Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (23): 4320-4327.doi: 10.3969/j.issn.2095-4344.2013.23.020
Previous Articles Next Articles
Wharton’s jelly mesenchymal stem cells: Biological characteristics and therapeutic implications for cardiovascular diseases
Zhang Wei, Liu Xiao-cheng
- Department of Cardiovascular Surgery, TEDA International Cardiovascular Disease Hospital, Cardiovascular Disease Clinical College of Tianjin Medical University, Tianjin 300457, China
-
Online:2013-06-04Published:2013-06-04 -
Contact:Liu Xiao-cheng, Master, Professor, Doctoral supervisor, Chief physician, Department of Cardiovascular Surgery, TEDA International Cardiovascular Disease Hospital, Cardiovascular Clinical College of Tianjin Medical University, Tianjin 300457, China -
About author:Zhang Wei☆, Studying for doctorate, Associate chief physician, Department of Cardiovascular Surgery, TEDA International Cardiovascular Disease Hospital, Cardiovascular Disease Clinical College of Tianjin Medical University, Tianjin 300457, China zhangw@tedaich.com
CLC Number:
Cite this article
Zhang Wei, Liu Xiao-cheng. Wharton’s jelly mesenchymal stem cells: Biological characteristics and therapeutic implications for cardiovascular diseases[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(23): 4320-4327.
share this article
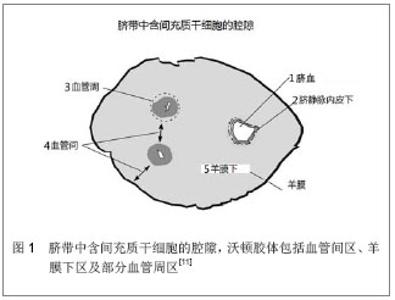
2.1 来源 目前已知人脐带中有4种间充质干细胞的来源。其一是沃顿胶体,其二是脐血管周,其三是脐带血,其四是脐静脉内皮下膜[30] ,见图1。其中,沃顿胶体是当前脐带源间充质干细胞的主要来源。原始的间充质干细胞在胚胎形成期间(在胚胎第10.5天前),当通过发育中的脐带向主动脉-性腺-中肾区第一次迁移和从胎盘再次通过早期脐带向胎儿肝脏第二次迁移时陷于结缔组织的基质中,并在妊娠期间始终存留于此,并最终构成沃顿胶体间充质干细胞[32] 。沃顿胶体间充质干细胞表达胚胎干细胞的标记物Tra-1-60、Tra-1-81、SSEA-1、SSEA-4、碱性磷酸酶甚至能在体外形成类胚体[33] 。此外,沃顿胶体间充质干细胞还能表达多潜能干细胞的标记物Oct-4、Sox-2和Nanog[18,28,34-35] ,但与胚胎干细胞相比水平相对较低[33,36] 。如此,沃顿胶体间充质干细胞不仅保持成体间充质干细胞的特性,而且与胚胎干细胞还具有相似性。 2.2 分离与体外培养 目前已有数种技术被用于沃顿胶体间充质干细胞的分离和原代培养。这些技术在一定程度上能影响所分离细胞的数量和质量。Salehinejad等[37]比较了4种分离细胞的方法:3种酶的方法;胶原酶-透明质酸酶-胰蛋白酶法,胶原酶-胰蛋白酶法和胰蛋白酶法,以及1种外植培养的方法。所有方法均能成功地分离出沃顿胶体间充质干细胞,胶原酶-胰蛋白酶法所分离的细胞密度高于其他组且多能干细胞标记(诸如C-kit和Oct-4)的表达比率更高,而外植细胞分离法所生成的细胞增殖速率和活性较其他方法更高。De Bruyn等[38]报道了一种不用酶解和解剖而仅基于间充质干细胞对塑料的黏附性而从沃顿胶体分离间充质干细胞的方法。此方法能从所有人脐带中分离出间充质干细胞,且第2代平均获得细胞1.4×108,而第3代则超过7×109。Nekanti等[39]发现DMEM-KO和DMEM-F12与其他支持沃顿胶体间充质干细胞在体外扩增的培养基相比更优。每条脐带 15 d内在DEMO-KO中增殖可超过1×108,且在培养基中加入碱性成纤维细胞生长因子不影响HLA-DR和CD44的表达水平[40] 。与常氧下培养的沃顿胶体间充质干细胞相比,在缺氧条件下沃顿胶体间充质干细胞的累积细胞群体倍增值与更短的细胞群体倍增时间表明其增殖能力更高[41] 。周建等[42]报道经50 Hz 3.6 mT的正弦交变电磁场处理亦能促进沃顿胶体间充质干细胞的增殖。 2.3 细胞形态与活性 在组织学上,沃顿胶体间充质干细胞为成纤维细胞样细胞,呈漩涡状生长,细胞核大而不规则,核仁明显。朱少芳等[43]报道在透射电镜下沃顿胶体间充质干细胞的细胞器丰富,核浆比例大,甚至可见多个核仁,表明细胞代谢活跃,分化程度低,且细胞表面见微绒毛,可增加细胞的吸附能力和吸收能力。陈丽等[44]在原子力显微镜下观察到沃顿胶体间充质干细胞以长梭形为主,细胞骨架丝明显,并形成网状连接,这与其强大的增殖、迁移、分化功能相适应。Nekanti等[41]报道,在缺氧下培养沃顿胶体间充质干细胞的早期和晚期传代细胞中大的、扁平的细胞比例较常氧培养下高。在缺氧下细胞大小增大可能是由于对低氧的自然反应,增加的表面积使氧的弥散率得以增高。 Garzon等[45]发现沃顿胶体间充质干细胞从第1代到第3代细胞活性逐渐减低,接着直到第6代活性又逐渐增高,最后从第6代到第10代再次减低。最高的细胞活性水平相当于第5代和第6代。基因表达分析证实,细胞活性与调节凋亡细胞死亡的基因显著相关,尤其是促凋亡的FASTKD2、BNIP3L基因和抗凋亡的TNFAIP8和BCL2L2基因,提示在多代培养中的沃顿胶体间充质干细胞中可能有着复杂的生死平衡状态。 2.4 基因表达与分泌蛋白组学 沃顿胶体间充质干细胞不仅表达作为多能干细胞标记的Oct-4、Nanog和Sox-2的mRNAs,而且还在mRNA和蛋白质水平表达起免疫调节作用的HLA-G[46] 。Nekanti等[41]通过比较缺氧和常氧下的沃顿胶体间充质干细胞的基因表达谱,证实了一种差异性基因表达的模式,数种干细胞标记和早期中胚层和内皮基因诸如DESMIN、CD34、ACTC在缺氧下被上调,提示在缺氧条件下体外培养沃顿胶体间充质干细胞导致向中胚层或内皮分化的结果。在早期和晚期的传代细胞中,沃顿胶体间充质干细胞较骨髓间充质干细胞高表达Nanog、DNMT3B和GABRB3等胚胎干细胞、多能干细胞和早期内胚层细胞的标记[47] 。沃顿胶体间充质干细胞与骨髓间充质干细胞间趋化因子和生长因子的表达亦存在差异。Balasubramanian等[48]发现沃顿胶体间充质干细胞上调具有谷氨酸-亮氨酸-精氨酸结构和ELR-阳性的CXC趋化因子基因的表达;同时,沃顿胶体间充质干细胞较骨髓间充质干细胞更强地表达VEGF-D、PDGF-AA、TGF-β、bFGF和HGF。这些结果提示沃顿胶体间充质干细胞可能较骨髓间充质干细胞更适于治疗缺血性病变及减轻纤维化和瘢痕形成。Hsieh等[49]也发现沃顿胶体间充质干细胞表达更多的与血管生成和生长相关的基因(如表皮生长因子和FLT1基因),而骨髓间充质干细胞表达更多诸如RUNX2,DLX5和NPR3的成骨基因。骨髓间充质干细胞的基因表达类型较沃顿胶体间充质干细胞更类似于成骨细胞。相比之下,沃顿胶体间充质干细胞因其与胚胎干细胞共有某些基因而更原始。沃顿胶体间充质干细胞表达富含VEGF和PI3K-NFκ途径的基因,而骨髓间充质干细胞表达涉及抗原呈递和趋化因子/细胞因子途径的基因。因此。骨髓间充质干细胞在成骨和成脂分化上更有效,而沃顿胶体间充质干细胞则在血管生成和增殖方面更佳。Angelucci等[50]对人沃顿胶体间充质干细胞的蛋白组学谱进行了详尽的研究。他们发现包括Shootin1,腺苷酸激酶5同工酶和纤溶酶原激活物抑制剂2在内的数个重要的蛋白在早期细胞体外复制后不再表达,可能与其持久的多能性逐渐减低有关。此外,在细胞扩增的终末,新生的蛋白如ERO1样蛋白α、天冬氨酰-tRNA合成酶和脯氨酰-4-羟化酶α1和α2,可能参与损害复制期间的细胞生存和分化时间。 2.5 免疫原性与免疫调节功能 沃顿胶体间充质干细胞表达免疫耐受基因HLA-E、HLA-G和HLA-F及免疫调节基因VEGF、TGFβ1、HGF、HMOX1、IL1β、IL-6、LIF、LGALS-1/3/8、COX1/2和PTGE,而不表达免疫应答相关的基因HLA-DR、HLA-DQ、HLA-DP、CD80、CD86、CD40和CD40L。在接受沃顿胶体间充质干细胞移植的心衰患者中观察到T淋巴细胞亚群和血浆IgG/IgM没有明显变化,提示沃顿胶体间充质干细胞的免疫特性是在缺乏免疫应答相关基因表达的同时表达免疫逃避与免疫调节基因[51] 。Zhou等[52]观察到沃顿胶体间充质干细胞与其培养上清均能抑制植物凝集素激发的人外周血淋巴细胞和小鼠脾细胞的增殖。此外,沃顿胶体间充质干细胞还能抑制人外周血淋巴细胞对转化生长因子β1和干扰素-γ的分泌。作者得出结论,沃顿胶体间充质干细胞的免疫调节作用可能与直接细胞接触和对人外周血淋巴细胞相关细胞因子的分泌有关。Valencic等[53]发现激活的淋巴细胞在沃顿胶体间充质干细胞面前虽然正常表达激活的标记物,但其增殖和细胞因子的生成均明显减低。当靶淋巴细胞被膜分开时抑制被减弱,但加入外源性L-色氨酸可部分恢复抑制,提示2、3-二氧生成酶具有重要的作用(可能的旁分泌效应)。免疫特许在接触与非接触背景下均能增强免疫抑制的效果。沃顿胶体间充质干细胞免疫特许的时机对免疫抑制作用来说是决定性的。预先单独激活24 h的淋巴细胞对未经免疫特许的沃顿胶体间充质干细胞的增殖反应是正常甚至是增强的,而另一方面,其增殖可被免疫特许的沃顿胶体间充质干细胞所强烈抑制。Najar等[54]研究了沃顿胶体间充质干细胞对白血病抑制因子的表达及其在间充质干细胞介导的抑制机制中的作用。证实在活化T细胞的刺激下,通过表达COX1/COX2酶和产生PGE2介导,沃顿胶体间充质干细胞对T细胞增殖产生强有力的剂量依赖性免疫抑制作用,沃顿胶体间充质干细胞较骨髓间充质干细胞在抑制淋巴细胞应答上更强有力,它们通过分泌高水平的白血病抑制因子介导这一效应。此外,沃顿胶体间充质干细胞维持和促进调节性T-细胞的扩增不依赖于间充质干细胞/T淋巴细胞比[55] 。 2.6 成瘤性 沃顿胶体间充质干细胞低水平表达包括POUF1、NANOG、SOX2和LIN28在内的多潜能胚胎干细胞标记,从而解释了其为何不生成畸胎瘤。沃顿胶体间充质干细胞明显上调数种细胞因子的表达,其中包括IL12A(与诱导凋亡相关),从而解释了其抗癌的特性[56] 。Gauthaman等[57]比较了人沃顿胶体间充质干细胞和胚胎干细胞在免疫缺陷小鼠中的肿瘤形成能力。经由3种途径:皮下、肌内和腹腔内注射人胚胎干细胞(2×106细胞/位点)和人沃顿胶体间充质干细胞(5×106细胞/位点)。所有接受人胚胎干细胞的动物在6周内均发生了肿瘤,而在接受人沃顿胶体间充质干细胞的动物没有肿瘤发生,甚至当观察20周时依然没有肿瘤发生。此外,接受人胚胎干细胞的动物呈现免疫应答,而接受人沃顿胶体间充质干细胞的动物却未出现。作者得出结论,人沃顿胶体间充质干细胞既不诱发肿瘤也不在移植时发生排斥反应,在细胞治疗上具有巨大的潜能。 2.7 向心肌细胞分化的能力 人沃顿胶体间充质干细胞表现出分化为各种不同组织细胞的多潜能性,诸如心血管组织、骨、脂肪、软骨和神经细胞等。Wang等[17]用5-氮杂胞苷处理沃顿胶体间充质干细胞3周后,可表达出心肌细胞的标记物心脏肌钙蛋白I、缝隙连接蛋白43和结蛋白,并显示出心肌细胞的形态学。5-氮杂胞苷可能是通过调控GATA4、Nkx2.5基因的表达促进人脐带间充质干细胞分化为心肌样细胞,并促进其成熟[58] ,10 μmol/L是较佳的诱导浓度[59] 。Pereira等[60]报道,当培养3周时,某些分离的沃顿胶体间充质干细胞发育为带有心脏细胞形态学特征的表型,形成肌管结构和出现自发搏动。 2.8 在心血管系统疾病中的研究 为了评估人沃顿胶体间充质干细胞与心脏组织的整合特性,将其与正常的或缺血的胚胎鼠心室切片共培养。对共培养制备的冰冻切片做免疫组织化学分析,在共培养中进行人类细胞的追踪。沃顿胶体间充质干细胞明显趋向心室切片,强烈整合到正常的与缺血的心脏组织的深部,在体外证实沃顿胶体间充质干细胞能定植于心脏组织[61] 。何红燕等[62]将用BrdU标记的人脐带间充质干细胞移植至心肌梗死的大鼠心肌,2周后取材检测,结果显示细胞移植后2周仍能存活,存活细胞能表达心肌细胞标记物心脏肌钙蛋白I和T。Wu等[63]在体外试验和大鼠心肌梗死模型中再次证实了沃顿胶体间充质干细胞能向心肌细胞分化,同时超声提示移植后2周和4周的左室功能较对照改善。Latifpour等[64]观察到,结扎冠状动脉前降支后30 d,接受人沃顿胶体间充质干细胞治疗动物的左室射血分数和短轴缩短率明显改善,瘢痕含量显著减低。组织病理学分析证明,在少数兔中,邻近和远离梗死区域的植入细胞表达肌钙蛋白I、F-actin和connexin-43,在梗死区内亦可见少数移植的细胞。"
| [1]Phinney DG. Building a consensus regarding the nature and origin of mesenchymal stem cells. J Cell Biochem Suppl. 2002;38:7-12.[2]Pittenger MF, Mackay AM, Beck SC,et al. Multilineage potential of adult human mesenchymal stem cells.Science. 1999;284(5411):143-147.[3]Campagnoli C, Roberts IA, Kumar S,et al. Identification of mesenchymal stem/progenitor cells in human first-trimester fetal blood, liver, and bone marrow.Blood. 2001;98(8):2396-2402.[4]in 't Anker PS, Noort WA, Scherjon SA,et al. Mesenchymal stem cells in human second-trimester bone marrow, liver, lung, and spleen exhibit a similar immunophenotype but a heterogeneous multilineage differentiation potential. Haematologica. 2003;88(8):845-852.[5]Zuk PA, Zhu M, Mizuno H,et al.Multilineage cells from human adipose tissue: implications for cell-based therapies.Tissue Eng. 2001;7(2):211-228.[6]Zuk PA, Zhu M, Ashjian P,et al.Human adipose tissue is a source of multipotent stem cells.Mol Biol Cell. 2002;13(12): 4279-4295.[7]Kuznetsov SA, Mankani MH, Gronthos S,et al. Circulating skeletal stem cells.J Cell Biol. 2001;153(5):1133-1140.[8]Shi S, Gronthos S. Perivascular niche of postnatal mesenchymal stem cells in human bone marrow and dental pulp.J Bone Miner Res. 2003;18(4):696-704.[9]Gronthos S, Mankani M, Brahim J,et al. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo.Proc Natl Acad Sci U S A. 2000;97(25):13625-13630.[10]Pierdomenico L, Bonsi L, Calvitti M,et al. Multipotent mesenchymal stem cells with immunosuppressive activity can be easily isolated from dental pulp.Transplantation. 2005;80(6): 836-842.[11]Igura K, Zhang X, Takahashi K,et al.Isolation and characterization of mesenchymal progenitor cells from chorionic villi of human placenta.Cytotherapy. 2004;6(6): 543-553.[12]Fukuchi Y, Nakajima H, Sugiyama D,et al.Human placenta-derived cells have mesenchymal stem/progenitor cell potential.Stem Cells. 2004;22(5):649-658.[13]You Q, Cai L, Zheng J,et al.Isolation of human mesenchymal stem cells from third-trimester amniotic fluid.Int J Gynaecol Obstet. 2008;103(2):149-152.[14]Tsai MS, Lee JL, Chang YJ,et al.Isolation of human multipotent mesenchymal stem cells from second-trimester amniotic fluid using a novel two-stage culture protocol.Hum Reprod. 2004;19(6):1450-1456.[15]Erices A, Conget P, Minguell JJ. Mesenchymal progenitor cells in human umbilical cord blood.Br J Haematol. 2000; 109(1):235-242.[16]Romanov YA, Svintsitskaya VA, Smirnov VN.Searching for alternative sources of postnatal human mesenchymal stem cells: candidate MSC-like cells from umbilical cord.Stem Cells. 2003;21(1):105-110.[17]Wang HS, Hung SC, Peng ST,et al. Mesenchymal stem cells in the Wharton's jelly of the human umbilical cord. Stem Cells. 2004;22(7):1330-1337.[18]Weiss ML, Medicetty S, Bledsoe AR, et al. Human umbilical cord matrix stem cells: preliminary characterization and effect of transplantation in a rodent model of Parkinson's disease. Stem Cells. 2006;24(3):781-792.[19]Sarugaser R, Lickorish D, Baksh D,et al.Human umbilical cord perivascular (HUCPV) cells: a source of mesenchymal progenitors.Stem Cells. 2005;23(2):220-229.[20]Hu Y, Liao L, Wang Q,et al.Isolation and identification of mesenchymal stem cells from human fetal pancreas.J Lab Clin Med. 2003;141(5):342-349.[21]da Silva Meirelles L, Chagastelles PC, Nardi NB. Mesenchymal stem cells reside in virtually all post-natal organs and tissues. J Cell Sci. 2006;119(Pt 11):2204-2213. [22]Horwitz EM, Le Blanc K, Dominici M,et al. Clarification of the nomenclature for MSC: The International Society for Cellular Therapy position statement.Cytotherapy. 2005;7(5):393-395.[23]Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315-317.[24]Campagnoli C, Roberts IA, Kumar S,et al. Identification of mesenchymal stem/progenitor cells in human first-trimester fetal blood, liver, and bone marrow. Blood. 2001;98(8): 2396-2402.[25]Götherström C, Ringdén O, Westgren M,et al. Immunomodulatory effects of human foetal liver-derived mesenchymal stem cells.Bone Marrow Transplant. 2003; 32(3):265-272.[26]Götherström C, West A, Liden J, et al. Difference in gene expression between human fetal liver and adult bone marrow mesenchymal stem cells. Haematologica. 2005;90(8): 1017-1026.[27]Guillot PV, Gotherstrom C, Chan J, et al. Human first-trimester fetal MSC express pluripotency markers and grow faster and have longer telomeres than adult MSC.Stem Cells. 2007;25(3):646-654.[28]Carlin R, Davis D, Weiss M, et al. Expression of early transcription factors Oct-4, Sox-2 and Nanog by porcine umbilical cord (PUC) matrix cells.Reprod Biol Endocrinol. 2006;4:8.[29]Friedman R, Betancur M, Boissel L, et al. Umbilical cord mesenchymal stem cells: adjuvants for human cell transplantation.Biol Blood Marrow Transplant. 2007; 13(12): 1477-1486.[30]Troyer DL, Weiss ML.Wharton's jelly-derived cells are a primitive stromal cell population.Stem Cells. 2008;26(3): 591-599.[31]Weiss ML, Medicetty S, Bledsoe AR, et al. Human umbilical cord matrix stem cells: preliminary characterization and effect of transplantation in a rodent model of Parkinson's disease. Stem Cells. 2006;24(3):781-792.[32]Wang XY, Lan Y, He WY, et al. Identification of mesenchymal stem cells in aorta-gonad-mesonephros and yolk sac of human embryos. Blood. 2008;111(4):2436-2443.[33]Fong CY, Richards M, Manasi N, et al. Comparative growth behaviour and characterization of stem cells from human Wharton's jelly. Reprod Biomed Online. 2007;15(6):708- 718.[34]Can A, Karahuseyinoglu S. Concise review: human umbilical cord stroma with regard to the source of fetus-derived stem cells. Stem Cells. 2007;25(11):2886-2895.[35]La Rocca G, Anzalone R, Corrao S,et al. Isolation and characterization of Oct-4+/HLA-G+ mesenchymal stem cells from human umbilical cord matrix: differentiation potential and detection of new markers. Histochem Cell Biol. 2009; 131(2): 267-282.[36]Fong CY, Chak LL, Biswas A, et al. Human Wharton's jelly stem cells have unique transcriptome profiles compared to human embryonic stem cells and other mesenchymal stem cells. Stem Cell Rev. 2011;7(1):1-16.[37]Salehinejad P, Alitheen NB, Ali AM,et al. Comparison of different methods for the isolation of mesenchymal stem cells from human umbilical cord Wharton's jelly. In Vitro Cell Dev Biol Anim. 2012;48(2):75-83.[38]De Bruyn C, Najar M, Raicevic G, et al. A rapid, simple, and reproducible method for the isolation of mesenchymal stromal cells from Wharton's jelly without enzymatic treatment. Stem Cells Dev. 2011;20(3):547-557.[39]Nekanti U, Rao VB, Bahirvani AG, et al. Long-term expansion and pluripotent marker array analysis of Wharton's jelly-derived mesenchymal stem cells. Stem Cells Dev. 2010; 19(1):117-130.[40]Nekanti U, Mohanty L, Venugopal P,et al. Optimization and scale-up of Wharton's jelly-derived mesenchymal stem cells for clinical applications. Stem Cell Res. 2010;5(3):244-254.[41]Nekanti U, Dastidar S, Venugopal P,et al. Increased proliferation and analysis of differential gene expression in human Wharton's jelly-derived mesenchymal stromal cells under hypoxia. Int J Biol Sci. 2010;6(5):499-512.[42]周建,葛宝丰,王家琪,等.不同处理时间50 Hz3.6 mT的正弦交变磁场对人脐带干细胞增殖与成骨性分化的研究[J]. 重庆医科大学学报,2012,37(1):51-55.[43]朱少芳,何援利,付霞霏.人脐带间充质干细胞的生物学特性和超微结构[J].中国医学科学院学报,2011,33(4):382-386.[44]陈丽,吴本清,朱华民.原子力显微镜观察人脐带间充质干细胞:生物学特性与超微结构的关系[J].中国组织工程研究与临床康复, 2010,14(6):996-1001.[45]Garzón I, Pérez-Köhler B, Garrido-Gómez J, et al. Evaluation of the cell viability of human Wharton's jelly stem cells for use in cell therapy.Tissue Eng Part C Methods. 2012;18(6): 408-419.[46]Kim MJ, Shin KS, Jeon JH, et al. Human chorionic-plate-derived mesenchymal stem cells and Wharton's jelly-derived mesenchymal stem cells: a comparative analysis of their potential as placenta-derived stem cells.Cell Tissue Res. 2011;346(1):53-64.[47]Nekanti U, Rao VB, Bahirvani AG,et al. Long-term expansion and pluripotent marker array analysis of Wharton's jelly-derived mesenchymal stem cells.Stem Cells Dev. 2010; 19(1):117-130.[48]Balasubramanian S, Venugopal P, Sundarraj S, et al. Comparison of chemokine and receptor gene expression between Wharton's jelly and bone marrow-derived mesenchymal stromal cells.Cytotherapy. 2012;14(1):26-33.[49]Hsieh JY, Fu YS, Chang SJ,et al. Functional module analysis reveals differential osteogenic and stemness potentials in human mesenchymal stem cells from bone marrow and Wharton's jelly of umbilical cord. Stem Cells Dev. 2010;19(12): 1895-1910.[50]Angelucci S, Marchisio M, Di Giuseppe F, et al. Proteome analysis of human Wharton's jelly cells during in vitro expansion. Proteome Sci. 2010;8:18.[51]Chen H, Zhang N, Li T,et al. Human umbilical cord Wharton's jelly stem cells: immune property genes assay and effect of transplantation on the immune cells of heart failure patients. Cell Immunol. 2012;276(1-2):83-90.[52]Zhou C, Yang B, Tian Y,et al. Immunomodulatory effect of human umbilical cord Wharton's jelly-derived mesenchymal stem cells on lymphocytes. Cell Immunol. 2011;272(1): 33-38.[53]Valencic E, Piscianz E, Andolina M,et al. The immunosuppressive effect of Wharton's jelly stromal cells depends on the timing of their licensing and on lymphocyte activation. Cytotherapy. 2010;12(2):154-160.[54]Najar M, Raicevic G, Boufker HI, et al. Mesenchymal stromal cells use PGE2 to modulate activation and proliferation of lymphocyte subsets: Combined comparison of adipose tissue, Wharton's Jelly and bone marrow sources.Cell Immunol. 2010; 264(2):171-179.[55]Najar M, Raicevic G, Boufker HI, et al. Adipose-tissue-derived and Wharton's jelly-derived mesenchymal stromal cells suppress lymphocyte responses by secreting leukemia inhibitory factor. Tissue Eng Part A. 2010;16(11):3537-3546.[56]Fong CY, Chak LL, Biswas A, et al. Human Wharton's jelly stem cells have unique transcriptome profiles compared to human embryonic stem cells and other mesenchymal stem cells. Stem Cell Rev. 2011;7(1):1-16.[57]Gauthaman K, Fong CY, Suganya CA, et al. Extra-embryonic human Wharton's jelly stem cells do not induce tumorigenesis, unlike human embryonic stem cells. Reprod Biomed Online. 2012;24(2):235-246.[58]阮忠宝,杨向军,陈各才,等.5-氮杂胞苷诱导人脐带间充质干细胞向心肌样细胞的分化[J].中国组织工程研究与临床康复,2011, 15(36):6705-6708.[59]唐欣,王岩,易海波,等.不同浓度5-氮胞苷体外诱导人脐带间充质干细胞向心肌样细胞的分化[J].中国组织工程研究与临床康复, 2011,15(40):7459-7462.[60]Pereira WC, Khushnooma I, Madkaikar M,et al. Reproducible methodology for the isolation of mesenchymal stem cells from human umbilical cord and its potential for cardiomyocyte generation.J Tissue Eng Regen Med. 2008;2(7):394-399.[61]Lupu M, Khalil M, Andrei E, et al. Integration properties of Wharton's jelly-derived novel mesenchymal stem cells into ventricular slices of murine hearts.Cell Physiol Biochem. 2011;28(1):63-76.[62]何红燕,林晓波,应文娟.人脐带间充质干细胞经体内定植并向心肌样细胞分化的研究[J].中国输血杂志,2009,22(3):188-191.[63]Wu KH, Mo XM, Zhou B, et al. Cardiac potential of stem cells from whole human umbilical cord tissue.J Cell Biochem. 2009;107(5):926-932.[64]Latifpour M, Nematollahi-Mahani SN, Deilamy M, et al. Improvement in cardiac function following transplantation of human umbilical cord matrix-derived mesenchymal cells. Cardiology. 2011;120(1):9-18.[65]Madonna R, De Caterina R, Willerson JT, et al. Biologic function and clinical potential of telomerase and associated proteins in cardiovascular tissue repair and regeneration. Eur Heart J. 2011;32(10):1190-1196.[66]Vassalli G, Moccetti T. Cardiac repair with allogeneic mesenchymal stem cells after myocardial infarction. Swiss Med Wkly. 2011;141:w13209.[67]Liu L, Chai J, Han Y,et al. Research progress of biological characteristics and advantages of Wharton's jelly-mesenchymal stem cells. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2011;25(6):745-749.[68]Fong CY, Subramanian A, Biswas A, et al. Derivation efficiency, cell proliferation, freeze-thaw survival, stem-cell properties and differentiation of human Wharton's jelly stem cells. Reprod Biomed Online. 2010;21(3):391-401.[69]Weiss ML, Anderson C, Medicetty S,et al. Immune properties of human umbilical cord Wharton's jelly-derived cells.Stem Cells. 2008;26(11):2865-2874. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [3] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [4] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [5] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [6] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [7] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [8] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [9] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [10] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [11] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [12] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [13] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [14] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [15] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||