Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (8): 1293-1298.doi: 10.3969/j.issn.2095-4344.2024
Previous Articles Next Articles
Exercises regulate bone marrow adipocytes derived from mesenchymal stem cells: roles and prospects
Qiao Xuesong1, Chen Ni2, Yang Fengying2, Niu Yanmei2
- 1Tianjin Chengjian University, Tianjin 300384, China; 2Department of Rehabilitation and Sports Medicine, Tianjin Medical University, Tianjin 300070, China
-
Received:2019-07-23Revised:2019-07-27Accepted:2019-09-17Online:2020-03-18Published:2020-01-23 -
Contact:Niu Yanmei, MD, Associate professor, Department of Rehabilitation and Sports Medicine, Tianjin Medical University, Tianjin 300070, China -
About author:Qiao Xuesong, Master, Lecturer, Tianjin Chengjian University, Tianjin 300384, China -
Supported by:the National Natural Science Foundation of China, No. 31671237 and 81601962; the Excellent Teacher Foundation of Tianjin Medical University, No. TYZY201822
CLC Number:
Cite this article
Qiao Xuesong, Chen Ni, Yang Fengying, Niu Yanmei. Exercises regulate bone marrow adipocytes derived from mesenchymal stem cells: roles and prospects [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1293-1298.
share this article
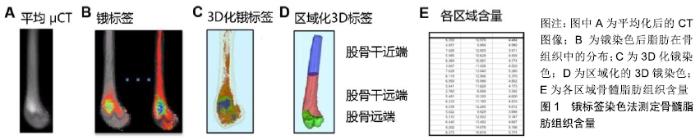
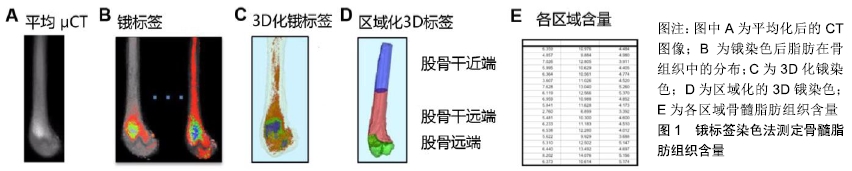
2.1 骨髓脂肪组织的来源、演变及功能 骨髓脂肪组织是存在于外周骨和远端骨中的特殊脂肪组织,来源于间充质干细胞群的分化。人类刚出生时,骨髓的主要成分是造血细胞,简称红髓。随着年龄增长,骨髓中的骨髓脂肪组织数量逐年增加,骨髓由红色变成黄色[1]。研究表明,骨髓脂肪组织在人群中的分布存在性别和年龄差异,成年男性椎骨、骶骨中的骨髓脂肪组织含量显著高于同年龄段女性[2]。然而55-65岁之间的女性椎骨中骨髓脂肪组织含量较高,这与绝经后雌激素失调或造血功能下降有关,雌激素失调能引起骨髓脂肪组织增多并可导致骨质疏松和骨折的发生[12],且伴随着皮下脂肪和腹部脂肪的聚集增多[2-3]。 作为骨髓中的重要组成部分,骨髓脂肪组织的功能与骨生成及造血有关。首先,骨髓脂肪组织含量与骨量呈负相关[7],且影响间充质干细胞与骨髓脂肪组织、成骨细胞之间的转化。间充质干细胞向骨髓脂肪组织的分化增多会影响骨的生成,而抑制骨髓脂肪组织则促进间充质干细胞向成骨细胞的分化。此外,骨髓脂肪组织还可刺激破骨细胞的分化和活性进而引起骨量减少[2]。研究表明,当机体缺乏甲状旁腺素受体时,骨髓脂肪组织可促进核因子κB受体激活因子的分泌从而增加破骨过程及骨的重吸收[2]。其次,骨髓脂肪组织也可影响造血过程,且它与造血细胞的关系具有两方面作用,一方面骨髓脂肪组织的增加引起造血干细胞数量减少,研究发现在胸椎和尾椎黄色骨髓中,由于骨髓脂肪组织与造血干细胞竞争生存空间导致造血干细胞的数量和活性降低[13];另一方面,骨髓脂肪组织对造血细胞的功能也有支持作用,过氧化物酶增殖活化受体γ激动剂通过促进骨髓脂肪组织含量增加,进而改善辐射损伤小鼠红系祖细胞和中性粒细胞的再生。总之,骨髓脂肪组织功能与骨量和造血细胞功能有关,但它们之间的具体调节机制还不甚清楚。 2.2 运动对骨髓脂肪组织的调节 众所周知,运动可促进骨质生成,并直接或间接参与骨组织的适应性改变来影响骨骼架构[4-6],但由于目前缺乏可重复的量化技术,因此探究运动对骨髓脂肪组织影响的研究很少。研究发现,高脂饮食情况下骨髓脂肪组织含量显著升高,而规律运动后骨髓脂肪组织含量则显著降低[14]。运动促进成骨作用可能与骨髓脂肪组织被抑制有关,这可能是由于热量从脂肪聚集处散失或间充质干细胞靶向分化为骨细胞而非脂肪细胞,抑或是两者的综合效应[14]。当给予小鼠罗格列酮(受体激动剂)干预后,小鼠股骨骨髓脂肪组织含量较对照组高出5倍,而运动干预可抑制正常小鼠股骨骨髓脂肪组织的聚集,且显著逆转了罗格列酮诱导的骨髓脂肪组织含量的增多[13],提示运动可通过促进间充质干细胞分化为成骨细胞而减少骨髓脂肪组织生成来满足对成骨作用的一种适应性反应[1-3,13]。 目前有关骨髓脂肪组织的生理学作用仍然不完全清楚,研究发现骨髓脂肪组织增加与骨生成受损和造血功能异常有关[14]。随年龄增长,人体内骨髓脂肪组织含量逐渐增多,至25岁时,骨髓腔内约70%都充满了骨髓脂肪组织[15]。 骨髓脂肪组织同时具有白色脂肪组织与棕色脂肪组织的特点[16-18]。白色脂肪组织是脂肪因子和炎症因子的重要来源,在机体能量代谢和心血管功能方面既有积极影响又有消极影响。腹腔内脏脂肪是白色脂肪组织典型的存在形式,它与能量代谢紊乱及心血管功能不全、皮质醇再生、成骨作用减低等密切相关[19]。棕色脂肪组织与白色脂肪组织的区别在于它含有一系列具有产热功能的蛋白,因此在机体产热方面发挥重要作用[20]。最近研究发现,相比于白色脂肪组织,骨髓脂肪组织可产生较多的脂联素,提示骨髓脂肪组织也具有内分泌功 能[19]。由于骨髓脂肪组织同时具有白色脂肪组织与棕色脂肪组织的特征,例如增加脂肪细胞中脂质存储、分泌脂肪因子、产生热量等功能。由于运动干预可影响白色脂肪组织和棕色脂肪组织的代谢,其可能是研究骨髓脂肪组织生理功能的一项重要干预措施。 2.3 运动对高脂饮食干预骨髓脂肪组织的调节 最新研究表明,短期高脂饲料喂养的啮齿动物中骨髓脂肪组织总量增加,进一步佐证了肥胖与骨髓脂肪组织之间存在确切的联系[19]。事实上,肥胖个体通常有更高的骨密度而少有骨质疏松性骨折的发生[7],然而骨髓脂肪组织又与低骨密度和骨折的风险增加有关[21],因此骨髓脂肪组织影响骨质量的机制尚不清楚。由于肥胖与骨密度增加和低骨折概率有关,在肥胖个体中骨髓脂肪组织可作为一个特殊的脂肪存储区,促进骨的合成代谢。 研究表明,运动改善肥胖所引起的疾病和死亡风险并不依赖于机体体质量的减轻和能量的消耗[22]。众所周知,运动可增强骨骼张力,减轻骨质疏松症[23-25]。研究发现,正常饮食和高脂饮食处理的小鼠经6周自主运动后均抑制了骨髓脂肪组织的积累[26]。与对照组相比,高脂饮食组小鼠骨小梁参数未发生变化,而运动组小鼠胫骨骨小梁的含量和厚度均显著增加。经短期实验研究发现,高脂饮食与运动干预均未改变小鼠的皮质骨参数[27]。因此推测,骨小梁比皮质骨可能更容易被运动影响。 2.4 运动对罗格列酮干预骨髓脂肪组织的调节 人体和动物模型研究均发现罗格列酮可促进骨髓脂肪组织的含量增加,其原因是过氧化物酶增殖活化受体γ可诱导间充质干细胞分化为脂肪细胞,从而促进骨髓脂肪组织含量增加[28]。体外研究发现,通过机械加载的方式模拟运动可抑制罗格列酮所致的间充质干细胞向脂肪细胞分化[29]。体内实验结果显示,罗格列酮干预的小鼠具有较强的生脂功能,而运动则能抑制罗格列酮介导骨髓脂肪组织的生成[28]。虽然罗格列酮显著增加实验小鼠胫骨近端的皮质骨疏松度,但并未影响骨密度和骨小梁的含量,而运动可增加骨小梁含量及其厚度,进一步证实了运动对骨骼的有益影响。 2.5 运动与机械刺激对骨密度及骨量的调节 运动的健康促进作用可归因于运动对神经系统调节的改善并可提高心血管系统的健康,减少炎症发生并降低患慢性疾病的风险。运动对机体最直接、显著的效应是增加肌肉、骨骼的质量[30]。通过对肌肉、骨骼持续施加负荷促进其合成代谢。事实上,骨组织可通过增加负荷促进骨重塑的方式来维持骨的承受能力。在细胞水平上,细胞外刺激和核内遗传物质共同介导机械刺激诱导的骨重塑,促进骨、软骨及肌源性生长因子的转录,从而抑制脂肪生成,这些调控的信号分子在Wnt/β-catenin信号通路传导中可上调Runx2和SOX9基因表达,促进成骨和软骨的生成[31]。机械刺激通过抑制脂肪生成和骨吸收途径既能调节肌肉骨骼的质量,也参与肌肉骨骼的合成代谢。 相比运动而言,低体力活动(长期卧床休息、微重力、体力活动减少)在很大程度上降低机体的机械性刺激,导致肌肉骨骼组织内稳态平衡紊乱,加速肌肉、肌腱、韧带的降解和骨质吸收过程,以上反应进一步诱发骨质疏松和骨折的风险。对于骨骼组织而言,长期低负荷效应是否促进骨髓脂肪组织的合成尚不完全清楚。然而,有研究显示卧床时间增加能够促进骨髓脂肪组织的合 成[32]。在缺乏机械刺激时,过氧化物酶增殖活化受体γ和核因子κB配体的受体激活因子在骨组织中表达增加,促进破骨细胞介导的骨吸收过程[13]。 2.6 运动对骨和脂肪前体细胞的影响 研究发现,间充质干细胞和早期未分化的祖细胞对机械刺激可表现出一定反应[33]。骨髓中的骨细胞、成骨细胞、前成骨细胞(MC3T3)是已知的机械感应细胞,有助于机械信号的传导并促进成骨基因的表达[34-36]。虽然机械刺激信号可以通过作用于骨细胞和间充质干细胞中RANKL的表达来抑制破骨细胞的生成及后期骨的重吸收,但对骨重建的影响还需通过激活Wnt/β-catenin信号通路促进间充质干细胞向成骨细胞的分化[37-39]。间充质干细胞的定向分化易受环境因素的影响,例如外源性的机械刺激、局部底物水平和信号因子的级联反应[40-41]。这些因素通过促进骨、软骨及肌肉组织而不是促进脂肪组织的形成,更好地维持机体骨骼-肌肉稳态。运动不仅能够促进间充质干细胞增殖分化,也可通过上调碱性磷酸酶活性提升成骨潜能,促使脂滴和脂肪细胞直径缩小[42]。研究显示,运动组小鼠体内脂肪组织减少与前成骨细胞增加有关[43]。 2.7 运动对骨髓脂肪组织棕色化的影响 运动开始后骨骼肌中脂质的摄入和氧化会逐渐增加。随着运动强度不断增大,机体对碳水化合物的摄取增加而对脂肪的利用减少,长时间的耐力训练却增大了机体对脂质的利 用[44-45]。在对冬眠动物和婴儿的研究中发现,棕色脂肪组织不通过颤栗性的生热方式促进能量消耗,并且在成年人的白色脂肪组织中发现了可诱导为棕色脂肪组织的米色脂肪[46]。在寒冷或β-肾上腺素刺激下,这些米色脂肪细胞可高表达线粒体解偶联蛋白,并且促进多泡脂肪细胞的形成,这些变化都是棕色脂肪的表现特征[47]。Irisin,一种运动诱导的肌源性激素,也可以激活线粒体解偶联蛋白并促进白色脂肪组织棕色化[48]。过氧化物酶增殖活化受体γ辅激活因子1α(peroxisome proliferator-activated receptor gamma co-activator 1α,PGC1α)可激活Irisin,在能量摄入和运动消耗不变的前提下,PGC1α 转基因小鼠展示了具有增加能量消耗的作用。总的来说,不同脂肪细胞为满足能量需求可进行相互转换,运动可以使体内存储的白色脂肪组织发生棕色化[49],因此可以设想运动也可能促使骨髓脂肪组织发生棕色化。事实上运动增加了骨组织中线粒体解偶联蛋白mRNA的表达[50-52],而线粒体解偶联蛋白存在于哺乳动物棕色脂肪组织线粒体内膜内上,因此线粒体解偶联蛋白可作为棕色脂肪组织的特异性标记物。运动使线粒体解偶联蛋白增加可能预示骨髓脂肪组织发生棕色化,但这还需要进一步确认。研究发现,运动诱导线粒体解偶联蛋白表达量增加可能与Irisin的增加有关,虽然Irisin在运动中的生理作用还不清楚[53]。最近研究表明Irisin除了对脂肪细胞有影响外,可能对骨组织也有直接影响[54],因此Irisin在骨骼健康方面的研究仍是一个热点。 2.8 骨髓脂肪组织的测量和定量方法 早期为了量化运动对骨髓脂肪组织的影响,研究者采用组织学方法检测脂肪细胞大小、骨髓脂肪组织的含量、与周围骨内膜的结合情况、细胞环境及分泌因子[55-59]。由于脂肪细胞体积较大,且细胞骨架缺乏刚性,因此在纯化和提取的过程中致使细胞易出现溶解。然而,最新采用的研究方法已显著避免了这种影响[60],在骨髓过滤过程中(孔隙大小为150 μm)只允许脂肪细胞及更小的细胞内容物通过,随后对悬浮脂肪进行离心,这种处理方式的可操作性较强[61-62]。然而,脂质和蛋白质仍能黏附在管壁从而堵塞仪器[63],而蛋白质荧光探针标记的应用使流式细胞仪中的抗体具有高亲和力,这使得对骨髓脂肪组织和骨髓中其他细胞的鉴别愈加具体。 为了进一步加深对骨髓脂肪组织的了解,目前已开发出新的原位可视化成像技术用以量化骨髓脂肪组织。虽然质子核磁共振谱已成功地量化人类椎骨中的骨髓脂肪组织,但仍不能用于实验动物研究[64]。MRI结合微型CT骨髓密度测量技术为测量啮齿动物骨组织中的骨髓脂肪组织提供了方向,该技术主要对骨进行锇染色(显示骨组织内脂肪的存在情况及其形态)并结合微型CT进行成像[65-66]。简而言之,股骨进行脱钙,在四氧化锇中浸泡并放入重铬酸钾中,随后使用微型CT扫描进行骨骼成像(分辨率10 μm×10 μm×10 μm)。由于锇密度比骨密度更大,因此骨密度可被定量。定量后的股骨被细分为不同的解剖区域(股骨干近端、股骨干远端和股骨远端),不同区域中锇含量再次被量化,见图1[65]。这种技术可重复量化骨髓脂肪组织并使其可视化,可用于量化饮食、运动所导致的骨髓脂肪组织含量改变。 "
| [1] BUKOWSKA J, FRAZIER T, SMITH S, et al. Bone Marrow Adipocyte Developmental Origin and Biology. Curr Osteoporos Rep. 2018;16(3): 312-319. [2] SEBO ZL, RENDINA-RUEDY E, ABLES GP, et al. Bone Marrow Adiposity: Basic and Clinical Implications. Endocr Rev. 2019;40(5): 1187-1206. [3] AMBROSI TH, SCHULZ TJ. The emerging role of bone marrow adipose tissue in bone health and dysfunction. J Mol Med (Berl). 2017;95(12): 1291-1301. [4] SAÑUDO B, DE HOYO M, DEL POZO-CRUZ J, et al. A systematic review of the exercise effect on bone health: the importance of assessing mechanical loading in perimenopausal and postmenopausal women. Menopause. 2017;24(10):1208-1216. [5] XU J, LOMBARDI G, JIAO W, et al. Effects of Exercise on Bone Status in Female Subjects, from Young Girls to Postmenopausal Women: An Overview of Systematic Reviews and Meta-Analyses. Sports Med. 2016;46(8):1165-1182. [6] KEMMLER W, ENGELKE K, VON STENGEL S. Long-Term Exercise and Bone Mineral Density Changes in Postmenopausal Women--Are There Periods of Reduced Effectiveness?J Bone Miner Res. 2016; 31(1): 215-222. [7] TOSA I, YAMADA D, YASUMATSU M, et al. Postnatal Runx2 deletion leads to low bone mass and adipocyte accumulation in mice bone tissues. Biochem Biophys Res Commun. 2019;516(4):1229-1233. [8] STYNER M, THOMPSON WR, GALIOR K, et al. Bone marrow fat accumulation accelerated by high fat diet is suppressed by exercise. Bone. 2014;64:39-46. [9] DOUCETTE CR, HOROWITZ MC, BERRY R, et al. A High Fat Diet Increases Bone Marrow Adipose Tissue (MAT) But Does Not Alter Trabecular or Cortical Bone Mass in C57BL/6J Mice. J Cell Physiol. 2015;230(9):2032-2037. [10] CHA YJ, KOO JS. Roles of omental and bone marrow adipocytes in tumor biology. Adipocyte. 2019;8(1):304-317. [11] BREDELLA MA, FAZELI PK, DALEY SM, et al. Marrow fat composition in anorexia nervosa. Bone. 2014;66:199-204. [12] HOROWITZ MC, TOMMASINI SM. Fat and Bone: PGC-1α Regulates Mesenchymal Cell Fate during Aging and Osteoporosis. Cell Stem Cell. 2018;23(2):151-153. [13] LI Z, HARDIJ J, BAGCHI DP, et al. Development, regulation, metabolism and function of bone marrow adipose tissues. Bone. 2018;110:134-140. [14] PICKE AK, SYLOW L, MØLLER LLV, et al. Differential effects of high-fat diet and exercise training on bone and energy metabolism. Bone. 2018; 116:120-134. [15] INGLÉS M, SERRA-AÑÓ P, GAMBINI J, et al. Active paraplegics are protected against exercise-induced oxidative damage through the induction of antioxidant enzymes. Spinal Cord. 2016;54(10):830-837. [16] HOROWITZ MC, BERRY R, HOLTRUP B, et al. Bone marrow adipocytes. Adipocyte. 2017;6(3):193-204. [17] ABDULLAHI A, CHEN P, STANOJCIC M, et al. IL-6 Signal From the Bone Marrow is Required for the Browning of White Adipose Tissue Post Burn Injury. Shock. 2017;47(1):33-39. [18] HARDOUIN P, RHARASS T, LUCAS S. Bone Marrow Adipose Tissue: To Be or Not To Be a Typical Adipose Tissue? Front Endocrinol (Lausanne). 2016;7:85. [19] BREWER CB, BENTLEY JP, DAY LB, et al. Resistance exercise and naproxen sodium: effects on a stable PGF2α metabolite and morphological adaptations of the upper body appendicular skeleton. Inflammopharmacology. 2015;23(6):319-327. [20] MONTANARI T, POŠĆIĆ N, COLITTI M. Factors involved in white-to-brown adipose tissue conversion and in thermogenesis: a review. Obes Rev. 2017;18(5):495-513. [21] YU EW, GREENBLATT L, EAJAZI A, et al. Marrow adipose tissue composition in adults with morbid obesity. Bone. 2017;97:38-42. [22] TUAZON MA, CAMPBELL SC, KLEIN DJ, et al. Effects of ovariectomy and exercise training intensity on energy substrate and hepatic lipid metabolism, and spontaneous physical activity in mice. Metabolism. 2018;83:234-244. [23] MOREIRA LD, OLIVEIRA ML, LIRANI-GALVÃO AP, et al. Physical exercise and osteoporosis: effects of different types of exercises on bone and physical function of postmenopausal women. Arq Bras Endocrinol Metabol. 2014;58(5):514-522. [24] HOWE TE, SHEA B, DAWSON LJ, et al. Exercise for preventing and treating osteoporosis in postmenopausal women. Cochrane Database Syst Rev. 2011;(7):CD000333. [25] Benedetti MG, Furlini G, Zati A, et al. The Effectiveness of Physical Exercise on Bone Density in Osteoporotic Patients. Biomed Res Int. 2018;2018:4840531. [26] RIDEOUT TC, MOVSESIAN C, TSAI YT, et al. Maternal Phytosterol Supplementation during Pregnancy and Lactation Modulates Lipid and Lipoprotein Response in Offspring of apoE-Deficient Mice. J Nutr. 2015; 145(8):1728-1734. [27] VIRTANEN KS, NIKKINEN P, LINDROTH L, et al. Myocardial fatty acid utilisation during exercise induced ischemia in patients with coronary artery disease. Nuklearmedizin. 2002;41(3):157-161. [28] BEEKMAN KM, VELDHUIS-VLUG AG, VAN DER VEEN A, et al. The effect of PPARγ inhibition on bone marrow adipose tissue and bone in C3H/HeJ mice. Am J Physiol Endocrinol Metab. 2019;316(1):E96-E105. [29] DAVID V, MARTIN A, LAFAGE-PROUST MH, et al. Mechanical loading down-regulates peroxisome proliferator-activated receptor gamma in bone marrow stromal cells and favors osteoblastogenesis at the expense of adipogenesis. Endocrinology. 2007;148(5):2553-2562. [30] BORGES LDA S, DERMARGOS A, DA SILVA JUNIOR EP, et al. Melatonin decreases muscular oxidative stress and inflammation induced by strenuous exercise and stimulates growth factor synthesis. J Pineal Res. 2015;58(2):166-172. [31] FERRARESI C, PARIZOTTO NA, PIRES DE SOUSA MV, et al. Light-emitting diode therapy in exercise-trained mice increases muscle performance, cytochrome c oxidase activity, ATP and cell proliferation. J Biophotonics. 2015;8(9):740-754. [32] LEWIS NC, BAILEY DM, DUMANOIR GR, et al. Conduit artery structure and function in lowlanders and native highlanders: relationships with oxidative stress and role of sympathoexcitation. J Physiol. 2014;592(5): 1009-1024. [33] EVERSON CA, FOLLEY AE, TOTH JM. Et al. Chronically inadequate sleep results in abnormal bone formation and abnormal bone marrow in rats. Exp Biol Med (Maywood). 2012;237(9):1101-1109. [34] XIAO Z, QUARLES LD. Physiological mechanisms and therapeutic potential of bone mechanosensing. Rev Endocr Metab Disord. 2015; 16(2):115-129. [35] LI Y, GE C, LONG JP, et al. Biomechanical stimulation of osteoblast gene expression requires phosphorylation of the RUNX2 transcription factor. J Bone Miner Res. 2012;27(6):1263-1274. [36] GU GL, KURATA K, CHEN Z, et al. Osteocyte: a Cellular Basis for Mechanotransduction in Bone. Journal of Biomechanical Science and Engineering. 2007; 2(4): 150-165. [37] NIU Q, LI F, ZHANG L, et al. Role of the Wnt/β-catenin signaling pathway in the response of chondrocytes to mechanical loading. Int J Mol Med. 2016;37(3):755-762. [38] DUAN P, BONEWALD LF. The role of the wnt/β-catenin signaling pathway in formation and maintenance of bone and teeth. Int J Biochem Cell Biol. 2016;77(Pt A):23-29. [39] ROBINSON JA, CHATTERJEE-KISHORE M, YAWORSKY PJ, et al. Wnt/beta-catenin signaling is a normal physiological response to mechanical loading in bone. J Biol Chem. 2006;281(42):31720-31728. [40] MACALUSO F, MORICI G, CATANESE P, et al. Effect of conjugated linoleic acid on testosterone levels in vitro and in vivo after an acute bout of resistance exercise. J Strength Cond Res. 2012;26(6):1667-1674. [41] STEWARD AJ, KELLY DJ. Mechanical regulation of mesenchymal stem cell differentiation. J Anat. 2015;227(6):717-731. [42] CAWTHORN WP, SCHELLER EL, LEARMAN BS, et al. Bone marrow adipose tissue is an endocrine organ that contributes to increased circulating adiponectin during caloric restriction. Cell Metab. 2014; 20(2):368-375. [43] MENUKI K, MORI T, SAKAI A, et al. Climbing exercise enhances osteoblast differentiation and inhibits adipogenic differentiation with high expression of PTH/PTHrP receptor in bone marrow cells. Bone. 2008; 43(3):613-620. [44] JOHNSON ML, ZARINS Z, FATTOR JA, et al. Twelve weeks of endurance training increases FFA mobilization and reesterification in postmenopausal women. J Appl Physiol (1985). 2010;109(6): 1573-1581. [45] FIKENZER K, FIKENZER S, LAUFS U, et al. Effects of endurance training on serum lipids. Vascul Pharmacol. 2018;101:9-20. [46] HOJSAK I, COLOMB V, BRAEGGER C, et al. ESPGHAN Committee on Nutrition Position Paper. Intravenous Lipid Emulsions and Risk of Hepatotoxicity in Infants and Children: a Systematic Review and Meta-analysis. J Pediatr Gastroenterol Nutr. 2016;62(5):776-792. [47] STEVENSON EJ, THELWALL PE, THOMAS K, et al. Dietary glycemic index influences lipid oxidation but not muscle or liver glycogen oxidation during exercise. Am J Physiol Endocrinol Metab.2009;296(5):E1140-1147. [48] ZHANG J, VALVERDE P, ZHU X, et al. Exercise-induced irisin in bone and systemic irisin administration reveal new regulatory mechanisms of bone metabolism. Bone Res. 2017;5:16056. [49] MOTIANI P, TEUHO J, SAARI T, et al. Exercise training alters lipoprotein particles independent of brown adipose tissue metabolic activity. Obes Sci Pract. 2019;5(3):258-272. [50] QIU S, BOSNYÁK E, TREFF G, et al. Acute exercise-induced irisin release in healthy adults: Associations with training status and exercise mode. Eur J Sport Sci. 2018;18(9):1226-1233. [51] NEDERGAARD J, CANNON B. The browning of white adipose tissue: some burning issues. Cell Metab. 2014;20(3):396-407. [52] LEHNIG AC, STANFORD KI. Exercise-induced adaptations to white and brown adipose tissue. J Exp Biol. 2018;221(Pt Suppl 1): jeb161570. [53] FOX J, RIOUX BV, GOULET EDB, et al. Effect of an acute exercise bout on immediate post-exercise irisin concentration in adults: A meta-analysis. Scand J Med Sci Sports. 2018;28(1):16-28. [54] MAALOUF GE, EL KHOURY D. Exercise-Induced Irisin, the Fat Browning Myokine, as a Potential Anticancer Agent. J Obes. 2019;2019: 6561726. [55] ACKERT-BICKNELL CL, SHOCKLEY KR, HORTON LG, et al. Strain- specific effects of rosiglitazone on bone mass, body composition, and serum insulin-like growth factor-I. Endocrinology. 2009;150(3):1330-1340. [56] BIELOHUBY M, MATSUURA M, HERBACH N, et al. Short-term exposure to low-carbohydrate, high-fat diets induces low bone mineral density and reduces bone formation in rats. J Bone Miner Res. 2010; 25(2):275-284. [57] SPATZ JM, ELLMAN R, CLOUTIER AM, et al. Sclerostin antibody inhibits skeletal deterioration due to reduced mechanical loading. J Bone Miner Res. 2013;28(4):865-874. [58] ROSEN CJ, ACKERT-BICKNELL CL, ADAMO ML, et al. Congenic mice with low serum IGF-I have increased body fat, reduced bone mineral density, and an altered osteoblast differentiation program. Bone. 2004; 35(5):1046-1058. [59] XIA L, LIU J, SUN Y, et al. Rosiglitazone Improves Glucocorticoid Resistance in a Sudden Sensorineural Hearing Loss by Promoting MAP Kinase Phosphatase-1 Expression. Mediators Inflamm. 2019;2019: 7915730. [60] NAVEIRAS O, NARDI V, WENZEL PL, et al. Bone-marrow adipocytes as negative regulators of the haematopoietic microenvironment. Nature. 2009;460(7252):259-263. [61] ROBEY PG, KUZNETSOV SA, RIMINUCCI M, et al. Bone marrow stromal cell assays: in vitro and in vivo. Methods Mol Biol. 2014;1130: 279-293. [62] MCDANIEL JS, ANTEBI B, PILIA M, et al. Quantitative Assessment of Optimal Bone Marrow Site for the Isolation of Porcine Mesenchymal Stem Cells. Stem Cells Int. 2017;2017:1836960. [63] LI X, KUO D, SCHAFER AL, et al. Quantification of vertebral bone marrow fat content using 3 Tesla MR spectroscopy: reproducibility, vertebral variation, and applications in osteoporosis. J Magn Reson Imaging. 2011;33(4):974-979. [64] PAGNOTTI GM, STYNER M. Exercise Regulation of Marrow Adipose Tissue. Front Endocrinol (Lausanne). 2016;7:94. [65] SCHELLER EL, TROIANO N, VANHOUTAN JN, et al. Use of osmium tetroxide staining with microcomputerized tomography to visualize and quantify bone marrow adipose tissue in vivo. Methods Enzymol. 2014; 537:123-139. [66] PAI VM, KOZLOWSKI M, DONAHUE D, et al. Coronary artery wall imaging in mice using osmium tetroxide and micro-computed tomography (micro-CT). J Anat. 2012;220(5):514-524. |
| [1] | Shen Jinbo, Zhang Lin. Micro-injury of the Achilles tendon caused by acute exhaustive exercise in rats: ultrastructural changes and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1190-1195. |
| [2] | Wang Mengting, Gu Yanping, Ren Wenbo, Qin Qian, Bai Bingyi, Liao Yuanpeng. Research hotspots of blood flow restriction training for dyskinesia based on visualization analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1264-1269. |
| [3] | Wang Yongsheng, Wu Yang, Li Yanchun. Effect of acute high-intensity exercise on appetite hormones in adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1305-1312. |
| [4] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [5] | Zhao Xiang, Wei Cuilan, Zhang Yeting. Neurogenesis and neuroinflammation under exercise: alteration and regulation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 813-820. |
| [6] | Chen Ziyang, Pu Rui, Deng Shuang, Yuan Lingyan. Regulatory effect of exosomes on exercise-mediated insulin resistance diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 4089-4094. |
| [7] | Jiang Xiaoyan, Zhu Haifei, Lin Haiqi, Lin Wentao. Cold therapy promotes self-limited recovery of delayed-onset muscle soreness [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3609-3613. |
| [8] | Bai Shengchao, Gao Yang, Wang Bo, Li Junping, Wang Ruiyuan. Dynamic changes of mitochondrial function of the skeletal muscle after acupuncture intervention in rats with heavy load exercise-induced injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3648-3653. |
| [9] | Lu Jie, Li Xue, Wang Lu, Fan Jia, Zhang Yeting, Lu Xiaobin, Yuan Qiongjia. Effects of different-intensity swimming exercises on spatial learning and memory ability and the expression of Orexin A in the rat cerebellum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3697-3703. |
| [10] | Wang Zhen, Lin Haiqi, He Fei, Lin Wentao. Exercise activates skeletal muscle satellite cells: exercise prevention and treatment for age-related sarcopenia and muscle injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3752-3759. |
| [11] | Wang Chaoge, Weng Xiquan, Lin Baoxuan, Chen Lina, Xu Guoqin. Exercises under cold exposure change fat type and function in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3162-3167. |
| [12] | Liu Bo, Chen Xianghe, Yang Kang, Sun Changliang, Yu Huilin, Lu Pengcheng. Epigenetic reprogramming and exercise regulation of bone metabolism disorders [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3210-3218. |
| [13] | Wang Jing, Wu Jiangbo. Irisin mediates protective effects on failing heart via integrin alpha V receptors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3219-3225. |
| [14] | Lu Yi, Deng Wenchong. Regulation and difference of different exercise styles on brain structure and cognitive function [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3252-3258. |
| [15] | Lin Haiqi, Chen Liang, Tang Lu, Weng Xiquan, Lin Wentao. Significance of urinary proteomics assessing pathological changes in the body [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3259-3266. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||