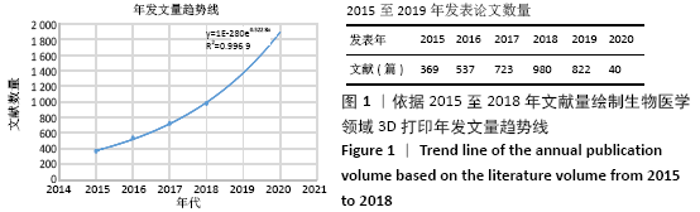
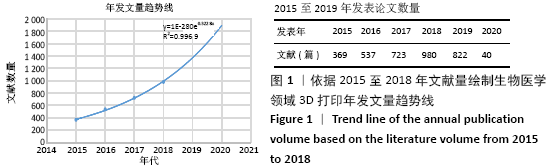
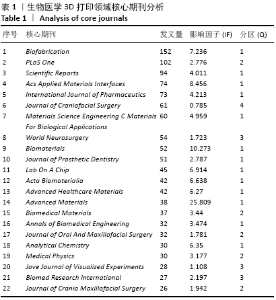
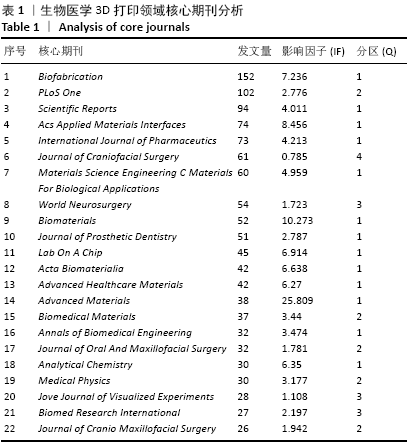
Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (15): 2385-2393.doi: 10.3969/j.issn.2095-4344.3818
Previous Articles Next Articles
Knowledge mapping of three-dimensional printing in biomedical field based on VOSviewer
Song Yahan1, Wu Yunxia2, Fan Daoyang2
- 1Library, 2Department of Orthopedics, Peking University Third Hospital, Beijing 100191, China
-
Received:2020-06-15Revised:2020-06-19Accepted:2020-07-23Online:2021-05-28Published:2021-01-05 -
Contact:Song Yahan, Library, Peking University Third Hospital, Beijing 100191, China -
About author:Song Yahan, Master, Library, Peking University Third Hospital, Beijing 100191, China
CLC Number:
Cite this article
Song Yahan, Wu Yunxia, Fan Daoyang. Knowledge mapping of three-dimensional printing in biomedical field based on VOSviewer[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(15): 2385-2393.
share this article

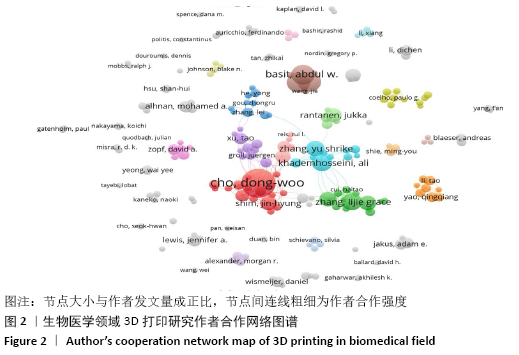
2.3 论文作者、机构及国家分布及合作网络分析 发文量排名前5位的作者分别是韩国浦项科技大学的CHO, DONG-WOO,英国伦敦大学学院药学院的 BASIT,ABDUL W、GAISFORD,SIMON和GOYANES,ALVARO,哈佛医学院的KHADEMHOSSEINI,ALI。各学者之间相互合作逐渐成为一种趋势,可在一定程度上促进该学科的发展。利用VOSVIEWER绘制作者共现图谱发现作者合作关系,见图2。通过图谱可以看出在生物医学3D打印领域,形成了以上述作者为核心的多个合作子网络,子网络内各学者之间均有较强的学术联系,但子网络外存在强度不一的合作关系,以BASIT,ABDUL W作者为核心的子网络与其他子网络合作较少,该子网络研究以药物制剂为主。通过作者合作网发现某领域的研究小团体,有助于拓展与自身研究方向不同的其他研究人员交流与合作。 "
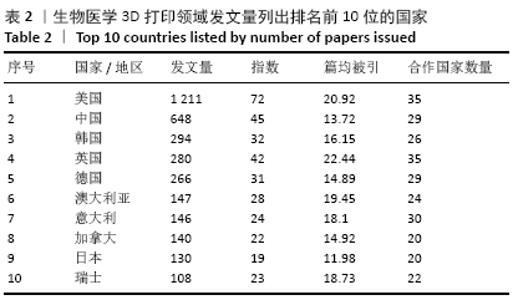
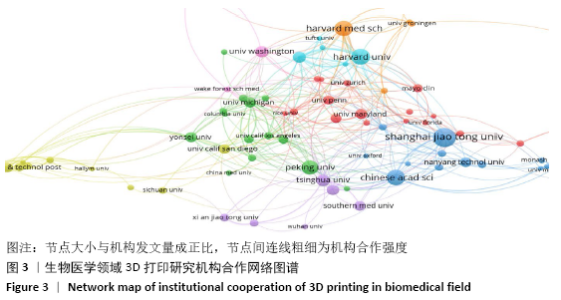
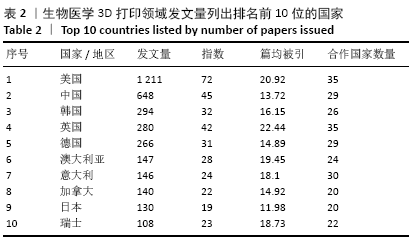
统计的3 471篇文献中共涉及59个国家或地区,按发文量列出排名前10位的国家,比较这些国家的发表论文量、h指数、篇均被引频次和合作度,以考察和分析这些国家在医学3 d研究中的科研实力及国际影响力,见表2。美国的论文数量、h指数、篇均被引频次以及合作国数量相比其他国家/地区有明显的优势,可见其雄厚的科研实力及较高的科研投入。中国虽然发文量排名第2位,但篇均被引频次较低,说明中国论文在质量上还有待提高,应引起国内研究者的重视,在注重论文数量的同时,也要着力提高论文质量。韩国与英国虽然发文量不及中国的一半,但h指数相当,篇均被引频次远高于中国,说明了这2个国家在该领域的深远学术影响力和一流科研水平。绘制国家合作图谱见图4,可看出中国与多国/地区之间建立了国际合作,根据连线粗细,与美国合作最多,依次是英国、韩国、德国、澳大利亚、新加坡、瑞士和加拿大等。 "

2.4 文献共被引分析 利用VOSviewer分析所有参考文献共82 321篇,设定最低被引次数为30次,对纳入的112篇参考文献进行共被引关联度分析和绘制可视化图谱,见图5。VOSviewer软件将上述参考文献分为4类,每类用不同的颜色表示。红色为聚类#1,包括45篇文献,主要研究内容为3D生物打印;绿色为聚类#2,包括35篇文献,主要研究内容为3D打印在临床外科中的应用;蓝色为聚类#3,包括19篇文献,主要研究内容为3D打印在药物制剂领域应用;黄色为聚类#4,包括13篇文献,主要研究内容为组织工程支架制造。选取每个聚类中关联强度和被引次数排名前3的非综述类文献为重点文献,见表3。表中的年代分布能大致展现出医学3D打印研究的发展进程,组织工程应用3D打印技术研究较早,并逐渐应用到临床,而3D生物打印和药物制剂领域应用则是近年的主要研究内容。其中,组织工程支架制造重点文献年代跨度从2002到2014年,说明持续不断的技术改进贯穿在整个研究过程中。 "

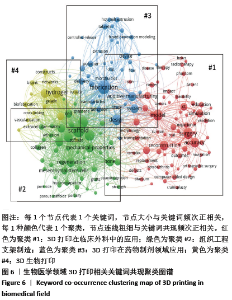
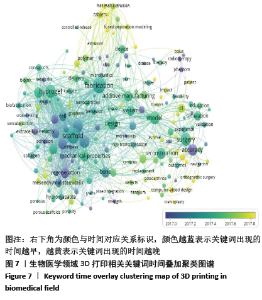
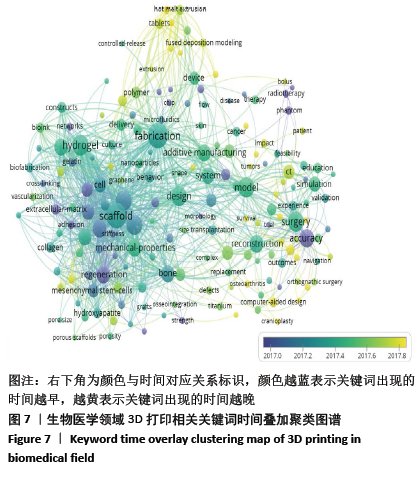
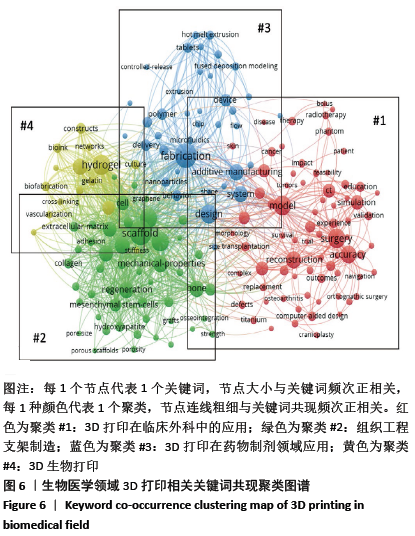
2.5 研究热点主题分析 文章获取了3 471篇文献,共获取13 036个关键词。关键词的准确性和频次是影响共现方法识别领域研究热点结果准确性的两个重要因素[20]。为使分析结果更准确,先对文献中的关键词进行清洗整理,主要包括对词的大小写及单复数,全称与缩写及同义词进行统一。 2.5.1 关键词聚类 通过VOSviewer对清洗整理后的关键词进行统计,设定频次大于等于20次为高频关键词,取消频次最高但无分析意义的关键词“3D printing”,建立共词矩阵并绘制关键词共现网络图谱,包括聚类图、时间叠加图及密度图,见图6-8。根据关键词共现聚类可把目前医学领域3D打印的研究方向分为4大类,分别为红色区域#1临床应用;绿色区域#2组织工程支架;蓝色区域#3药物制剂领域的应用;黄色区域#4 3D生物打印。 将文献发表时间叠加到关键词共现网络中获取关键词时间叠加图见图7,即不同的颜色对应着关键词在文献中出现的平均年份,从图7可发现该领域研究演化趋势,#1临床应用和#3药物制剂领域的应用聚类中黄色节点较多,是医学领域3D打印最新研究热点。关键词密度图谱上每一点都会根据该点周围元素的密度来填充颜色,密度大小依赖于周围元素的数量以及这些元素的权重大小,蓝色表示密度低端区域,红色代表密度高的区域。密度视图可以快速发现某一研究领域的主要研究内容,从图8可以看到医学领域3D打印研究重点包括外科、支架、水凝胶、聚合物、再生、骨、间充质干细胞和机械性能等方向。 2.5.2 3D打印在临床应用 红色区域为#1聚类,共74个词,以“模型”和“外科手术”为核心主题,主要研究3D打印的临床应用,包括关节置换术、下颌骨重建、颅骨成形术、正颌外科、钛、植入、癌症、放射治疗、术前计划、立体光刻、快速成型和教育等关键词。3D打印假体和植入物的研究起步较早,且发展迅速,已应用于骨科、颌面外科、颅外科和脊柱等外科手术中,包括用于重建颅骨和面部骨骼的颅面植入物,下颌假体和钛髋关节[21-23]。由于钛及钛合金具有良好的机械性能和生物相容性,成为植入物的主要打印材料。如何通过改变打印材料的各种参数来调节植入物的性能,使植入物从外形到力学性能与人体自身骨达到双重适配,需要进一步的深入研究[24-26]。在解剖模型方面,由于人体结构复杂且个体差异大,外科手术风险极高,利用3D打印可准确再现病变局部的复杂解剖结构及其空间关系,利于医生做充分的术前规划[27]。此外,3D打印解剖模型也逐渐应用于医学生的基础教育[28-30],3D打印解剖模型在术前规划及教育的应用效果有待于进一步评估。3D打印的个性化导航模板可用于引导穿刺、切割、固定和重建等,临床医生根据手术导板的导向作用精确进行手术操作,缩短手术时间并降低手术风 险[31-33]。在近距离治疗恶性肿瘤过程中,利用3D打印模板联合CT引导可增强125I放射性粒子组织间植入的准确性和安全性[34-35]。建立模型结构是打印模型的基础,可以通过计算机辅助设计软件(CAD)直接绘制,或者基于核磁共振成像(MRI)或计算机断层扫面(CT)技术获取医学影像,再利用三维重建软件建立打印结构模型[36]。3D打印的解剖模型彻底改变了外科医生手术方式,各种植入物及假体的应用,提升了临床诊疗效果。随着新材料的开发与打印技术的发展,3D打印将能够解决更多的临床疑难问题。 2.5.3 组织工程支架 绿色区域为#2聚类,共49个词,以“组织工程”和“支架”为核心主题,主要研究3D打印组织工程支架材料及应用,包括机械性能、多孔支架、间充质干细胞、羟基磷灰石、聚已内酯、胶原蛋白、细胞外基质、石墨烯、纳米纤维、骨组织工程、血管生成和再生等关键词。支架是组织工程的核心内容之一,3D打印支架技术是一个正在快速发展的研究领域。打印的组织工程支架既要有结构支撑作用,还要有利于细胞的黏附、增殖和分化,诱导缺损组织的修复或再生。因此支架的材料选择和设计是研究核心。近年来,支架材料呈现从单一同种向多种复合的发展趋势,通过取长补短的材料性质来改善其生物学性能和力学性能,且不同组织亦需要选择不同的支架材料。以羟基磷灰石和磷酸钙为主的生物陶瓷支架具有良好的生物相容性、生物降解性、高机械强度和固有的骨诱导能力,被广泛用于3D打印。纳米羟基磷灰石/壳聚糖/聚己内酯多孔复合支架材料,具有适度的抗压强度、一定的孔隙率、适宜的降解速度和吸水率,具备细胞生长的环境要求[37]。羟基磷灰石/聚己内酯支架内部经扫描电镜显示为整齐排列的纤维网状结构,纤维粗细均匀,并具有三维立体结构,支架具有适当的结构参数,包括最佳的孔隙率和孔径,以及孔的连通性,以促进细胞迁移和营养物质流通。这些特点都与3D打印技术能够精确调控支架内部结构密不可分[38-39]。基于石墨烯的3D复合支架具有良好的生物相容性、机械性能、比表面积高,能促进细胞黏附、增殖和分化等特点,近年来成为了骨组织工程领域的研究热点。氧化石墨烯与天然聚合物壳聚糖、聚已内酯的复合支架均能够改善支架的生物相容性,增强细胞迁移及整体分布,从而影响细胞的附着和渗透,促进前成骨细胞增殖和分化[40-42]。 大量研究表明,支架的微观结构,包括孔隙率、孔径和相互连接的孔结构,在增强细胞活力和促进组织生长方面发挥着关键作用。有足够孔隙率的支架在保持力学性能的同时能够促进骨生成[43]。孔径350 μm和500 μm的植入物在细胞生长、迁移和黏附方面有较好的生物相容性,但350 μm孔径支架在成骨基因的表达水平上有所增加[44]。在增加孔隙率和孔径加速细胞生长的同时,还要保障支架的机械性能,以达到良好的平衡。组织工程支架正在从实验研究向临床应用不断发展,3D打印支架的材料选择和微观结构设计仍将是组织工程的研究热点。 2.5.4 3D打印在药物制剂领域的应用 蓝色区域为#3聚类,共38个词,以“药物输送”和“药物释放”为核心主题,主要研究3D打印在药物制剂领域的应用,包括热熔挤出(HME)、熔融沉积成型(FDM)、个性化医疗、控制释放、剂型、聚合物、纳米粒子、微流体元件、形状和稳定性等关键词。2015-08-03首款由3D打印技术研发制造的左乙拉西坦(levetiracetam,SPRITAM)速溶片获得美国食品药品监督管理局(FDA)批准于2016年正式上市,加快了3D打印技术在药物制剂领域的应用研究[45]。3D打印技术可以打印出不同几何形状、支架孔洞通道与强度密度、药物浓度梯度或离散分布、多药在同一药剂中准确定位的各类型片剂,从而实现准确控制药物释放速率、释放时间、释药量及体内释药位置。热熔挤出(HME)、熔融沉积成型(FDM)是高频关键词,是药物制剂打印技术主要研究方向。通过FDM打印技术可以实现药物释放速率的控制,同时可根据患者个人特点来调整片剂释药速率,实现个性化药品设计[46-47]。 近年来,各种研究通过控制3D打印产品的形状、大小及孔隙来进行缓、控释制剂的进一步开发[48-49]。此外,聚合物、纳米粒子、微流控做为较高频关键词,也是3D打印药物领域的研究重点。聚合物是3D打印药品所需的关键材料,有研究通过改变材料来获取不同的药物释放行为[50]。这就需要在了解聚合物或聚合物活性药物成分组合性能的基础上,研发更多适于基于挤出的3D打印药物的聚合物。将3D打印和纳米技术同时应用在药物制剂领域,生产载有药物的纳米胶囊的固体剂型,开发新型药物递送系统,是个性化药物发展新方向[51]。在药物分析领域,3D打印技术片可明显缩短传统制备微流控芯片的时间,并且能批量制造[52]。由此可见,在精准医疗模式下,定制个性化药物将是3D打印药物的长期研究方向。 2.5.5 3D生物打印 黄色区域为#4聚类,共15个词。以“3D生物打印”和“生物墨水”为核心主题,包括水凝胶、海藻酸盐、交联、刚度和干细胞等关键词。3D生物打印基于3D打印技术,是将活细胞与凝胶类支架材料混合成“生物墨水”同时置于打印机的喷头中,在数字三维模型驱动下,按照增材制造原理定位打印。3D生物打印对生物墨水要求较高,需具备稳定的生物相容性和合适的力学性能、可降解性及生物功能等。生物墨水既要满足所打印支架的要求,还需要确保支架中细胞的活性。 目前研究的3D生物打印支架材料主要可分为:①天然材料,如海藻酸盐、明胶、纤维蛋白、胶原、丝素蛋白、透明质酸钠、壳聚糖和脱细胞外基质等;②合成材料,如聚己酸内酯、聚乙二醇及聚乳酸一羟基乙酸共聚物等;③复合支架材料,如聚乳酸/壳聚糖、磷酸钙/纤维蛋白胶原等;④新型材料,如纳米生物材料及可控性智能材料等[53-55]。 水凝胶具有良好亲水性、锁水性、组织黏附性和生物降解性,并可以给细胞提供良好的生长环境,是常用的打印材料之一,包括天然的海藻酸盐水凝胶以及人工合成的聚乙二醇和丙烯酰胺等。海藻酸盐与其他材料交联可提高其性能,将海藻酸钠与聚乙二醇交联,打印的支架具有更好的弹性[56]。相比海藻酸盐和海藻酸盐/明胶,纤维蛋白和胶原蛋白具有适合的降解性及可打印性,支持与血管生成相关的内皮细胞增殖,这类水凝胶最适合内皮细胞的生物打印[57]。此外,藻酸盐溶液的浓度和水凝胶的刚度也是影响细胞迁移和形态的因素[58]。应用3D生物打印已制造出具有一定结构和功能的组织器官。以生物相容性较好的静电纺纳米纤维和壳聚糖为材料打印了人工血管支架[59]。以胶原、水凝胶为支架材料,人脐静脉内皮细胞为种子材料打印出具有灌注功能的血管模型,支持邻近组织活力,并显示出对血浆蛋白和高分子量葡聚糖分子的屏障功能,可较好地模仿人体内微循环、血液渗透等生命过程[60]。海藻酸盐与纳米纤维素交联可以提升黏度,更加有利于细胞黏附。这两种材料形成的生物墨水加入软骨细胞可以打印出人耳和半月板[61]。将干细胞作为种子细胞种植在多孔生物材料支架中,能促进软骨细胞增殖[62]。3D生物打印干细胞是新的研究焦点,现阶段主要是体外研究。目前可用于3D生物打印的材料有限,合理选取材料并加工成适于打印的生物墨水、打印的支架材料可以成形并具有一定力学性能和精细结构、种子细胞可以黏附于支架并存活和发挥作用是3D生物打印的研究热点,并且要进一步研究打印后的组织器官随着时间变化而产生的结构、力学性能变化。 "
| [1] BERMAN B. 3-d printing: the new industrial revolution. Bus Horiz. 2012;55(2):155-162. [2] CAI H, LIU Z, WEI F, et al. 3D printing in spine surgery. Adv Exp Med Biol. 2018;1093:345-359. [3] DAWOOD A, MARTI MB, SAURET-JACKSON V, et al. 3D printing in dentistry. Br Dent J. 2015;219(11):521-529. [4] ZHU W, MA X, GOU M, et al. 3D printing of functional biomaterials for tissue engineering. Curr Opin Biotechnol. 2016;40:103-112. [5] TRENFIELD SJ, AWAD A, MADLA CM, et al. Shaping the future: recent advances of 3D printing in drug delivery and healthcare. Expert Opin Drug Deliv. 2019;16(10):1081-1094. [6] VAN ECK NJ. Software suvey: VOSviewer, a computer program for bibliometric mapping. Scientometrics. 2010;84(2):523-538. [7] 刘金花,崔金梅.基于VOSviewer的领域性热门研究主题挖掘[J].情报探索,2016(2):13-16. [8] KOLESKY DB, TRUBY RL, GLADMAN AS, et al. 3D bioprinting of vascularized, heterogeneous cell-laden tissue constructs. Adv Mater. 2014;26(19):3124-3130. [9] PATI F, JANG J, HA DH, et al. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat Commun. 2014;5:3935. [10] KANG HW, LEE SJ, KO IK, et al. A 3D bioprinting system to produce human-scale tissue constructs with structural integrity. Nat Biotechnol. 2016;34(3):312-319. [11] MANNOOR MS, JIANG Z, JAMES T, et al. 3D printed bionic ears. Nano Lett. 2013;13(6):2634-2639. [12] ZOPF DA, HOLLISTER SJ, NELSON ME, et al. Bioresorbable airway splint created with a three-dimensional printer. N Engl J Med. 2013; 368(21):2043-2045. [13] ZEIN NN, HANOUNEH IA, BISHOP PD, et al. Three-dimensional print of a liver for preoperative planning in living donor liver transplantation. Liver Transpl. 2013;19(12):1304-1310. [14] GOYANES A, ROBLES MP, BUANZ A, et al. Effect of geometry on drug release from 3D printed tablets. Int J Pharm. 2015;494(2):657-663. [15] GOYANES A, BUANZ AB, HATTON GB, et al. 3D printing of modified-release aminosalicylate (4-ASA and 5-ASA) tablets. Eur J Pharm Biopharm. 2015;89:157-162. [16] GOYANES A, BUANZ AB, BASIT AW, et al. Fused-filament 3D printing (3DP) for fabrication of tablets. Int J Pharm. 2014;476(1-2):88-92. [17] ZEIN I, HUTMACHER D W, TAN K C, et al. Fused deposition modeling of novel scaffold architectures for tissue engineering applications. Biomaterials. 2002;23(4):1169-1185. [18] INZANA JA, OLVERA D, FULLER SM, et al. 3D printing of composite calcium phosphate and collagen scaffolds for bone regeneration. Biomaterials. 2014;35(13):4026-4034. [19] WILLIAMS JM, ADEWUNMI A, SCHEK RM, et al. Bone tissue engineering using polycaprolactone scaffolds fabricated via selective laser sintering. Biomaterials. 2005;26(23):4817-4827. [20] 潘玮,郑鹏,黄锦泉,等.基于数据清洗“DEAN”流程的健康信息领域研究热点探测[J]. 现代情报,2018,38(10):73-77. [21] PARK EK, LIM JY, YUN IS, et al. Cranioplasty enhanced by three-dimensional printing: custom-made three-dimensional-printed titanium implants for skull defects. J Craniofac Surg. 2016;27(4):943-949. [22] RACHMIEL A, SHILO D, BLANC O, et al. Reconstruction of complex mandibular defects using integrated dental custom-made titanium implants. Br J Oral Maxillofac Surg. 2017;55(4):425-427. [23] PERTICARINI L, ZANON G, ROSSI S M, et al. Clinical and radiographic outcomes of a trabecular titanium acetabular component in hip arthroplasty: results at minimum 5 years follow-up. BMC Musculoskelet Disord. 2015;16:375. [24] SHAH FA, SNIS A, MATIC A, et al. 3D printed Ti6Al4V implant surface promotes bone maturation and retains a higher density of less aged osteocytes at the bone-implant interface. Acta Biomater. 2016;30: 357-367. [25] 沃金,袁峰,李威,等.钛支架微孔参数对成骨细胞黏附、增殖、分化的影响[J].中华实验外科杂志,2019,36(3):499-501. [26] ARABNEJAD S, JOHNSTON B, TANZER M, et al. Fully porous 3D printed titanium femoral stem to reduce stress-shielding following total hip arthroplasty. J Orthop Res. 2017;35(8):1774-1783. [27] MACKEY A, NG JI, CORE J, et al. Three-dimensional-printed uterine model for surgical planning of a cesarean delivery complicated by multiple myomas. Obstet Gynecol. 2019;133(4):720-724. [28] GARAS M, VACCAREZZA M, NEWLAND G, et al. 3D-Printed specimens as a valuable tool in anatomy education: a pilot study. Ann Anat. 2018; 219:57-64. [29] PLOCH CC, MANSI C, JAYAMOHAN J, et al. Using 3D Printing to Create Personalized Brain Models for Neurosurgical Training and Preoperative Planning. World Neurosurg. 2016;90:668-674. [30] 田晓军,邱敏,颜野,等.3D打印肾脏模型和CT影像在住院医师教学过程中的对比应用[J].中国高等医学教育,2018,(9):74-75. [31] LI P, XUAN M, LIAO C, et al. Application of intraoperative navigation for the reconstruction of mandibular defects with microvascular fibular flaps-preliminary clinical experiences. J Craniofac Surg. 2016;27(3): 751-755. [32] HUANG H, HSIEH M F, ZHANG G, et al. Improved accuracy of 3D-printed navigational template during complicated tibial plateau fracture surgery. Australas Phys Eng Sci Med. 2015;38(1):109-117. [33] WEISSLER J M, SOSIN M, DORAFSHAR A H, et al. Combining virtual surgical planning, intraoperative navigation, and 3-dimensional printing in prosthetic-based bilateral microtia reconstruction. J Oral Maxillofac Surg. 2017;75(7):1491-1497. [34] JIANG Y, JI Z, GUO F, et al. Side effects of CT-guided implantation of (125)I seeds for recurrent malignant tumors of the head and neck assisted by 3D printing non co-planar template. Radiat Oncol. 2018;13(1):18. [35] LIU JY, MAN QW, MA YQ, et al. I(125) brachytherapy guided by individual three-dimensional printed plates for recurrent ameloblastoma of the skull base. Br J Oral Maxillofac Surg. 2017;55(7): e38-e40. [36] 鞠尔男,力武,李欣芯,等.生物医疗领域三维打印的研究与应用[J].中国组织工程研究,2018,22(30):4906-4912. [37] 余和东,张丽,夏凌云,等.3D打印成型纳米羟基磷灰石/壳聚糖/聚己内酯三元复合支架材料的构建及表征[J].中国组织工程研究, 2020,24(10):1496-1501. [38] 余程,刘凯,侯长江,等.3D打印纳米羟基磷灰石/聚己内酯支架生物相容性[J].青岛大学医学院学报,2017,53(1):1-4. [39] HASSANAJILI S, KARAMI-POUR A, ORYAN A, et al. Preparation and characterization of PLA/PCL/HA composite scaffolds using indirect 3D printing for bone tissue engineering. Mater Sci Eng C Mater Biol Appl. 2019;104:109960. [40] JAKUS AE, SHAH RN. Multi and mixed 3D-printing of graphene-hydroxyapatite hybrid materials for complex tissue engineering. J Biomed Mater Res A. 2017;105(1):274-283. [41] HERMENEAN A, CODREANU A, HERMAN H, et al. Chitosan-graphene oxide 3d scaffolds as promising tools for bone regeneration in critical-size mouse calvarial defects. Sci Rep. 2017;7(1):16641. [42] UNAGOLLA JM, JAYASURIYA AC. Enhanced cell functions on graphene oxide incorporated 3D printed polycaprolactone scaffolds. Mater Sci Eng C Mater Biol Appl. 2019;102:1-11. [43] CAVO M, SCAGLIONE S. Scaffold microstructure effects on functional and mechanical performance: Integration of theoretical and experimental approaches for bone tissue engineering applications. Mater Sci Eng C Mater Biol Appl. 2016;68:872-879. [44] YANG F, CHEN C, ZHOU Q, et al. Laser beam melting 3D printing of Ti6Al4V based porous structured dental implants: fabrication, biocompatibility analysis and photoelastic study. Sci Rep. 2017;7:45360. [45] ANONYMOUS. Spritam-a new formulation of levetiracetam for epilepsy. Med Lett Drugs Ther. 2016;58(1497):78-79. [46] ARAFAT B, QINNA N, CIESZYNSKA M, et al. Tailored on demand anti-coagulant dosing: an in vitro and in vivo evaluation of 3D printed purpose-designed oral dosage forms. Eur J Pharm Biopharm. 2018; 128:282-289. [47] GOYANES A, FINA F, MARTORANA A, et al. Development of modified release 3D printed tablets (printlets) with pharmaceutical excipients using additive manufacturing. Int J Pharm. 2017;527(1-2):21-30. [48] SKOWYRA J, PIETRZAK K, ALHNAN MA. Fabrication of extended-release patient-tailored prednisolone tablets via fused deposition modelling (FDM) 3D printing. Eur J Pharm Sci. 2015;68:11-17. [49] GOYANES A, ROBLES MP, BUANZ A, et al. Effect of geometry on drug release from 3D printed tablets. Int J Pharm. 2015;494(2):657-663. [50] KEMPIN W, FRANZ C, KOSTER LC, et al. Assessment of different polymers and drug loads for fused deposition modeling of drug loaded implants. Eur J Pharm Biopharm. 2017;115:84-93. [51] BECK R, CHAVES PS, GOYANES A, et al. 3D printed tablets loaded with polymeric nanocapsules: An innovative approach to produce customized drug delivery systems. Int J Pharm. 2017;528(1-2):268-279. [52] HE Y, GAO Q, WU WB, et al. 3D Printed Paper-Based Microfluidic Analytical Devices. Micromachines (Basel). 2016;7(7):108. [53] 张慧,孟桐辉,刘琳,等.3D生物打印材料在生物医学领域中的应用及研究进展[J].中华临床医师杂志(电子版),2019,13(2):157-160. [54] 史建峰,王涵,赵蕾,等.3D生物打印支架材料的研究进展[J].中国药事,2018,32(10):1406-1411. [55] CHIMENE D, LENNOX KK, KAUNAS RR, et al. Advanced bioinks for 3d printing: a materials science perspective. Ann Biomed Eng. 2016;44(6): 2090-2102. [56] HONG S, SYCKS D, CHAN HF, et al. 3D Printing: 3D Printing of Highly Stretchable and Tough Hydrogels into Complex, Cellularized Structures. Adv Mater. 2015;27(27):4034. [57] BENNING L, GUTZWEILER L, TRONDLE K, et al. Assessment of hydrogels for bioprinting of endothelial cells. J Biomed Mater Res A. 2018;106(4): 935-947. [58] SHI P, LAUDE A, YEONG WY. Investigation of cell viability and morphology in 3D bio-printed alginate constructs with tunable stiffness. J Biomed Mater Res A. 2017;105(4):1009-1018. [59] LEE SJ, HEO DN, PARK JS, et al. Characterization and preparation of bio-tubular scaffolds for fabricating artificial vascular grafts by combining electrospinning and a 3D printing system. Phys Chem Chem Phys. 2015; 17(5):2996-2999. [60] LEE VK, KIM DY, NGO H, et al. Creating perfused functional vascular channels using 3D bio-printing technology. Biomaterials. 2014;35(28): 8092-8102. [61] MARKSTEDT K, MANTAS A, TOURNIER I, et al. 3D Bioprinting human chondrocytes with nanocellulose-alginate bioink for cartilage tissue engineering applications. Biomacromolecules. 2015;16(5):1489-1496. [62] APELGREN P, AMOROSO M, LINDAHL A, et al. Chondrocytes and stem cells in 3D-bioprinted structures create human cartilage in vivo. PLoS One. 2017;12(12):e189428. |
| [1] | Lu Dezhi, Mei Zhao, Li Xianglei, Wang Caiping, Sun Xin, Wang Xiaowen, Wang Jinwu. Digital design and effect evaluation of three-dimensional printing scoliosis orthosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1329-1334. |
| [2] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [3] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [4] | Liu Zhengpeng, Wang Yahui, Zhang Yilong, Ming Ying, Sun Zhijie, Sun He. Application of 3D printed interbody fusion cage for cervical spondylosis of spinal cord type: half-year follow-up of recovery of cervical curvature and intervertebral height [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 849-853. |
| [5] | Xu Junma, Yu Yuechao, Liu Zhi, Liu Yu, Wang Feitong. Application of 3D-printed coplanar template combined with fixed needle technique in percutaneous accurate biopsy of small pulmonary nodules [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 761-764. |
| [6] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [7] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [8] | Li Li, Ma Li. Immobilization of lactase on magnetic chitosan microspheres and its effect on enzymatic properties [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 576-581. |
| [9] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [10] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [11] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [12] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [13] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [14] | Fan Yinuo, Guan Zhiying, Li Weifeng, Chen Lixin, Wei Qiushi, He Wei, Chen Zhenqiu. Research status and development trend of bibliometrics and visualization analysis in the assessment of femoroacetabular impingement [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 414-419. |
| [15] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||