Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (16): 2492-2497.doi: 10.12307/2022.246
Previous Articles Next Articles
Influence of lay-down angles on mechanical properties of three-dimensional printed polycaprolactone scaffolds
Liu Jiali1, Suo Hairui1, 2, Yang Han1, Wang Ling1, 2, Xu Mingen1, 2
- 1College of Automation, Hangzhou Dianzi University, Hangzhou 310018, Zhejiang Province, China; 2Zhejiang Provincial Key Lab of Medical Information and Three-Dimensional Bio-Printing, Hangzhou 310018, Zhejiang Province, China
-
Received:2020-12-01Revised:2020-12-05Accepted:2021-01-23Online:2022-06-08Published:2021-10-29 -
Contact:Xu Mingen, Professor, College of Automation, Hangzhou Dianzi University, Hangzhou 310018, Zhejiang Province, China; Zhejiang Provincial Key Lab of Medical Information and Three-Dimensional Bio-Printing, Hangzhou 310018, Zhejiang Province, China -
About author:Liu Jiali, Master candidate, College of Automation, Hangzhou Dianzi University, Hangzhou 310018, Zhejiang Province, China -
Supported by:the National Natural Science Foundation of China, No. 61675059 (to WL); the National Key Research & Development Program of China, No. 2017YFC1103400 (to XME)
CLC Number:
Cite this article
Liu Jiali, Suo Hairui, Yang Han, Wang Ling, Xu Mingen. Influence of lay-down angles on mechanical properties of three-dimensional printed polycaprolactone scaffolds[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(16): 2492-2497.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

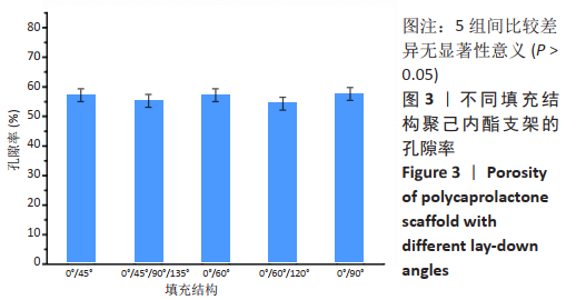
2.1 支聚己内酯架形貌观察 图2显示了0°/45°、0°/45°/90°/135°、0°/60°、0°/60°/120°和0°/90°五种填充结构的XY、YZ、XZ平面的聚己内酯支架形貌。从图中可以看出,不同填充结构的聚己内酯支架结构规则,细丝直径粗细均匀、间距相等,丝径约为344 μm,孔径约为379 μm,无闭孔。在XY平面,0°/45°组、0°/60°组、0°/90°组呈现规则的孔结构,0°/60°/120°组角度变换次数为3,构建了均匀规则的等边三角形孔结构,而0°/45°/90°/135°组则呈现出更小的孔;在XZ平面,0°/45°/90°/135°组孔结构较为明显,但分布不均匀,孔大小也不等,0°/45°组、0°/60°组、0°/60°/120°组呈现侧孔不明显,是由于旋转角度促使相邻层细丝方向相近,上层细丝遮挡了侧孔;在YZ平面,5种结构均存在明显的侧孔,0°/45°组、0°/60°组、0°/90°组侧孔多却小,而0°/60°/120°组、0°/45°/90°/135°组侧孔数量较少但是孔隙大,这是因为多次的角度变换致使相同方向的细丝间相隔层数增加,比如0°/90°组中两个0°方向的细丝中间相隔1层,0°/60°/120°组中两个0°方向的细丝相隔2层,而0°/45°/90°/135°组中两个0°方向的细丝相隔3层。"

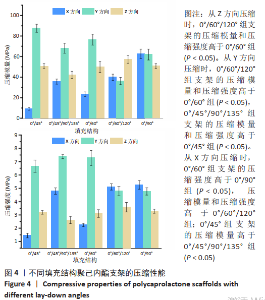
2.3 聚己内酯支架的力学性能 图4为不同填充结构聚己内酯支架的压缩性能。从Z方向压缩时,在拉力机的量程内,不同填充结构的支架呈现相同的趋势,即线弹性阶段、屈服阶段、塑性变形阶段。0°/45°组、0°/60°组、0°/90°组支架的压缩模量和压缩强度比较差异无显著性意义(P > 0.05),这是因为Z轴压缩时影响力学性能的主要因素为相邻层间的交点个数[21]、交点面积和孔隙率,填充结构对其影响很小;0°/60°/120°组支架的压缩模量和压缩强度较0°/60°组更强 (P < 0.05),这是因为0°/60°/120°组相邻3层的受力结点交于一点,且相邻层填充角度幅度减小,接触面积更大,形成的纵向柱状结构更粗[19];5种支架结构中,0°/45°/90°/135°组支架的压缩强度和压缩模量最小,这是因为该组以45°旋转4次,相邻4层的受力结点未集中到一点,不能形成纵向上的柱状结构,而其他4种支架结构相邻层受力结点交于一点,形成了受力方向的柱状结构[24]。"
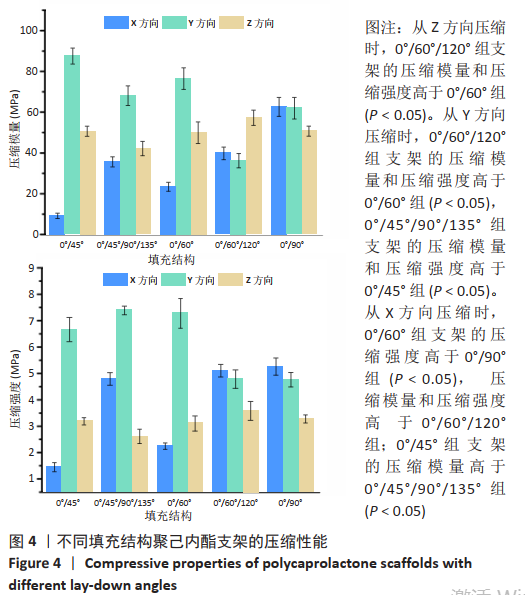
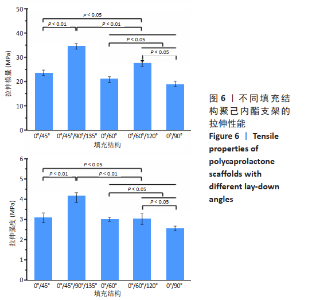
0°/45°组、0°/60°组、0/90°组支架的压缩模量和压缩强度随填充角度增加,力学性能增强,这是因为填充角度越大细丝越接近力的方向,当填充角度为90°时细丝方向与力方向一致,此时可以看作力施加在了圆柱结构上,不易变形,当填充角度逐渐减小,此时可看作力施加在了弹簧结构上,容易变形[18],其中0°/45°组支架变形最为明显;0°/60°/120°组支架的压缩模量是0°/60°组的1.71倍(P < 0.05),压缩强度是0°/60°组的2.25倍(P < 0.05),这是因为0°/60°/120°组形成的三角形结构具有更高的稳定性;0°/45°/90°/135°组支架的压缩模量和压缩强度较0/45°组更强(P < 0.05),这是因为0°/45°/90°/135°组在4次旋转中会形成沿力方向的细丝。 从X方向压缩时,0°/45°组、0°/60°组、0°/90°组支架的压缩模量随填充角度增加,力学性能较弱,这是因为X方向与Y方向呈现相反的关系,即在Y方向填充角度越大,那么在X方向填充角度越小,0°/45°组、0°/60°组支架的压缩强度比较差异无显著性意义(P > 0.05),变化较为平稳,0°/60°组支架的压缩强度比0°/90°组大2.51 MPa(P < 0.05);0°/60°/120°组支架的压缩模量和压缩强度较0°/60°组弱 (P < 0.05),原因是0°/60°组支架沿力的方向的细丝有10层,而0°/60°/120°组支架沿力的方向的细丝仅有7层(共20层),这些沿力方向的细丝构成了阻止变形的柱形主体。由于分别受到柱形主体和三角形结构的支撑,0°/60°/120°组支架在X方向和Y方向上的压缩性能较接近;0°/45°组支架的压缩模量较0°/45°/90°/135°组更大(P < 0.05),原因是0°/45°组支架沿力的方向的细丝有10层,而0°/45°/90°/135°组支架沿力的方向的细丝仅有5层,虽然0°/45°组支架的压缩强度较0°/45°/90°/135°组小,但差异无显著性意义(P > 0.05)。 从X、Y、Z方向压缩,0°/45°组、0°/45°/90°/135°组、0°/60°组支架的压缩强度和压缩模量各不相同,而0°/90°组、0°/60°/120°组的压缩力学性能相近。值得指出的是,0°/90°组在Z方向上的压缩模量和压缩强度比X、Y方向上的略小,这是因为从不同方向压缩0°/90°组时受力载荷传递机制不同,当从Z方向压缩,受力主体为层与层之间的重叠区域,当从X、Y方向压缩时受力主体为沿力的方向的细丝,刚性更强、变形更小。 图6为不同填充结构聚己内酯支架的拉伸性能。0°/45°组、0°/60°组、0°/90°组支架拉伸模量和拉伸强度随填充角度增加而降低,这是因为填充角度越小细丝的方向越接近于拉力的方向,当填充角度为0°时相邻两层细丝重合,相当于拉伸并行的圆柱。在5种填充结构的支架中,0°/45°/90°/135°组支架的拉伸模量(34.6±1.55) MPa和强度(4.16±0.37) MPa最大,而0°/90°组支架的拉伸模量(18.88±0.98) MPa和强度(2.56±0.23) MPa最小,0°/45°/90°/135°组支架拉伸模量是0°/90°组的1.83倍,拉伸强度是0°/90°组的1.37倍,这说明可以通过改变填充结构而改变支架的拉伸力学性能。"
| [1] JI WC, ZHANG XW, QIU YS. Selected suitable seed cell, scaffold and growth factor could maximize the repair effect using tissue engineering method in spinal cord injury. World J Exp Med. 2016;6(3): 58-62. [2] O’KEEFE RJ, MAO J. Bone tissue engineering and regeneration: from discovery to the clinic-an overview introduction. Tissue Eng Part B Rev. 2011;17(6):389-392. [3] 佘荣峰,张一,陈龙,等.丝素蛋白-壳聚糖支架复合骨髓间充质干细胞体内构建组织工程化软骨的生物相容性[J].中国组织工程研究,2020,24(1):27-32. [4] SUH JK, MATTHEW HW. Application of chitosan-based polysaccharide biomaterials in cartilage tissue engineering: a review. Biomaterials. 2000;21(24):2589-2598. [5] NG AMH, BIN SAIM A, TAN KK, et al. Comparison of bioengineered human bone construct from four sources of osteogenic cells. J Orthop Sci. 2005;10(2):192-199. [6] HSIEH CY, HSIEH HJ, LIU HC, et al. Fabrication and release behavior of a novel freeze-gelled chitosan/gamma-PGA scaffold as a carrier for rhBMP-2. Dent Mater. 2006;22(7):622-629. [7] LIU Y, NELSON T, CROMEENS B, et al. HB-EGF embedded in PGA/PLLA scaffolds via subcritical CO2 augments the production of tissue engineered intestine. Biomaterials.2016;103:150-159. [8] RAFIEI M, JOOYBAR E, ABDEKHODAIE MJ, et al. Construction of 3D fibrous PCL scaffolds by coaxial electrospinning for protein delivery. Mater Sci Eng C-Biomimetic Supramol Syst. 2020;113:110913. [9] 潘璐,程亭亭,徐岚.聚己内酯/聚乙二醇大孔径纳米纤维膜的制备及其在组织工程支架中的应用[J] . 纺织学报,2020,41(9): 167-173. [10] OH SH, PARK IK, KIM JM, et al. In vitro and in vivo characteristics of PCL scaffolds with pore size gradient fabricated by a centrifugation method. Biomaterials. 2007;28(9):1664-1671. [11] SIDDIQUI N, ASAWA S, BIRRU B, et al. PCL-Based composite scaffold matrices for tissue engineering applications. Mol Biotechnol. 2018; 60(7):506-532. [12] HWANG S, TODO M. Characterization of compressive deformation behavior of multi-layer porous composite materials for articular tissue engineering. J Mech Sci Technol. 2012;26(7):1999-2004. [13] DENG Y, JIANG C, LI C, et al. 3D printed scaffolds of calcium silicate-doped beta-TCP synergize with co-cultured endothelial and stromal cells to promote vascularization and bone formation. Sci Rep. 2017;7: 5588. [14] DIAO J, OUYANG J, DENG T, et al. 3D-Plotted beta-tricalcium phosphate scaffolds with smaller pore sizes improve in vivo bone regeneration and biomechanical properties in a critical-sized calvarial defect rat model. Adv Healthc Mater. 2018;7(17):1800441. [15] ROOHANI-ESFAHANI SI, NEWMAN P, ZREIQAT H. Design and fabrication of 3D printed scaffolds with a mechanical strength comparable to cortical bone to repair large bone defects. Sci Rep. 2016;6:19468. [16] SHAO H, YANG X, HE Y, et al. Bioactive glass-reinforced bioceramic ink writing scaffolds: sintering, microstructure and mechanical behavior. Biofabrication. 2015;7(3):035010. [17] ANG KC, LEONG KF, CHUA CK, et al. Investigation of the mechanical properties and porosity relationships in fused deposition modelling-fabricated porous structures. Rapid Prototyping J. 2006;12(2):100-105. [18] ZHANG B, GUO L, CHEN H, et al. Finite element evaluations of the mechanical properties of polycaprolactone/hydroxyapatite scaffolds by direct ink writing: Effects of pore geometry. J Mech Behav Biomed Mater. 2020;104:103665. [19] DOMINGOS M, INTRANUOVO F, RUSSO T, et al. The first systematic analysis of 3D rapid prototyped poly(epsilon-caprolactone) scaffolds manufactured through BioCell printing: the effect of pore size and geometry on compressive mechanical behaviour and in vitro hMSC viability. Biofabrication. 2013;5(4):045004. [20] KORPELA J, KOKKARI A, KORHONEN H, et al. Biodegradable and bioactive porous scaffold structures prepared using fused deposition modeling. J Biomed Mater Res B Appl Biomater. 2013;101B(4): 610-619. [21] 李相成,索海瑞,王玲,等.基于3D打印羟基磷灰石支架的填充结构与力学性能研究[J].中国生物医学工程学报,2020,39(1):91-96. [22] LEVINGSTONE TJ, MATSIKO A, DICKSON GR, et al. A biomimetic multi-layered collagen-based scaffold for osteochondral repair. Acta Biomater. 2014;10(5):1996-2004. [23] O’LEARY C, CAVANAGH B, UNGER RE, et al. The development of a tissue-engineered tracheobronchial epithelial model using a bilayered collagen-hyaluronate scaffold. Biomaterials. 2016;85:111-127. [24] GLEADALL A, VISSCHER D, YANG J, et al. Review of additive manufactured tissue engineering scaffolds: relationship between geometry and performance. Burns Trauma. 2018;6:19. [25] SHEPHERD DE, SEEDHOM BB. The ‘instantaneous’ compressive modulus of human articular cartilage in joints of the lower limb. Rheumatology (Oxford). 1999;38(2):124-132. [26] ATHANASIOU KA, AGARWAL A, DZIDA FJ. Comparative study of the intrinsic mechanical properties of the human acetabular and femoral head cartilage. J Orthop Res. 1994;12(3):340-349. [27] CHEN SS, FALCOVITZ YH, SCHNEIDERMAN R, et al. Depth-dependent compressive properties of normal aged human femoral head articular cartilage: relationship to fixed charge density. Osteoarthritis Cartilage. 2001;9(6):561-569. [28] KAPLAN SJ, HAYES WC, STONE JL, et al. Tensile strength of bovine trabecular bone. J Biomech. 1985;18(9):723-727. [29] WILLIAMS JL, LEWIS JL. Properties and an anisotropic model of cancellous bone from the proximal tibial epiphysis. J Biomech Eng. 1982;104(1):50-56. [30] WONG BL, SAH RL. Mechanical asymmetry during articulation of tibial and femoral cartilages: Local and overall compressive and shear deformation and properties. J Biomech Eng. 2010;43(9): 1689-1695. [31] AKIZUKI S, MOW VC, MULLER F, et al. Tensile properties of human knee joint cartilage: I. Influence of ionic conditions, weight bearing, and fibrillation on the tensile modulus. J Orthop Res. 1986;4(4): 379-392. [32] TELLIS BC, SZIVEK JA, BLISS CL, et al. Trabecular scaffolds created using micro CT guided fused deposition modeling. Mater. Sci. Eng. C-Biomimetic Supramol. Syst. 2008;28(1):171-178. [33] HUTMACHER DW, SCHANTZ T, ZEIN I, et al. Mechanical properties and cell cultural response of polycaprolactone scaffolds designed and fabricated via fused deposition modeling. J Biomed Mater Res. 2001;55(2):203-216. [34] ZHANG B, GUO L, CHEN H, et al. Finite element evaluations of the mechanical properties of polycaprolactone/hydroxyapatite scaffolds by direct ink writing: Effects of pore geometry. J Mech Behav Biomed Mater. 2020;104:103665. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Yu Chengxiang, Liu Lehong, Li Wenbo, Chen Jinshi, Ran Chunlei, Wang Zhongping. Correlation between spine-pelvic sagittal parameters and prognosis of vertebroplasty in the treatment of thoracolumbar osteoporotic vertebral compression fractures [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1412-1417. |
| [3] | Xu Xinzhong, Wu Zhonghan, Yu Shuisheng, Zhao Yao, Xu Chungui, Zhang Xin, Zheng Meige, Jing Juehua. Biomechanical analysis of different ways of inserting Steinmann Pins into the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1313-1317. |
| [4] | Gao Lei, Qin Xinyuan, Nie Xin, Wang Lei, Wang Jiangning. Extracorporeal circulation compression perfusion in the reconstruction of limb microcirculation from the mechanism of mechanical and chemical signal transduction [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1334-1340. |
| [5] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [6] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [7] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [8] | Wang Xinmin, Liu Fei, Xu Jie, Bai Yuxi, Lü Jian. Core decompression combined with dental pulp stem cells in the treatment of steroid-associated femoral head necrosis in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1074-1079. |
| [9] | Song Yuxin, Zhang Tongtong, Niu Jianxiong, Wang Zengping, Wen Jie, Zhang Qunli, Xue Wen, Liu Lin. Precise screw placement of 3D printing model and orthopedic robot in spinal deformity [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 904-907. |
| [10] | Peng Kun. Improvement of the treatment effect of osteoporotic fractures: research status and strategy analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 980-984. |
| [11] | Zhang Jianguo, Chen Chen, Hu Fengling, Huang Daoyu, Song Liang. Design and biomechanical properties of dental implant pore structure based on three-dimensional finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 585-590. |
| [12] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [13] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [14] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [15] | Wang Ruanbin, Cheng Liqian, Chen Kai. Application and value of polymer materials in three-dimensional printing biological bones and scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 610-616. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||