Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (22): 3576-3582.doi: 10.3969/j.issn.2095-4344.2017.22.022
Previous Articles Next Articles
Bioceramics in bone tissue engineering
- Department of Oral Craniomaxillofacial, Shanghai Ninth People’s Hospital, Shanghai 200000, China
-
Received:2017-04-27Online:2017-08-08Published:2017-09-01 -
Contact:Wang Xu-dong, Chief physician, Professor, Doctoral supervisor, Department of Oral Craniomaxillofacial, Shanghai Ninth People’s Hospital, Shanghai 200000, China -
About author:Lu Chen-pei, Studying for master’s degree, Department of Oral Craniomaxillofacial, Shanghai Ninth People’s Hospital, Shanghai 200000, China
CLC Number:
Cite this article
Lu Chen-pei, Wang Xu-dong, Shen Guo-fang. Bioceramics in bone tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(22): 3576-3582.
share this article

2.1 骨组织工程材料所需性能 骨组织工程支架应能够支持活细胞的黏附、增殖与分化,并且对受体组织没有毒害性,此即生物相容性。同时该载体还须具有骨诱导,骨传导及诱导血管生成的能力[2]。当用于生物体内修复缺损时,理想化的支架在诱导组织再生的同时自身能以可控的速率逐步降解,为新生的骨组织提供可容空间,最终用完全的再生组织来修复缺损,这就要求组织支架具备可降解性[2]。同时,骨细胞的生长需要适宜的营养及氧分要求,这就要求支架必须多孔化,以利于营养成分的输送。 此外,骨组织作为生物体内受力的组织,其对组织的机械强度也有相当的要求。用作缺损修复和重建的骨组织工程材料应当能够能承担生理功能下的机械应力,与周围本体组织的机械性能相适应[2]。 2.2 骨组织工程中常用的生物陶瓷材料及其特点 人体正常骨组织由细胞、纤维和基质构成。纤维为骨胶原纤维,基质中含有大量的固体无机盐。骨盐的主要成分是磷酸钙和碳酸钙,其主要形式为羟基磷灰石结晶(分子式:Ca10(PO4)6(OH)2)和无定型的磷酸钙混合物。这些无机成分是骨能维持一定强度和刚性的基础。生物陶瓷是指用作特定的生物或生理功能的一类陶瓷材料,即直接用于人体或与人体直接相关的生物、医用、生物化学等的陶瓷材料。骨组织工程中常用的生物陶瓷大致上可分为钙磷灰石和生物活性玻璃两类。由于它们的组成与人体正常骨组织中的结构和化学成分十分相近,成为骨组织工程中支架的良好材料,见表1。"

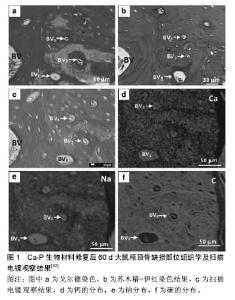
2.2.1 无机磷酸钙 临床上常用的无机磷酸钙包括羟基磷灰石及β-磷酸三钙。羟基磷灰石的主要形式为Ca10(PO4)6(OH)2,其本身是正常骨组织骨盐的主要成分,因此具有极佳的生物相容性。在体外和生物体内实验中,羟基磷灰石都被证明有良好的骨诱导和骨结合能力,同时能够较好地支持成骨细胞的黏附、增殖及分 化[3]。但羟基磷灰石在人体中的降解速度十分缓慢,Marcacci等[4]将羟基磷灰石支架植入人体内用于长骨修复,在六七年间的临床随访中,支架没有出现生物降解。相比之下,β-磷酸三钙在具有良好骨诱导性能的同时有良好的可吸收性[5]。与此同时,β-磷酸三钙在降解时可释放钙离子及磷酸盐离子,有助于新骨形成。β-磷酸三钙修复小面积缺损的骨愈合能力明显高于羟基磷灰石,但在修复大面积缺损时,由于β-磷酸三钙的降解速度过快导致缺损部位不能充分的被新骨填充[6]。有学者研究羟基磷灰石及相关无机磷酸钙材料的溶出度,提示其溶解速度顺序:无定型Cap>无定型羟基磷灰石>晶体Cap>晶体羟基磷灰石[7]。因此,研究者们结合羟基磷灰石和磷酸三钙形成双相磷酸钙,针对不同缺损修复大小的要求,通过调整两种材料的复合比例来控制材料降解及骨诱导生成的平衡,达到良好修复缺损的目的。 在机械强度方面,正常人体密质骨抗压强度为130-180 MPa,抗张强度为50-151 MPa,弹性模量为12-18 GPa,断裂韧性(断裂韧性:指材料阻止宏观裂纹失稳扩展能力的度量,也是材料抵抗脆性破坏的韧性参数)为6-8 MPa;正常人体松质骨抗压强度为4-12 MPa,弹性模量为0.1-0.5 GPa。单纯羟基磷灰石的抗压强度 > 400 MPa,抗张强度在40 MPa,弹性模量为 100 GPa,断裂韧性<1.0 MPa。无机磷酸钙的抗压强度为20-900 MPa,抗张强度为30-200 MPa,弹性模量为30-103 GPa,断裂韧性<1.0 MPa[8]。可见,单纯生物陶瓷材料脆性较大,其断裂韧性远小于正常密质骨,其抗张抗压强度也不能完全模仿生理骨组织,这导致了单一材料不能适应于承重骨或大面积骨缺损的替代重建。此外材料的多孔性也影响了材料的机械性能[9]。82%- 86%的多孔羟基磷灰石抗张强度为0.21-0.41 MPa,弹性模量为0.83-1.6 GPa[7]。已有研究证明,生物陶瓷材料的硬度和刚度和孔隙数目呈反比。Lane等[10-11]的研究指出,连接空隙的大小至少要达到100 μm直径才能进行有利的营养输送,且不同大小的孔隙(250-350 μm和<8 μm)互存对于羟基磷灰石支架上的骨形态蛋白发生、骨诱导和机械强度有益。Woodard等[12]对多微孔支架和单微孔支架的压力实验证明,单微孔支架的强度和刚性下降了46%,而多微孔支架的强度下降了30%,刚性下降了31%。由此可见,在强度和刚度上,非多孔支架>单微孔支架>多微孔支架。 为了解决这一问题,高分子聚合物和生物陶瓷的复合材料应运而生。因高分子聚合物具有与体内骨胶原相似的极性分子结构及优秀的机械性能,从而被运用于改善单纯磷灰石材料的性能。Zhang等[13]的研究证明陶瓷聚合支架材料较松质骨强度低,但其生物可降解性、弹性都较单纯生物陶瓷材料好。此外,在羟基磷灰石材料中加入直径15 nm、长度10-20 μm的纯纳米碳罐也可增加其承重的抗压性能[9]。也有学者通过将羟基磷灰石/磷酸三钙与生物活性玻璃结合,生物活性玻璃充当烧结助剂,增加材料密合度和机械强度[14]。 生理性骨组织除主要组成成分羟基磷灰石外,还包含有许多离子,比如碳酸根离子、钠离子、镁离子、锌离子等。为了充分模拟生理骨组织中离子交换情况,近年来,有许多学者将各种离子与替代羟基磷灰石中的部分钙离子,使得离子能在羟基磷灰石支架中缓释,发挥其生理活性功能。例如,Si已被证实在骨组织矿化和基因激活中起重要作用。生物体内实验表明,含硅的羟基磷灰石颗粒成骨细胞的成骨情况显著优于纯羟基磷灰石[15]。 2.2.2 生物活性玻璃 生物活性玻璃在1970年被发现,Hench总结了它在诱导成骨方面的特点,它能够和活体组织紧密结合营造稳定的界面,还可触发一系列生物反应,在自身吸收降解的同时一道组织再生和血管 化[16]。在机体内,它通过在生物活性玻璃表面形成碳化羟基磷灰石层使植入物和骨组织形成良好的连接,与此同时,其释放的Si、Na、Ca、磷酸根离子能刺激细胞生成新的组织。 临床上最常用的生物活性玻璃为45S5 Bioglass®,其构成主要是Na2O-CaO-SiO2-P2O5。45S5 Bioglass®抗压强度为500 MPa,抗张强度为42 MPa,弹性模量为35 GPa,断裂韧性为0.5-1.0 MPa[7]。它已被证实具有促进成骨细胞黏附、分化和诱导间充质细胞分化为成骨细胞的能力[17]。不仅如此,研究者还发现它具有提高蛋白活性,系促进血管新生的作用[18]。近年来的研究发现,生物活性玻璃通过释放Si、Ca、P等离子促进成骨细胞的有丝分裂和一型胶原纤维的合成,此外,离子的聚集还能够上调某些成骨相关基因的表达[19]。 2.3 生物陶瓷作为骨组织工程支架的体外实验研究 很多实验已证实,间充质干细胞、成骨细胞等在羟基磷灰石、磷酸三钙支架、生物玻璃支架上都能保持良好的细胞活性,进行细胞的黏附聚集、增殖、分化等过程。此外,在生物陶瓷材料表面涂布生长因子、骨形成蛋白2、金属离子等也可促进生物陶瓷材料的成骨活性[20-22]。Rahman等[23]将MC3T3-E1细胞定植于多孔连接羟基磷灰石-陶瓷支架,并用成纤维细胞生长因子2和褪黑激素诱导,发现成纤维细胞生长因子2和褪黑激素增强了MC3T3-E1细胞的增殖,上调了OCN和OPN的表达,并且提高了碱性磷酸酶的活性。Chen等[24]将MG63细胞定植于多孔生物玻璃-陶瓷支架上,观测细胞功能及细胞增殖情况,细胞能够在渗透进支架中心生长,复合材料的晶体成分在细胞增殖过程中降解后,黏附的细胞及分泌的胶原修补晶体溶解过程中产生的表面微裂隙,使得支架的强度仍能够维持。在羟基磷灰石/阮藻酸盐复合支架上,人间充质干细胞成骨基因的表达较单纯阮藻酸盐更高,产生的基质及骨矿化数量也较单纯阮藻酸盐支架更高[25]。 此外,近年来的许多研究表明,用特定的离子取代羟基磷灰石中的部分离子,形成替代羟基磷灰石,能够更大程度模仿生理骨组织的组成[26-27]。离子取代对材料的性能、晶体结构、形态、溶解度等都有显著影响。有学者分别用Pb2+、Sr2+、Co2+、Zn2+、Fe2+、Cu2+取代羟基磷灰石中一个Ca离子,形成X羟基磷灰石(X为取代钙离子者),然后测定其细胞相容性,发现除Zn-羟基磷灰石外,所有替代羟基磷灰石都出现细胞凋亡增多的现象。其中Mg-羟基磷灰石、Co-羟基磷灰石的细胞凋亡最甚[28]。Mg-羟基磷灰石支架在新西兰大白兔股骨缺损的模型实验中表现出了良好的骨诱导活性和高材料降解活性,缺损股骨1个月内就可见编织骨形成,3个月后编织骨完全被成熟板层骨取代[29]。 2.4 生物陶瓷作为骨组织工程支架的的体内实验 除体外细胞研究,生物陶瓷材料结合3D打技术、激光烧结技术在动物体内实验中也表现出了良好的诱导骨组织再生及血管化的能力。Smeets等[30]运用选择性激光熔融技术制备聚丙交酯/β-磷酸三钙多孔径支架,经体外细胞实验证实其生物相容性和成骨活性后,用于标准大小颌骨及颅顶骨缺损的大鼠模型,实验组大鼠在30 d后出现新骨置换,其骨形成表现优于自体骨修复的对照组。Fernández等[31]用Ca-P生物材料修复大鼠颅顶骨缺损,发现Ca-P在初期以60 μm/d(0.8 mg/d)的速度显著加速编织骨的形成和矿化,组织学检测发现早期骨单位形成,提示早期纤维组织中的血管形成,见图1。Suba 等[32]在20例缺牙患者上比较了上颌窦提升术后分别植入自体骨粉和β-磷酸三钙后的骨形成情况,术后6个月双侧上颌窦底骨组织学检测结果显示,植入β-磷酸三钙的一侧的骨形成情况与对照侧无明显差异,新生骨密度与对照侧相当。β-磷酸三钙具有良好的骨诱导、骨结合和骨再生能力,可形成适合牙种植的骨床。 除了能够支持细胞的增殖分化外,生物陶瓷材料还能在成骨基因的表达、骨组织血管化中起到促进作用。Meagher等[33]将羟基磷灰石/胶原支架在小鼠动物模型中皮下异位植入,12周的观察显示,支架上的血管密度、基质沉积及矿化情况与支架中羟基磷灰石体积比有剂量依赖关系。 2.5 生物陶瓷支架在骨缺损重建的现有临床应用 生物陶瓷在骨组织工程中的各项研究都显示出了其在骨修复重建领域广阔的前景,与此同时,生物陶瓷也已部分运用于临床,为患者带来福音。在骨科中,Marcacci等[4]利用3D 打印技术,将患者体内分离的骨髓基质干细胞置于羟基磷灰石支架上,用以修复大面积胫骨缺损,在长达6.5年的随访中,赝复体与宿主骨组织结合良好,患者肢体功能大部分恢复。 生物陶瓷结合3D打印技术和计算机辅助设计在颅面部个性化定制缺损重建上也有许多运用。Li等[34]运用计算机辅助设计及快速成型技术,以纳米羟基磷灰石/聚酰胺为材料,对髁突切除术后面部外形不良的患者进行个性化下颌骨髁突修复,最终恢复面部外观和稳定的颞下颌关节功能。Engstrand等[35]运用β-磷酸三钙等生物陶瓷材料结合计算机辅助技术,制成8 mm×8 mm× 4 mm大小的方块,用钛网连接形成马赛克样式的植入体,用以修复颅颞骨大面积缺损,24个月随访显示颅骨缺损处新骨形成。利摩日大学医院神经外科及颌面外科学部以8例患者为临床研究对象,运用立体平板印刷技术,以羟基磷灰石为生物材料定制赝复体,修复颅面部复杂性骨缺损(缺损面积25 cm2),术后随访12个月未发现并发症,患者对外形效果满意[36-37]。在耳鼻咽喉科学方面,Huang等[38]对14例因鼓室硬化置换生物陶瓷人工听小骨的患者随访发现,术后3-24个月内语音频率及音听阈均值有明显改善。此外生物陶瓷还运用于眼眶的修复[39]。 2.6 生物活性陶瓷作为骨组织工程支架表面涂层的体内外实验及部分临床应用 生物活性陶瓷不仅可作为主体材料在细胞黏附增殖、骨组织形成、缺损重建中起到重要作用,还可通过等离子喷射、溶胶-凝胶法、电沉积法等形成表面涂层,改善其他材料的生物活性。许多研究表明,在钛植入物表面覆盖Ca-P涂层可改善植入物的生物功能[40-42]。Orsini等[43]比较了上颌骨后部的羟基磷灰石涂层钛种植体和单纯钛种植体,发现羟基磷灰石涂层的钛种植体平均骨-种植体接触大于对照组,证明种植体表面的涂层能够缩短种植体骨整合时间,有利于减小微小移动,缩短治疗时间。Johansson等[44]将羟基磷灰石纳米微粒涂层的聚醚支架植入12只兔股骨缺损模型中,发现骨-植入物的接触面面积大于无涂层的对照组面积,植入物螺纹处的骨修复也较对照组多,证明羟基磷灰石涂层能显著改善聚醚支架的成骨性能。 此外,羟基磷灰石用作金属支架涂层,可用于改善金属合金表面的抗腐蚀性[45],Zhang等[46]将Ca-P和BGCC/Ca-P分别涂层至纯镁支架上,分析其微结构、形态、矿化能力等发现,两种涂层都能显著加速骨诱导矿化的成核现象且BGCC/Ca-P涂层的镁支架降解速率比纯镁支架降低10倍。提示BGCC/Ca-P能抵抗镁金属表面的腐蚀,增强骨-支架表面的连接。钛表面的晶体羟基磷灰石涂层还能增加细胞的反应活性[47]。 临床上,羟基磷灰石还用作全髋关节置换体表面涂层,Capellpo等[48]在316例羟基磷灰石涂层全髋关节置换患者术后的临床随访显示,羟基磷灰石涂层的髋关节修复体促进了骨组织的内向生长和表面生长,并且不伴有骨质溶解现象。 "
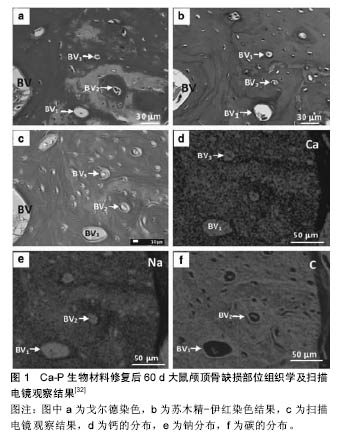
| [1]Harada N,Watanabe Y,Sato K,et al.Bone regeneration in a massive rat femur defect through endochondral ossification achieved with chondrogenically differentiated MSCs in a degradable scaffold. Biomaterials. 2014;35(27):7800-7810.[2]Bose S,Roy M,Bandyopadhyay A.Recent advances in bone tissue engineering scaffolds. Trends Biotechnol.2012;30(10): 546-554.[3]Boccaccini AR,Blaker JJ.Bioactive composite materials for tissue engineering scaffolds.Expert Rev Med Devices.2005; 2(3):303-317.[4]Marcacci M,Kon E,Moukhachev V,et al.Stem cells associated with macroporous bioceramics for long bone repair: 6- to 7-year outcome of a pilot clinical study.Tissue Eng.2007; 13(5):947-955.[5]Jensen SS,Yeo A,Dard M,et al.Evaluation of a novel biphasic calcium phosphate in standardized bone defects: a histologic and histomorphometric study in the mandibles of minipigs.Clin Oral Implants Res. 2007;18(6):752-760.[6]von Arx T,Cochran DL,Hermann JS,et al.Lateral ridge augmentation and implant placement: an experimental study evaluating implant osseointegration in different augmentation materials in the canine mandible.Int J Oral Maxillofac Implants. 2001;16(3):343-354.[7]Rezwan K,Chen QZ,Blaker JJ,et al.Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering.Biomaterials. 2006;27(18): 3413-3431.[8]Thavornyutikarn B,Chantarapanich N,Sitthiseripratip K,et al.Bone tissue engineering scaffolding: computer-aided scaffolding techniques.Prog Biomater.2014;3: 61-102.[9]Khalid P,Hussain MA,Rekha PD,et al.Carbon nanotube-reinforced hydroxyapatite composite and their interaction with human osteoblast in vitro.Hum Exp Toxicol.2015;34(5):548-556.[10]Lane JM.Bone graft substitutes.West J Med.1995;163(6): 565-566.[11]Gross JS.Bone grafting materials for dental applications: a practical guide.Compend Contin Educ Dent.1997; 18(10): 1013-1018,1020-1022,1024.[12]Woodard JR,Hilldore AJ,Lan SK,et al.The mechanical properties and osteoconductivity of hydroxyapatite bone scaffolds with multi-scale porosity.Biomaterials.2007; 28(1): 45-54.[13]Zhang K,Wang Y,Hillmyer MA,et al.Processing and properties of porous poly(L-lactide)/bioactive glass composites. Biomaterials.2004;25(13):2489-2500.[14]Bellucci D,Sola A,Cannillo V.Hydroxyapatite and tricalcium phosphate composites with bioactive glass as second phase: State of the art and current applications. J Biomed Mater Res A.2016; 104(4):1030-1056.[15]Fielding G,Bose S.SiO2 and ZnO dopants in three-dimensionally printed tricalcium phosphate bone tissue engineering scaffolds enhance osteogenesis and angiogenesis in vivo. Acta Biomater.2013; 9(11):9137-9148.[16]Jones JR.Review of bioactive glass: from Hench to hybrids. Acta Biomater.2013;9(1): 4457-4486.[17]Gatti AM,Valdre G,Andersson OH.Analysis of the in vivo reactions of a bioactive glass in soft and hard tissue. Biomaterials.1994;15(3):208-212.[18]Day RM,Boccaccini AR,Shurey S,et al.Assessment of polyglycolic acid mesh and bioactive glass for soft-tissue engineering scaffolds.Biomaterials. 2004;25(27): 5857-5866.[19]Xynos ID,Edgar AJ,Buttery LD,et al.Gene-expression profiling of human osteoblasts following treatment with the ionic products of Bioglass 45S5 dissolution.J Biomed Mater Res. 2001;55(2):151-157.[20]Wang H, Wu G,Zhang J,et al.Osteogenic effect of controlled released rhBMP-2 in 3D printed porous hydroxyapatite scaffold.Colloids Surf B Biointerfaces.2016;141:491-498.[21]An SH,Matsumoto T,Miyajima H,et al.Porous zirconia/hydroxyapatite scaffolds for bone reconstruction. Dent Mater.2012;28(12):1221-1231.[22]Nasseri F,Gholami GA,Kadkhodazadeh M.Effect of bioactive ceramic and recombinant human transforming growth factor-beta (rhTGF-beta) on regeneration of parietal bone defects in rat.J Long Term Eff Med Implants.2011;21(1): 71-78.[23]Rahman MZ,Shigeishi H,Sasaki K,et al.Combined effects of melatonin and FGF-2 on mouse preosteoblast behavior within interconnected porous hydroxyapatite ceramics - in vitro analysis.J Appl Oral Sci.2016;24(2):153-161. [24]Chen QZ,Efthymiou A,Salih V,et al.Bioglass-derived glass-ceramic scaffolds: study of cell proliferation and scaffold degradation in vitro.J Biomed Mater Res A.2008; 84(4):1049-1060.[25]Wang MO,Bracaglia L,Thompson JA,et al.Hydroxyapatite doped alginate beads as scaffolds for the osteoblastic differentiation of mesenchymal stem cells.J Biomed Mater Res A.2016; 104(9):2325-2333.[26]Ratnayake JT,Mucalo M,Dias GJ.Substituted hydroxyapatites for bone regeneration: A review of current trends. J Biomed Mater Res B Appl Biomater.2016.doi: 10.1002/jbm.b.33651. [Epub ahead of print][27]Gao J,Wang M,Shi C,et al.Synthesis of trace element Si and Sr codoping hydroxyapatite with non-cytotoxicity and enhanced cell proliferation and differentiation.Biol Trace Elem Res.2016;174(1):208-217. [28]de Lima IR,Alves GG,Soriano CA,et al.Understanding the impact of divalent cation substitution on hydroxyapatite: an in vitro multiparametric study on biocompatibility.J Biomed Mater Res A.2011;98(3):351-358.[29]Landi E,Logroscino G,Proietti L,et al.Biomimetic Mg-substituted hydroxyapatite: from synthesis to in vivo behaviour.J Mater Sci Mater Med.2008;19(1):239-247.[30]Smeets R,Barbeck M,Hanken H,et al.Selective laser-melted fully biodegradable scaffold composed of poly(d,l-lactide) and beta-tricalcium phosphate with potential as a biodegradable implant for complex maxillofacial reconstruction: In vitro and in vivo results.J Biomed Mater Res B Appl Biomater, 2016.doi: 10.1002/jbm.b.33660. [Epub ahead of print][31]Fernández T,Olave G,Valencia CH,et al.Effects of calcium phosphate/chitosan composite on bone healing in rats: calcium phosphate induces osteon formation.Tissue Eng Part A.2014;20(13-14):194819-194860.[32]Suba Z,Hrabák K,Huys L,et al.Histologic and histomorphometric study of bone regeneration induced by beta-tricalcium phosphate (multicenter study).Orv Hetil.2004; 145(27):1431-1437.[33]Meagher MJ,Weiss-Bilka HE,Best ME,et al.Acellular Hydroxyapatite-Collagen Scaffolds Support Angiogenesis and Osteogenic Gene Expression in an Ectopic Murine Model: Effects of Hydroxyapatite Volume Fraction.J Biomed Mater Res A.2016;104(9):2178-2188.[34]Li J,Hsu Y,Luo E,et al.Computer-aided design and manufacturing and rapid prototyped nanoscale hydroxyapatite/polyamide (n-HA/PA) construction for condylar defect caused by mandibular angle ostectomy. Aesthetic Plast Surg.2011;35(4):636-640.[35]Engstrand T,Kihlström L,Neovius E,et al.Development of a bioactive implant for repair and potential healing of cranial defects.J Neurosurg.2014;120(1):273-277.[36]Brie J,Chartier T,Chaput C,et al.A new custom made bioceramic implant for the repair of large and complex craniofacial bone defects.J Craniomaxillofac Surg.2013; 41(5):403-407.[37]Staffa G,Barbanera A,Faiola A,et al.Custom made bioceramic implants in complex and large cranial reconstruction: a two-year follow-up.J Craniomaxillofac Surg.2012;40(3): e65-70.[38]Huang ZY,Zhou FH,Xie NP,et al.Therapeutic effect of ossicular reconstruction with bioceramic or porous macromolecular polyethylene partial ossicular replacement prosthesis in patients with tympanosclerosis.Nan Fang Yi Ke Da Xue Xue Bao.2010;30(9):2181-2184.[39]Jordan DR,Klapper SR,Gilberg SM,et al.The bioceramic implant: evaluation of implant exposures in 419 implants. Ophthal Plast Reconstr Surg.2010;26(2):80-82.[40]Le Guehennec L,Soueidan A,Layrolle P,et al.Surface treatments of titanium dental implants for rapid osseointegration. Dent Mater.2007;23(7):844-854.[41]Yang GL,He FM,Hu JA,et al.Effects of biomimetically and electrochemically deposited nano-hydroxyapatite coatings on osseointegration of porous titanium implants.Oral Surg Oral Med Oral Pathol Oral Radiol Endod.2009;107(6):782-789.[42]Yang GL,He FM,Hu JA,et al.Biomechanical comparison of biomimetically and electrochemically deposited hydroxyapatite-coated porous titanium implants.J Oral Maxillofac Surg.2010;68(2):420-427.[43]Orsini G,Piattelli M,Scarano A,et al.Randomized, controlled histologic and histomorphometric evaluation of implants with nanometer-scale calcium phosphate added to the dual acid-etched surface in the human posterior maxilla.J Periodontol.2007;78(2):209-218.[44]Johansson P,Jimbo R,Naito Y,et al.Polyether ether ketone implants achieve increased bone fusion when coated with nano-sized hydroxyapatite: a histomorphometric study in rabbit bone.Int J Nanomedicine.2016;11: 1435-1442.[45]Say Y,Aksakal B.Effects of hydroxyapatite/Zr and bioglass/Zr coatings on morphology and corrosion behaviour of Rex-734 alloy.J Mater Sci Mater Med.2016;27(6):105.[46]Zhang X,Li XW,Li JG,et al.Preparation and characterizations of bioglass ceramic cement/Ca-P coating on pure magnesium for biomedical applications.ACS Appl Mater Interfaces.2014; 6(1):513-525.[47]Nasiri N,Ceramidas A,Mukherjee S,et al.Ultra-Porous Nanoparticle Networks: A Biomimetic Coating Morphology for Enhanced Cellular Response and Infiltration.Sci Rep.2016;6: 24305.[48]Capello WN,D'Antonio JA,Manley MT,et al.Hydroxyapatite in total hip arthroplasty. Clinical results and critical issues.Clin Orthop Relat Res.1998;(355):200-211.[49]Choi JW,Kim N.Clinical application of three-dimensional printing technology in craniofacial plastic surgery.Arch Plast Surg.2015;42(3):267-277.[50]Chia HN,Wu BW.Recent advances in 3D printing of biomaterials.J Biol Eng.2015;9:4.[51]Ciocca L,De Crescenzio F,Fantini M,et al.CAD/CAM and rapid prototyped scaffold construction for bone regenerative medicine and surgical transfer of virtual planning: a pilot study.Comput Med Imaging Graph.2009;33(1):58-62.[52]Liu A,Xue GH,Sun M,et al.3D Printing Surgical Implants at the clinic: A Experimental Study on Anterior Cruciate Ligament Reconstruction.Sci Rep.2016;6:21704.[53]Grayson WL,Fröhlich M,Yeager K,et al.Engineering anatomically shaped human bone grafts.Proc Natl Acad Sci U S A.2010;107(8):3299-3304.[54]Tarafder S,Davies NM,Bandyopadhyay A,et al.3D printed tricalcium phosphate scaffolds: Effect of SrO and MgO doping on osteogenesis in a rat distal femoral defect model.Biomater Sci.2013;1(12):1250-1259.[55]Inzana JA,Olvera D,Fuller SM,et al.3D printing of composite calcium phosphate and collagen scaffolds for bone regeneration. Biomaterials.2014;35(13):4026-4034.[56]Tarafder S,Bose S.Polycaprolactone-coated 3D printed tricalcium phosphate scaffolds for bone tissue engineering: in vitro alendronate release behavior and local delivery effect on in vivo osteogenesis. ACS Appl Mater Interfaces.2014;6(13): 9955-9965. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [3] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [4] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [5] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [6] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [7] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [8] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [9] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [10] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [11] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [12] | Guan Jian, Jia Yanfei, Zhang Baoxin , Zhao Guozhong. Application of 4D bioprinting in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 446-455. |
| [13] | Huang Bo, Chen Mingxue, Peng Liqing, Luo Xujiang, Li Huo, Wang Hao, Tian Qinyu, Lu Xiaobo, Liu Shuyun, Guo Quanyi . Fabrication and biocompatibility of injectable gelatin-methacryloyl/cartilage-derived matrix particles composite hydrogel scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2600-2606. |
| [14] | Liu Jiali, Suo Hairui, Yang Han, Wang Ling, Xu Mingen. Influence of lay-down angles on mechanical properties of three-dimensional printed polycaprolactone scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2612-2617. |
| [15] | Li Xuan, Sun Yimin, Li Longbiao, Wang Zhenming, Yang Jing, Wang Chenglin, Ye Ling. Manufacturing of nano-modified polycaprolactone microspheres and its biological effects in dental pulp cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1530-1536. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||