Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (29): 7749-7754.doi: 10.12307/2026.277
Previous Articles Next Articles
Mendelian randomization analysis identifies potential drug targets for spinal osteoarthritis
Zhao Ruikai1, Wang Yu1, Guo Xiaohui1, Sun Zehua2, Wang Xu1
- 1Department of Orthopedics, Tangshan Second Hospital, Tangshan 063000, Hebei Province, China; 2Department of Rehabilitation, Fengnan District Hospital of Traditional Chinese Medicine, Tangshan 063000, Hebei Province, China
-
Received:2025-08-14Revised:2025-12-15Online:2026-10-18Published:2026-03-09 -
Contact:Wang Xu, Chief physician, Department of Orthopedics, Tangshan Second Hospital, Tangshan 063000, Hebei Province, China -
About author:Zhao Ruikai, MS, Department of Orthopedics, Tangshan Second Hospital, Tangshan 063000, Hebei Province, China
CLC Number:
Cite this article
Zhao Ruikai, Wang Yu, Guo Xiaohui, Sun Zehua, Wang Xu. Mendelian randomization analysis identifies potential drug targets for spinal osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7749-7754.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
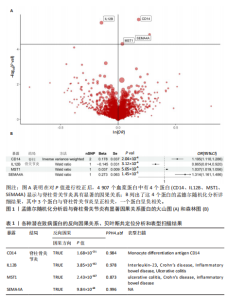
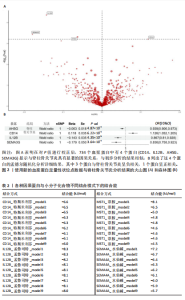
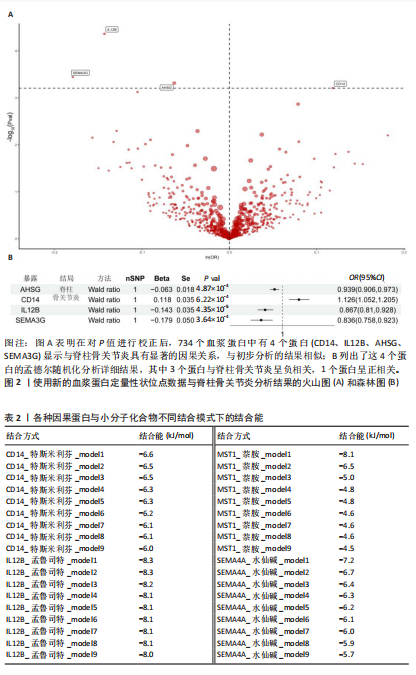
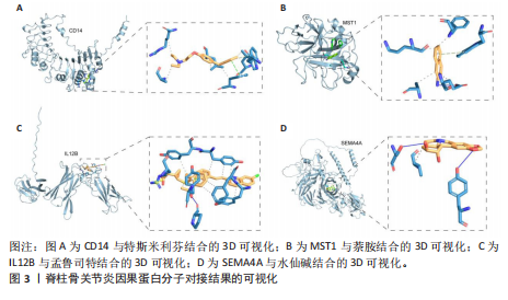
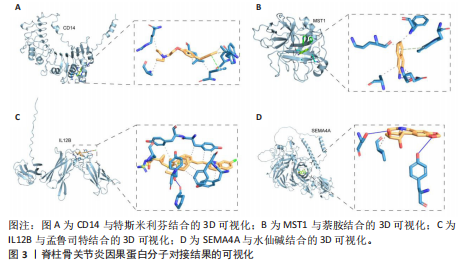
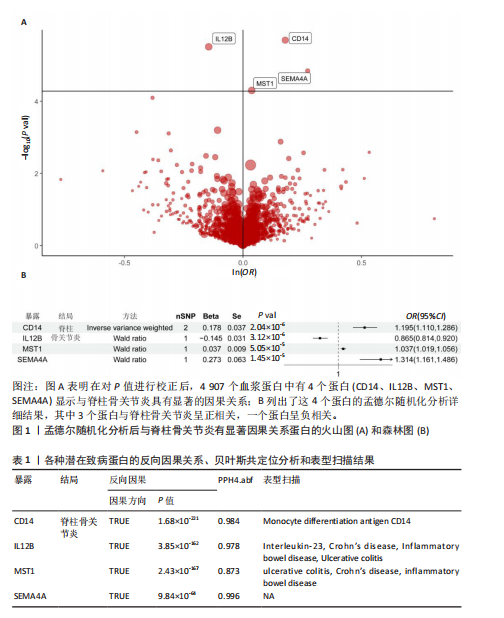
2.1 1 553个血浆蛋白对脊柱骨关节炎的因果影响 图1A显示,在孟德尔随机化分析后共有4种蛋白满足Bonferroni多重校正标准(P < 0.05/1 553=3.22×10-5),分别为单核细胞分化抗原CD14(CD14)、白细胞介素12β亚基(IL12B)、肝细胞生长因子样蛋白(MST1)以及Semaphorin-4A(SEMA4A)。这4种蛋白与脊柱骨关节炎存在不同的因果关系(图1B)。其中,IL12B的表达与脊柱骨关节炎呈负相关,OR值为0.865,提示IL12B水平每增加一个单位可使骨关节炎风险降低至原来的86.5%。而其余3种蛋白均表现为正向关联,即蛋白的表达水平升高与疾病风险增加有关。值得关注的是,SEMA4A的正相关最强,OR值达到1.314,表明该蛋白水平每上升一个单位,发病风险将增加1.314倍。 2.2 敏感性分析 表1展示了反向因果检验、共定位分析和表型扫描的结果。首先,反向因果分析的结果显示,4种蛋白质的P值均远远低于0.05,证实了因果关系的单向性。其次,共定位分析发现,CD14(PPH4.abf=0.984)、IL12B(PPH4.abf=0.978)、MST1(PPH4.abf=0.873)和SEMA4A(PPH4.abf=0.996)均达到显著的共定位阈值,支持它们可能与疾病风险共享相同的致病单核苷酸多态性位点。 最后,使用PhenoScanner V2搜索了与这些蛋白质显著相关的工具变量是否与其他表型相关。结果表明,与CD14相关的单核苷酸多态性(rs5744454)仅与其自身的蛋白质水平相关。与IL12B(rs10043720)和MST1(rs1131095)相关的单核苷酸多态性先前与克罗恩病、炎症性肠病和溃疡性结肠炎等免疫相关疾病有关。然而,与SEMA4A(rs7695)相对应的单核苷酸多态性与其他性状没有显著关联。尽管一些已鉴定的单核苷酸多态性与免疫系统疾病有关,但目前还没有令人信服的证据表明这些疾病与脊柱骨关节炎之间存在直接的因果关系。因此,它们不太可能代表混淆的主要来源。 2.3 外部验证 外部验证结果表明,CD14与脊柱骨关节炎正相关,IL12B与脊柱骨关节炎负相关,这与之前的结果一致(图2)。然而,新的血浆蛋白数据虽然是来自5个全基因组关联研究的meta分析,但其所包含的蛋白数量仍达不到初步孟德尔随机化分析中用做暴露的蛋白数据,因此缺乏MST1和SEMA4A的遗传数据,未能进行重复验证。 2.4 小分子药物的筛选和分子对接 通过Enrichr分析工具,对4种因果蛋白(CD14、IL12B、MST1以及SEMA4A)筛选了靶向它们的小分子化合物。此次研究根据结果中P值< 0.05进行排除后,分别获得了38个、42个、2个和1个与靶向上述4种蛋白的小分子化合物。"
| [1] FELSON DT, LAWRENCE RC, DIEPPE PA, et al. Osteoarthritis: new insights. Part 1: the disease and its risk factors. Ann Intern Med. 2000;133(8): 635-646. [2] CHO HJ, MOREY V, KANG JY, et al. Prevalence and Risk Factors of Spine, Shoulder, Hand, Hip, and Knee Osteoarthritis in Community-dwelling Koreans Older Than Age 65 Years. Clin Orthop Relat Res. 2015;473(10):3307-3314. [3] IORDACHE S, CURSARU A, MARINESCU A, et al. Magnetic Resonance Imaging Features and Functional Score in Patients Requiring Total Knee Arthroplasty. Cureus. 2024;16(9):e68595. [4] LUO H, LAI Y, TANG W, et al. Mitochondrial transplantation: a promising strategy for treating degenerative joint diseases. J Transl Med. 2024; 22(1):941. [5] TANG L, DING J, YANG K, et al. New insights into the mechanisms and therapeutic strategies of chondrocyte autophagy in osteoarthritis. J Mol Med (Berl). 2024;102(10):1229-1244. [6] CIOROIANU GO, FLORESCU A, FLORESCU LM, et al. Knee Osteoarthritis-Current Diagnosis and Treatment Options-A Narrative Review. Curr Health Sci J. 2024;50(2):163-169. [7] EMILSSON V, GUDMUNDSDOTTIR V, GUDJONSSON A, et al. Coding and regulatory variants are associated with serum protein levels and disease. Nat Commun. 2022;13(1):481. [8] GUDJONSSON A, GUDMUNDSDOTTIR V, AXELSSON GT, et al. A genome-wide association study of serum proteins reveals shared loci with common diseases. Nat Commun. 2022;13(1):480. [9] LIU F, YANG H, YANG T, et al. Dysregulated proteasome activity and steroid hormone biosynthesis are associated with mortality among patients with acute COVID-19. J Transl Med. 2024; 22(1):626. [10] MAHIN A, SOMAN SP, MODI PK, et al. Meta-analysis of the serum/plasma proteome identifies significant associations between COVID-19 with Alzheimer’s/Parkinson’s diseases. J Neurovirol. 2024;30(1):57-70. [11] GU Y, JIN Q, HU J, et al. Causality of genetically determined metabolites and metabolic pathways on osteoarthritis: a two-sample mendelian randomization study. J Transl Med. 2023;21(1): 357. [12] TERCIC D, BOZIC B. The basis of the synovial fluid analysis. Clin Chem Lab Med. 2001;39(12):1221-1226. [13] PIERCE BL, BURGESS S. Efficient design for Mendelian randomization studies: subsample and 2-sample instrumental variable estimators. Am J Epidemiol. 2013;178(7):1177-1184. [14] HARTWIG FP, DAVIES NM, HEMANI G, et al. Two-sample Mendelian randomization: avoiding the downsides of a powerful, widely applicable but potentially fallible technique. Int J Epidemiol. 2016;45(6):1717-1726. [15] LIN J, ZHOU J, XU Y. Potential drug targets for multiple sclerosis identified through Mendelian randomization analysis. Brain. 2023;146(8): 3364-3372. [16] REAY WR, CAIRNS MJ. Advancing the use of genome-wide association studies for drug repurposing. Nat Rev Genet. 2021;22(10):658-671. [17] EMDIN CA, KHERA AV, KATHIRESAN S. Mendelian Randomization. JAMA. 2017;318(19):1925-1926. [18] FERKINGSTAD E, SULEM P, ATLASON BA, et al. Large-scale integration of the plasma proteome with genetics and disease. Nat Genet. 2021; 53(12):1712-1721. [19] ZHENG J, HABERLAND V, BAIRD D, et al. Phenome-wide Mendelian randomization mapping the influence of the plasma proteome on complex diseases. Nat Genet. 2020;52(10):1122-1131. [20] BOER CG, HATZIKOTOULAS K, SOUTHAM L, et al. Deciphering osteoarthritis genetics across 826,690 individuals from 9 populations. Cell. 2021; 184(24):6003-6005. [21] ZHANG J, DUTTA D, KÖTTGEN A, et al. Plasma proteome analyses in individuals of European and African ancestry identify cis-pQTLs and models for proteome-wide association studies. Nat Genet. 2022;54(5):593-602. [22] WANG Q, DAI H, HOU T, et al. Dissecting Causal Relationships Between Gut Microbiota, Blood Metabolites, and Stroke: A Mendelian Randomization Study. J Stroke. 2023;25(3):350-360. [23] YAN W, JIANG M, HU W, et al. Causality Investigation between Gut Microbiota, Derived Metabolites, and Obstructive Sleep Apnea: A Bidirectional Mendelian Randomization Study. Nutrients. 2023;15(21):4544. [24] BOWDEN J, DEL GRECO MF, MINELLI C, et al. Assessing the suitability of summary data for two-sample Mendelian randomization analyses using MR-Egger regression: the role of the I2 statistic. Int J Epidemiol. 2016;45(6):1961-1974. [25] CHENG J, YANG L, YE Y, et al. Mendelian Randomisation Analysis of Causal Association between Lifestyle, Health Factors, and Keratoconus. Bioengineering (Basel). 2024; 11(3):221. [26] BURGESS S, BUTTERWORTH A, THOMPSON SG. Mendelian randomization analysis with multiple genetic variants using summarized data. Genet Epidemiol. 2013;37(7):658-665. [27] HEMANI G, TILLING K, DAVEY SMITH G. Orienting the causal relationship between imprecisely measured traits using GWAS summary data. PLoS Genet. 2017;13(11):e1007081. [28] CULLELL N, GALLEGO-FÁBREGA C, CÁRCEL-MÁRQUEZ J, et al. ICA1L Is Associated with Small Vessel Disease: A Proteome-Wide Association Study in Small Vessel Stroke and Intracerebral Haemorrhage. Int J Mol Sci. 2022;23(6):3161. [29] GIAMBARTOLOMEI C, VUKCEVIC D, SCHADT EE, et al. Bayesian test for colocalisation between pairs of genetic association studies using summary statistics. PLoS Genet. 2014;10(5):e1004383. [30] KAMAT MA, BLACKSHAW JA, YOUNG R, et al. PhenoScanner V2: an expanded tool for searching human genotype-phenotype associations. Bioinformatics. 2019;35(22):4851-4853. [31] BURLEY SK, BHIKADIYA C, BI C, et al. RCSB Protein Data Bank: powerful new tools for exploring 3D structures of biological macromolecules for basic and applied research and education in fundamental biology, biomedicine, biotechnology, bioengineering and energy sciences. Nucleic Acids Res. 2021;49(D1):D437-d451. [32] MONTGOMERY SB, DERMITZAKIS ET. From expression QTLs to personalized transcriptomics. Nat Rev Genet. 2011;12(4):277-282. [33] OPPMANN B, LESLEY R, BLOM B, et al. Novel p19 protein engages IL-12p40 to form a cytokine, IL-23, with biological activities similar as well as distinct from IL-12. Immunity. 2000;13(5):715-725. [34] TENG MW, BOWMAN EP, MCELWEE JJ, et al. IL-12 and IL-23 cytokines: from discovery to targeted therapies for immune-mediated inflammatory diseases. Nat Med. 2015;21(7):719-729. [35] EK WE, KARLSSON T, HÖGLUND J, et al. Causal effects of inflammatory protein biomarkers on inflammatory diseases. Sci Adv. 2021;7(50): eabl4359. [36] WRIGHT SD, RAMOS RA, TOBIAS PS, et al. CD14, a receptor for complexes of lipopolysaccharide (LPS) and LPS binding protein. Science. 1990;249(4975): 1431-1433. [37] SETOGUCHI M, NASU N, YOSHIDA S, et al. Mouse and human CD14 (myeloid cell-specific leucine-rich glycoprotein) primary structure deduced from cDNA clones. Biochim Biophys Acta. 1989; 1008(2):213-222. [38] ZANONI I, TAN Y, DI GIOIA M, et al. By Capturing Inflammatory Lipids Released from Dying Cells, the Receptor CD14 Induces Inflammasome-Dependent Phagocyte Hyperactivation. Immunity. 2017;47(4):697-709.e693. [39] FURNKRANZ A, SCHOBER A, BOCHKOV VN, et al. Oxidized phospholipids trigger atherogenic inflammation in murine arteries. Arterioscler Thromb Vasc Biol. 2005;25(3):633-638. [40] SHARYGIN D, KONIARIS LG, WELLS C, et al. Role of CD14 in human disease. Immunology. 2023;169(3):260-270. [41] FUENTELSAZ-ROMERO S, BARRIO-ALONSO C, GARCÍA CAMPOS R, et al. The Macrophage Reprogramming Ability of Antifolates Reveals Soluble CD14 as a Potential Biomarker for Methotrexate Response in Rheumatoid Arthritis. Front Immunol. 2021;12:776879. [42] NAKAMIZO S, DUTERTRE CA, KHALILNEZHAD A, et al. Single-cell analysis of human skin identifies CD14+ type 3 dendritic cells co-producing IL1B and IL23A in psoriasis. J Exp Med. 2021;218(9):e20202345. [43] WONG SS, OSHANSKY CM, GUO XJ, et al. Activated CD4(+) T cells and CD14(hi)CD16(+) monocytes correlate with antibody response following influenza virus infection in humans. Cell Rep Med. 2021;2(4):100237. [44] SHI Z, ZHOU Z. MST kinases in innate immune signaling. Cell Stress. 2017;2(1):4-13. [45] PRASKOVA M, XIA F, AVRUCH J. MOBKL1A/MOBKL1B phosphorylation by MST1 and MST2 inhibits cell proliferation. Curr Biol. 2008;18(5): 311-321. [46] ABUKAR Y, RAMCHANDRA R, HOOD SG, et al. Increased cardiac sympathetic nerve activity in ovine heart failure is reduced by lesion of the area postrema, but not lamina terminalis. Basic Res Cardiol. 2018;113(5):35. [47] LI T, WEN Y, LU Q, et al. MST1/2 in inflammation and immunity. Cell Adh Migr. 2023;17(1):1-15. [48] ZONG F, ZHAO Y. Alkaloid leonurine exerts anti-inflammatory effects via modulating MST1 expression in trophoblast cells. Immun Inflamm Dis. 2021;9(4):1439-1446. [49] ZHOU C, YAO S, FU F, et al. Morroniside attenuates nucleus pulposus cell senescence to alleviate intervertebral disc degeneration via inhibiting ROS-Hippo-p53 pathway. Front Pharmacol. 2022; 13:942435. [50] LIU C, CHU X, BIAO Y, et al. Association between lipid-lowering agents with intervertebral disc degeneration, sciatica and low back pain: a drug-targeted mendelian randomized study and cross-sectional observation. Lipids Health Dis. 2024; 23(1):327. [51] QUAN M, LV H, LIU Z, et al. MST1 Suppresses Disturbed Flow Induced Atherosclerosis. Circ Res. 2022;131(9):748-764. [52] IYER AS, CHAPOVAL SP. Neuroimmune Semaphorin 4A in Cancer Angiogenesis and Inflammation: A Promoter or a Suppressor? Int J Mol Sci. 2018; 20(1):124. [53] NKYIMBENG-TAKWI E, CHAPOVAL SP. Biology and function of neuroimmune semaphorins 4A and 4D. Immunol Res. 2011;50(1):10-21. [54] KUMANOGOH A, MARUKAWA S, SUZUKI K, et al. Class IV semaphorin Sema4A enhances T-cell activation and interacts with Tim-2. Nature. 2002; 419(6907):629-633. [55] WANG L, SONG G, ZHENG Y, et al. Expression of Semaphorin 4A and its potential role in rheumatoid arthritis. Arthritis Res Ther. 2015; 17(1):227. [56] KUMANOGOH A, KIKUTANI H. Immune semaphorins: a new area of semaphorin research. J Cell Sci. 2003;116(Pt 17):3463-3470 |
| [1] | Wu Zhilin, , He Qin, Wang Pingxi, Shi Xian, Yuan Song, Zhang Jun, Wang Hao . DYRK2: a novel therapeutic target for rheumatoid arthritis combined with osteoporosis based on East Asian and European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1569-1579. |
| [2] | Liu Hongtao, Wu Xin, Jiang Xinyu, Sha Fei, An Qi, Li Gaobiao. Causal relationship between age-related macular degeneration and deep vein thrombosis: analysis based on genome-wide association study data [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1602-1608. |
| [3] | Guo Ying, Tian Feng, Wang Chunfang. Potential drug targets for the treatment of rheumatoid arthritis: large sample analysis from European databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1549-1557. |
| [4] | Gao Zengjie, , Pu Xiang, Li Lailai, Chai Yihui, Huang Hua, Qin Yu. Increased risk of osteoporotic pathological fractures associated with sterol esters: evidence from IEU-GWAS and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1302-1310. |
| [5] | Liu Fengzhi, Dong Yuna, Tian Wenyi, Wang Chunlei, Liang Xiaodong, Bao Lin. Gene-predicted associations between 731 immune cell phenotypes and rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1311-1319. |
| [6] | Zhang Cuicui, Chen Huanyu, Yu Qiao, Huang Yuxuan, Yao Gengzhen, Zou Xu. Relationship between plasma proteins and pulmonary arterial hypertension and potential therapeutic targets [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1331-1340. |
| [7] | Zeng Hao, Sun Pengcheng, Chai Yuan, Huang Yourong, Zhang Chi, Zhang Xiaoyun. Association between thyroid function and osteoporosis: genome-wide data analysis of European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1019-1027. |
| [8] | Rong Xiangbin, , Zheng Haibo, Mo Xueshen, Hou Kun, Zeng Ping, . Plasma metabolites, immune cells, and hip osteoarthritis: causal inference based on GWAS data from European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1028-1035. |
| [9] | He Qiwang, , , Chen Bo, Liang Fuchao, Kang Zewei, Zhou Yuan, Ji Anxu, Tang Xialin, . Relationship between Alzheimer’s disease and sarcopenia and body mass index: analysis of GWAS datasets for European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1036-1046. |
| [10] | Ding Yu, Chen Jingwen, Chen Xiuyan, Shi Huimin, Yang Yudie, Zhou Meiqi, Cui Shuai, . Circulating inflammatory proteins and myocardial hypertrophy: large sample analysis of European populations from GWAS Catalog and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1047-1057. |
| [11] | Zhao Feifan, Cao Yujing. An artificial neural network model of ankylosing spondylitis and psoriasis shared genes and machine learning-based mining and validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 770-784. |
| [12] | Liu Chu, Qiu Boyuan, Tong Siwen, He Linyuwei, Chen Haobo, Ou Zhixue. A genetic perspective reveals the relationship between blood metabolites and osteonecrosis: an analysis of information from the FinnGen database in Finland [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 785-794. |
| [13] | Tian Xuanhe, Tong Siyu, Teng Fei, Zhong Shuai, Zhao Xiaohu, Zhang Yuya, Liu Yuan, Jiang Ping. Potential targets and drug prediction for gout: identification of druggable genes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7706-7714. |
| [14] | Wei Bingqi, Zhang Xinyue, Ren Xingyue, Sun Jiahui, Chen Liu, Li Yijing, Qi Yifan, Wang Shangzeng. Zinc finger DHHC-type containing 2 emerges as a novel therapeutic target in osteoarthritis pathogenesis: genome-wide data analysis in European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7715-7723. |
| [15] | Wei Qiuyu, Yu Shaoyong, Zhou Zheyi, Wu Gang . Causal relationship between plasma metabolites and osteonecrosis: a large sample analysis based on genome-wide association study database and FinnGen database [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7732-7738. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||