Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (30): 4882-4887.doi: 10.3969/j.issn.2095-4344.0989
Previous Articles Next Articles
Functional metal ions-containing bioactive glass: characteristics, functions and applications
Wang Qing1, Wang De-ping2, Huang Wen-chan2, Jia Wei-tao1
- 1Department of Orthopedics, Sixth People’s Hospital of Shanghai Jiao Tong University, Shanghai 200233, China; 2School of Materials Science and Engineering, Tongji University, Shanghai 201804, China
-
Received:2018-05-21Online:2018-10-28Published:2018-10-28 -
Contact:Jia Wei-tao, MD, Associate chief physician, Master’s supervisor, Department of Orthopedics, Sixth People’s Hospital of Shanghai Jiao Tong University, Shanghai 200233, China -
About author:Wang Qing, Master candidate, Department of Orthopedics, Sixth People’s Hospital of Shanghai Jiao Tong University, Shanghai 200233, China -
Supported by:the National Natural Science Foundation of China, No. 81572105; Shanghai Municipal Education Commission-Gaofeng Clinical Medicine Grant Support, No. 20172026; Shanghai’s Subsidy Funding Project for Talent Development, No. 2017035
CLC Number:
Cite this article
Wang Qing, Wang De-ping, Huang Wen-chan2, Jia Wei-tao. Functional metal ions-containing bioactive glass: characteristics, functions and applications[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(30): 4882-4887.
share this article

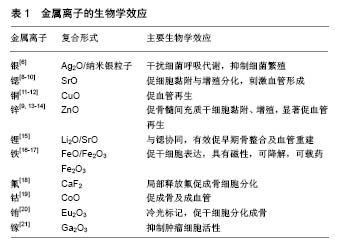
2.1 复合银 移植物本身所造成的感染率上升一直是亟待解决的难题,生物材料吸引细胞锚定的同时,也为细菌提供了生长平面,如表葡菌常在植入的医疗器械表面形成致密生物膜,导致抗生素耐药和免疫逃逸[3],使感染顽固而难以治愈。由于抗生素的广泛应用,细菌耐药性明显增加,用药也出现诸多限制,并且全身应用抗生素并不能保证在感染部位达到有效抗菌浓度,长期应用还会带来不良反应,因此具有抗菌作用的生物玻璃植入材料将具有极大的发展空间。 银离子是性能良好的广谱抗菌剂,可结合细菌呼吸相关酶,形成强烈结合键导致蛋白变性,使微生物窒息而亡[4]。王会等[5]采用熔融法将Ag2O、SiO2、P2O3、B2O3、MO、R2O等玻璃组分按一定配比浇铸淬冷制备含银硼酸盐生物玻璃支架,在模拟体液中浸泡后记录pH及质量变化,30 d后做X射线衍射,图谱表明支架表面的晶体主要产物为Ca10(PO4)6(OH)2,说明硼酸盐生物玻璃支架在逐渐降解并转化生成人体骨骼的无机组成羟基磷灰石。材料支架与大肠杆菌和金葡菌共培养后,在支架周围均观察到较为明显的抑菌圈,且抑菌效果可维持3 d以上。此外,将含银支架与MC3T3-E1细胞共培养3 d后,含银生物玻璃组的细胞活性及碱性磷酸酶活性与单纯生物玻璃组相比无显著性差异。这表明含银硼酸盐生物玻璃支架除具有良好生物活性和降解性外,还可以发挥持续的抗菌活性。Boccaccini等[6]将纳米银粒子、生物活性玻璃微粒子和聚醚醚酮粉末悬浮于乙醇溶液,经搅拌、超声匀浆、电泳、热处理,制备成致密生物涂层,可降低金属移植物的硬度以减轻应力遮挡效应的影响。在后续针对大肠杆菌的抗菌实验中,该涂层还表现出与银剂量相关的强抗菌活性。Zhu等[7]分别将1 g M58S和SM58S加入硝酸银溶液中搅拌,待银离子吸附达到平衡后过滤、冲洗、干燥,得到Ag-M58S和Ag-SM58S生物玻璃,在这种改良多孔58S生物玻璃支架(SM58S)表面添加KH-550氨基官能团硅烷后可明显提高了银装载量,实验进一步证实Ag-SM58S可促进成骨细胞增殖分化,并且银离子的缓释在模拟体液中可维持768 h,有效抑制大肠杆菌和金黄色葡萄球菌增殖,1个月后抗菌率仍在100%,这表明含银SM58S可作为一种有效的骨组织修复生物材料,发挥促骨再生和抗菌的双重作用(表1)。 2.2 复合锶 锶是人体骨骼和牙齿的正常组成部分,99%的锶都存在于骨骼中,尤其集中于骨代谢循环进行比较旺盛的区域。在骨代谢进程中锶的机制主要是通过Wnt/catenin信号通路激活β-链蛋白表达,促进骨髓间充质干细胞增殖分化,进而刺激骨重建[22],另外还可以通过抑制核因子κB受体活化因子配体的表达来抑制破骨细胞分化,抑制骨吸收[23]。 掺杂锶的常见手段是以氧化锶替换生物玻璃中的氧化钙成分,Arepalli等[8]首次尝试用氧化锶替代生物玻璃中的二氧化硅成分,以石英、碳酸钠、碳酸钙、碳酸锶和正磷酸二氢铵为原料以熔融法制备出具有良好骨诱导活性的硅酸盐生物活性玻璃。锶替代部分硅后,玻璃支架的弹性模量有所下降。在模拟体液中,生物活性玻璃粉末浓度达到500 g/L时,含摩尔百分比为2% SrO组的细胞生存率仍有86%,细胞生长良好并与人红细胞相容,表明这种生物玻璃可作为骨组织修复的替代材料。Jia等[10]和Zhao等[9]通过3D打印技术制备了新型的含锶生物玻璃三维支架,体外实验证实该支架可以促进MC3T3-E1细胞黏附、增殖、分化和分泌碱性磷酸酶,上调成骨基因表达,动物骨缺损模型应用研究发现锶可以促进生物玻璃表面羟基磷灰石层形成,从而加骨缺损区域血管再生及成骨,证明了含锶多孔生物玻璃在骨缺损修复的临床应用潜能(表1)。 2.3 复合铜 血管化在骨再生过程中发挥至关重要的作用,丰富的血管可以运送养分、排出代谢废物,促进骨组织再生。有研究发现,在体内细胞招募、细胞分化和血管构建方面,缺氧微环境发挥了重要作用[24],其中最为关键的、受缺氧环境直接诱导的蛋白为缺氧诱导因子1,在缺氧条件下,缺氧诱导因子1α亚基和缺氧诱导因子1β亚基结合后,启动内皮细胞中血管内皮生长因子的表达,刺激血管形成[25]。 Wu等[11]经熔融法合成出具有良好促血管生成效应的含铜多孔生物玻璃,实验发现该含铜生物玻璃可以显著促进人骨髓间充质干细胞血管内皮生长因子基因的表达,原因是支架中释放的铜离子可以模拟缺氧环境,诱导细胞出现缺氧应激反应,从而发挥促血管再生、促骨生成作用,是修复骨组织的优良材料。Wang等[12]通过熔融法制备含铜硼酸盐生物玻璃支架,植入鼠颅骨缺损区,8周后的组织学定量分析显示含铜生物玻璃组的血管密度比单纯生物玻璃组明显增高,并且血管分布区域更广,CD31免疫组化染色同样支持这一结论,证实含铜生物玻璃可以刺激血管再生,从而促进骨修复(表1)。 2.4 复合锌 骨组织中锌元素的含量在衰老、绝经后均有下降,妇女绝经后常因骨质流失出现骨质疏松。然而由于乳腺癌与雌激素的强关联性,若使用雌激素治疗会增加患乳腺癌的风险,而使用含锌生物材料在局部促进骨质沉积则能有效规避这一风险。有研究表明,锌可以激活Runx2基因表达,抑制骨髓间充质细胞向破骨细胞样细胞转化,还可以启动成熟破骨细胞凋亡来抑制破骨细胞骨吸收进程[26] (表1)。 骨相关疾病中(如骨质疏松)大多存在氧化应激反应,活性氧若过多聚集对骨组织代谢活动威胁很大。Samira等[13]将SiO2、CaO、Na2O、P2O5、ZnO按一定比例混合、熔融制备出含锌生物玻璃,并植入鼠股骨缺损模型,90 d后,抗氧化物成分如超氧化物歧化酶、过氧化氢酶、谷胱甘肽过氧化物酶活性均有所增加。在降解过程中,含锌生物玻璃-骨界面结合强度测试证实与股骨髁接近,组织学参数如骨小梁数目、宽度、间隙等均有明显改善。Mohammad-Reza等[14]通过溶胶-凝胶方法将含锌纳米生物玻璃粉末和双相磷酸钙复合来加强后者的机械强度,在与鼠颅骨源性成骨细胞共培养后发现成骨细胞分化早期标记物碱性磷酸酶含量明显升高,说明含锌生物玻璃-双向磷酸钙复合物可以促进成骨细胞分化和增殖。Wang等[9]通过传统熔炼铸造技术,制备出与人骨小梁有着类似微结构的含锌硼酸盐生物玻璃支架,体外细胞实验证实含锌生物玻璃可以促进人间充质干细胞增殖、分化和碱性磷酸酶分泌表达,当培养液中锌浓度在10-250 μmol/L时,破骨细胞活性明显受到抑制。鼠颅骨缺损修复实验进一步证实含锌生物玻璃组骨再生能力显著高于不含锌玻璃组。 2.5 复合锂 Wnt/β-catenin信号转导通路是调控骨髓间充质干细胞增殖及分化的枢纽,其上游的糖原合成酶激酶3β则是通路“开关”。研究表明,锂剂可以通过糖原合成酶激酶3β来激活Wnt/β-catenin信号转导通路,刺激新骨再生[27] (表1)。 Khan等[15]将SiO2、CaCO3、Na2CO3、Na2B4O7•10H2O、TiO2,NH4H2PO4、Li2CO3和SrCO3等成分按比例配置,以传统熔融法制备出含有锶/锂两种成分的生物玻璃,并用兔股骨缺损模型分别评估含锂或锶单一金属元素、含锶/锂两种元素时生物玻璃的骨修复效应。植入2个月后,含锶组成骨明显,特征为出现大量哈弗氏小管,成骨细胞健康且核质比正常,骨基质由血管浸润,内有红细胞。含锂组与之相似,呈现生长良好的骨组织结构。含锶/锂组不仅骨组织结构生长良好,且有大量血管分支浸润,在骨缺陷边缘区成骨及破骨细胞活跃。此外,含锶/锂组的新生骨形成的密度比纯生物玻璃组、含锂玻璃组和含锶玻璃组更高,新生骨面积更广,表明存在更快速、效率更高的骨重建。因此,锶、锂均可以加速早期骨修复,两种元素联合作用效果更为明显。 2.6 复合磁性粒子 作为磁性分子的四氧化三铁在生物医疗界应用广泛,磁性分子在外部磁场下可以产热,当局部温度>43 ℃时可以杀死肿瘤细胞,而其它正常细胞正常存活[28]。在骨肿瘤切除后使用复合磁性粒子的生物玻璃支架既促进骨缺损修复,又可在必要时通过磁性粒子发热来杀死局部残留肿瘤恶性细胞,避免复发。 超顺磁性材料被认为是局部肿瘤磁热治疗中最为有效的材料[29]。Goh等[16]以快速碱溶胶凝胶技术合成出双向硫酸钙生物玻璃,又以溶胶凝胶法添加铁离子。含 摩尔百分比为5%铁生物玻璃组对外界磁场呈消极反应,说明不适合进行磁热治疗,而摩尔百分比为20% Fe组滞后回线极窄,几乎无矫顽磁力及剩磁,提示具有天然超级顺磁性,对外界磁场响应良好,可以作为肿瘤细胞热疗领域的理想候选材料。Wu等[17]添加三价铁离子替换部分钙离子将玻璃成分搅拌熔融制备出含铁多孔生物玻璃,赋予其磁性。体外细胞实验中,含铁生物玻璃支架表面细胞黏附生长良好,细胞数量和线粒体活性,以及碱性磷酸酶、骨钙素基因表达较单纯生物玻璃组显著增加,表明新合成的含铁磁性生物玻璃具有良好的骨诱导生物活性,可应用于骨科恶性肿瘤术后的骨缺损修复中,结合磁热疗法和局部释放药物来发挥治疗作用(表1)。 2.7 复合其他功能离子 2.7.1 氟 多项体外及动物实验表明,氟可以调节骨吸收,直接或间接影响RANKL/OPG通路、调节骨形态发生蛋白/Smads信号通路、抑制破骨相关NFATc1基因表达以降低破骨细胞活性[30]。氟广泛用于口腔修复材料中,局部输送氟可降低牙本质及珐琅质中无机矿物质的溶解速率[31]。Gentleman等[18]以熔融法制备含氟生物玻璃,经体外培养及细胞定量实验发现,在含氟比例较高的生物玻璃表面,细胞群形成了高密度骨结节,碱性磷酸酶活性明显高于氟含量低的生物玻璃组。这些效应表明含氟生物玻璃可以促进成骨细胞的增殖及成骨矿化来修复骨缺损。此外,实验还发现,在Saos-2细胞与含摩尔百分比为17.76% CaF2生物玻璃组共培养7 d后,细胞分泌的白细胞介素6含量显著高于其他含量组及对照组。结合其他研究,白细胞介素6基因敲除小鼠的骨折愈合及骨矿化进程与野生型相比有显著延后[32]。并且有文献报道用甲状旁腺激素治疗的骨质疏松患者体内成骨细胞分泌的白细胞介素6有所增加[33]。由此表明含氟生物玻璃或许起到类似的促骨形成作用(表1)。 2.7.2 钴 钴离子常用于模拟缺氧环境,通过调节缺氧诱导因子1来激活体内数条与血管再生相关的信号通路。Hoppe等[19]通过熔融技术与泡沫复制技术制备出含钴1393生物玻璃,将MG-63细胞系与1393生物玻璃共培养后,在不同时间间隔下观察含钴生物玻璃组和纯玻璃组的细胞黏附及扩散行为,发现二者并无明显差别。体外实验发现,含1%钴1393生物玻璃对人真皮毛细血管内皮细胞吞噬低密度脂蛋白的细胞活动没有影响,而含5%钴1393生物玻璃组中内皮细胞不再吞噬低密度脂蛋白,产生细胞毒性,表明含1%钴1393生物玻璃具有良好的细胞相容性。流式细胞学检测结果显示含1%钴生物玻璃组中血管内皮细胞表现出高功能性,血管性血友病因子、血小板-内皮细胞粘附分子(CD31)、血管内皮细胞生长因子受体表达均有显著增高,表明含1%钴生物玻璃在应用于骨组织修复时,通过模拟低氧微环境上调血管再生相关基因表达,促进血管发生及骨再生(表1)。 2.7.3 铕 铕属于稀土元素之一,能长时间保持激发态,可应用于生物鉴定及生物荧光成像。铕多用于标记纳米分子,在药物释放过程中通过探测纳米材料中荧光量的改变来追踪药物释放情况[34]。铕在体外能够与羟基磷灰石结合,因此也可用于标记组织工程支架,可以用来监测支架降解[35]。另外,免疫反应在骨再生过程中也起到重要作用,如M1和M2型巨噬细胞就与组织修复及骨形成有着重要联系[36]。而铕可以通过促进巨噬细胞分泌炎性物质如白细胞介素6、白细胞介素18、抑瘤素M等来影响破骨细胞分化、抑制骨吸收,同时加强成骨细胞活性刺激骨再生[37]。Wu等[20]制备了含铕的可发光多孔生物玻璃支架,在与组织细胞共培养时,随着支架降解,组织细胞不断吞噬游离铕来达到标记的目的,支架中铕的含量不同,荧光密度亦不相同。此外,含铕支架能够显著加快碱性磷酸酶、Ⅰ型胶原蛋白、Runx 2等成骨基因的表达,加快骨髓间充质干细胞增殖分化。表明含铕生物玻璃支架具备生物标记和促进骨再生的双重功效(表1)。 2.7.4 镓 快速分化的肿瘤细胞中铁的代谢置换较普通细胞更为旺盛,镓可以竞争性抑制铁的吸收[38]。Keenan等[21]将纳米58S粉末加入含镓N-甲基-2-吡咯烷酮溶液,经不断过滤干燥得到含镓纳米58S生物玻璃粉,并通过称量不同重量的镓及纳米58S粉末,得到不同含量的含镓58S生物玻璃。材料抗压强度随镓含量升高而增加(0-0.5%),当镓含量达到1%时,材料强度开始下降。将材料在生理模拟液浸泡7 d后,材料表面有磷灰石层形成,钙磷比为1.69,与天然骨组织类似。以MG-63骨肉瘤细胞系测试材料抗肿瘤特性,发现细胞在含16 mol%镓生物玻璃组的浸提液中生长30 d后,细胞活力较单纯生物玻璃组明显下降。而在以往研究中证实,含镓生物玻璃对MC3T3-E1细胞相容性良好[39],表明镓可以在抗肿瘤细胞的同时,不影响到正常细胞增殖,进一步表明含镓生物玻璃可应用于骨肿瘤术后的骨缺损修复(表1)。 "
| [1] Kaur G, Pandey OP, Singh K, et al. A review of bioactive glasses: Their structure, properties, fabrication and apatite formation. J Biomed Mater Res A. 2014;102(1):254-274. [2] Wang X, Li X, Ito A, et al. Synthesis and characterization of hierarchically macroporous and mesoporous CaO-MO-SiO(2)-P(2)O(5) (M=Mg, Zn, Sr) bioactive glass scaffolds. Acta Biomater. 2011;7(10):3638-3644. [3] Qin Z, Zhang J, Hu Y, et al. Organic compounds inhibiting S. epidermidis adhesion and biofilm formation. Ultramicroscopy. 2009;109(8):881-888.[4] Percival SL, Bowler PG, Russell D. Bacterial resistance to silver in wound care. J Hosp Infect. 2005;60(1):1-7.[5] 王会,黄文旵,罗仕华,等.具有抗菌及骨修复的硼酸盐玻璃支架的性能研究[J].稀有金属材料与工程,2014,43(S1):54-58.[6] Seuss S, Heinloth M, Boccaccini AR. Development of bioactive composite coatings based on combination of PEEK, bioactive glass and Ag nanoparticles with antibacterial properties. Surf Coat Technol. 2016;301:100-105.[7] Zhu H, Hu C, Zhang F, et al. Preparation and antibacterial property of silver-containing mesoporous 58S bioactive glass. Mater Sci Eng C Mater Biol Appl. 2014;42:22-30. [8] Arepalli SK, Tripathi H, Hira SK, et al. Enhanced bioactivity, biocompatibility and mechanical behavior of strontium substituted bioactive glasses. Mater Sci Eng C Mater Biol Appl. 2016;69:108-116. [9] Zhao S, Zhang J, Zhu M, et al. Three-dimensional printed strontium-containing mesoporous bioactive glass scaffolds for repairing rat critical-sized calvarial defects. Acta Biomater. 2015;12:270-280. [10] Jia W, Lau GY, Huang W, et al. Bioactive Glass for Large Bone Repair. Adv Healthc Mater. 2015;4(18):2842-2848. [11] Wu C, Zhou Y, Xu M, et al. Copper-containing mesoporous bioactive glass scaffolds with multifunctional properties of angiogenesis capacity, osteostimulation and antibacterial activity. Biomaterials. 2013;34(2):422-433. [12] Wang H, Zhao S, Xiao W, et al. Influence of Cu doping in borosilicate bioactive glass and the properties of its derived scaffolds. Mater Sci Eng C Mater Biol Appl. 2016;58:194-203.[13] Samira J, Saoudi M, Abdelmajid K, et al. Accelerated bone ingrowth by local delivery of Zinc from bioactive glass: oxidative stress status, mechanical property, and microarchitectural characterization in an ovariectomized rat model. Libyan J Med. 2015;10(1):28572. [14] Badr-Mohammadi MR, Hesaraki S, Zamanian A. Mechanical properties and in vitro cellular behavior of zinc-containing nano-bioactive glass doped biphasic calcium phosphate bone substitutes. J Mater Sci Mater Med. 2014;25(1):185-197. [15] Khan PK, Mahato A, Kundu B, et al. Influence of single and binary doping of strontium and lithium on in vivo biological properties of bioactive glass scaffolds. Sci Rep. 2016;6: 32964.[16] Goh YF, Akram M, Alshemary AZ, et al. Synthesis, characterization and in vitro study of magnetic biphasic calcium sulfate-bioactive glass. Mater Sci Eng C Mater Biol Appl. 2015;53:29-35. [17] Wu C, Fan W, Zhu Y, et al. Multifunctional magnetic mesoporous bioactive glass scaffolds with a hierarchical pore structure. Acta Biomater. 2011;7(10):3563-3572.[18] Gentleman E, Stevens MM, Hill RG, et al. Surface properties and ion release from fluoride-containing bioactive glasses promote osteoblast differentiation and mineralization in vitro. Acta Biomater. 2013;9(3):5771-5779. [19] Hoppe A, Brandl A, Bleiziffer O, et al. In vitro cell response to Co-containing 1,393 bioactive glass. Mater Sci Eng C Mater Biol Appl. 2015;57:157-163.[20] Wu C, Xia L, Han P, et al. Europium-Containing Mesoporous Bioactive Glass Scaffolds for Stimulating in Vitro and in Vivo Osteogenesis. ACS Appl Mater Interfaces. 2016;8(18): 11342-11354.[21] Keenan TJ, Placek LM, Coughlan A, et al. Structural characterization and anti-cancerous potential of gallium bioactive glass/hydrogel composites. Carbohydr Polym. 2016; 153:482-491. [22] Yang F, Yang D, Tu J, et al. Strontium enhances osteogenic differentiation of mesenchymal stem cells and in vivo bone formation by activating Wnt/catenin signaling. Stem Cells. 2011;29(6):981-991. [23] Marie PJ, Felsenberg D, Brandi ML. How strontium ranelate, via opposite effects on bone resorption and formation, prevents osteoporosis. Osteoporos Int. 2011;22(6): 1659-1667. [24] Basini G, Grasselli F, Bussolati S, et al. Hypoxia stimulates the production of the angiogenesis inhibitor 2-methoxyestradiol by swine granulosa cells. Steroids. 2011;76(13):1433-1436. [25] Ahluwalia A, Tarnawski AS. Critical role of hypoxia sensor--HIF-1α in VEGF gene activation. Implications for angiogenesis and tissue injury healing. Curr Med Chem. 2012;19(1):90-97.[26] Shepherd DV, Kauppinen K, Brooks RA, et al. An in vitro study into the effect of zinc substituted hydroxyapatite on osteoclast number and activity. J Biomed Mater Res A. 2014;102(11):4136-4141. [27] Ferensztajn-Rochowiak E, Rybakowski JK. The effect of lithium on hematopoietic, mesenchymal and neural stem cells. Pharmacol Rep. 2016;68(2):224-230. [28] Andronescu E, Ficai M, Voicu G, et al. Synthesis and characterization of collagen/hydroxyapatite: magnetite composite material for bone cancer treatment. J Mater Sci Mater Med. 2010;21(7):2237-2242. [29] Yang J K, Yu JH, Kim J, et al. Preparation of superparamagnetic nanocomposite particles for hyperthermia therapy application. Mater Sci Eng A. 2007;449:477-479.[30] Pei J, Li B, Gao Y, et al. Fluoride decreased osteoclastic bone resorption through the inhibition of NFATc1 gene expression. Environ Toxicol. 2014;29(5):588-595. [31] Burke FM, Ray NJ, McConnell RJ. Fluoride-containing restorative materials. Int Dent J. 2006;56(1):33-43.[32] Yang X, Ricciardi BF, Hernandez-Soria A, et al. Callus mineralization and maturation are delayed during fracture healing in interleukin-6 knockout mice. Bone. 2007;41(6): 928-936. [33] Li NH, Ouchi Y, Okamoto Y, et al. Effect of parathyroid hormone on release of interleukin 1 and interleukin 6 from cultured mouse osteoblastic cells. Biochem Biophys Res Commun. 1991;179(1):236-242.[34] Miao G, Chen X, Mao C, et al. Synthesis and characterization of europium-containing luminescent bioactive glasses and evaluation of in vitro bioactivity and cytotoxicity. J Solgel Sci Technol. 2014;69(2):250-259.[35] Mawani Y, Orvig C. Improved separation of the curcuminoids, syntheses of their rare earth complexes, and studies of potential antiosteoporotic activity. J Inorg Biochem. 2014;132: 52-58. [36] Shi M, Chen Z, Farnaghi S, et al. Copper-doped mesoporous silica nanospheres, a promising immunomodulatory agent for inducing osteogenesis. Acta Biomater. 2016;30:334-344. [37] Shi M, Xia L, Chen Z, et al. Europium-doped mesoporous silica nanosphere as an immune-modulating osteogenesis/ angiogenesis agent. Biomaterials. 2017;144:176-187. [38] Minandri F, Bonchi C, Frangipani E, et al. Promises and failures of gallium as an antibacterial agent. Future Microbiol. 2014;9(3):379-397. [39] Keenan TJ, Placek LM, Mcginnity TL, et al. Relating ion release and pH to in vitro cell viability for gallium-inclusivebioactive glasses. J Mater Sci. 2016;51(2): 1107-1120.[40] Hardes J, Ahrens H, Gebert C, et al. Lack of toxicological side-effects in silver-coated megaprostheses in humans. Biomaterials. 2007;28(18):2869-2875.[41] Hung YH, Bush AI, Cherny RA. Copper in the brain and Alzheimer's disease. J Biol Inorg Chem. 2010;15(1):61-76. [42] Zamani A, Omrani GR, Nasab MM. Lithium's effect on bone mineral density. Bone. 2009;44(2):331-334. [43] Balena R, Kleerekoper M, Foldes JA, et al. Effects of different regimens of sodium fluoride treatment for osteoporosis on the structure, remodeling and mineralization of bone. Osteoporos Int. 1998;8(5):428-435. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Cheng Yanan, Wu Yucong, Mao Qiuhua, Chen Ling, Lu Liying, Xu Pu. Restoration effect and stability of resin infiltration combined with bioactive glass on demineralized tooth enamel [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3522-3526. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||