Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (4): 637-642.doi: 10.12307/2022.104
Previous Articles Next Articles
Electrospinning for rotator cuff repair
Gao Cangjian1, 2, Yang Zhen1, 2, Liu Shuyun1, Li Hao1, 2, Fu Liwei1, 2, Zhao Tianyuan1, 2, Chen Wei1, 2, Liao Zhiyao1, 2, Li Pinxue1, 2, Sui Xiang1, Guo Quanyi1#br#
- 1Institute of Orthopedics, the First Medical Center, Chinese PLA General Hospital, Beijing Key Laboratory of Regenerative Medicine in Orthopedics, Key Laboratory of Musculoskeletal Trauma & War Injuries, PLA, Beijing 100853, China; 2Medical College of Nankai University, Tianjin 300071, China
-
Received:2020-08-26Revised:2020-08-28Accepted:2020-09-26Online:2022-02-08Published:2021-12-06 -
Contact:Guo Quanyi, Institute of Orthopedics, the First Medical Center, Chinese PLA General Hospital, Beijing Key Laboratory of Regenerative Medicine in Orthopedics, Key Laboratory of Musculoskeletal Trauma & War Injuries, PLA, Beijing 100853, China -
About author:Gao Cangjian, Master candidate, Institute of Orthopedics, the First Medical Center, Chinese PLA General Hospital, Beijing Key Laboratory of Regenerative Medicine in Orthopedics, Key Laboratory of Musculoskeletal Trauma & War Injuries, PLA, Beijing 100853, China; Medical College of Nankai University, Tianjin 300071, China -
Supported by:the National Key Research and Development Plan Project, No. 2019YFA0110600 (to GQY)
CLC Number:
Cite this article
Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
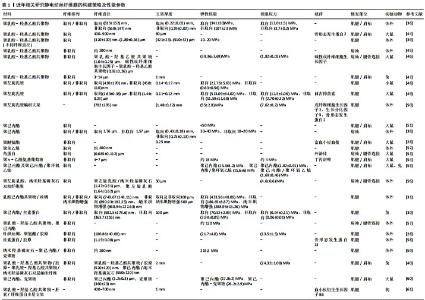
2.1 材料选择 合成材料与天然材料均被广泛应用于静电纺丝技术与组织工程修复,在选择静电纺丝材料时往往需要考虑材料的亲水性、纤维直径、机械性能、生物降解性等。在合成材料中,聚乳酸、聚乙醇酸、聚乳酸-羟基乙酸共聚物、聚己内酯由于其优秀的生物相容性和可降解性,是静电纺丝肌腱再生最常用的聚合物[1,15]。其中聚乙醇酸为亲水材料,有益于细胞黏附及增殖,韧性最高,但其降解时间过快(4-6周)[21-23],肌腱胶原纤维的重建常于损伤后6-8周后开始[20],单纯以聚乙醇酸材料修复肌腱或肩袖损伤显然不是最好的选择。对于聚乳酸和聚乙醇酸来说,可以通过改变浓度及结晶度来调节共聚物的性能,从而影响聚合物的力学性能和降解时间[24],相比于聚乳酸和聚乙醇酸,聚己内酯及聚乳酸-羟基乙酸共聚物的降解时间更慢,但疏水性较高[23,25]。TAYLOR等[26]对电纺丝纳米纤维再生肩袖肌腱进行了体内外相关研究,发现聚乳酸-羟基乙酸共聚物电纺纳米纤维在体外可促进细胞增殖,支架植入体内8周后的生物力学测试结果显示修复效果优于对照组。 除了合成聚合物,天然聚合物如丝素蛋白、胶原蛋白和壳聚糖也被用于研究肌腱组织再生,然而它们都有比较相似的缺点:较低的机械性能、较差的加工性能和低效率的可再生性[27]。为了克服这些缺陷,研究常将天然材料与合成材料混合,构建的静电纺丝材料既具有良好的生物相容性又兼备优良的生物力学性能[28-34]。高超等[34]将聚己内酯与Ⅰ型胶原混纺制备出含25%Ⅰ型胶原含量的聚己内酯/Ⅰ型胶原电纺纤维膜,将兔肌腱干细胞与电纺纤维膜共培养3 d后发现,相比于纯聚己内酯的对照组,聚己内酯/Ⅰ型胶原电纺纤维膜具有更好的细胞黏附效果和细胞活性。 2.2 纳米纤维的取向性 肌腱结构是由排列整齐的纤维束形成的,胶原纤维束的直径为20 nm,肌腱纤维束的直径为3 mm[15],如何使静电纺丝制成的纳米纤维从直径和形态上模仿肌腱的显微结构,是研究者应当首先考虑的问题。肩袖损伤难以自愈且无法恢复至原初状态,其主要原因是肌腱自愈过程中形成的瘢痕组织中胶原纤维组织结构紊乱,这也是造成愈合肌腱力学性能差的主要原因[35-36]。取向电纺丝纳米纤维具有较高的抗拉强度和拉应力,可以模仿肌腱组织中天然胶原纤维的结构和功能;非取向电纺丝纳米纤维比定向电纺丝纳米纤维具有更高的延展性,可以模仿腱骨处靠近骨侧的细胞外基质[31,36-37]。 研究人员就如何取舍取向结构与非取向结构展开了一系列的研究。MOFFAT等[36]对电纺纳米纤维的趋取向性与肩袖再生关系进行了探究,分别将人肩袖组织成纤维细胞接种于取向和非取向的聚乳酸-羟基乙酸共聚物静电纺丝纤维膜,取向电纺丝纳米纤维膜的力学性能高于非取向纤维膜,细胞黏附对纤维膜的弹性模量无影响,但降低了材料的极限拉应力和屈服强度;虽然纤维的取向性与细胞生长和增殖之间未见显著差异,但取向纤维相比于非取向纤维增加了基质沉积、改善了接种细胞和胶原基质方向的一致性、提高了相关基因的表达。ORR等[38]在体外研究了多层排列纳米纤维基质对肩袖再生的影响,将取向支架与人类脂肪来源干细胞结合,发现腱调节蛋白和胶原的水平增加,与随机取向的支架相比,取向结构支架的力学性能有显著提高。ERISKEN等[39]制备了取向的聚乳酸-羟基乙酸共聚物静电纺丝纤维膜修复肩袖损伤,支架的弹性模量为(421±23)-(510±32) MPa,抗拉强度为10-15 MPa。Chen及其研究团队[30]制备了取向及非取向的两种聚己内酯/丝素蛋白静电纺丝纤维膜并应用于肌腱组织工程,分别对其弹性模量进行测定,非取向电纺纤维膜的弹性模量为(3.93±0.05) MPa,取向电纺纤维膜为(70.52±2.83) MPa,二者具有显著差异。其他相关研究制备的取向静电纺丝支架弹性模量均在50-300 MPa之间,普遍较非取向纳米纤维膜的弹性模量高。综上所述,如何选择纤维排列的取向与否需要进行多方面的考虑:如纤维排列的显微结构、宏观的力学性质等。 2.3 改性与载药 由于单一性质的纳米纤维支架无法模仿肩袖复杂的解剖结构,静电纺丝过程中在电纺溶液中混入少量其他物质(如纳米羟基磷灰石、透明质酸、金属氧化物等)可改变电纺纤维膜的性质。这些物质不能单独电纺,混合电纺后被纳米纤维包裹,可以改变支架的原有性能,并赋予支架特殊性质,如促进细胞增殖、分化,提升力学性能,抗粘连等。一些研究将电纺纤维进行矿物梯度基质改性,并进行了相关研究[26]。SUN等[40]制备了用于兔肩袖大面积撕裂愈合的双纳米纤维基质,其目标是开发一种理想的基质,能够同时支持肌腱和骨组织再生,修复大面积的肩袖撕裂;将纳米羟基磷灰石混入电纺溶液中,构建了聚羟基乙酸/聚己内酯-胶原/纳米羟基磷灰石的共电纺双纳米纤维,将聚己内酯/纳米羟基磷灰石部分复合骨组织,将聚乳酸-羟基乙酸共聚物/胶原部分包裹在损伤腱残端并连接腱与骨之间的断端。研究发现,聚乳酸-羟基乙酸共聚物/胶原-聚己内酯/纳米羟基磷灰石可以支持细胞生长、提高生存能力,以及促进胶原分泌和成骨细胞矿化;12周后,在基质中可见肌腱样组织和新骨形成,构建的双相纳米纤维基质具有良好生物相容性和生物力学性能,为肩袖组织工程原位再生提供了可靠的思路。LIU等[41]将透明质酸加入到聚己内酯支架中,发现随着透明质酸浓度的增加,支架的抗拉强度下降(透明质酸百分比从0%增加到12%时,抗拉强度相应从 2.13 MPa下降到1.55 MPa),相反,拉伸模量随着透明质酸百分比的增加而增加;体外接种细胞共培养3 d显示,多能性小鼠干细胞附着在聚己内酯/透明质酸支架上,并随着透明质酸含量的增加促增殖作用明显;鸡跟腱断裂修复实验表明,与聚己内酯支架相比,12%聚己内酯/透明质酸支架的修复效果最好,创面无组织粘连,肌腱愈合评分最高[41]。CHAINANI等[42]利用机械、化学等方法制备猪肌腱细胞外基质,并将其涂在聚己内酯电纺支架表面,以没有细胞外基质涂层的聚己内酯作为对照组,体外接种培养人脂肪干细胞28 d后,细胞增殖、细胞外基质(主要为Ⅰ型胶原)分泌和相关成腱基因表达均高于对照组,接种细胞后支架的屈服强度也有所提高。纳米纤维改性也可用于辅助体内研究。MARTIN等[43]介绍了一种制造植入后放射成像可见的支架,将氧化锆纳米颗粒分子加入电纺丝溶液中制得的纳米纤维膜,在体外小鼠椎间盘和体外牛半月板撕裂模型中具有良好的生物相容性,具体表现为纤维组织覆盖、无钙化沉积。重要的是,使用微型计算机断层扫描可以使植入体内的支架成像,监测组织的愈合、支架的位移和与周围组织的碰撞,并且在需要翻修时可以更容易地定位支架。近年相关研究静电纺丝纤维膜的构建策略及性能参数,见表1。 静电纺丝纳米纤维还是良好的药物载体,由于电纺材料的可降解性,被包裹在纳米纤维中的药物随着材料的降解而逐渐释放,达到缓释药物的效果。纳米纤维的药物释放通过从表面解吸、气孔扩散、基质降解等多种因素,这些药物释放过程都可能受到材料(聚合物或其他材料)的选择、纳米纤维的孔隙度、形态和几何形状的影响[62]。一些研究集中在促进和调节细胞行为的替代方法上,如使用小分子对纳米纤维进行修饰。ZHANG等[49]使用曲古抑菌素A改善干细胞的成腱分化能力,负载曲古抑菌素A的电纺丝纳米纤维可提高修复肌腱的生物力学性能。ZHAO等[46]将碱性成纤维细胞生长因子加入到纳米纤维中,在最初的48 h内碱性成纤维细胞生长因子的突释率为15.4%,相比于无因子对照组,试验组体外接种人成纤维细胞增殖更快,并且在接种于非生物活性对照支架上的细胞上保持了成纤维细胞表型。LUI等[63]将不同比例的聚左旋乳酸和聚己内酯溶解在六氟异丙醇中,并将萘普生混入电纺溶液中,制备了搭载萘普生的纳米纤维支架,萘普生是一种防止肌腱瘢痕组织产生、抗肌腱组织粘连的药物;他们发现电纺膜中聚左旋乳酸与聚己内酯的比例和萘普生释放率呈负相关,且药物释放可以持续长达3周;体外接种小鼠细胞系表明,萘普生对细胞存活率或细胞形态没有影响。"

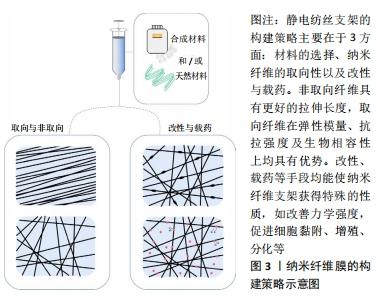
2.4 腱-骨界面的仿生 如前所述,肩袖损伤修复过程中最难以实现的是腱-骨界面的再生,质地均匀、单一的静电纺丝支架显然无法实现腱骨连接处过渡组织的结构仿生。组织工程支架对腱-骨界面的仿生策略主要包括两种:分层支架,即在腱、腱骨及骨区域分别采用适宜的构造方式,并将3部分结合在一起;梯度支架,即支架的结构由腱到骨呈现出渐变的趋势,模拟腱骨区域过渡的组织结构[4,20]。Olvera及其团队[64]应用分层支架策略成功制备修复韧带-骨界面缺损的多相支架:在取向聚己内酯静电纺丝纤维膜的基础上,分别在韧带区域及骨区域以共价结合的方式附着一层韧带细胞外基质与软骨细胞外基质,并在软骨细胞外基质包覆纤维的远端区域添加羟基磷灰石层,以促进软骨内成骨,体内实验结果表明,当修复如骨-韧带界面时,组织特异性细胞外基质的衍生成分可作为指导间充质干细胞分化的线索。KISHAN等[65]通过双喷头混合静电纺丝,利用两种不同硬度的聚氨酯脲分别制备了取向纤维和非取向纤维的组分梯度支架,使用荧光染色验证了支架中两种成分含量的过渡结构,拉伸实验证实了电纺膜力学性能随纤维排列的变化呈现梯度改变,体外实验显示梯度的纤维排列诱导了干细胞的相应排列。 2.5 传统静电纺丝的限制与改进 如图3所示。"
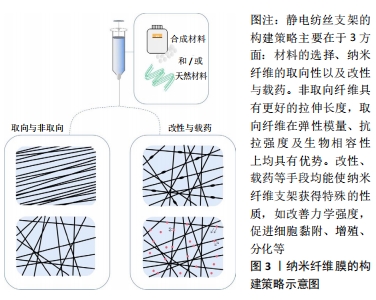

静电纺丝支架的构建策略主要在于3方面:材料的选择、纳米纤维的取向性及改性与载药,尽管不同研究构建策略不同,它们都围绕着同一根本目标而进行——模拟肩袖的组织力学结构与细胞生长微环境。虽然静电纺丝技术于肩袖组织工程修复领域已展现巨大潜力,但依旧存有不足之处:静电纺丝制得的纳米纤维膜厚度均在1.5 mm以下,在厚度上无法满足正常肌腱的需求,在修复肌腱断裂类损伤时需要后续处理,设计出三维立体结构[66];静电纺丝纤维膜虽然具有良好的比表面积与合适的纤维直径,但其纤维结构过于致密,细胞只能生长于支架的表面,而不能进入支架[67];静电纺丝材料大部分为可降解材料,支架移植体内后随时间逐步降解,对支架的机械性能产生影响,使其弹性模量、抗拉强度等显著降低,探索材料随时间降解的关系至关重要,降解速率须与组织再生和成熟的速度相匹配,以确保整体修复结构的机械强度在整个组织愈合周期均足够强大,以防止再次断裂的发生[17];静电纺丝载药的相关研究是近年热点话题,静电纺丝使用的有机溶剂是否会降低或破坏药物的活性,相关问题尚待研究,以及静电纺丝载药时药物突释的问题需要更好的解决办法[68]。 近年来,多喷头静电纺丝相关研究的兴起可能解决以上所述的不足,其与单纯混合多种材料电纺的区别在于:多喷头静电纺丝支架具有多种纳米纤维结构,多组分纳米纤维比传统的纳米纤维具有更大的优势,因为多组分纳米纤维具有多种组分组合而来的新特性,避免了单一组分纳米纤维由于生物降解而导致的力学性能突发性降低[60]。同轴静电纺丝又称核-壳电纺,作为多组分纳米纤维制备手段的一种,它可以使一种材料的纳米纤维被另一种材料包裹,呈现出“内核-外壳”状结构,不仅具有独特的机械性能,其纳米纤维外层结构所构成的保护套也有效防止了载药时药物的突释[68]。SHEN等[69]对同轴电纺在肩袖组织工程中的应用进行了初步探索,通过同轴静电纺丝在聚乳酸-羟基乙酸共聚物纤维表面包裹一层碱性壳聚糖纤维,用以中和聚乳酸-羟基乙酸共聚物在生物降解过程中的酸性产物,避免了修复过程中因环境过酸而导致的无菌性炎症的发生。体外模拟酸性环境中培养人体真皮成纤维细胞时,壳聚糖介导的酸性中和作用可在体外显著减少炎症因子的分泌并下调相关炎症基因的表达;体外生物相容性评价表明,壳聚糖/聚乳酸-羟基乙酸共聚物纤维的细胞黏附能力较聚乳酸-羟基乙酸共聚物纤维差,但细胞相容性较高,有促进细胞迁移和胶原分泌的能力。"

| [1] ZHAO S, SU W, SHAH V, et al. Biomaterials based strategies for rotator cuff repair. Colloids Surf B Biointerfaces. 2017;157:407-416. [2] LONGO UG, BERTON A, PAPAPIETRO N, et al. Epidemiology, genetics and biological factors of rotator cuff tears. Med Sport Sci. 2012;57:1-9. [3] TELLADO SF, BALMAYOR ER, VAN GRIENSVEN M. Strategies to engineer tendon/ligament-to-bone interface: Biomaterials, cells and growth factors. Adv Drug Deliv Rev. 2015;94:126-140. [4] SMITH L, XIA Y, GALATZ L M, et al. Tissue-engineering strategies for the tendon/ligament-to-bone insertion. Connect Tissue Res. 2012;53(2):95-105. [5] CHO NS, RHEE YG. The factors affecting the clinical outcome and integrity of arthroscopically repaired rotator cuff tears of the shoulder. Clin Orthop Surg. 2009;1(2):96-104. [6] 冉北航,张芬,尹文芳.羽毛球运动中发生手腕和肩袖损伤的相关因素调查[J].中国临床康复,2006,10(44):41-43. [7] VITALE MA, VITALE MG, ZIVIN JG, et al. Rotator cuff repair: an analysis of utility scores and cost-effectiveness. J Shoulder Elbow Surg. 2007;16(2):181-187. [8] THOMOPOULOS S, WILLIAMS G, SOSLOWSKY L. Tendon to bone healing: differences in biomechanical, structural, and compositional properties due to a range of activity levels. J Biomech Eng. 2003;125(1):106-113. [9] GALATZ LM, BALL CM, TEEFEY SA, et al. The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J Bone Joint Surg Am. 2004;86(2):219-224. [10] NERI B R, CHAN K W, KWON Y W. Management of massive and irreparable rotator cuff tears. J Shoulder Elbow Surg. 2009;18(5):808-818. [11] 陈疾忤,陈世益, MURRELL GA.肩袖损伤的治疗进展[J].国外医学(骨科学分册),2004,25(2):92-94. [12] 赵晨,王蕾.组织工程修复肩袖损伤促进腱骨愈合的研究进展[J].中国骨与关节杂志,2015,4(11):868-872. [13] YEA JH, BAE TS, KIM BJ, et al. Regeneration of the rotator cuff tendon-to-bone interface using umbilical cord-derived mesenchymal stem cells and gradient extracellular matrix scaffolds from adipose tissue in a rat model. Acta Biomater. 2020;114:104-116. [14] JIANG X, WU S, KUSS M, et al. 3D printing of multilayered scaffolds for rotator cuff tendon regeneration. Bioact Mater. 2020;5(3):636-643. [15] WU Y, HAN Y, WONG YS, et al. Fibre‐based scaffolding techniques for tendon tissue engineering. J Tissue Eng Regen Med. 2018;12(7):1798-1821. [16] TAMAYOL A, AKBARI M, ANNABI N, et al. Fiber-based tissue engineering: Progress, challenges, and opportunities. Biotechnol Adv. 2013t;31(5):669-687. [17] LI WJ, LAURENCIN CT, CATERSON EJ, et al. Electrospun nanofibrous structure: a novel scaffold for tissue engineering. J Biomed Mater Res. 2002; 60(4):613-621. [18] BOGNITZKI M, CZADO W, FRESE T, et al. Nanostructured fibers via electrospinning. Adv Mater. 2001;13(1):70-72. [19] KIM JH, CHOI YJ, YI HG, et al. A cell-laden hybrid fiber/hydrogel composite for ligament regeneration with improved cell delivery and infiltration. Biomed Mater. 2017;12(5):055010. [20] SAVEH-SHEMSHAKI N, NAIR L S, LAURENCIN CT. Nanofiber-based matrices for rotator cuff regenerative engineering. Acta biomater. 2019;94:64-81. [21] CZAPLEWSKI SK, TSAI TL, DUENWALD-KUEHL SE, et al. Tenogenic differentiation of human induced pluripotent stem cell-derived mesenchymal stem cells dictated by properties of braided submicron fibrous scaffolds. Biomaterials. 2014;35(25):6907-6917. [22] AGHDAM RM, NAJARIAN S, SHAKHESI S, et al. Investigating the effect of PGA on physical and mechanical properties of electrospun PCL/PGA blend nanofibers. J Appl Polym Sci. 2012;124(1):123-131. [23] ZAPLOTNIK R, VESEL A, PRIMC G, et al. Rapid hydrophilization of model polyurethane/urea (PURPEG) polymer scaffolds using oxygen plasma treatment. Polymers. 2016;8(4):144. [24] GENTILE P, CHIONO V, CARMAGNOLA I, et al. An overview of poly (lactic-co-glycolic) acid (PLGA)-based biomaterials for bone tissue engineering. Int J Mol Sci. 2014;15(3):3640-3659. [25] WOODRUFF MA, HUTMACHER DW. The return of a forgotten polymer—Polycaprolactone in the 21st century. Prog Polym Sci. 2010;35(10):1217-1256. [26] TAYLOR ED, NAIR LS, NUKAVARAPU SP, et al. Novel nanostructured scaffolds as therapeutic replacement options for rotator cuff disease. J Bone Joint Surg Am. 2010; 92(Suppl 2):170-179. [27] YAMANE S, IWASAKI N, MAJIMA T, et al. Feasibility of chitosan-based hyaluronic acid hybrid biomaterial for a novel scaffold in cartilage tissue engineering. Biomaterials. 2005;26(6):611-619. [28] SHARIFI-AGHDAM M, FARIDI-MAJIDI R, DERAKHSHAN MA, et al. Preparation of collagen/polyurethane/knitted silk as a composite scaffold for tendon tissue engineering. Proc Inst Mech Eng H. 2017;231(7):652-662. [29] CHILDERS EP, DREGER NZ, ELLENBERGER AB, et al. Enhanced rotator-cuff repair using platelet-rich plasma adsorbed on branched poly (ester urea) s. Biomacromolecules. 2018;19(7):3129-3139. [30] CHEN CH, CHEN SH, KUO CY, et al. Response of dermal fibroblasts to biochemical and physical cues in aligned polycaprolactone/silk fibroin nanofiber scaffolds for application in tendon tissue engineering. Nanomaterials. 2017;7(8):219. [31] YANG C, DENG G, CHEN W, et al. A novel electrospun-aligned nanoyarn-reinforced nanofibrous scaffold for tendon tissue engineering. Colloids Surf B Biointerfaces. 2014;122:270-276. [32] YANG G, LIN H, ROTHRAUFF BB, et al. Multilayered polycaprolactone/gelatin fiber-hydrogel composite for tendon tissue engineering. Acta Biomater. 2016;35:68-76. [33] 郭岗岗,庞亚博,杨建华,等.采用静电纺丝技术制备丝素纤维蛋白/聚已内酯临时肩袖补片[J].中国组织工程研究,2018,22(34):5501-5509. [34] 高超,李超明,徐玉龙,等.静电纺丝聚己内酯/Ⅰ型胶原纳米纤维取向性补片用于肩袖修复[J].中国修复重建外科杂志,2019,33(5):628-633. [35] KOVACEVIC D, RODEO SA. Biological augmentation of rotator cuff tendon repair. Clin Orthop Relat Res. 2008;466(3):622-633. [36] MOFFAT KL, KWEI ASP, SPALAZZI JP, et al. Novel nanofiber-based scaffold for rotator cuff repair and augmentation. Tissue Eng Part A. 2009;15(1):115-126. [37] RICOTTI L, POLINI A, GENCHI GG, et al. Proliferation and skeletal myotube formation capability of C2C12 and H9c2 cells on isotropic and anisotropic electrospun nanofibrous PHB scaffolds. Biomed Mater. 2012;7(3):035010. [38] ORR SB, CHAINANI A, HIPPENSTEEL KJ, et al. Aligned multilayered electrospun scaffolds for rotator cuff tendon tissue engineering. Acta Biomater. 2015;24:117-126. [39] ERISKEN C, ZHANG X, MOFFAT KL, et al. Scaffold fiber diameter regulates human tendon fibroblast growth and differentiation. Tissue Eng Part A. 2013;19(3-4):519-528. [40] SUN Y, HAN F, ZHANG P, et al. A synthetic bridging patch of modified co-electrospun dual nano-scaffolds for massive rotator cuff tear. J Mater Chem B. 2016;4(45):7259-7269. [41] LIU S, ZHAO J, RUAN H, et al. Biomimetic sheath membrane via electrospinning for antiadhesion of repaired tendon. Biomacromolecules. 2012;13(11):3611-3619. [42] CHAINANI A, HIPPENSTEEL KJ, KISHAN A, et al. Multilayered electrospun scaffolds for tendon tissue engineering. Tissue Eng Part A. 2013;19(23-24): 2594-2604. [43] MARTIN JT, MILBY AH, IKUTA K, et al. A radiopaque electrospun scaffold for engineering fibrous musculoskeletal tissues: Scaffold characterization and in vivo applications. Acta biomater. 2015;26:97-104. [44] LIPNER J, SHEN H, CAVINATTO L, et al. In vivo evaluation of adipose-derived stromal cells delivered with a nanofiber scaffold for tendon-to-bone repair. Tissue Eng Part A. 2015;21(21-22):2766-2774. [45] LIU W, LIPNER J, XIE J, et al. Nanofiber scaffolds with gradients in mineral content for spatial control of osteogenesis. ACS Appl Mater Interfaces. 2014; 6(4):2842-2849. [46] ZHAO S, ZHAO J, DONG S, et al. Biological augmentation of rotator cuff repair using bFGF-loaded electrospun poly (lactide-co-glycolide) fibrous membranes. Int J Nanomedicine. 2014;92:373. [47] INUI A, KOKUBU T, MIFUNE Y, et al. Regeneration of rotator cuff tear using electrospun poly (d, l-lactide-co-glycolide) scaffolds in a rabbit model. Arthroscopy. 2012;28(12):1790-1799. [48] YIN Z, CHEN X, CHEN JL, et al. The regulation of tendon stem cell differentiation by the alignment of nanofibers. Biomaterials. 2010;31(8):2163-2175. [49] ZHANG C, WANG X, ZHANG E, et al. An epigenetic bioactive composite scaffold with well-aligned nanofibers for functional tendon tissue engineering. Acta biomater. 2018;66:141-156. [50] BARBER JG, HANDORF AM, ALLEE TJ, et al. Braided nanofibrous scaffold for tendon and ligament tissue engineering. Tissue Eng Part A. 2013;19(11-12): 1265-1274. [51] HUEGEL J, KIM DH, CIRONE JM, et al. Autologous tendon‐derived cell‐seeded nanofibrous scaffolds improve rotator cuff repair in an age‐dependent fashion. J Orthop Res. 2017;35(6):1250-1257. [52] POPIELARCZYK TL, NAIN AS, BARRETT JG. Aligned nanofiber topography directs the tenogenic differentiation of mesenchymal stem cells. Appl Sci. 2017;7(1):59. [53] SEVIVAS N, TEIXEIRA FG, PORTUGAL R, et al. Mesenchymal stem cell secretome improves tendon cell viability in vitro and tendon-bone healing in vivo when a tissue engineering strategy is used in a rat model of chronic massive rotator cuff tear. Am J Sports Med. 2018;46(2):449-459. [54] NUSS C, HUEGEL J, BOORMAN-PADGETT J, et al. Poly-N-acetyl glucosamine (sNAG) enhances early rotator cuff tendon healing in a rat model. Ann Biomed Eng. 2017;45(12):2826-2836. [55] BEASON DP, CONNIZZO BK, DOURTE LM, et al. Fiber-aligned polymer scaffolds for rotator cuff repair in a rat model. J Shoulder Elbow Surg. 2012;21(2):245-250. [56] LI X, CHENG R, SUN Z, et al. Flexible bipolar nanofibrous membranes for improving gradient microstructure in tendon-to-bone healing. Acta Biomater. 2017;61:204-216. [57] LI X, XIE J, LIPNER J, et al. Nanofiber scaffolds with gradations in mineral content for mimicking the tendon-to-bone insertion site. Nano Lett. 2009; 9(7):2763-2768. [58] MAGHDOURI-WHITE Y, PETROVA S, SORI N, et al. Electrospun silk–collagen scaffolds and BMP-13 for ligament and tendon repair and regeneration. Biomed Phys Eng Express. 2018;4(2):025013. [59] WU G, DENG X, SONG J, et al. Enhanced biological properties of biomimetic apatite fabricated polycaprolactone/chitosan nanofibrous bio-composite for tendon and ligament regeneration. J Photochem Photobiol B. 2018;178:27-32. [60] ZHAO S, ZHAO X, DONG S, et al. A hierarchical, stretchable and stiff fibrous biotemplate engineered using stagger-electrospinning for augmentation of rotator cuff tendon-healing. J Mater Chem B. 2015;3(6):990-1000. [61] MANNING C, SCHWARTZ A, LIU W, et al. Controlled delivery of mesenchymal stem cells and growth factors using a nanofiber scaffold for tendon repair. Acta Biomater. 2013;9(6):6905-6914. [62] GOONOO N, BHAW-LUXIMON A, JHURRY D. Drug loading and release from electrospun biodegradable nanofibers. J Biomed Nanotechnol. 2014;10(9): 2173-2199. [63] LUI YS, LEWIS MP, LOO SJ. Sustained‐release of naproxen sodium from electrospun‐aligned PLLA–PCL scaffolds. J Tissue Eng Regen Med. 2017; 11(4): 1011-1021. [64] OLVERA D, SATHY BN, KELLY DJ. Spatial Presentation of Tissue-Specific Extracellular Matrix Components along Electrospun Scaffolds for Tissue Engineering the Bone–Ligament Interface. ACS Biomater Sci Eng. 2020;6(9):5145-5161. [65] KISHAN A, ROBBINS A, MOHIUDDIN S, et al. Fabrication of macromolecular gradients in aligned fiber scaffolds using a combination of in-line blending and air-gap electrospinning. Acta Biomater. 2017;56:118-128. [66] BLAKENEY BA, TAMBRALLI A, ANDERSON JM, et al. Cell infiltration and growth in a low density, uncompressed three-dimensional electrospun nanofibrous scaffold. Biomaterials. 2011;32(6):1583-1590. [67] CHEN Y, SHAFIQ M, LIU M, et al. Advanced fabrication for electrospun three-dimensional nanofiber aerogels and scaffolds. Bioact Mater. 2020;5(4):963-979. [68] MONFARED M, TAGHIZADEH S, ZARE-HOSEINABADI A, et al. Emerging frontiers in drug release control by core–shell nanofibers: A review. Drug Metab Rev. 2019;51(4):589-611. [69] SHEN Y, TU T, YI B, et al. Electrospun acid-neutralizing fibers for the amelioration of inflammatory response. Acta Biomater. 2019;97:200-215. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Jiang Huanchang, Zhang Zhaofei, Liang De, Jiang Xiaobing, Yang Xiaodong, Liu Zhixiang. Comparison of advantages between unilateral multidirectional curved and straight vertebroplasty in the treatment of thoracolumbar osteoporotic vertebral compression fracture [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1407-1411. |
| [3] | Xue Yadong, Zhou Xinshe, Pei Lijia, Meng Fanyu, Li Jian, Wang Jinzi . Reconstruction of Paprosky III type acetabular defect by autogenous iliac bone block combined with titanium plate: providing a strong initial fixation for the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1424-1428. |
| [4] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [5] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [6] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [7] | Li Wei, Zhu Hanmin, Wang Xin, Gao Xue, Cui Jing, Liu Yuxin, Huang Shuming. Effect of Zuogui Wan on bone morphogenetic protein 2 signaling pathway in ovariectomized osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1173-1179. |
| [8] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [9] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [10] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [11] | Wu Bingshuang, Wang Zhi, Tang Yi, Tang Xiaoyu, Li Qi. Anterior cruciate ligament reconstruction: from enthesis to tendon-to-bone healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1293-1298. |
| [12] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [13] | Hu Wei, Xie Xingqi, Tu Guanjun. Exosomes derived from bone marrow mesenchymal stem cells improve the integrity of the blood-spinal cord barrier after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 992-998. |
| [14] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [15] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||