Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (25): 4007-4013.doi: 10.3969/j.issn.2095-4344.0927
Previous Articles Next Articles
Autologous bone marrow mesenchymal stem cell transplantation for the treatment of early femoral head necrosis
Chen Xiang1, Wang Wei-zhuo2, Xue Jian-li2, Xia Li-feng2, Li Tao3, Qiu Ling4, Song Huan-jin2
- 1First Clinical Medical College of Shaanxi University of Chinese Medicine, Xianyang 712046, Shaanxi Province, China; 2Department of Orthopaedics, Second Affiliated Hospital of Xi’an Jiaotong University, Xi’an 710004, Shaanxi Province, China; 3Department of Orthopedics, Xi’an Honghui Hospital, Xi’an 710054, Shaanxi Province, China; 4Department of Orthopaedics, Xi’an First Hospital, Xi’an 710002, Shaanxi Province, China
-
Revised:2018-06-21Online:2018-09-08Published:2018-09-08 -
Contact:Song Huan-jin, M.D., Associate chief physician, Department of Orthopaedics, Second Affiliated Hospital of Xi’an Jiaotong University, Xi’an 710004, Shaanxi Province, China -
About author:Chen Xiang, Master, Physician, First Clinical Medical College of Shaanxi University of Chinese Medicine, Xianyang 712046, Shaanxi Province, China -
Supported by:the Natural Science Basic Research Project of Shaanxi Province, No. 2016JM8129
CLC Number:
Cite this article
Chen Xiang, Wang Wei-zhuo, Xue Jian-li, Xia Li-feng, Li Tao, Qiu Ling, Song Huan-jin. Autologous bone marrow mesenchymal stem cell transplantation for the treatment of early femoral head necrosis[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(25): 4007-4013.
share this article
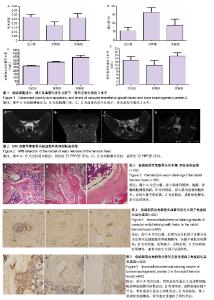
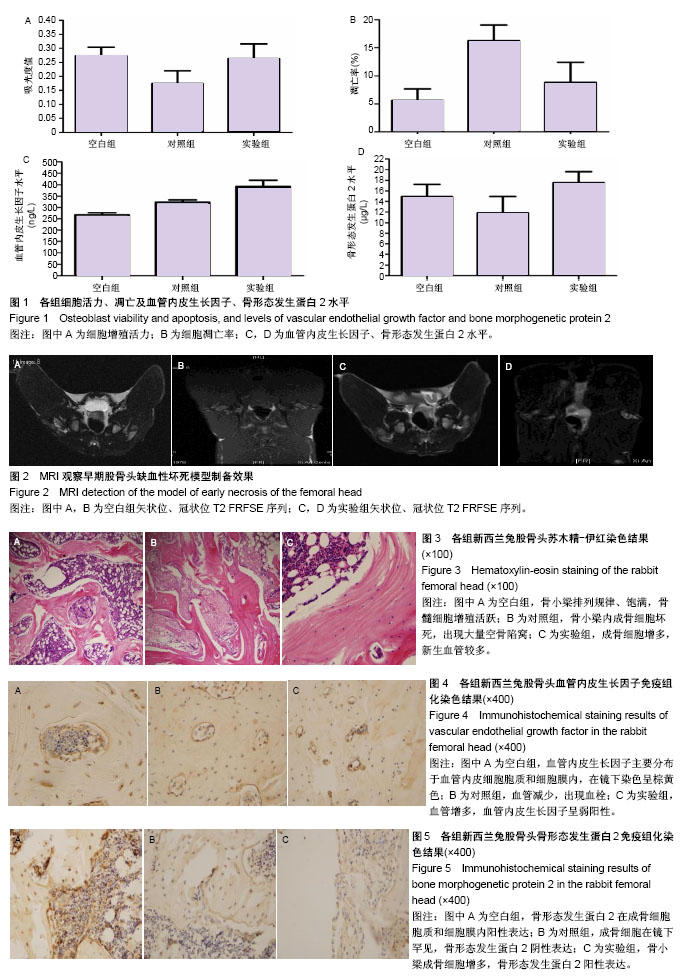
2.1 成骨细胞活力、凋亡及血管内皮生长因子、骨形态发生蛋白2水平 空白组成骨细胞吸光度值为0.273 2± 0.030 8,对照组成骨细胞吸光度值为0.174 6±0.045 6,实验组成骨细胞吸光度值为0.263 6±0.052 6。对照组与实验组吸光度值差异有显著性意义(P < 0.05),提示成骨细胞与BMSCs共培养时,可以促使成骨细胞活力增强(图1A)。 空白组成骨细胞总凋亡率为(5.6±2.1)%,对照组总凋亡率为(16.3±2.8)%,实验组总凋亡率为(8.8±3.6)%。各组间比较差异有显著性意义(P < 0.05),提示成骨细胞与BMSCs共培养时,可减少成骨细胞凋亡(图1B)。 空白组、对照组、实验组血管内皮生长因子水平分别为(266.4±10.2),(322.1±10.3),(391.7±18) ng/L;空白组、对照组、实验组骨形态发生蛋白2水平分别为(14.95± 2.26),(11.80±3.17),(17.58±2.01) μg/L。组间比较行方差分析:①血管内皮生长因子组间比较F=668.86,P=0.000,进一步行Dunnett T检验,每两组间比较P=0.000,提示每两组的血管内皮生长因子水平差异有显著性意义;②骨形态发生蛋白2组间比较F=473.847,P=0.000,进一步行Dunnett T检验,每两组间比较P=0.000,提示每两组的骨形态发生蛋白2水平差异有显著性意义(图1C,D)。 2.2 体内实验结果 造模4周后动物双后肢活动减少;治疗后观察手术动物一般情况良好,伤口无红肿、无渗液;治疗后3 d患肢无主动活动,1周后患肢开始自主活动,期间无动物死亡。 2.2.1 MRI检测造模效果 行兔股骨头冠状位(OA)及矢状位(OC)MRI检查,MRI结果T2加权抑脂序列可见:空白组兔股骨头区信号均一,未有异常信号表现(图2A,B);实验组可见兔股骨头区呈现不均匀的低信号,关节腔内可见少量关节积液,实验组MRI检查符合早期股骨头缺血性坏死期表现,提示造模成功(图2C,D)。 2.2.2 苏木精-伊红染色结果 获取标本时,按原手术切口逐层切开,各组动物切口周围组织纤维化粘连,无流脓、无溢液,取兔股骨头区域制备切片行苏木精-伊红染色(图3)。空白组股骨头骨小梁排列规律,骨小梁饱满,细胞核较大位于中央,骨小梁中罕见空骨凹陷,骨髓细胞增殖活跃,骨小梁边缘可见成骨细胞依附。对照组股骨头骨小梁内成骨细胞坏死,有大量空骨陷窝,骨髓脂肪细胞增大,主要分布在髓芯减压孔道区;实验组股骨头骨小梁结构较紊乱,成骨细胞增多,新生血管较多,骨髓脂肪细胞增多,但较对照组脂肪细胞增大;骨小梁中仍有骨细胞,空骨陷窝较空白组宽大,细胞核周可见空晕,但空骨陷窝较对照组明显减少;骨小梁周围成骨细胞较空白组减少,较对照组增多。 每组每只动物标本随机选取3张切片,每张切片随机选取5个视野进行计数。空白组、对照组、实验组血管数分别为(8.4±1.3)条,(3.5±1.2)条,(6.2±1.5)条,空骨陷窝率分别为(13.9±7.5)%,(58.3±7.3)%,(37.4±6.6)%。各组血管数、空骨陷窝率比较,差异有显著性意义(P < 0.05)。 2.2.3 免疫组化染色结果 血管内皮生长因子染色阳性细胞在镜下呈棕黄色,其主要分布于血管内皮细胞胞质和细胞膜内。空白组血管内皮生长因子阳性表达(图4A)。对照组血管减少,出现血栓,血管内皮生长因子呈阴性表达(图4B);实验组新生血管增多,血管内皮生长因子呈弱阳性,提示骨髓间充质干细胞移植可使兔股骨头区血管内皮生长因子表达增加(图4C)。 空白组、对照组、实验组血管内皮生长因子免疫组化染色吸光度值分别为0.285 9±0.066 0,0.187 2±0.037 5,0.250 0±0.058 5,组间比较差异有显著性意义(P < 0.05)。 骨形态发生蛋白2阳性染色表达在成骨细胞胞质和细胞膜内,成骨细胞主要存在于骨小梁周围边缘。空白组可见骨小梁周围成骨细胞胞质骨形态发生蛋白2阳性表达(图5A)。对照组成骨细胞罕见,骨小梁周围成骨细胞胞质骨形态发生蛋白2阴性表达(图5B)。实验组骨小梁成骨细胞增多,细胞胞质骨形态发生蛋白2阳性表达(图5C),提示骨髓间充质干细胞移植可使兔股骨头坏死区骨形态发生蛋白2表达增加。 空白组、对照组、实验组骨形态发生蛋白2免疫组化染色吸光度值分别为0.238 9±0.033 5,0.132 0±0.037 3,0.213 1±0.051 3,组间比较差异有显著性意义(P < 0.05)。"
| [1] 李莉莉,钟佩茹.非创伤性股骨头坏死发病相关因素及信号通路机制的研究进展[J].医学综述,2018,24(1):22-27.[2] 蔡伟斌,范积平,吕扬阳,等. 髓芯减压术、自体髂骨移植术与骨髓间充质干细胞移植术联合治疗战创伤致早期成人股骨头坏死[J]. 广东医学,2016,37(1):106-108.[3] Ehlinger M, Moser T, Adam P, et al. Early prediction of femoral head avascular necrosis following neck fracture.Orthop Traumatol Surg Res. 2011;97(1):79-88.[4] Bastian JD, Turina M, Siebenrock KA, et al. Long-term outcome after traumatic anterior dislocation of the hip.Arch Orthop Trauma Surg. 2011;131(9):1273-1278.[5] Song HJ, Lan BS, Cheng B, et al. Treatment of early avascular necrosis of femoral head by small intestinal submucosal matrix with peripheral blood stem cells. Transplant Proc. 2011;43(5):2027-2032.[6] Vaishya R, Agarwal AK, Gupta N, et al. Sartorius muscle pedicle iliac bone graft for the treatment of avascular necrosis of femur head.J Hip Preserv Surg. 2016;3(3):215-222.[7] Xiao D, Ye M, Li X, et al. Biomechanics analysis for the treatment of ischemic necrosis of the femoral head by using an interior supporting system.Int J ClinExp Med. 2015;8(3): 4551-4556.[8] Yuan HF, Zhang J, Guo CA, et al. Clinical outcomes of osteonecrosis of the femoral head after autologous bone marrow stem cell implantation: a meta-analysis of seven case-control studies.Clinics (Sao Paulo). 2016;71(2):110-113.[9] 张洪雨,马志强,刘建宇.干细胞治疗股骨头坏死的相关研究[J].东南大学学报:医学版, 2015,34(2):291-294.[10] Fu Q, Tang NN, Zhang Q, et al. Preclinical Study of Cell Therapy for Osteonecrosis of the Femoral Head with Allogenic Peripheral Blood-Derived Mesenchymal Stem Cells.Yonsei Med J. 2016;57(4):1006-1015.[11] Hernigou P, Flouzat-Lachaniette CH,Delambre J, et al. Osteonecrosis repair with bone marrow cell therapies: state of the clinical art.Bone. 2015;70:102-109.[12] 樊天,余方圆, 温振涛,等. 非创伤性股骨头坏死的机制及早期治疗的研究进展[J]. 现代生物医学进展, 2016, 16(21):4170-4173.[13] 王天胜,滕寿发,张英霞,等. 骨髓基质干细胞移植在激素性股骨头坏死治疗中的作用机制研究[J]. 临床和实验医学杂志, 2015, 14(1):10-12.[14] Wang T, Teng S, Zhang Y, et al. Role of mesenchymal stem cells on differentiation in steroid-induced avascular necrosis of the femoral head.ExpTher Med. 2017;13(2):669-675.[15] Tian L, Wen Q, Dang X, et al. Immune response associated with Toll-like receptor 4 signaling pathway leads to steroid-induced femoral head osteonecrosis.BMC Musculoskelet Disord. 2014;15:18.[16] 满达,赵建民,刘瑞,等. 股骨头坏死非手术治疗的研究进展[J]. 中国当代医药, 2017, 24(2):13-15.[17] Tripathy SK, Goyal T, Sen RK.Management of femoral head osteonecrosis: Current concepts.Indian J Orthop. 2015;49(1): 28-45.[18] 石正松,李强,宁寅宽,等. hBMP-2基因转染兔骨髓间充质干细胞体内移植后hBMP-2的活性检测[J]. 中国骨质疏松杂志, 2015, 21(11):1333-1337.[19] García-Gareta E, Hua J, Blunn GW.Osseointegration of acellular and cellularizedosteoconductive scaffolds: is tissue engineering using mesenchymal stem cells necessary for implant fixation?J Biomed Mater Res A. 2015;103(3): 1067-1076.[20] Wyles CC, Houdek MT, Crespo-Diaz RJ, et al. Adipose-derived Mesenchymal Stem Cells Are Phenotypically Superior for Regeneration in the Setting of Osteonecrosis of the Femoral Head.Clin Orthop Relat Res. 2015;473(10): 3080-3090.[21] Chen C, Qu Z, Yin X, et al. Efficacy of umbilical cord-derived mesenchymal stem cell-based therapy for osteonecrosis of the femoral head: A three-year follow-up study.Mol Med Rep. 2016;14(5):4209-4215.[22] Veronesi F, Torricelli P, Della Bella E, et al. In vitro mutual interaction between tenocytes and adipose-derived mesenchymal stromal cells.Cytotherapy. 2015;17(2):215-223.[23] Ferrara N, Henzel WJ.Pituitary follicular cells secrete a novel heparin-binding growth factor specific for vascular endothelial cells.BiochemBiophys Res Commun. 1989;161(2):851-858.[24] Veronesi F, Torricelli P, Della Bella E, et al. In vitro mutual interaction between tenocytes and adipose-derived mesenchymal stromal cells.Cytotherapy. 2015;17(2):215-223.[25] Daltro GC, Fortuna V, de Souza ES, et al. Efficacy of autologous stem cell-based therapy for osteonecrosis of the femoral head in sickle cell disease: a five-year follow-up study.Stem Cell Res Ther. 2015;6:110.[26] 王伟,刘丹平,张德志,等. 自体骨髓间充质干细胞骨泥治疗早期股骨头缺血性坏死近期疗效分析[J]. 医学与哲学, 2016, 37(8): 47-50.[27] Ma XW, Cui DP, Zhao DW.Vascular endothelial growth factor/bone morphogenetic protein-2 bone marrow combined modification of the mesenchymal stem cells to repair the avascular necrosis of the femoral head.Int J Clin Exp Med. 2015;8(9):15528-15534.[28] Liu X, Li Q, Niu X, et al. Exosomes Secreted from Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells Prevent Osteonecrosis of the Femoral Head by Promoting Angiogenesis.Int J Biol Sci. 2017;13(2):232-244.[29] Hernigou P, Trousselier M, Roubineau F, et al. Stem Cell Therapy for the Treatment of Hip Osteonecrosis: A 30-Year Review of Progress.Clin Orthop Surg. 2016;8(1):1-8.[30] 李民,李书奎,陈汉文,等. 髓芯减压后自体MCS移植治疗早期股骨头坏死效果[J]. 齐鲁医学杂志, 2016,31(3):340-341.[31] 王冠. 骨髓间充质干细胞治疗股骨头坏死的临床分析[J]. 中国卫生标准管理, 2016, 7(3):32-33.[32] Piuzzi NS, Chahla J, Jiandong H, et al. Analysis of Cell Therapies Used in Clinical Trials for the Treatment of Osteonecrosis of the Femoral Head: A Systematic Review of the Literature.J Arthroplasty. 2017;32(8):2612-2618.[33] Papakostidis C, Tosounidis TH, Jones E, et al. The role of "cell therapy" in osteonecrosis of the femoral head.A systematic review of the literature and meta-analysis of 7 studies. Acta Orthop. 2016;87(1):72-78.[34] 孙明林,徐辉,王景贵. 重组人骨形态发生蛋白2/纤维蛋白胶植入联合髓芯减压治疗兔股骨头缺血性坏死[J].中国组织工程研究与临床康复,2009,13(12):2257-2260.[35] 薛元锁,时述山,李亚非,等.激素性股骨头坏死病程中骨形态发生蛋白-2的改变及其意义[J].中华实验外科杂志, 2000,17(5): 455-456.[36] Hernigou P, Flouzat-Lachaniette CH, Delambre J, et al. Osteonecrosis repair with bone marrow cell therapies: state of the clinical art.Bone. 2015;70:102-109.[37] Villa JC, Husain S, van der List JP, et al. Treatment of Pre-Collapse Stages of Osteonecrosis of the Femoral Head: a Systematic Review of Randomized Control Trials.HSS J. 2016;12(3):261-271.[38] Banno K, Yoder MC.Tissue regeneration using endothelial colony-forming cells: promising cells for vascular repair. Pediatr Res. 2018;83(1-2):283-290.[39] Corre I, Paris F, Huot J.The p38 pathway, a major pleiotropic cascade that transduces stress and metastatic signals in endothelial cells.Oncotarget. 2017;8(33):55684-55714.[40] Liao S, Luo C, Cao B, et al. Endothelial Progenitor Cells for Ischemic Stroke: Update on Basic Research and Application.Stem Cells Int. 2017;2017:2193432. [41] Tenreiro MM, Correia ML, Brito MA.Endothelial progenitor cells in multiple myeloma neovascularization: a brick to the wall.Angiogenesis. 2017;20(4):443-462.[42] Samee M, Kasugai S, Kondo H, et al. Bone morphogenetic protein-2 (BMP-2) and vascular endothelial growth factor (VEGF) transfection to human periosteal cells enhances osteoblast differentiation and bone formation.J Pharmacol Sci. 2008;108(1):18-31.[43] Bianconi V, Sahebkar A, Kovanen P, et al. Endothelial and cardiac progenitor cells for cardiovascular repair: A controversial paradigm in cell therapy.PharmacolTher. 2018; 181:156-168.[44] Schlegel F, Appler M, Halling M, et al. Reprogramming Bone Marrow Stem Cells to Functional Endothelial Cells in a Mini Pig Animal Model.Med SciMonit Basic Res. 2017;23:285-294.[45] Carmona MD, Cañadillas S, Romero M, et al. Intramyocardial bone marrow mononuclear cells versus bone marrow-derived and adipose mesenchymal cells in a rat model of dilated cardiomyopathy.Cytotherapy. 2017;19(8):947-961.[46] Fukunishi T, Best CA, Ong CS, et al. Role of Bone Marrow Mononuclear Cell Seeding for Nanofiber Vascular Grafts. Tissue Eng Part A. 2018;24(1-2):135-144. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [3] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [6] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [7] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [8] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [9] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [10] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [11] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| [12] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [13] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [14] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [15] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||