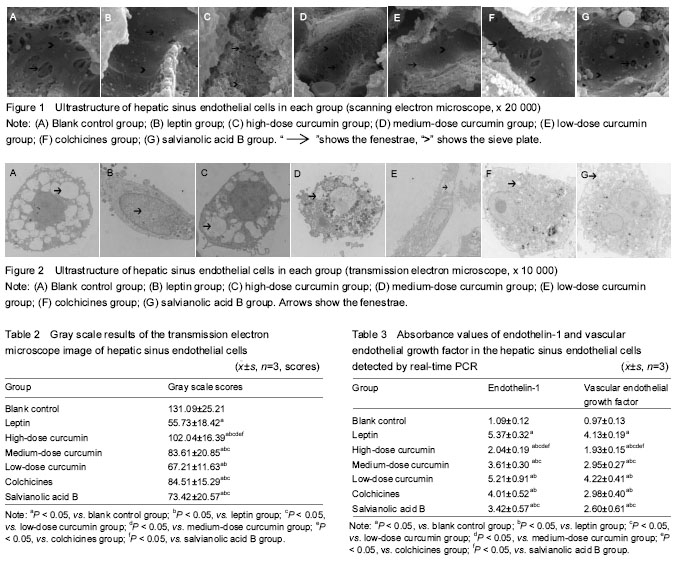
Ultrastructural changes in the HSECs The results of scanning electron microscope showed that the cell wall of HSECs in the blank control group was mesoporous, with abundant fenestrae of uneven size (the large diameter was about 1 μm and the small one was about 0.1 μm), especially the large fenestrae (Figure 1A). The cell wall of HSECs in the leptin group showed less or none fenestrae, and its shape changes from honeycomb to flat plate (Figure 1B). Compared with the leptin group, the number of fenestrae in HSECs in the high-dose curcumin group was significantly increased, and the sinus wall returned to mesoporous shape with more large fenestrae (the diameter > 1 μm) (Figure 1C). The number of fenestrae in the medium-dose curcumin group was more than that in the leptin group, but lower than those in the high-dose curcumin group (Figure 1D). There was no significant difference in the number of fenestrae between low-dose curcumin and leptin groups. Compared with the medium- and high-dose curcumin groups, in the low-dose curcumin group, the number of fenestrae was less and with smaller diameter (Figure 1E).The number of fenestrae in the HSEC wall in the colchicines and salvianolic acid B groups was obviously more than that in the blank control and low-dose curcumin group, and the size of the fenestrae was larger, but compared with the medium- and high-dose curcumin groups, the number and size of fenestrae were reduced. The number and size of fenestrae did not differ significantly between colchicines and salvianolic acid groups (Figure 1F, G). Results of transmission electron microscope revealed that there were a large number of hollow light-colored areas in the HSECs of the blank control group, and the fenestrae vwere dense and large in area, the monolayer cell membrane was thin, without basement membrane formation (Figure 2A). In the leptin group, the light-translucent area in HSECs almost disappeared, the number and area of fenestrae were reduced, the thickened cell membrane showed two-to-three layers, and the light-colored basement membrane area was obvious and continuous (Figure 2B). In the high-dose curcumin group, compared with the leptin group, the light translucent area within the HSECs increased significantly, the number and area of visible fenestrae were also increased, hollow-intensive sinus wall was almost sieve-shaped; thin membrane was observed, and the light-colored basement membrane area was formed outside the wall (Figure 2C). In the medium-dose curcumin group, compared with the leptin group, the light translucent area within the HSECs was enlarged, with a large number of fenestrae; two layers of the cell membrane could be still observed, but the light-colored basement membrane outside the cell wall was unobvious. However, compared with the high-dose curcumin group, the intracellular light-colored fenestrae area was smaller, and the extracellular basement membrane area existed (Figure 2D). Compared with the leptin group, in the low-dose curcumin group, the light-colored translucent area in the HSECs increased, the number of fenestrae increased; two distinct layers of the cell membrane were visible, obvious light-colored basement membrane area appeared outside the cell wall. Compared with the medium- and high-dose curcumin groups, the light-colored fenestrae area in the cells of the low-dose curcumin group was obviously smaller, and the light-colored basement membrane area was clearly seen (Figure 2E). Compared with the leptin group, in the colchicine and salvianolic acid B groups, the light translucent area in the HSECs was significantly expanded, with increased number and area of fenestrae; the cell membrane was thinned, and it was hard to detect the two layers, and the light-colored basement membrane area was nearly observed outside the cell wall. There were no significant differences in the number and area of fenestrae, the thickness of the cell membrane and the basement membrane outside the cell wall between two control drug groups. Compared with the medium-and high-dose curcumin groups, the number and area of fenestrae in the two drug control groups were lower, and the formation of extracellular basement membrane was more obvious. Compared with the low-dose curcumin group, the light-colored fenestrae area in the two drug control groups was larger with unobvious cell membrane thickening, and unclear basement membrane (Figure 2F, G). Gray scale results of the HSECs transmission electron microscope images showed that the gray scale score of HSECs in the high-dose curcumin group was the highest (P < 0.05; Table 2). mRNA and protein expression levels of endothelin-1 and VEGF in each group RT-PCR and EILSA results are shown in Tables 3, 4."
| [1] Ford AJ, Jain G, Rajagopalan P. Designing a fibrotic microenvironment to investigate changes in human liver sinusoidal endothelial cell function. Acta Biomater. 2015;24:220-227.[2] Poynard T, Munteanu M, Deckmyn O, et al. Validation of liver fibrosis biomarker (FibroTest) for assessing liver fibrosis progression: proof of concept and first application in a large population. J Hepatol. 2012;57(3):541-548.[3] Fomin ME, Zhou Y, Beyer AI, et al. Production of factor viii byhuman liver sinusoidal endothelial cells transplanted in immunodeficient uPA mice. PLoS One. 2013;8:e77255.[4] Feng YQ, Yang YS, Cui SL, et al. Advance in the development and application of experimental animal models of liver fibrosis. Guangdong Yaoxueyuan Xuebao. 2013;29(5):1-4.[5] Xie G, Wang X, Wang L, et al. Role of differentiation of liver sinusoidal endothelial cells in progression and regression of hepatic fibrosis in rats. Gastroenterology. 2012;142:918-927.[6] Liang S, Kisseleva T, Brenner DA. The role of NADPH oxidases (NOXs) in liver fibrosis and the activation of myofibroblasts. Front Physiol. 2016;7:17.[7] Connolly BM, Choi EY, Gadsvoll H, et al. Selective abrogation of the uPA-uPAR interaction in vivo reveals a novel role in sup-pression of fibrin-associated inflammation. Blood. 2010;116(9):1593-1603.[8] Du W, San YL, Liao QL, et al. Clinical significances of HA, PⅢNP, LN and CⅣ in the diagnosis of chronic liver disease and liver fibrosis. Chongqing Yike Daxue Xuebao. 2013;10(5):530-533.[9] Wu XL, Wang LK, Xue J, et al. Correlation of expressions of DNA binding differentiation inhibitory proteins-1 and vascular endothelial growth factor with its microvascular generation. Zhongguo Linhcunag Yaolixue Zazhi. 2016;05:430-436.[10] Yin F, Wang S, Hao Y, et al. Progress in pharmacology and mechanism research of curcumin. Hebei Lianhe Daxue Xuebao. 2013;15(11):798-802.[11] Wang XX, Liu WD. Progress in pharmacology research of curcumin. Beifang Yaoxue. 2010;7(2):30-34.[12] Wu SH, Wang YS. Progress of curcumin. Shuli Yiyaoxue Zazhi. 2013;9(3):122-125.[13] Huang MF, Liu DY, Xu R. Action mechanism of curcumin appreciated and analysis on clinical application. Zhonghua Zhongyiyao Xuekan. 2014;11(8):86-92.[14] Peng Y, Miao WN, Zhao TJ, et al. The research on toxicity and its mechanism of human hepatic stellate cells by Plumbagin. Shizhen Guoyi Guoyao. 2016;1(27):5-8. [15] Wei YF, Liu XM, Tang AC, et al. Effects of plumbagozeylanica (plumbago) on expression of transforming growth factorbeta1 in human hepatic stellate cells activated by lepti. Zhongguo Yaolixue Tongbao. 2010;26(6):421-424.[16] Xue DY, Hong JH, Xu LM. Effects of salvianol ic acid B on signal transduction induced by transforming growth factor-BB and plateletderived growth factor-β1 in hepatic stellate cells of rats. Zhongxiyi Jiehe Zazhi. 2006;26(5):439-442.[17] Zhang AL, Yang Z, Xiao XM, et al. Expression of p-moesin in sinusoidal endothelial cell of schistosomal hepatic fibrosis mice. Weixunhuanxue Zazhi. 2012;3:1-8.[18] Mormone E, George J, Nieto N. Molecular pathogenesis of hepatic fibrosis and current therapeutic approaches. Chem Biol Interact. 2011;19(3):225-231.[19] Yang ZX, Han DW, Xing HY, et al. Pathogenesis of hepatic sinusoidal capillarization in hepatic cirrhosis. World J Digestol. 2009;17(30):3101-3108.[20] Huebert Robert C, Shah Vijay H. sinusoidal endothelial cells direct traffic at the intersection of regeneration andfibrosis. Hepatology. 2014;60(2):754-756.[21] Jacobs F, Wisse E, De Geest B. The role of liver sinusoidal cells in hepatocyte-directed gene transfer. Am J Pathol. 2010;176(1):14-21.[22] Tian BF. Progress in the clinical application of colchicines. Kunming Yike Daxue Xuebao. 2009;30(2B):107-111. [23] Xu CS, Zhang H, Ca DH. Advances in clinical application of colchicine. Zhongguo Linchuang Yaoxue Zazhi. 2001;10(3):202.[24] Rambaldi A, Gluud C. Colchicine for alcoholic and non-alcoholic liver fibrosis and cirrhosis. Cochrane Database Syst Rev. 2005;18(2):CD002148.[25] Qu GW, Xie FX, Xue XD, et al. The distribution of salvianolic acid B and tanshinone Ⅱ A in Salvia Miltiorrhiza Bge. Zhongugo Xiandai Yiyao. 2005;7(1):11-13.[26] Zhu YQ, Zong W, Guo XY, et al. Sixty-three cases of fibrosis in patients with chronic hepatitis B treated with telbivudine compared with salvianolate. Shaanxi Yixue Zazhi. 2012;41(7):873-875.[27] Zhu YQ, Yang JS, Zong W, et al. Treatment of 61 cases of hepatitis B cirrhosis with Salvia miltiorrhiza citrate. Shaanxi Zhongyi. 2012;33(5):515-517.[28] Xu XY, Chen J, Hou XH, et al. Antioxidant effects of salvianolic acid B on pancreatic fibrosis. Zhonghua Xiaohua Zazhi. 2007;27(6):409-410.[29] Luo TS, Li HW, Liu ZW, et al. Advances in studies on extraction, separation, and pharmacological action of curcumin. Xiandai Yaowu yu Linchuang. 2011;26(2):102-107.[30] Di JB, Gu ZL, Zhao XD, et al. Advances in studies on antioxidant and anti-inflammation of curcumin. Zhongcaoyao. 2014;5(4):134-138.[31] Zhao ZD. Experimental study on curcuminon anti-liver fibrosis and its mechanism. Zhongguo Shiyan Fangjixue Zazhi. 2010;16(3):122-125.[32] Zhong Y, Zhong XH, Sun YM, et al. Protective effect of curcumin on liver fibrosis induced by carbon tetrachloride in rats. Zhongguo Gonggong Weisheng. 2015;31(4):447-449.[33] Liu XM, Li GY, Deng Y, et al. The effects of Guangxi Curcuma extract to the function of secretory collagen protein and MMPs of human hepatic stellate cell. Shizhen Guoyi Guoyao. 2013;34(4):35-39.[34] Maslak E, Gregorius A, Chlopicki S. Liver sinusoidal endothelial cells (LSECs) function and NAFLD; NO-based therapy targeted to the liver. Pharmacol Rep. 2015;67(4):689-694.[35] Rowe IA, Galsinh SK, Wilson GK, et al. Paracrine signals from liver sinusoidal endothelium regulate hepatitis C virus replication. Hepatology. 2014;59(2):375-384. [36] Li JM, Yan J, Lu WL, et al. Mechanism of liver sinusoidal endothelial cells involved in liver fibrosis. Dalian Yike Daxue Xuebao. 2013;35(1):81-82. [37] Shafiei MS, Lui S, Rockey DC. Integrin-linked kinase regulates endothelial cell nitric oxide synthase expression in hepatic sinusoidal endothelial cells. Liver Int. 2015;35(4):1213-1221.[38] DeLeve LD. Liver sinusoidal endothelial cells in hepatic fibrosis. Hepatology. 2015;61(5):1740-1746.[39] Yang WJ, Lei ZS, Li DY. Effects of nitric oxide and endouthelin-1 on hepatic damage induced by endotoxin in rats. Gandan Waike Zazhi. 2002;10(1):68-70.[40] Zhang J, Zhang ZT, Wang Y, et al. Effect of endothelin-1 and its antagonists on the expression of endothelin receptors mRNA in HSC-T6 cells. Zhonghua Waike Zazhi. 2005;43(21):68-70.[41] Arriazu E, de Galarreta MR, Cubero FJ, et al. Extracellular matrix and liver disease. Antioxid Redox Signal. 2014;21(7):1078-1097.[42] Li XH, Yang PF, Qi HX, et al. The changes and significance of serum TGF-β1, VEGF in patients with chronic hepatitis B, liver fibrosis and liver cirrhosis. Linchuang Xueyaobing Zazhi. 2011;23(3):144-145.[43] Tang X, Yang J, Li J. Sensitization of human hepatic stellate cells to tumor necrosis factorrelated apoptosis-inducing ligand-induced apoptosis by leflunomide. Biol Pharm Bul. 2009;32(6):963-967.[44] Teratani T, Tomita K, Suzuki T, et al. A high-cholesterol diet exacerbates liver fibrosis in micevia accumulation of free cholesterol in hepatic stellate cells. Gastroenterology. 2012;142(1):152-164.[45] Li L, Wang JY, Yang CQ, et al. Effect of RhoA on transforming growth factor β1-induced rat hepatic stellate cell migration. Liver Int. 2012;32(7):1093-1102.[46] Liu J. Chitosan nanoparticles enfolding antisense endothelin converting enzyme RNA expression plasmid and airway hyperresponsiveness in asthmatic mouse models. Zhongguo Zazhi Gongcheng Yanjiu. 2015;19(43):6913-6917. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Chen Jiming, Wu Xiaojing, Liu Tianfeng, Chen Haicong, Huang Chengshuo. Effects of silymarin on liver injury and bone metabolism induced by carbon tetrachloride in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1224-1228. |
| [3] | Wang Hanyue, Li Furong, Yang Xiaofei, Hu Chaofeng. Direct reprogramming hepatocytes into islet-like cells by efficiently targeting and activating the endogenous genes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1056-1063. |
| [4] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [5] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [6] | Liu Yang, Gong Yi, Fan Wei. Anti-hepatoma activity of targeted Pluronic F127/formononetin nanocomposite system in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 526-531. |
| [7] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [8] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [9] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [10] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [11] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [12] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [13] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [14] | Hao Xiaona, Zhang Yingjie, Li Yuyun, Xu Tao. Bone marrow mesenchymal stem cells overexpressing prolyl oligopeptidase on the repair of liver fibrosis in rat models [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3988-3993. |
| [15] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||