Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (29): 7706-7714.doi: 10.12307/2026.273
Previous Articles Next Articles
Potential targets and drug prediction for gout: identification of druggable genes
Tian Xuanhe1, Tong Siyu2, Teng Fei3, Zhong Shuai1, Zhao Xiaohu1, Zhang Yuya1, Liu Yuan1, Jiang Ping1
- 1First Clinical College, Shandong University of Traditional Chinese Medicine, Jinan 250014, Shandong Province, China; 2College of Traditional Chinese Medicine, Shandong University of Traditional Chinese Medicine, Jinan 250355, Shandong Province, China; 3Third Affiliated Hospital, Beijing University of Chinese Medicine, Beijing 100036, China
-
Received:2025-08-04Revised:2025-11-28Online:2026-10-18Published:2026-03-07 -
Contact:Jiang Ping, PhD, Professor, First Clinical College, Shandong University of Traditional Chinese Medicine, Jinan 250014, Shandong Province, China Co-corresponding author: Liu Yuan, PhD candidate, First Clinical College, Shandong University of Traditional Chinese Medicine, Jinan 250014, Shandong Province, China -
About author:Tian Xuanhe, MS candidate, First Clinical College, Shandong University of Traditional Chinese Medicine, Jinan 250014, Shandong Province, China -
Supported by:National Natural Science Foundation of China, No. 82274481 (to JP); Joint Scientific Research Project of the Science and Technology Department of the National Administration of Traditional Chinese Medicine, No. GZY-KJS-SD-2023-041 (to JP); Shandong Provincial Natural Science Foundation, No. ZR2022LZY004 (to JP); Construction Project of Shandong Health Technology Innovation Team, No. 2024sdskctd-03 (to JP); 2024 Doctoral Research Quality Enhancement and Innovation Project of Shandong University of Traditional Chinese Medicine, No. YJSTZCX2024010 (to LY); 2025 Postgraduate Quality Improvement and Innovation Project of Shandong University of Traditional Chinese Medicine, No. YJSTZCX2025120 (to TXH)
CLC Number:
Cite this article
Tian Xuanhe, Tong Siyu, Teng Fei, Zhong Shuai, Zhao Xiaohu, Zhang Yuya, Liu Yuan, Jiang Ping. Potential targets and drug prediction for gout: identification of druggable genes[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7706-7714.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

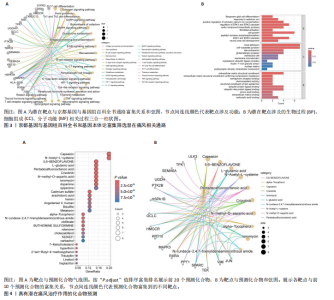
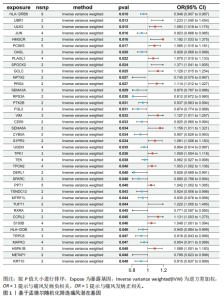
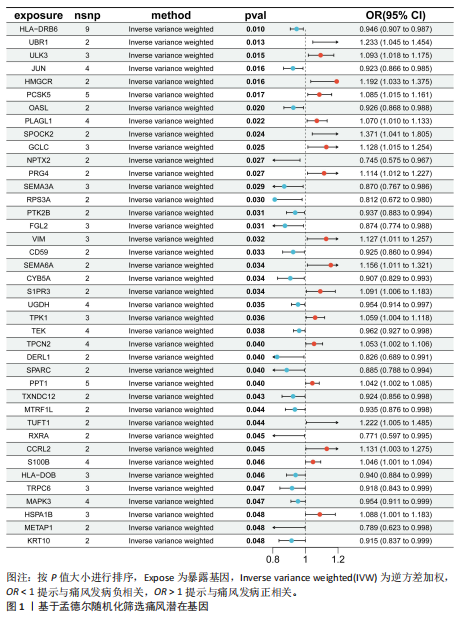
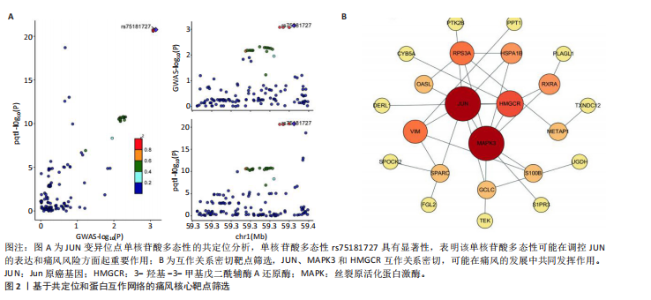
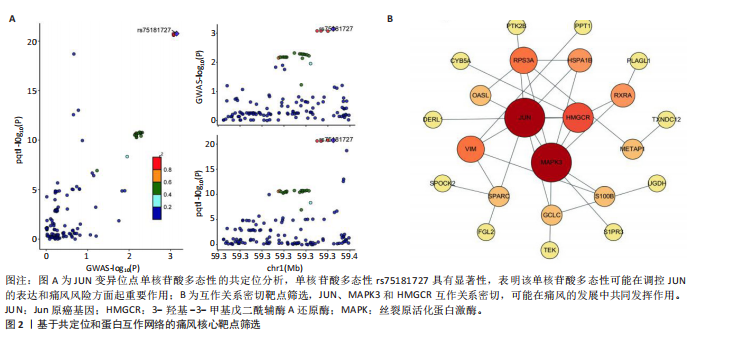
2.1 孟德尔随机化分析 经筛选获得7 417个强关联单核苷酸多态性作为工具变量,所有单核苷酸多态性的F值均> 10。结果显示,在采用逆方差加权方法进行的孟德尔随机化分析中,OR < 1的基因共有22个,包括NPTX2、RXRA、METAP1、RPS3A、DERL1、SEMA3A、FGL2、SPARC、CYB5A、KRT10、TRPC6、JUN、TXNDC12、CD59、OASL、MTRF1L、PTK2B、HLA-DOB、HLA-DRB6、MAPK3、UGDH、TEK等,与痛风存在显著的负相关因果关系;OR > 1的基因共有18个,包括PPT1、S100B、TPCN2、TPK1、PLAGL1、PCSK5、HSPA1B、S1PR3、ULK3、PRG4、VIM、GCLC、CCRL2、SEMA6A、HMGCR、TUFT1、UBR1、SPOCK2等,与痛风存在显著的正相关因果关系(表2,图1)。 2.2 核心靶点筛选 共定位分析显示40个基因中JUN(SNP.PP.H4=0.733)与痛风关系显著,为痛风关键易感基因(图2A);蛋白互作网络显示JUN、MAPK3和HMGCR互作关系最为密切(图2B)。 2.3 京都基因与基因组百科全书和基因本体论通路富集分析 京都基因与基因组百科全书富集结果显示共富集到25条通路,包括Th17、"

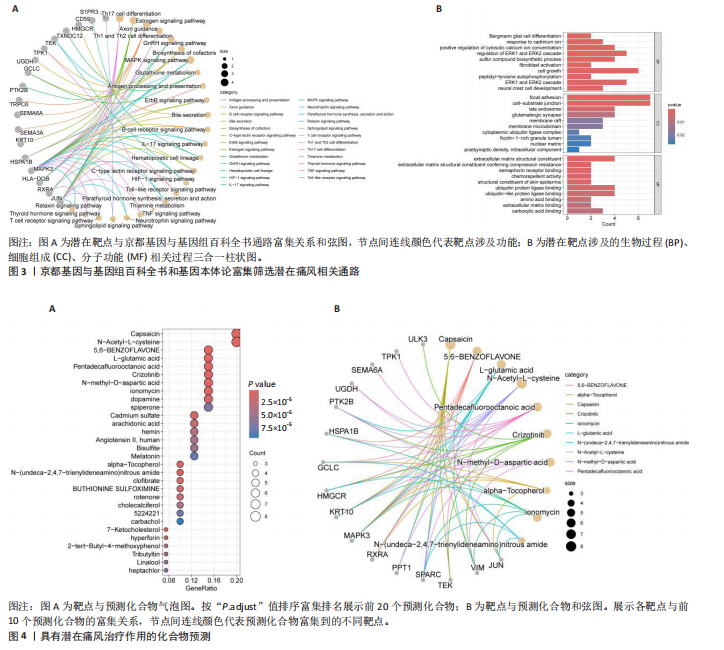
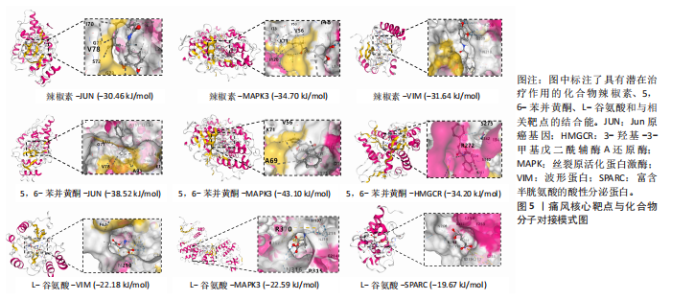
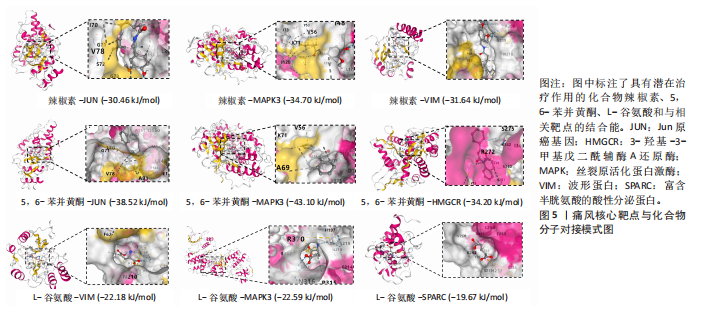
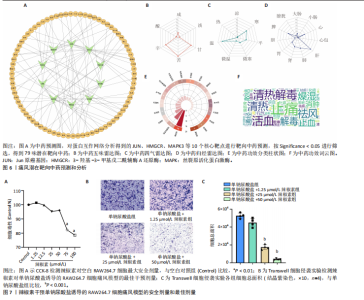
Th1和Th2细胞分化、MAPK、TNF、ErbB、白细胞介素17、缺氧诱导因子1、Toll样受体等信号通路(图3A);基因本体论富集显示生物过程类别中共富集到72条显著通路和过程,如抗原加工和呈递、ERK1/2级联反应的正向调控、MAPK级联反应的正向调控和谷胱甘肽代谢过程等;细胞组成类别中共富集到24条显著通路,如谷胱甘肽突触调节等;分子功能类别中共富集到17条显著通路,如泛素蛋白的调节等(图3B)。 2.4 药物预测富集分析 在DGIbd数据库中获取了2022年2月发布的“类别数据”同时从FINAN等[20]的综述中获取可用于药物治疗的基因列表。共预测出372种具有潜在治疗作用的化合物,包括槲皮素、和厚朴酚、山柰酚、肉桂醛、穿心莲内酯和异黄酮等多种中药单体;按“P.adjust”值排序富集排名前3的化合物为辣椒素、5,6-苯并黄酮和L-谷氨酸,涉及的作用靶点包括辣椒素:JUN、VIM、MAPK3、TEK、SPARC、PPT1、RXRA、KRT10;5,6-苯并黄酮:JUN、HMGCR、MAPK3、GCLC、HSPA1B、KRT10;L-谷氨酸:VIM、SPARC、MAPK3、PTK2B、PPT1、KRT10(图4)。 2.5 预测药物分子对接 结合“2.2”中筛选的核心靶点和“2.4”中化合物富集靶点,运用分子对接技术分析辣椒素与JUN、VIM、MAPK3,5,6-苯并黄酮与JUN、HMGCR、MAPK3,L-谷氨酸与VIM、SPARC、MAPK3的结合程度(图5)。 2.6 中药预测分析 将筛选出的核心靶点进行预测和筛选中药,共选出包括苍术、厚朴、土茯苓、泽泻、丹参、黄连等79味中药(图6A),其中苦味药38种,甘味药31种,辛味药29种,甘味药31种,咸味药10种,酸味药4种,淡味药3种(图6B);寒性药19种,微寒药9种,凉性药2种,热性药1种,温性药27种,微温药6种,平性药15种(图6C);"
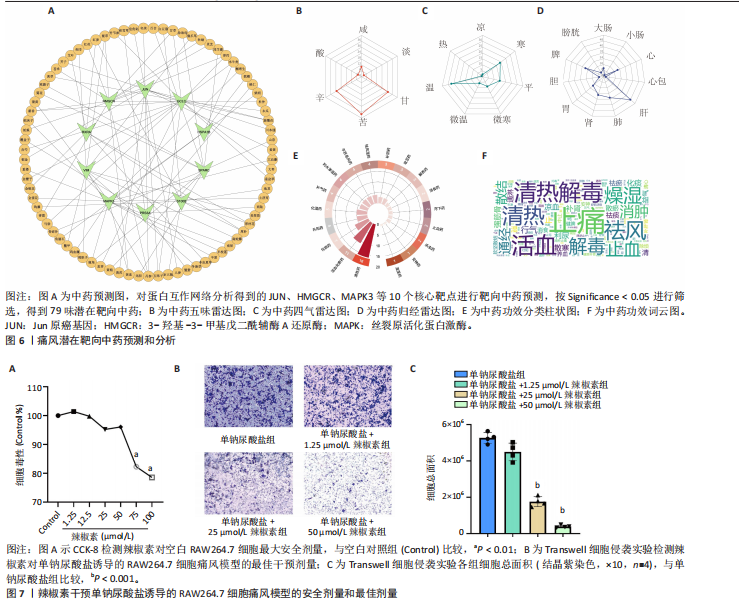
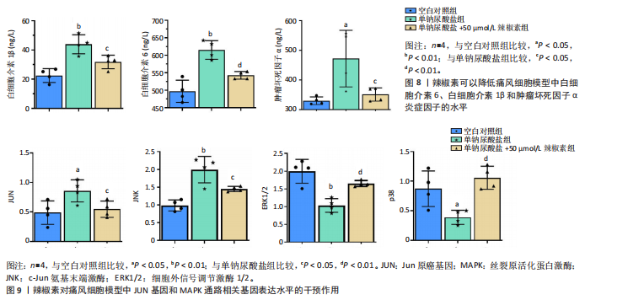
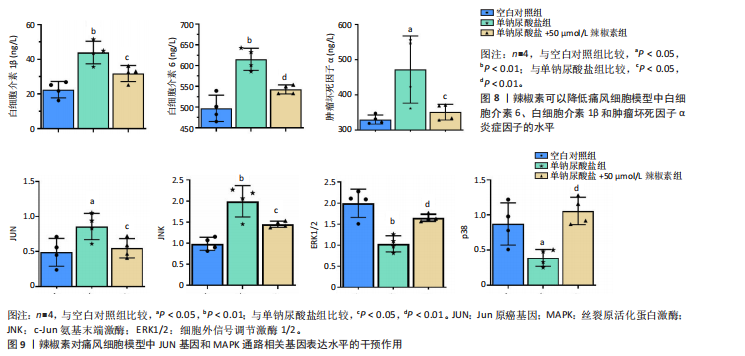
归肝经48种,肺经30种,脾经27种,肾经26种,心经21种,胃经19种,大肠经11种,膀胱经6种,胆经6种,心包经3种,小肠经2种(图6D);包含清热药16种,活血化瘀药11种,化痰药7种,补阳药5种,化湿药5种,补气药4种,利水渗湿药4种,平肝息风药4种,祛风湿药4种,补阴药3种,收涩药3种,解表药2种,消食药2种,泻下药2种,止血药2种,杀虫药2种,安神药1种,温里药1种(图6E),涵盖了清热解毒、祛风止痛、活血化瘀、利水渗湿、补益肝肾等多种功效(图6F)。 2.7 细胞实验验证辣椒素对单钠尿酸盐诱导的RAW264.7细胞痛风模型的干预作用 2.7.1 最佳给药浓度筛选 CCK-8结果显示当给药剂量超过75 μmol/L时出现明显细胞毒性(图7A),Transwell细胞侵袭实验结果显示不同浓度辣椒素对单钠尿酸盐诱导的RAW264.7细胞迁移呈梯度抑制,其中50 μmol/L抑制效果最佳(图7B,C),因此确定50 μmol/L为最佳给药浓度。 2.7.2 炎症因子水平 ELISA结果显示单钠尿酸盐诱导的细胞中白细胞介素6、白细胞介素1β和肿瘤坏死因子α的水平显著升高(P < 0.05),提示使用200 μg/mL 单钠尿酸盐晶体刺激细胞12 h造模成功,同时给予50 μmol/L辣椒素干预可以显著降低炎症因子水平(P < 0.05)(图8)。 2.7.3 关键通路基因表达 实时荧光定量反转录PCR结果显示模型组中JUN基因水平和MAPK通路相关基因JNK、ERK1/2、p38水平上调,辣椒素可以降低痛风细胞模型中JUN基因表达,下调MAPK通路相关mRNA的表达(P < 0.05),见图9。"
| [1] DALBETH N, GOSLING A L, GAFFO A, et al. Gout. Lancet. 2021;397(10287):1843-1855. [2] RAGAB G, ELSHAHALY M, BARDIN T. Gout: An old disease in new perspective - A review. J Adv Res. 2017;8(5):495-511. [3] SINGH JA, GAFFO A. Gout epidemiology and comorbidities. Semin Arthritis Rheum. 2020; 50(3S):S11-S16. [4] 彭紫凝, 邓茜, 孟凡雨, 等. 痛风“湿-热-寒-瘀-虚”复杂病机网络及生物学基础研究进展[J]. 世界中医药,2024,19(23):3725-3730. [5] 张娟娟, 朱美红, 刘娜, 等. 家庭医生签约制度下临床药师对痛风患者的管理实践[J]. 中国药业,2025,34(1):21-24. [6] YANG S, LIU H, FANG X, et al. Signaling pathways in uric acid homeostasis and gout: From pathogenesis to therapeutic interventions. Int Immunopharmacol. 2024;132:111932. [7] SO AK, MARTINON F. Inflammation in gout: mechanisms and therapeutic targets. Nat Rev Rheumatol. 2017;13(11):639-647. [8] 郎吉瑞, 曾瑾, 陈世龙, 等. 中医经典方剂治疗痛风的研究进展[J]. 中药药理与临床,2023, 39(1):104-111. [9] SCHLESINGER N, PILLINGER MH, SIMON LS, et al. Interleukin-1β inhibitors for the management of acute gout flares: a systematic literature review. Arthritis Res Ther. 2023;25(1):128. [10] 饶嘉, 陈楠, 李成江. Anakinra对急性痛风的作用机制与治疗研究进展[J]. 赣南医学院学报, 2019,39(10):1003-1006. [11] 熊逸凡, 赵东宝. 托珠单抗改善难治性痛风患者骨破坏2例报告[J]. 中国实用内科杂志, 2018,38(12):1210-1213. [12] 王佳亮, 樊慧芳, 贺奥城, 等. 中医药治疗痛风的研究进展[J]. 中国实验方剂学杂志,2023, 29(21):220-227. [13] GUO JW, LIN GQ, TANG XY, et al. Therapeutic potential and pharmacological mechanisms of Traditional Chinese Medicine in gout treatment. Acta Pharmacol Sin. 2025;46(5):1156-1176. [14] SEKULA P, DEL GRECO MF, PATTARO C, et al. Mendelian Randomization as an Approach to Assess Causality Using Observational Data. J Am Soc Nephrol. 2016;27(11):3253-3265. [15] SONG J, LI J, ZHANG G, et al. Identification of potential biomarkers and therapeutic targets for cerebral venous thrombosis. Neurol Res. 2025: 1-15. doi: 10.1080/01616412.2025.2532039. [16] FRESHOUR SL, KIWALA S, COTTO KC, et al. Integration of the Drug-Gene Interaction Database (DGIdb 4.0) with open crowdsource efforts. Nucleic Acids Res. 2021;49(D1):D1144-D1151. [17] KURKI M I, KARJALAINEN J, PALTA P, et al. FinnGen provides genetic insights from a well-phenotyped isolated population. Nature. 2023;613(7944): 508-518. [18] CANNON M, STEVENSON J, STAHL K, et al. DGIdb 5.0: rebuilding the drug-gene interaction database for precision medicine and drug discovery platforms. Nucleic Acids Res. 2024;52(D1): D1227-D1235. [19] 李华燕, 苏培培, 王鑫, 等. 基于miR-146a调控TLR4/NF-κB信号通路探讨加味四妙散对尿酸钠晶体诱导的RAW264.7细胞炎症的作用机制[J]. 中国比较医学杂志,2025,35(6):41-49. [20] FINAN C, GAULTON A, KRUGER FA, et al. The druggable genome and support for target identification and validation in drug development. Sci Transl Med. 2017;9(383): eaag1166. [21] LI Y, HUANG C, XIE Y, et al. JUN and ATF3 in Gout: Ferroptosis-related potential diagnostic biomarkers. Heliyon. 2024;10(22):e39957. [22] CHOE J, PARK K, KIM S. Monosodium Urate in the Presence of RANKL Promotes Osteoclast Formation through Activation of c-Jun N-Terminal Kinase. Mediators Inflamm. 2015; 2015: 597512. [23] PAN J, ZHANG C, SHI M, et al. Ethanol extract of Liriodendron chinense (Hemsl.) Sarg barks attenuates hyperuricemic nephropathy by inhibiting renal fibrosis and inflammation in mice. J Ethnopharmacol. 2021;264:113278. [24] WU C, CHEN S, LIU Y, et al. Cynarin suppresses gouty arthritis induced by monosodium urate crystals. Bioengineered. 2022;13(5): 11782-11793. [25] EFFERTH T, OESCH F. The immunosuppressive activity of artemisinin-type drugs towards inflammatory and autoimmune diseases. Med Res Rev. 2021;41(6):3023-3061. [26] XU L, CHENG J, LU J, et al. Integrating network pharmacology and experimental validation to clarify the anti-hyperuricemia mechanism of cortex phellodendri in mice. Front Pharmacol. 2022;13:964593. [27] LIU M, YIN N, ZHU Y, et al. Associations between lipid-lowering drugs and urate and gout outcomes: a Mendelian randomization study. Front Endocrinol (Lausanne). 2024;15: 1398023. [28] LACHER SM, BRUTTGER J, KALT B, et al. HMG-CoA reductase promotes protein prenylation and therefore is indispensible for T-cell survival. Cell Death Dis. 2017;8(5):e2824. [29] SONG S, LOU Y, MAO Y, et al. Alteration of Gut Microbiome and Correlated Amino Acid Metabolism Contribute to Hyperuricemia and Th17-Driven Inflammation in Uox-KO Mice. Front Immunol. 2022;13:804306. [30] ZHAO L, WANG H, GAO H, et al. Increase in different peripheral effector T subsets in acute and chronic gout. Transpl Immunol. 2023;76:101763. [31] WU C, XU X, SHI Y, et al. Neutrophil Extracellular Trap Formation Model Induced by Monosodium Urate and Phorbol Myristate Acetate: Involvement in MAPK Signaling Pathways. Int J Mol Sci. 2024; 26(1):143. [32] LEE C, CHIANG S, KO AM, et al. ALPK1 phosphorylates myosin IIA modulating TNF-α trafficking in gout flares. Sci Rep. 2016;6:25740. [33] LI Y, DENG W, WU L, et al. Anti-Inflammatory Effects of Polyphenols from Plum (Prunus salicina Lindl) on RAW264.7 Macrophages Induced by Monosodium Urate and Potential Mechanisms. Foods. 2023;12(2):254. [34] WU M, ZHANG M, MA Y, et al. Chaetocin attenuates gout in mice through inhibiting HIF-1α and NLRP3 inflammasome-dependent IL-1β secretion in macrophages. Arch Biochem Biophys. 2019;670:94-103. [35] JOOSTEN LAB, ABDOLLAHI-ROODSAZ S, DINARELLO CA, et al. Toll-like receptors and chronic inflammation in rheumatic diseases: new developments. Nat Rev Rheumatol. 2016;12(6): 344-357. [36] SILVA CR, SARAIVA AL, ROSSATO MF, et al. What do we know about Toll-Like Receptors Involvement in Gout Arthritis? Endocr Metab Immune Disord Drug Targets. 2023;23(4):446-457. [37] DALBETH N, GOSLING AL, GAFFO A, et al. Gout. Lancet. 2021;397(10287): 1843-1855. [38] CHENG Z, HUANG M, LI W, et al. HECTD3 inhibits NLRP3 inflammasome assembly and activation by blocking NLRP3-NEK7 interaction. Cell Death Dis. 2024;15(1):86. [39] FENG W, ZHONG X, ZHENG X, et al. Study on the effect and mechanism of quercetin in treating gout arthritis. Int Immunopharmacol. 2022;111: 109112. [40] MIAO L, YUAN Z, ZHANG S, et al. Honokiol alleviates monosodium urate-induced gouty pain by inhibiting voltage-gated proton channels in mice. Inflammopharmacology. 2024;32(4): 2413-2425. [41] LI N, CHEN S, DENG W, et al. Kaempferol Attenuates Gouty Arthritis by Regulating the Balance of Th17/Treg Cells and Secretion of IL-17. Inflammation. 2023;46(5):1901-1916. [42] KIYANI MM, SADIQ S, SARFRAZ M, et al. Therapeutic Potential of Cinnamaldehyde Nanoparticles as an Antihyperuricemic, Anti-Inflammatory, and Antioxidant Agent Against Monosodium Urate Crystals-Mediated Gouty Arthritis in BALB/c Mice. Altern Ther Health Med. 2025;31(3):34-41. [43] LO C, LII C, HONG J, et al. Andrographolide inhibits IL-1β release in bone marrow-derived macrophages and monocyte infiltration in mouse knee joints induced by monosodium urate. Toxicol Appl Pharmacol. 2021;410:115341. [44] LI Y, FRENZ CM, LI Z, et al. Virtual and in vitro bioassay screening of phytochemical inhibitors from flavonoids and isoflavones against xanthine oxidase and cyclooxygenase-2 for gout treatment. Chem Biol Drug Des. 2013;81(4):537-544. [45] QU Y, FU Y, LIU Y, et al. The role of TRPV1 in RA pathogenesis: worthy of attention. Front Immunol. 2023;14:1232013. [46] ZHANG W, ZHANG Y, FAN J, et al. Pharmacological activity of capsaicin: Mechanisms and controversies (Review). Mol Med Rep. 2024; 29(3):38. [47] MORA-BOGA R. [Use of topical capsaicin for pain management: Review and evidence-based indications]. Semergen. 2025;51(1):102312. [48] KHAN BA, NAZ F, ALQAHTANI A, et al. A nanocomposite competent to overcome solubility and permeation issues of capsaicin and thiocolchicoside simultaneously in gout management: Fabrication of nanocubosomes. Saudi Pharm J. 2024;32(5):102050. [49] 刘大旭, 高晓波, 祁永华, 等. 杜仲有效成分β-萘黄酮对UVB诱导人皮肤成纤维细胞光老化保护作用研究[J]. 中医药学报,2016,44(3):41-43. [50] HOANG DH, SONG M, KOVALE LM, et al. Beta-naphthoflavone and doxorubicin synergistically enhance apoptosis in human lung cancer cells by inducing doxorubicin accumulation, mitochondrial ROS generation, and JNK pathway signaling. Biochem Biophys Res Commun. 2022;635:37-45. [51] LUBET RA, HECKMAN BM, DE FLORA SL, et al. Effects of 5,6-benzoflavone, indole-3-carbinol (I3C) and diindolylmethane (DIM) on chemically-induced mammary carcinogenesis: is DIM a substitute for I3C? Oncol Rep. 2011;26(3): 731-736. [52] LEE C, LIU KH, SINGER G, et al. High-Throughput Production of Diverse Xenobiotic Metabolites with Cytochrome P450-Transduced Huh7 Hepatoma Cell Lines. Drug Metab Dispos. 2022;50(9):1182-1189. [53] 房凤玲, 丁劲松. L-谷氨酸的胃肠道保护作用机制及药物开发[J]. 药学学报,2017,52(4):517-523. [54] 梁浩瀚, 崔伟, 叶来生, 等. 土茯苓及其活性成分防治痛风性关节炎作用与机制研究进展[J]. 中药材,2023,46(10):2628-2639. [55] SHI M, LIU X, MA X, et al. Study on the effect and mechanism of ZeXie decoction in treating MSU-induced acute gouty arthritis model through PI3K-AKT-mTOR signaling pathway. Int Immunopharmacol. 2025;150:114214. [56] YUE H, YANG Z, OU Y, et al. Tanshinones inhibit NLRP3 inflammasome activation by alleviating mitochondrial damage to protect against septic and gouty inflammation. Int Immunopharmacol. 2021;97:107819. [57] WU J, LUO Y, JIANG Q, et al. Coptisine from Coptis chinensis blocks NLRP3 inflammasome activation by inhibiting caspase-1. Pharmacol Res. 2019;147: 104348. [58] 王艺涵, 孙文宇, 郭玉杰, 等. 中医药治疗急性痛风性关节炎的证据图谱[J]. 中国中医急症, 2024,33(10):1699-1703. [59] 张翱, 王晓倩. 中医药治疗痛风的临床文献多源融合分析与用药规律挖掘[J]. 中国实验方剂学杂志,2025,31(8):244-249. [60] 钱爱, 黄传兵, 李明, 等. 基于多元数据挖掘探析专利中药复方治疗痛风性关节炎的用药规律[J]. 上海中医药杂志,2024,58(12):16-21. [61] 刘江腾, 阮智超, 张华, 等. 基于数据挖掘探讨中药复方专利治疗痛风的用药规律[J]. 中国医院用药评价与分析,2025,25(3):274-278. [62] 曾浩, 孙鹏程, 柴源, 等. 甲状腺功能和骨质疏松症的关联:欧洲人群全基因组数据分析[J]. 中国组织工程研究,2026,30(4):1019-1027. |
| [1] | Wu Zhilin, , He Qin, Wang Pingxi, Shi Xian, Yuan Song, Zhang Jun, Wang Hao . DYRK2: a novel therapeutic target for rheumatoid arthritis combined with osteoporosis based on East Asian and European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1569-1579. |
| [2] | Zhang Qingfeng, Wang Chaoyi, Yang Jingyan, Li Hanyu, Zhao Yuyang, Hao Huatao, Yu Dong. Potential target genes for spondylolisthesis: drugable genome analysis based on the European population-based biodatabase [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1592-1601. |
| [3] | Liu Hongtao, Wu Xin, Jiang Xinyu, Sha Fei, An Qi, Li Gaobiao. Causal relationship between age-related macular degeneration and deep vein thrombosis: analysis based on genome-wide association study data [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1602-1608. |
| [4] | Guo Ying, Tian Feng, Wang Chunfang. Potential drug targets for the treatment of rheumatoid arthritis: large sample analysis from European databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1549-1557. |
| [5] | Gao Zengjie, , Pu Xiang, Li Lailai, Chai Yihui, Huang Hua, Qin Yu. Increased risk of osteoporotic pathological fractures associated with sterol esters: evidence from IEU-GWAS and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1302-1310. |
| [6] | Liu Fengzhi, Dong Yuna, Tian Wenyi, Wang Chunlei, Liang Xiaodong, Bao Lin. Gene-predicted associations between 731 immune cell phenotypes and rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1311-1319. |
| [7] | Zhang Cuicui, Chen Huanyu, Yu Qiao, Huang Yuxuan, Yao Gengzhen, Zou Xu. Relationship between plasma proteins and pulmonary arterial hypertension and potential therapeutic targets [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1331-1340. |
| [8] | Zeng Hao, Sun Pengcheng, Chai Yuan, Huang Yourong, Zhang Chi, Zhang Xiaoyun. Association between thyroid function and osteoporosis: genome-wide data analysis of European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1019-1027. |
| [9] | Rong Xiangbin, , Zheng Haibo, Mo Xueshen, Hou Kun, Zeng Ping, . Plasma metabolites, immune cells, and hip osteoarthritis: causal inference based on GWAS data from European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1028-1035. |
| [10] | He Qiwang, , , Chen Bo, Liang Fuchao, Kang Zewei, Zhou Yuan, Ji Anxu, Tang Xialin, . Relationship between Alzheimer’s disease and sarcopenia and body mass index: analysis of GWAS datasets for European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1036-1046. |
| [11] | Ding Yu, Chen Jingwen, Chen Xiuyan, Shi Huimin, Yang Yudie, Zhou Meiqi, Cui Shuai, . Circulating inflammatory proteins and myocardial hypertrophy: large sample analysis of European populations from GWAS Catalog and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1047-1057. |
| [12] | Zhao Feifan, Cao Yujing. An artificial neural network model of ankylosing spondylitis and psoriasis shared genes and machine learning-based mining and validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 770-784. |
| [13] | Liu Chu, Qiu Boyuan, Tong Siwen, He Linyuwei, Chen Haobo, Ou Zhixue. A genetic perspective reveals the relationship between blood metabolites and osteonecrosis: an analysis of information from the FinnGen database in Finland [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 785-794. |
| [14] | Wei Bingqi, Zhang Xinyue, Ren Xingyue, Sun Jiahui, Chen Liu, Li Yijing, Qi Yifan, Wang Shangzeng. Zinc finger DHHC-type containing 2 emerges as a novel therapeutic target in osteoarthritis pathogenesis: genome-wide data analysis in European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7715-7723. |
| [15] | Wei Qiuyu, Yu Shaoyong, Zhou Zheyi, Wu Gang . Causal relationship between plasma metabolites and osteonecrosis: a large sample analysis based on genome-wide association study database and FinnGen database [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7732-7738. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||