Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (7): 1123-1129.doi: 10.3969/j.issn.2095-4344.2018
Previous Articles Next Articles
Tissue repair using mesenchymal stem cells via mitochondrial transfer
Wang Zhangling1, Yu Limei1, Zhao Chunhua1, 2
- 1Key Laboratory of Cell Engineering in Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China; 2Center for Tissue Engineering Research, Institute of Basic Medical Sciences of Chinese Academy of Medical Sciences & School of Basic Medicine of Peking Union Medical College, Beijing 100730, China
-
Received:2019-08-08Revised:2019-08-10Accepted:2019-09-17Online:2020-03-08Published:2020-01-20 -
Contact:Zhao Chunhua, MD, Chief physician, Key Laboratory of Cell Engineering in Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China; Center for Tissue Engineering Research, Institute of Basic Medical Sciences of Chinese Academy of Medical Sciences & School of Basic Medicine of Peking Union Medical College, Beijing 100730, China Yu Limei, MD, Chief physician, Key Laboratory of Cell Engineering in Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
About author:Wang Zhangling, Key Laboratory of Cell Engineering in Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81260507; the Major Project of Guizhou Province, No. [2011]6002
CLC Number:
Cite this article
Wang Zhangling, Yu Limei, Zhao Chunhua. Tissue repair using mesenchymal stem cells via mitochondrial transfer [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1123-1129.
share this article
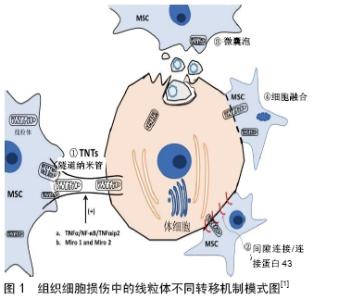
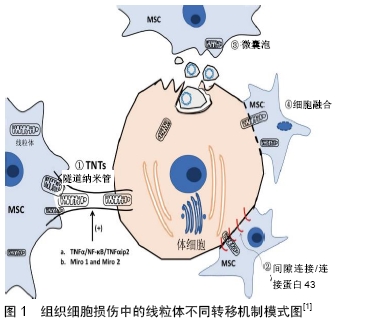
2.1 组织修复中间充质干细胞的常见作用机制 总的来说,间充质干细胞的治疗效果可归因于3个关键作用机制:第一是“归巢”,静脉滴注而全身应用的间充质干细胞,由于细胞因子的趋化或招募作用,随血循环迁移到组织损伤部位而发挥治疗作用。已知间充质干细胞的迁移、归巢及定植过程中,涉及趋化因子SDF-1及其受体CXCR4等信号途径的转导,也涉及血管细胞黏附分子1和G蛋白偶联受体的信号传导,且与局部组织中的炎症因子、免疫细胞、血管内皮细胞等相互作用;第二是分化成多种特定功能细胞,替换受损的组织细胞,恢复组织结构与功能;第三是分泌多种生物活性物质,影响局部和全身的病理生理过程[8],减轻炎症损伤,促进再生修复。因此,间充质干细胞被应用于多种疾病治疗,如脑卒中[9]、心肌梗死[10]、急性肾功能衰竭[11]、肺动脉高压[12]、肝衰竭等[11]。此外,在对骨髓、人脐带等来源的间充质干细胞研究中,还证明了间充质干细胞可通过促血管生成、功能细胞增殖等作用,减轻心脏、肝脏、肾脏、肺脏、骨髓、视网膜和脑等组织损伤[13]。另外,间充质干细胞来源外泌体也能明显保护上述多种组织器官并减轻组织器官损伤[14] 。 2.2 间充质干细胞组织修复的新机制:线粒体转移 组织损伤初期,线粒体损伤会加重组织损伤的严重程度[15],而线粒体损伤与活性氧簇累积、钙离子失衡和能量供给不足有关[16]。健康线粒体复制、分裂、融合、降解及细胞间转移的生理特性为消除和替换受损线粒体提供了可能[17]。诸多研究表明,细胞间细胞器的通信有助于维持细胞稳态,特别是参与能量代谢和生物调节等生物功能的维持[18-19]。细胞之间的线粒体转移可在体外和体内发生,有趣的是干细胞可将其自身的正常线粒体转移到其周围线粒体受损的靶细胞,从而影响受损细胞的增殖、分化、组织稳态、发育和衰老等过程[20]。间充质干细胞的线粒体转移已在心血管、呼吸、神经和肾脏系统中得到广泛研究,间充质干细胞可将线粒体转移至受损靶细胞的首次证据来自对人骨髓间充质干细胞与人A549 rho上皮细胞的共培养研究,发现了来自供体间充质干细胞的线粒体DNA,而A549 rho细胞内并无此线粒体DNA[21]。通过DsRed2标记技术也证实了上皮细胞可通过转移线粒体而维持正常的细胞呼吸功能[22]。众所周知,功能失调的线粒体可导致细胞损伤和细胞凋亡,由此推断,将健康的线粒体转移入线粒体功能失调的细胞中,似乎是减轻疾病状况的有效手段。间充质干细胞的线粒体转移以替代患病细胞/组织中功能失调的线粒体,为线粒体功能障碍疾病的治疗奠定了重要理论基础。此后多种实验不断证实,不同组织来源的间充质干细胞确实能够将线粒体转移到角膜上皮细胞[23]、皮肤成纤维细胞[24]、心肌细胞等各种受损细胞[25]。在心血管、呼吸、神经和肾脏系统中的体内外实验证实来自骨髓间充质干细胞、诱导多能干细胞的线粒体转移在细胞修复和再生中发挥关键作用[1],并在相关受体细胞和应激条件下,观察到了不同的线粒体转移模式,如在心肌细胞[26]、肺细胞[27]、肾细胞[28]、角膜上皮细胞[23]、大脑皮质细胞 中[29],急性缺血损伤条件下线粒体转移主要通过隧道纳米管实现;免疫炎症条件下,线粒体转移则主要通过微囊泡形成实现[30];在心脏细胞中观察到祖细胞的细胞融合和重编程[31]。间充质干细胞的此种线粒体转移过程不仅通过直接的细胞间连接,还可通过间接分泌作用来拯救受损的体细胞。间充质干细胞将其正常线粒体捐献给受损细胞以抵抗氧化应激,改善细胞代谢状态的同时,还吞噬和降解来自受损体细胞的线粒体,即促进受损细胞的增殖,也增强了抗凋亡能力[32]。因此,细胞间线粒体转移为使用干细胞作为载体治愈线粒体功能障碍疾病提供了一种新策略和新思路。 2.2.1 线粒体转移机制 线粒体转移是指源于干细胞的健康线粒体可通过隧道纳米管形成、间隙连接、微囊泡、细胞融合和分离等几种不同模式转移到线粒体功能障碍的细胞中,恢复其有氧呼吸功能而发挥治疗作用[33],见图1。SPEES等[22]在研究间充质干细胞将功能正常线粒体转移至线粒体功能障碍的细胞时,证明了隧道纳米管和微囊泡形成介导线粒体转移的机制,且提出细胞融合并非线粒体转移的主要机制。隧道纳米管是通过细胞之间的连接,构建运输细胞组分(细胞质、离子、脂滴病毒、细菌病原体、细胞器等)的通道,以此作为间充质干细胞与受损细胞之间线粒体转移的主要方式[34]。隧道纳米管广泛存在于血管平滑肌细胞、内皮细胞、间充质干细胞和癌细胞等多种细胞中,通过隧道纳米管的交换功能,可以使线粒体受损的细胞或组织一定程度地恢复线粒体功能,从而发挥间充质干细胞对疾病的治疗效应[18],改善受损细胞和组织的能量供应,对于血运丰富且能量敏感的脑、心脏和肾脏等组织可能具有重要意义。Miro1是一种与微管中线粒体运动至关重要的钙敏感衔接蛋白,它在隧道纳米管与间充质干细胞转移线粒体中发挥重要作用,间充质干细胞中Miro1的过表达可增强线粒体转移和修复受损肺上皮细胞,而敲除Miro1则可消除这一效应[21]。Miro1在其他辅助蛋白如TRAK 1和TRAK 2,Myo 10和Myo 19的帮助下将线粒体与KLF 5驱动蛋白结合,从而帮助线粒体在隧道纳米管上运输并调节其运动。用细胞松弛素B处理诱导多能干细胞时,由于抑制了肌动蛋白聚合和隧道纳米管的形成,可抑制诱导多能干细胞和心肌细胞之间线粒体转移的发生。通过诱导多能干细胞和骨髓间充质干细胞的比较发现,Miro1和肿瘤坏死因子α诱导蛋白2(recombinant tumor necrosis factor alpha induced protein 2,TNFαip2)过表达的诱导多能干细胞呈现更多的隧道纳米管形成和线粒体转移,且赋予诱导多能干细胞高效率的线粒体转移[35]。在隧道纳米管形成期间,NF-κB亚基p-IκB的磷酸化水平随着肿瘤坏死因子α和TNFαip2水平的升高而显著增加,而NF-κB抑制剂SC-514显著减弱隧道纳米管的形成,表明NF-κB介导的信号转导机制在隧道纳米管形成中具有重要作用[36],NF-κB是与炎症密切相关的重要信号分子,这也提示在炎症反应条件下,可能有利于间充质干细胞通过隧道纳米管机制发生线粒体转移而产生抗炎的治疗效应,值得深入探讨。现有的证据表明,线粒体转移主要通过隧道纳米管和微囊泡的形成而不是通过间隙连接所介导,这也可能是因为大多数研究是在体外进行的,使得隧道纳米管形成容易被观察到[36]。在骨髓间充质干细胞治疗气道灌注大肠杆菌脂多糖所致的急性肺损伤小鼠模型实验研究中[37],连接蛋白43表达功能丧失的骨髓间充质干细胞不能黏附到肺泡上皮,也不能转移线粒体,可见连接蛋白43对于线粒体从骨髓间充质干细胞转移至肺泡上皮细胞进而增加上皮ATP具有重要作用[38]。还有研究在比较骨髓、肺实质、支气管肺泡灌洗液不同来源间充质干细胞与支气管上皮细胞线粒体转移模式时发现,用细胞松弛素B和Dynasore抑制剂分别阻断隧道纳米管和微囊泡形成,可以使线粒体转移现象完全消除,且不受连接蛋白43抑制剂Gap26的影响,而当所有抑制剂化合物加在一起时却使线粒体转移现象显著减弱,表明从骨髓、肺组织和支气管肺泡灌洗液中分离的间充质干细胞向支气管上皮细胞提供细胞质内容物和线粒体,这种快速单向的线粒体转移涉及了微泡、隧道纳米管在内的多种机制,而非连接蛋白43间隙连接[39],也有证据表明线粒体转移模式之间可能存在协同作用。细胞溶质钙离子、ATP和葡萄糖也被认为是调节线粒体转移的主要因素[38];同时受损细胞与间充质干细胞的微环境很可能成为调节线粒体转移模式的促成因素。因此,为促进疾病的治疗,需要进一步全面而深入地了解线粒体转移机制及该过程中涉及的信号转导途径。 "

2.2.2 间充质干细胞经线粒体转移改善心脏功能 在心脏疾病如心肌缺血、心肌梗死和其他心肌病中发现,提供健康线粒体可改善功能失调的线粒体[40],验证了影响受损心肌线粒体动力学的细胞过程在心血管系统疾病治疗中也起着重要作用[41]。线粒体功能障碍被认为是心肌缺血再灌注损伤的突出特征[42],而间充质干细胞可通过隧道纳米管将线粒体转移至受损心肌细胞,从而改善或恢复受损细胞功能[43],而不仅仅是间充质干细胞的定向分化、旁分泌及免疫调节等功能。内皮祖细胞之间经隧道纳米管模式转移的线粒体也可促进受损心肌的再生与修复[44],间充质干细胞的功能性线粒体转移与血管平滑肌细胞之间也是通过隧道纳米管进行细胞间的通讯[45]。此外,ACQUISTAPACE等[31]将人脂肪干细胞与小鼠心肌细胞共培养证实了细胞间F-肌动蛋白连接的形成,也显示了线粒体通过部分细胞融合参与细胞功能恢复的过程,进一步表明间充质干细胞的线粒体转移是多种心脏疾病治疗的新机制[46-47]。 2.2.3 间充质干细胞通过线粒体转移在肺和脑损伤的作用 ISLAM等[37]在用脂多糖处理的小鼠急性肺损伤模型中,通过荧光显微镜观察到线粒体从骨髓间充质干细胞向肺泡上皮转移,并导致肺泡ATP水平增加。该实验还表明了连接蛋白43在调节线粒体转移中起重要作用。此外,诱导多能干细胞来源的间充质干细胞减轻了由香烟烟雾诱发的肺损伤大鼠肺泡破坏和纤维化的严重程 度[27]。特别是在香烟烟雾介质中,线粒体不仅能从诱导多能干细胞来源的间充质干细胞递送至受损的支气管上皮细胞,还能传递给气道平滑肌细胞[48],改善人肺细胞和小鼠肺部的炎症和气道高反应性。同时在鱼藤酮引起的肺损伤和过敏原引起哮喘的肺损伤疾病模型中也证实,间充质干细胞通过上调Miro1促进线粒体转移至受损的肺泡上皮细胞而发挥保护作用[49-50]。此外,有研究通过骨髓间充质干细胞与星形胶质细胞和神经元样嗜铬细胞瘤细胞的体外共培养体系观察到线粒体转移,线粒体从骨髓间充质干细胞向星形胶质细胞的转移受到氧化应激的刺激,活性氧水平增加,向神经元样细胞的转移恢复了细胞有氧呼吸和增殖[51]。Miro1的过表达可促进线粒体从间充质干细胞到大鼠皮质神经元的转移,致使脑源性神经营养因子增加,表明了间充质干细胞的显著神经保护行为[52]。还有研究用DsRed2标记骨髓间充质干细胞并追踪移植的线粒体,然后颈动脉注射至脑缺血再灌注大鼠体内受损的脑血管系统,不但观察到移植线粒体的转移,且进一步证实其对损伤的脑微血管系统具有保护作用。因此,干细胞移植治疗缺血性脑卒中的有益作用主要是通过线粒体转移来保护受损的大脑微血管系统,而不仅仅是通过旁分泌或转分化机制[53]。 2.2.4 间充质干细胞经线粒体转移对角膜损伤和肾损伤的影响 角膜的化学灼伤包括碱性损伤,是导致严重角膜损伤和视力损害的常见原因之一。集中且高浓度的碱性损伤会破坏角膜的所有层,但较少浓度的碱性损伤也会对视力造成威胁,因为氧化应激和炎症引起的角膜线粒体损伤通常会延迟角膜修复。在角膜化学灼伤的急性期,治疗抗炎、抗血管生成因子和增强上皮愈合对于角膜修复至关重要。在角膜严重损伤的情况下,角膜干细胞移植成为用于角膜再生和瘢痕形成预防的新策略。近年来,大量体内外研究证据表明,间充质干细胞主要是通过旁分泌、抗炎作用、角膜细胞分化或细胞替换而预防角膜瘢痕形成和促进角膜伤口愈合。干细胞治疗可加速角膜上皮细胞的再生,并对治疗角膜化学灼伤、角膜瘢痕、视网膜退行性疾病等与眼睛有关的损伤同样有效[54-56]。与正常的角膜上皮细胞相比,鱼藤酮处理的角膜上皮细胞产生ATP、静息呼吸和最大呼吸速率等生物能量参数的比率较低。所有这些参数在与诱导多能干细胞来源间充质干细胞共培养的角膜上皮细胞中得到明显改善;线粒体从诱导多能干细胞来源间充质干细胞转移到受损的角膜上皮细胞,使其免受鱼藤酮诱导的细胞凋亡和死亡,表明间充质干细胞的线粒体转移是间充质干细胞治疗角膜损伤的重要机制[23]。此外,在大鼠肾小管细胞和间充质干细胞的共培养系统中,观察到间充质干细胞的线粒体可向肾小管细胞转移[28]。在骨髓间充质干细胞移植治疗大鼠链脲佐菌素诱导的糖尿病肾病时,不仅大鼠的血糖水平得到有效控制,肾脏损害明显减轻,还观察到间充质干细胞线粒体向肾近端小管上皮细胞转移,在高糖条件下共培养骨髓间充质干细胞和肾小管上皮细胞NRK-52E,骨髓间充质干细胞通过隧道纳米管方式将其线粒体转移至受损的肾近端小管上皮细胞,明显抑制了肾近端小管上皮细胞凋亡,还可增强SOD2和Bcl-2的表达,显著抑制活性氧的产生[57]。 2.2.5 间充质干细胞经线粒体转移对皮肤损伤修复的影响 作为高度动态变化的细胞器,线粒体在裂变和融合形态之间的动力学变化中,表现为线粒体功能和代谢的不断改变。过度的裂变与膜电位降低、ATP合成、氧消耗和质子泄漏增加有关,过度融合则导致相反膜电位升高,ATP合成、耗氧量减少和质子泄漏减少[58]。间充质干细胞与来自线粒体疾病患者的皮肤成纤维细胞共培养,观察到皮肤成纤维细胞异常的线粒体形态从裂变状态被拯救到融合状态。体内实验证实,间充质干细胞处理后宿主组织中线粒体丰度和线粒体氧消耗率升高,可推测间充质干细胞在病理状态下有调节线粒体的潜力。间充质干细胞经线粒体转移,减少了成纤维细胞中的基线片段化,部分恢复了共培养宿主细胞的线粒体功能[24],然而间充质干细胞影响宿主线粒体动力学的确切作用及能量改变过程和具体作用机制尚需深入研究。 2.2.6 间充质干细胞来源线粒体转移在肿瘤相关疾病中的作用 线粒体是细胞生物能量学和新陈代谢的关键调节剂,与肿瘤进展密切相关,也是癌症治疗的潜在靶标[59]。有研究证实了骨髓间充质干细胞的线粒体通过隧道纳米管转移至急性髓性白血病细胞并促进其增殖,导致其对化疗的敏感性降低[60]。没有线粒体DNA且呼吸功能缺陷的转移性黑素瘤细胞不能形成肿瘤,但通过水平基因转移从宿主Balb/c小鼠中分离小鼠间充质干细胞获得完整的线粒体及其DNA后,这种黑色素瘤细胞呼吸功能得以恢复[61]。最近的一项研究发现,通过抑制细胞间黏附分子1阻断隧道纳米管形成后,可阻断线粒体从间充质干细胞转移到Jurkat细胞中(T淋巴细胞白血病细胞系),并最终诱导Jurkat细胞死亡,提示通过防止隧道纳米管形成选择性抑制线粒体转移可作为癌症的潜在疗法[1]。但鉴于糖酵解在癌细胞生长、侵袭中的重要性和复杂性,全面了解改变间充质干细胞线粒体向癌细胞转移的具体作用机制及其影响癌细胞命运的意义仍有很长的路要走[62]。 2.2.7 间充质干细胞线粒体转移的抗菌活性 间充质干细胞通过将线粒体转移到巨噬细胞发挥其抗感染作用,该机制可能是通过隧道纳米管调节巨噬细胞的吞噬能力和增强其生物能量学来使ATP活性增加,而有助于巨噬细胞更有效地清除细菌[30]。为了确定间充质干细胞的抗微生物作用是否由肺泡巨噬细胞所介导,在正常大肠杆菌中测定了间充质干细胞的作用,发现在肺泡巨噬细胞耗尽的小鼠中施用间充质干细胞不能恢复参与抗炎作用的几种细胞因子的水平,但间充质干细胞可以恢复正常小鼠的细胞因子水平,这表明间充质干细胞的抗微生物活性是由巨噬细胞介导的。有趣的是,在单核细胞或嗜中性粒细胞中未观察到类似间充质干细胞线粒体转移所介导的巨噬细胞吞噬作用或间充质干细胞与巨噬细胞的融合,这表明线粒体转移可能是增强巨噬细胞吞噬功能和清除细菌的唯一手段。线粒体通过微囊泡或外泌体的非接触线粒体转移显著增强巨噬细胞的吞噬指数也在实验中被观察到,表明线粒体转移增强抗感染作用也是巨噬细胞的另一个关键作用机制[63]。因此,线粒体在抗微生物和免疫调节中的作用也是今后十分值得探讨的新课题。 "

| [1] LI C, CHEUNG MKH, HAN S, et al. Mesenchymal stem cells and their mitochondrial transfer: a double-edged sword. Biosci Rep. 2019;39(5):BSR20182417. [2] GRONTHOS S, MANKANI M, BRAHIM J, et al. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci U S A. 2000;97(25):13625-13630. [3] KLIMCZAK A, KOZLOWSKA U. Mesenchymal Stromal Cells and Tissue-Specific Progenitor Cells: Their Role in Tissue Homeostasis. Stem Cells Int. 2016;2016:4285215. [4] LIANG X, DING Y, ZHANG Y, et al. Paracrine mechanisms of mesenchymal stem cell-based therapy: current status and perspectives. Cell Transplant. 2014;23(9):1045-1059. [5] KANJI S, DAS H. Advances of Stem Cell Therapeutics in Cutaneous Wound Healing and Regeneration. Mediators Inflamm. 2017;2017:5217967. [6] LI CJ, CHEN PK, SUN LY, et al. Enhancement of Mitochondrial Transfer by Antioxidants in Human Mesenchymal Stem Cells. Oxid Med Cell Longev. 2017;2017:8510805. [7] TAN DQ, SUDA T. Reactive Oxygen Species and Mitochondrial Homeostasis as Regulators of Stem Cell Fate and Function. Antioxid Redox Signal. 2018;29(2):149-168. [8] VIZOSO FJ, EIRO N, CID S, et al. Mesenchymal Stem Cell Secretome: Toward Cell-Free Therapeutic Strategies in Regenerative Medicine. Int J Mol Sci. 2017;18(9): E1852. [9] BANG OY, LEE JS, LEE PH, et al. Autologous mesenchymal stem cell transplantation in stroke patients. Ann Neurol. 2005;57(6): 874-882. [10] LEE RH, PULIN AA, SEO MJ, et al. Intravenous hMSCs improve myocardial infarction in mice because cells embolized in lung are activated to secrete the anti-inflammatory protein TSG-6. Cell Stem Cell. 2009;5(1):54-63. [11] TÖGEL F, HU Z, WEISS K, et al. Administered mesenchymal stem cells protect against ischemic acute renal failure through differentiation-independent mechanisms. Am J Physiol Renal Physiol. 2005;289(1):F31-42. [12] LUAN Y, ZHANG ZH, WEI DE, et al. Implantation of mesenchymal stem cells improves right ventricular impairments caused by experimental pulmonary hypertension. Am J Med Sci. 2012; 343(5):402-406. [13] PARK SS, MOISSEIEV E, BAUER G, et al. Advances in bone marrow stem cell therapy for retinal dysfunction. Prog Retin Eye Res. 2017;56:148-165. [14] SPEES JL, LEE RH, GREGORY CA. Mechanisms of mesenchymal stem/stromal cell function. Stem Cell Res Ther. 2016;7(1):125. [15] PATANANAN AN, WU TH, CHIOU PY, et al. Modifying the Mitochondrial Genome. Cell Metab. 2016;23(5):785-796. [16] WANG J, LI H, YAO Y, et al. Stem cell-derived mitochondria transplantation: a novel strategy and the challenges for the treatment of tissue injury. Stem Cell Res Ther. 2018;9(1):106. [17] CHERNORUDSKIY AL, ZITO E. Regulation of Calcium Homeostasis by ER Redox: A Close-Up of the ER/Mitochondria Connection. J Mol Biol. 2017;429(5):620-632. [18] VIGNAIS ML, CAICEDO A, BRONDELLO JM, et al. Cell Connections by Tunneling Nanotubes: Effects of Mitochondrial Trafficking on Target Cell Metabolism, Homeostasis, and Response to Therapy. Stem Cells Int. 2017;2017:6917941. [19] SINHA P, ISLAM MN, BHATTACHARYA S, et al. Intercellular mitochondrial transfer: bioenergetic crosstalk between cells. Curr Opin Genet Dev. 2016;38:97-101. [20] CHUANG YC, LIOU CW, CHEN SD, et al. Mitochondrial Transfer from Wharton's Jelly Mesenchymal Stem Cell to MERRF Cybrid Reduces Oxidative Stress and Improves Mitochondrial Bioenergetics. Oxid Med Cell Longev. 2017;2017: 5691215. [21] AHMAD T, MUKHERJEE S, PATTNAIK B, et al. Miro1 regulates intercellular mitochondrial transport & enhances mesenchymal stem cell rescue efficacy. EMBO J. 2014;33(9):994-1010. [22] SPEES JL, OLSON SD, WHITNEY MJ, et al. Mitochondrial transfer between cells can rescue aerobic respiration. Proc Natl Acad Sci U S A. 2006;103(5):1283-1288. [23] JIANG D, GAO F, ZHANG Y, et al. Mitochondrial transfer of mesenchymal stem cells effectively protects corneal epithelial cells from mitochondrial damage. Cell Death Dis. 2016;7(11): e2467. [24] NEWELL C, SABOUNY R, HITTEL DS, et al. Mesenchymal Stem Cells Shift Mitochondrial Dynamics and Enhance Oxidative Phosphorylation in Recipient Cells. Front Physiol. 2018;9:1572. [25] KOYANAGI M, BRANDES RP, HAENDELER J, et al. Cell-to-cell connection of endothelial progenitor cells with cardiac myocytes by nanotubes: a novel mechanism for cell fate changes? Circ Res. 2005;96(10):1039-1041. [26] CSELENYÁK A, PANKOTAI E, HORVÁTH EM, et al. Mesenchymal stem cells rescue cardiomyoblasts from cell death in an in vitro ischemia model via direct cell-to-cell connections. BMC Cell Biol. 2010;11:29. [27] LI X, ZHANG Y, YEUNG SC, et al. Mitochondrial transfer of induced pluripotent stem cell-derived mesenchymal stem cells to airway epithelial cells attenuates cigarette smoke-induced damage. Am J Respir Cell Mol Biol. 2014;51(3):455-465. [28] PLOTNIKOV EY, KHRYAPENKOVA TG, GALKINA SI, et al. Cytoplasm and organelle transfer between mesenchymal multipotent stromal cells and renal tubular cells in co-culture. Exp Cell Res. 2010;316(15):2447-2455. [29] HAYAKAWA K, ESPOSITO E, WANG X, et al. Transfer of mitochondria from astrocytes to neurons after stroke. Nature. 2016;535(7613):551-555. [30] JACKSON MV, MORRISON TJ, DOHERTY DF, et al. Mitochondrial Transfer via Tunneling Nanotubes is an Important Mechanism by Which Mesenchymal Stem Cells Enhance Macrophage Phagocytosis in the In Vitro and In Vivo Models of ARDS. Stem Cells. 2016;34(8):2210-2223. [31] ACQUISTAPACE A, BRU T, LESAULT PF, et al. Human mesenchymal stem cells reprogram adult cardiomyocytes toward a progenitor-like state through partial cell fusion and mitochondria transfer. Stem Cells. 2011;29(5):812-824. [32] MAHROUF-YORGOV M, AUGEUL L, Da Silva CC, et al. Mesenchymal stem cells sense mitochondria released from damaged cells as danger signals to activate their rescue properties. Cell Death Differ. 2017;24(7):1224-1238. [33] MURRAY LMA, KRASNODEMBSKAYA AD. Concise Review: Intercellular Communication Via Organelle Transfer in the Biology and Therapeutic Applications of Stem Cells. Stem Cells. 2019; 37(1):14-25. [34] TANG BL. MIRO GTPases in Mitochondrial Transport, Homeostasis and Pathology. Cells. 2015;5(1): E1. [35] ZHANG Y, YU Z, JIANG D, et al. iPSC-MSCs with High Intrinsic MIRO1 and Sensitivity to TNF-α Yield Efficacious Mitochondrial Transfer to Rescue Anthracycline-Induced Cardiomyopathy. Stem Cell Reports. 2016;7(4):749-763. [36] WANG X, GERDES HH. Transfer of mitochondria via tunneling nanotubes rescues apoptotic PC12 cells. Cell Death Differ. 2015; 22(7):1181-1191. [37] ISLAM MN, DAS SR, EMIN MT, et al. Mitochondrial transfer from bone-marrow-derived stromal cells to pulmonary alveoli protects against acute lung injury. Nat Med. 2012;18(5): 759-765. [38] TORRALBA D, BAIXAULI F, SÁNCHEZ-MADRID F. Mitochondria Know No Boundaries: Mechanisms and Functions of Intercellular Mitochondrial Transfer. Front Cell Dev Biol. 2016;4:107. [39] SINCLAIR KA, YERKOVICH ST, HOPKINS PM, et al. Characterization of intercellular communication and mitochondrial donation by mesenchymal stromal cells derived from the human lung. Stem Cell Res Ther. 2016;7(1):91. [40] NAIR V, MADAN H, SOFAT S, et al. Efficacy of stem cell in improvement of left ventricular function in acute myocardial infarction--MI3 Trial. Indian J Med Res. 2015;142(2):165-174. [41] IKEDA Y, SHIRAKABE A, BRADY C, et al. Molecular mechanisms mediating mitochondrial dynamics and mitophagy and their functional roles in the cardiovascular system. J Mol Cell Cardiol. 2015;78:116-122. [42] KURIAN GA, RAJAGOPAL R, VEDANTHAM S, et al. The Role of Oxidative Stress in Myocardial Ischemia and Reperfusion Injury and Remodeling: Revisited. Oxid Med Cell Longev. 2016;2016: 1656450. [43] ZHANG J, ZHANG J, ZHAO L, et al. Differential roles of microtubules in the two formation stages of membrane nanotubes between human mesenchymal stem cells and neonatal mouse cardiomyocytes. Biochem Biophys Res Commun. 2019;512(3): 441-447. [44] PACAK CA, PREBLE JM, KONDO H, et al. Actin-dependent mitochondrial internalization in cardiomyocytes: evidence for rescue of mitochondrial function. Biol Open. 2015;4(5): 622-626. [45] VALLABHANENI KC, HALLER H, DUMLER I. Vascular smooth muscle cells initiate proliferation of mesenchymal stem cells by mitochondrial transfer via tunneling nanotubes. Stem Cells Dev. 2012;21(17):3104-3113. [46] FIGEAC F, LESAULT PF, LE COZ O, et al. Nanotubular crosstalk with distressed cardiomyocytes stimulates the paracrine repair function of mesenchymal stem cells. Stem Cells. 2014;32(1): 216-230. [47] HAN H, HU J, YAN Q, et al. Bone marrow-derived mesenchymal stem cells rescue injured H9c2 cells via transferring intact mitochondria through tunneling nanotubes in an in vitro simulated ischemia/reperfusion model. Mol Med Rep. 2016;13(2): 1517-1524. [48] LI X, MICHAELOUDES C, ZHANG Y, et al. Mesenchymal stem cells alleviate oxidative stress-induced mitochondrial dysfunction in the airways. J Allergy Clin Immunol. 2018;141(5):1634-1645. [49] 彭懿,舒畅,符州.骨髓间充质干细胞治疗组织损伤的线粒体转移机制[J].中国组织工程研究,2014,18(41):6708-6713. [50] HAN J, SONG Y, ZHU F. Mitochondrial transfer mechanism of stem cells for therapy of lung injury. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2018;30(1):88-90. [51] BABENKO VA, SILACHEV DN, POPKOV VA, et al. Miro1 Enhances Mitochondria Transfer from Multipotent Mesenchymal Stem Cells (MMSC) to Neural Cells and Improves the Efficacy of Cell Recovery. Molecules. 2018;23(3):E687. [52] BABENKO VA, SILACHEV DN, ZOROVA LD, et al. Improving the Post-Stroke Therapeutic Potency of Mesenchymal Multipotent Stromal Cells by Cocultivation With Cortical Neurons: The Role of Crosstalk Between Cells. Stem Cells Transl Med. 2015;4(9): 1011-1020. [53] LIU K, GUO L, ZHOU Z, et al. Mesenchymal stem cells transfer mitochondria into cerebral microvasculature and promote recovery from ischemic stroke. Microvasc Res. 2019;123:74-80. [54] CEJKA C, HOLAN V, TROSAN P, et al. The Favorable Effect of Mesenchymal Stem Cell Treatment on the Antioxidant Protective Mechanism in the Corneal Epithelium and Renewal of Corneal Optical Properties Changed after Alkali Burns. Oxid Med Cell Longev. 2016;2016:5843809. [55] BASU S, HERTSENBERG AJ, FUNDERBURGH ML, et al. Human limbal biopsy-derived stromal stem cells prevent corneal scarring. Sci Transl Med. 2014;6(266):266ra172. [56] WONG IY, POON MW, PANG RT, et al. Promises of stem cell therapy for retinal degenerative diseases. Graefes Arch Clin Exp Ophthalmol. 2011;249(10):1439-1448. [57] KONARI N, NAGAISHI K, KIKUCHI S, et al. Mitochondria transfer from mesenchymal stem cells structurally and functionally repairs renal proximal tubular epithelial cells in diabetic nephropathy in vivo. Sci Rep. 2019;9(1):5184. [58] SEBASTIÁN D, PALACÍN M, ZORZANO A. Mitochondrial Dynamics: Coupling Mitochondrial Fitness with Healthy Aging. Trends Mol Med. 2017;23(3):201-215. [59] CAICEDO A, FRITZ V, BRONDELLO JM, et al. MitoCeption as a new tool to assess the effects of mesenchymal stem/stromal cell mitochondria on cancer cell metabolism and function. Sci Rep. 2015;5:9073. [60] MOSCHOI R, IMBERT V, NEBOUT M, et al. Protective mitochondrial transfer from bone marrow stromal cells to acute myeloid leukemic cells during chemotherapy. Blood. 2016;128(2): 253-264. [61] DONG LF, KOVAROVA J, BAJZIKOVA M, et al. Horizontal transfer of whole mitochondria restores tumorigenic potential in mitochondrial DNA-deficient cancer cells. Elife. 2017;6: e22187. [62] LIBERTI MV, LOCASALE JW. The Warburg Effect: How Does it Benefit Cancer Cells? Trends Biochem Sci. 2016;41(3):287. [63] MORRISON TJ, JACKSON MV, CUNNINGHAM EK, et al. Mesenchymal Stromal Cells Modulate Macrophages in Clinically Relevant Lung Injury Models by Extracellular Vesicle Mitochondrial Transfer. Am J Respir Crit Care Med. 2017;196(10): 1275-1286. [64] HSU YC, WU YT, YU TH, et al. Mitochondria in mesenchymal stem cell biology and cell therapy: From cellular differentiation to mitochondrial transfer. Semin Cell Dev Biol. 2016;52:119-131. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [14] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [15] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||