Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (25): 4021-4027.doi: 10.3969/j.issn.2095-4344.0959
Previous Articles Next Articles
Effects of exosomes derived from canine umbilical cord mesenchymal stem cells on canine skin wound
Zhan Xiao-shu, Luo Dong-zhang, Wang Bing-yun, Lu Yu, Xian Wei-hang, Lan Yi-qi, Guo Jian-bo, Luo Hui-na, Bai Yin-shan, Ji Hui-qin, Chen Sheng-feng, Chen Zhi-sheng, Liu Can-ying
- School of Life Science and Engineering, Foshan University, Foshan 528231, Guangdong Province, China
-
Revised:2018-06-22Online:2018-09-08Published:2018-09-08 -
Contact:Wang Bing-yun, M.D., Professor, School of Life Science and Engineering, Foshan University, Foshan 528231, Guangdong Province, China -
About author:Zhan Xiao-shu, Master candidate, School of Life Science and Engineering, Foshan University, Foshan 528231, Guangdong Province, China -
Supported by:the Natural Science Foundation of Guangdong Province, No. 2017A030313171; the Project of the Key Laboratory of Preventive Veterinary Medicine of Guangdong Provincial Education Department, No. 2014KTSPT037; the National Undergraduate Innovation and Entrepreneurship Training Program; the Academic Foundation of Foshan University in 2017
CLC Number:
Cite this article
Zhan Xiao-shu, Luo Dong-zhang, Wang Bing-yun, Lu Yu, Xian Wei-hang, Lan Yi-qi, Guo Jian-bo, Luo Hui-na, Bai Yin-shan, Ji Hui-qin, Chen Sheng-feng, Chen Zhi-sheng, Liu Can-ying. Effects of exosomes derived from canine umbilical cord mesenchymal stem cells on canine skin wound[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(25): 4021-4027.
share this article
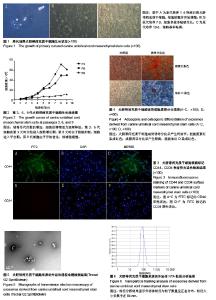
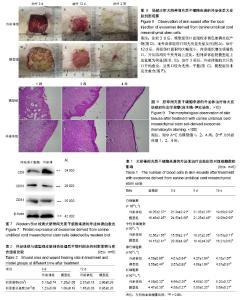
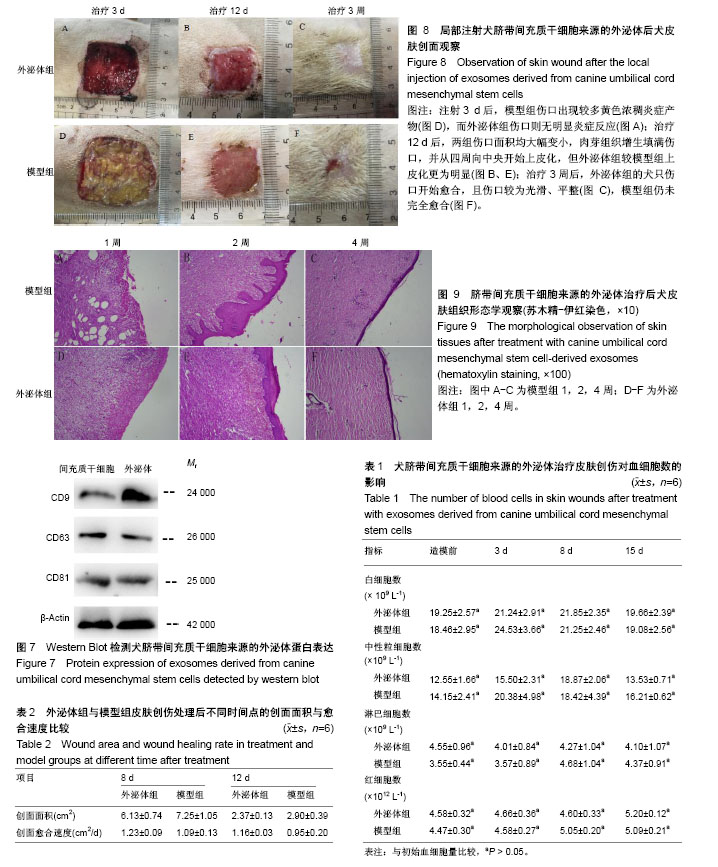
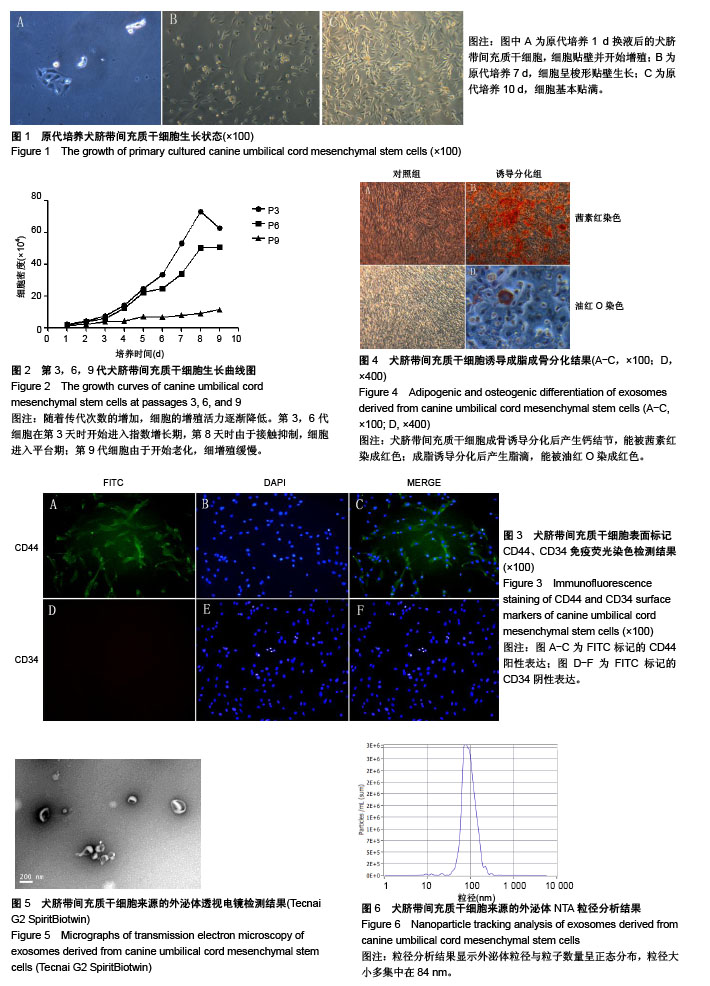
2.1 犬脐带间充质干细胞的培养与鉴定 细胞贴壁24 h后进行第1次换液,可见散在的短梭形细胞贴壁,部分增殖形成细胞集落,呈玫瑰花状(图1A)。原代培养7 d后,细胞增殖速度加快,密度明显增加,此时细胞呈长梭形生长(图1B)。培养至第10天时,细胞长满至90%,此时细胞形态均一,活性良好,可进行传代(图1C)。传代后细胞快速增殖,2∶1传代时每一两天可传代1次。由生长曲线图可看出,犬脐带间充质干细胞在P3代时活性最好,增殖最快,随着代数的增加,其增殖速率降低(图2)。P3、P6、P9群体倍增时间分别为23 h、37 h和70 h。免疫荧光检测,犬脐带间充质干细胞的细胞表面CD44呈阳性表达(图3A-C),CD34呈阴性表达(图3D-F),符合间充质干细胞特性;将P3代细胞进行成骨分化10 d后形成大量钙结节,用茜素红染色可染成红色(图4B),成脂分化21 d后形成透亮脂滴,用油红O可染成红色(图4D)。 2.2 犬UC-MSC-EXO的鉴定结果 透视电镜下观察,分离和提纯的犬UC-MSC-EXO呈杯托状(图5),NTA粒径检测显示其大小在30-200 nm,集中在84 nm(图6)。Western Blotting结果显示,犬UC-MSC-EXO表达特异性的外泌体表面蛋白CD81、CD63和CD9(图7)。以上特性表明,分离获得的囊泡体即为外泌体。 2.3 犬UC-MSC-EXO治疗犬皮肤创伤情况 犬全层皮肤损伤模型造模后,未见明显红肿热痛现象,饮食和精神状态均正常。在第3天时,模型组伤口处出现较多黄色浓稠炎症产物,而外泌体组伤口则无明显炎症反应;治疗12 d后,两组伤口面积均大幅变小,肉芽组织增生填满伤口,并从四周向中央开始上皮化,但外泌体组较模型组上皮化更为明显;治疗3周后,外泌体组的犬只伤口开始愈合,且伤口较为光滑、平整(图8)。 2.4 犬UC-MSC-EXO治疗后犬血液常规指标的变化 从表1可知,造模前两组白细胞总数无差异。造模后3 d 模型组的白细胞数迅速上升,较造模前提高了33%,而外泌体组只升高了10.3%;从第8天开始,模型组的白细胞总数下降,以后一直与外泌体组相近;模型组中性粒细胞数在造模第3天快速升高,而后缓慢降低,而外泌体组在治疗过程中升高较缓慢;两组在治疗过程中淋巴细胞数量和红细胞数量均无显著差异(见表1)。 2.5 犬UC-MSC-EXO对犬皮肤创伤面积和愈合速度的影响 从表2可见,从伤口愈合情况来看,在治疗第8和12天时,外泌体组创面面积较模型组小,愈合速度较快,但两组的创面面积和愈合速度无显著差异(P > 0.05)。 2.6 犬UC-MSC-EXO治疗后犬皮肤创伤病理组织学观察 从图9可见,第1周时伤口均被结缔组织增生形成的肉芽组织填满,此时增生组织中存在大量纤维细胞,开始形成形状较不规则的单个胶原纤维,由于表皮组织的再生能力远弱于结缔组织,此时还未见表皮层形成。模型组增生组织较外泌体组生长状况差,表面粗糙不平,有较多炎性细胞堆积,并且开始有脂肪细胞出现;第2周时两组伤口表皮均开始生长,均未见角质层及毛囊、皮脂腺、汗腺等皮肤附属结构,但外泌体组伤口再上皮化程度优于模型组;第4周时两组伤口均完全愈合,形成真皮层和表皮层,外泌体组胶原纤维排列成束较模型组更规则,外泌体组可见有汗腺导管再生,两组均未见明显皮脂腺、毛囊再生和角质层形成。"
| [1] 李晓康,王舒,于杨,等.皮肤创伤修复研究进展[J].中国中西医结合皮肤性病学杂志,2016,15(1):62-65.[2] Friedenstein AJ, Gorskaja JF, Kulagina NN. Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Exp Hematol. 1976;4(5):267-274. [3] Russell KA, Chow NH, Dukoff D, et al. Characterization and Immunomodulatory Effects of Canine Adipose Tissue- and Bone Marrow-Derived Mesenchymal Stromal Cells. PLoS One. 2016;11(12): e0167442. [4] 李艳琪,王洪一,姚尧,等.人脐带源和胎盘源间充质干细胞的生物学特性比较[J].军事医学,2015,39(6):418-422.[5] 蔡金宏,林春博,杨渊.间充质干细胞分离方法的研究与进展[J].中国组织工程研究,2014,18(45):7375-7380.[6] 韩之波,杨舟鑫,池颖,等.人脐带、胎盘绒毛膜来源间充质干细胞的生物学特性比较研究[J].中国实验血液学杂志, 2012;20(3):692-696.[7] Jiang W, Tan Y, Cai M, et al. Human Umbilical Cord MSC-Derived Exosomes Suppress the Development of CCl4-Induced Liver Injury through Antioxidant Effect. tem Cells Int. 2018;2018:6079642. [8] 范冬梅,张子祥,赵英新,等.人脐带来源间充质干细胞向HepG2肝癌细胞原位移植瘤的归巢及分化[J].中国肿瘤生物治疗杂志, 2017;24(6):595-600.[9] Mooney DJ, Vandenburgh H. Cell delivery mechanisms for tissue repair. Cell Stem Cell. 2008;2(3):205-213. [10] Young HE, Duplaa C, Katz R, et al. Adult-derived stem cells and their potential for use in tissue repair and molecular medicine. J Cell Mol Med. 2005;9(3):753-769. [11] Satake K, Lou J, Lenke LG. Migration of mesenchymal stem cells through cerebrospinal fluid into injured spinal cord tissue. Spine (Phila Pa 1976). 2004;29(18):1971-1979. [12] Formigli L, Paternostro F, Tani A, et al. MSCs seeded on bioengineered scaffolds improve skin wound healing in rats. Wound Repair Regen. 2015;23(1):115-123. [13] Arno AI, Amini-Nik S, Blit PH, et al. Human Wharton's jelly mesenchymal stem cells promote skin wound healing through paracrine signaling. Stem Cell Res Ther. 2014;5(1):28. [14] Kim YJ, Yoo SM, Park HH, et al. Exosomes derived from human umbilical cord blood mesenchymal stem cells stimulates rejuvenation of human skin. Biochem Biophys Res Commun. 2017;493(2):1102-1108. [15] Isakson M, de Blacam C, Whelan D, et al. Mesenchymal Stem Cells and Cutaneous Wound Healing: Current Evidence and Future Potential. Stem Cells Int. 2015;2015:831095. [16] Phinney DG. Functional heterogeneity of mesenchymal stem cells: implications for cell therapy. J Cell Biochem. 2012;113(9):2806-2812. [17] Anzalone R, Lo Iacono M, Corrao S, et al. New emerging potentials for human Wharton's jelly mesenchymal stem cells: immunological features and hepatocyte-like differentiative capacity. Stem Cells Dev. 2010;19(4): 423-438. [18] Dominici M, Le BK, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315. [19] L Ramos T, Sánchez-Abarca LI, Muntión S, et al. MSC surface markers (CD44, CD73, and CD90) can identify human MSC-derived extracellular vesicles by conventional flow cytometry. Cell Commun Signal. 2016;14:2. [20] Marquez-Curtis LA, Janowska-Wieczorek A, Mcgann LE, et al. Mesenchymal stromal cells derived from various tissues: Biological, clinical and cryopreservation aspects. Cryobiology. 2015;71(2):181-197. [21] Farrell E, Wielopolski P, Pavljasevic P, et al. Effects of iron oxide incorporation for long term cell tracking on MSC differentiation in vitro and in vivo. Biochem Biophys Res Commun. 2008;369(4):1076-1081. [22] Li X, Bai J, Ji X, et al. Comprehensive characterization of four different populations of human mesenchymal stem cells as regards their immune properties, proliferation and differentiation. Int J Mol Med. 2014;34(3): 695-704. [23] Boukouris S, Mathivanan S. Exosomes in bodily fluids are a highly stable resource of disease biomarkers. Proteomics Clin Appl. 2015;9(3-4): 358-367. [24] Phinney DG, Pittenger MF. Concise Review: MSC-Derived Exosomes for Cell-Free Therapy. Stem Cells. 2017;35(4):851-858. [25] 张静,易阳艳.干细胞外泌体生物学功能及临床应用前景[J].中国美容医学, 2017,26(4):136-140.[26] Yamashita T, Takahashi Y, Nishikawa M, et al. Effect of exosome isolation methods on physicochemical properties of exosomes and clearance of exosomes from the blood circulation. Eur J Pharm Biopharm. 2016;(98):1-8. [27] Li P, Kaslan M, Lee SH, et al. Progress in Exosome Isolation Techniques. Theranostics. 2017;7(3):789-804. [28] Ueda K, Ishikawa N, Tatsuguchi A, et al. Antibody-coupled monolithic silica microtips for highthroughput molecular profiling of circulating exosomes. Sci Rep. 2014;(4):6232. [29] Rood IM, Deegens JK, Merchant ML, et al. Comparison of three methods for isolation of urinary microvesicles to identify biomarkers of nephrotic syndrome. Kidney Int. 2010;78(8):810-816. [30] Timmers L, Lim SK, Arslan F, et al. Reduction of myocardial infarct size by human mesenchymal stem cell conditioned medium. Stem Cell Res. 2007;1(2):129-137. [31] Lai RC, Arslan F, Lee MM, et al. Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res. 2010;4(3): 214-222. [32] Martinez MC, Andriantsitohaina R. Microparticles in angiogenesis: therapeutic potential. Circ Res. 2011;109(1):110-119. [33] Yang Y, Ye Y, Su X, et al. MSCs-Derived Exosomes and Neuroinflammation, Neurogenesis and Therapy of Traumatic Brain Injury. Front Cell Neurosci. 2017;11:55. [34] Yu B, Shao H, Su C, et al. Exosomes derived from MSCs ameliorate retinal laser injury partially by inhibition of MCP-1. Sci Rep. 2016;(6): 34562. [35] Lee C, Mitsialis SA, Aslam M, et al. Exosomes mediate the cytoprotective action of mesenchymal stromal cells on hypoxia-induced pulmonary hypertension. Circulation. 2012;126(22):2601-2611. [36] Li T, Yan Y, Wang B, et al. Exosomes derived from human umbilical cord mesenchymal stem cells alleviate liver fibrosis. Stem Cells Dev. 2013; 22(6):845-854. [37] Fang S, Xu C, Zhang Y, et al. Umbilical Cord-Derived Mesenchymal Stem Cell-Derived Exosomal MicroRNAs Suppress Myofibroblast Differentiation by Inhibiting the Transforming Growth Factor-β/SMAD2 Pathway During Wound Healing. Stem Cells Transl Med. 2016;5(10): 1425-1439. [38] Zhang B, Wang M, Gong A, et al. HucMSC-Exosome Mediated-Wnt4 Signaling Is Required for Cutaneous Wound Healing. Stem Cells. 2015; 33(7):2158-2168. [39] 张斌.脐带MSC来源exosome在皮肤组织再生中的作用及机制[D].江苏大学, 2016. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Li Jing, Xie Jianshan, Cui Huilin, Cao Ximei, Yang Yanping, Li Hairong. Expression and localization of diacylglycerol kinase zeta and protein kinase C beta II in mouse back skin with different coat colors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1196-1200. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [6] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [7] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [8] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [9] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [10] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [11] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [12] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [13] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [14] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [15] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||