Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (23): 3751-3755.doi: 10.3969/j.issn.2095-4344.2693
Previous Articles Next Articles
Vitrification-based cryopreservation of tissues: strengths and existing problems
Zhang Yuan1, Zeng Min2, Zhai Bo1
- 1Department of Interventional Oncology, Renji Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200127, China; 2Shanghai Huicun Medical Technology Co., Ltd., Shanghai 201203, China
-
Received:2019-10-26Revised:2019-10-30Accepted:2019-12-20Online:2020-08-18Published:2020-07-30 -
Contact:Zhai Bo, MD, Chief physician, Professor, Department of Interventional Oncology, Renji Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200127, China -
About author:Zhang Yuan, Master, Physician, Department of Interventional Oncology, Renji Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200127, China
CLC Number:
Cite this article
Zhang Yuan, Zeng Min, Zhai Bo. Vitrification-based cryopreservation of tissues: strengths and existing problems[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(23): 3751-3755.
share this article
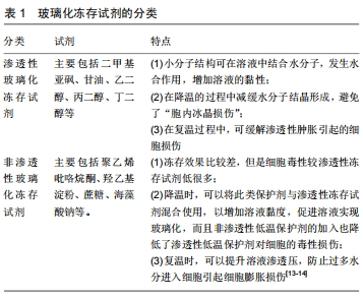
2.1 玻璃化冻存的生物学原理 低温冻存条件下,细胞内的生化代谢反应受到极大抑制,细胞可以长期保持低代谢甚至停止代谢的稳定状态,其生物学活性也因此而得到保留。大多数细胞在温度不低于-10 ℃时,细胞受质膜保护,可处于过冷状态而不结冰。在温度降到-10 ℃以下,过冷胞质极易形成冰晶。形成冰晶的危险温区介于-15 ℃至-50 ℃之间,采取适当的冷冻方法和优化的冻存液成分配比可以使细胞在冻存和复苏过程中快速安全通过危险温区,有效避免冰晶的产生[12]。玻璃化冷冻保存就是利用这些高浓度的低温保护剂组合成玻璃化冻存液,通过与水分子发生强烈的水合作用,增加溶液黏性,降低冰晶形成速度,从而使细胞在快速降温或复温过程中得以保护。 2.2 玻璃化冻存试剂的分类 见表1。①渗透性玻璃化冻存试剂:二甲基亚砜被认为是渗透性玻璃化冻存试剂中最优选的细胞保护剂[13]。由于玻璃化冻存试剂需要保持较高浓度,在冻存液导入及洗脱过程中对细胞及组织有毒性损害,所以细胞冻存后必须进行洗涤,清除降低冻存试剂的毒性影响。②非渗透性玻璃化冻存试剂:这类冻存试剂的冻存效果比较差,但是细胞毒性较渗透性冻存试剂低很多。 "


2.3 组织的玻璃化冻存 组织冻存不同于细胞冻存,在降温或复温时,由于组织结构复杂,各部温度变化不同步,可能出现热应力释放导致的结构破坏,导致冻存效果不近人意[15-16]。这一问题可以通过使用浓度更高、具有更强玻璃化能力的冻存试剂来缓解,但这种方法的代价是对组织的高毒性和高渗透性损害。为了降低整体损伤风险,可以混合使用多种低毒冻存试剂,添加非穿透性冻存试剂,或者添加可直接抑制冰核产生或重结晶的冻存试剂[17]。 2.3.1 卵巢的玻璃化冻存 卵巢组织的冻存与移植是实现癌症妇女患者在放化疗前生育能力保存最有希望的方法[18-20]。自体冻存卵巢移植成功后,该方法已经于2004年首次在人类临床上得到应用,至今共有120多个婴儿通过这种方法顺利出生[21-22]。SILBER等[23]进行了新鲜卵巢移植与冷冻卵巢移植后婴儿出生率的对照研究,11名新鲜卵巢移植的接受者顺利产下11名健康婴儿,另有13名冷冻卵巢移植的接受者产下9名健康婴儿。DONNEZ等[24]报道了60例冷冻卵巢组织原位移植的研究结果,60例中有11例受孕,其中6例产下12例健康婴儿。可见,玻璃化冻存对卵巢组织和卵泡细胞仍具有一定的毒性损伤作用,冷冻技术还未达到完全成熟的水平,玻璃化冻存卵巢技术还需进一步的优化和提升。KONG等[25]在研究中发现抗冻蛋白对再结晶有抑制作用,在玻璃化小鼠卵巢组织升温过程中添加抗冻蛋白可改善卵巢的组织活性与功能。NATEGHI等[26]通过对比乙二醇、丙二醇、二甲基亚砜对冻存蛋鸡卵巢的影响,发现乙二醇和二甲基亚砜联合应用可减轻玻璃化蛋鸡卵巢的冷冻损伤和凋亡基因的表达。BRITO等[27]分别采用浓度为0.1 mol/L和0.5 mol/L的蔗糖、海藻糖、棉籽糖混合40%乙二醇对成年猫的卵巢进行玻璃化,实验发现海藻糖混合乙二醇冻存复苏卵巢后的正常卵泡百分率最接近新鲜对照组(62.9±4.1)%。如果在现有的研究基础上对冷冻保存作进一步优化处理,将有望实现卵巢的高效保存和功能保留。 2.3.2 皮肤的玻璃化冻存 目前人造真皮和人造复合皮已经被广泛应用于烧伤后瘢痕修补、开放性创面植皮等多个方面[28-29],但只有对供体皮肤进行有效的保存,才能保证有充足的移植资源。玻璃化冻存技术可应用于供体皮肤长期的活性保存,有助于提高移植皮肤的存活率。WILSON等[30]进行了一项冻存皮肤移植治疗骨软组织开放性创面的有效性和安全性研究,所有15例患者均进行标准的冻存皮肤移植治疗,其中14例(93.3%)伤口完全愈合,创面覆盖肉芽组织的平均时间为36.14 d,创面完全愈合的平均时间为133 d,平均移植次数为2次,术后无与移植相关的不良反应。HOLZER等[31]对猪的新鲜皮肤移植和冷冻皮肤移植进行了横向对比研究,实验结果发现在移植早期二者可观察到显著的表观差异,但各组的总存活时间无差异。WOOD等[32]对比研究了甘油化保存、包含二甲基亚砜的冷冻保存和不包含二甲基亚砜的冷冻保存对人自体皮肤移植的组织力学变化。实验结果表明,甘油化和冷冻保存都会引起皮肤显著的组织力学改变,其中包含二甲基亚砜的冷冻保存引起皮肤的弹性纤维损伤,而甘油化则导致了真皮-表皮分离和弹力蛋白断裂,无二甲基亚砜的冷冻保存对皮肤的组织力学改变最大。NAALDIJK等[33]发现羟乙基淀粉可代替二甲基亚砜对真皮成纤维细胞和角质形成细胞进行冷冻保存,显著降低了冻存毒性,取得了良好的冻存效果。进一步完善冻存方法和冻存试剂的成分配比有利于提高皮肤的冻存效果。 2.3.3 角膜的玻璃化冻存 供移植角膜的短缺现象在许多国家日益严重。因此,角膜的长期保存是一个亟待解决的问题。早在20世纪六七十年代,就有学者对供体角膜的冷冻保存展开了研究,随访发现当年冻存的16例移植角膜有4例(25%)在27年后仍保持良好的功能状态,表明冷冻保存可以实现角膜的功能保留[34]。后来不断有学者对冷冻试剂进行探索和改良。然而,甘油、二甲基亚砜、甲酰胺、丙二醇、丁二醇等一系列冻存试剂的改良实验均以失败告终,角膜损伤严重,上皮细胞损伤明显[35-36]。后来又有学者对角膜上皮细胞进行冻存研究,采用10%甘油或二甲基亚砜将角膜在-80 ℃或-196 ℃条件下储存4或12周。实验结果表明,二甲基亚砜冻存的角膜上皮细胞在-80 ℃时的结构损伤更为明显,而甘油冷冻保存的角膜上皮细胞在-196 ℃条件下仅观察到微小的形态学变化[37]。可见,角膜的毒性损伤承受能力较弱,低毒性是冻存角膜试剂的必要条件。由于角膜的冻存技术要求很高,目前仍处于试验阶段。 2.3.4 肿瘤组织的玻璃化冻存 传统的组织标本保存会使活肿瘤组织完全失去活性,导致大量宝贵的临床活肿瘤样本因无法得到及时利用和长期活性保存而被浪费。医学研究的发展需要高质量的疾病资源支持,虽然国内不同层次的临床生物样本库相继建立,但是实际上临床标本资源并没有得到及时有效的利用[38]。有研究证明玻璃化冻存可实现肿瘤组织的活性保存,冻存前后肿瘤组织的生物学活性和组织学特征均未发生显著改变[39-40]。通过玻璃化冻存技术可以大幅提升活性肿瘤组织的利用率,对是实验标本的保存给予坚实的技术支持。 2.3.5 骨关节软骨的玻璃化冻存 骨关节软骨冻存技术常用于实验标本和软骨移植物的保存,在不加保护剂的情况下,软骨中的黏多糖成分浓度容易发生显著改变,导致软骨的硬度下降,进而影响了骨软骨移植的功能和长期存活的能力[41]。由于骨软骨的自我再生潜力非常有限,其功能常受限于骨关节炎、骨创伤等病变,极大地影响了患者的生活质量。软骨移植是治疗关节软骨缺损的有效方法,但由于不能长时间保存软骨,其应用受到限制[42]。近年来,有学者通过植入冷冻保存的活骨软骨同种异体移植物以促进膝关节软骨修复,疗效较为满意[43]。另有学者通过注射低温冻存骨髓基质细胞修复椎间盘,实验结果表明其治疗潜力巨大[44]。RAJAGOPAL等[45]则通过髂关节软骨细胞冻存实验,成功证实了冻存技术安全可行,软骨细胞的生物学活性和特征未发生显著改变。随着冻存技术的不断优化,冻存软骨细胞修复关节软骨损伤有望在实验与临床治疗中得到同步推进。 "

| [1]李建国,李盼,尹合勇,等.五种玻璃化冻存试剂单独或组合应用对髓核细胞的毒性分析[J].中国组织工程研究, 2016,20(11):1570-1576. [2]SOLANKI PK,RABIN Y.Analysis of polarized-light effects in glass-promoting solutions with applications to cryopreservation and organ banking. PloS one.2018;13(6): e0199155. [3]窦蒙家,张明宽,饶伟,等.人体低温保存——通向未来“永生”之路[J].科学(上海),2017,69(6):1-4. [4]FINGER EB, BISCHOF JC. Cryopreservation by vitrification: a promising approach for transplant organ banking.Curr Opin Organ Transplant.2018;23(3):353-360. [5]刘威,郭明伟,郭治宇,等.胞内冰形成机理研究进展[J].制冷学报, 2018, 39(3):126-134. [6]邱佳裔,贾晓青,黄岗,等.细胞,组织冻存方法及应用的研究进展[J].中国生物制品学杂志,2017,30(5):546-550. [7]袁海涵,马玉珍.玻璃化冻存卵巢的研究进展[J].世界最新医学信息文摘(电子版), 2016(81):12-13. [8]ONOFRE J, FAES K, KADAM P, et al. What is the best protocol to cryopreserve immature mouse testicular cell suspensions.Reprod Biomed Online. 2018;37(1):6-17. [9]TAJIMI H, YAMAZAKI T, OIKE S, et al. Vitrification for bovine embryos with low-quality grade.Anim Sci J. 2018;89(8):1194-1200. [10]HOSSEINI A, KHALILI MA, TALEBI AR, et al. Cryopreservation of Low Number of Human Spermatozoa; Which is Better: Vapor Phase or Direct Submerging in Liquid Nitrogen.Hum Fertil (Camb). 2019;22(2): 126-132. [11]DONNEZ J, GARCIA-SOLARES J, DOLMANS MM. Fertility preservation in women with ovarian endometriosis. Clin Med Insights Reprod Health. 2019;13:1179558119873386. [12]秦廷武,莫湘涛,杨志明.生物活性产品玻璃化冷冻保存研究进展[J].生物医学工程学杂志,2005,22(5):1070-1074. [13]KILBRIDE P, MORRIS GJ. Viscosities encountered during the cryopreservation of dimethyl sulphoxide systems.Cryobiology. 2017; 76:92-97. [14]SULTANI AB, MARQUEZ-CURTIS LA, ELLIOTT JA, et al. Improved Cryopreservation of Human Umbilical Vein Endothelial Cells:A Systematic Approach.Sci Rep.2016;6: 34393. [15]曾玉翠,吴瑞芳.卵巢组织冻存方案的优化研究进展[J].国际生殖健康/计划生育杂志,2015, 34(2):141-144. [16]EHRLICH LE, FAHY GM, WOWK BG, et al. Thermal Analyses of a Human Kidney and a Rabbit Kidney During Cryopreservation by Vitrification.J Biomech Eng. 2018;140(1). [17]WOWK B, FAHY GM, AHMEDYAR S, et al. Vitrification tendency and stability of DP6-based vitrification solutions for complex tissue cryopreservation.Cryobiology. 2018;82:70-77. [18]MOHSENZADEH M, SALEHI-ABARGOUEI A, TABIBNEJAD N, et al. Effect of vitrification on human oocyte maturation rate during in vitro maturation procedure:A systematic review and meta-analysis. Cryobiology. 2018;83:84-89. [19]杨云,周新丽,周楠峰,等.卵母细胞冷冻保存技术新进展[J].生殖与避孕, 2015,35(3):179-184. [20]CHAPUT L, GREMEAU AS, VORILHON S, et al. Fertility preservation in oncology.Bull Cancer.2018; 105(1):99-110. [21]FISCH B, ABIR R. Female fertility preservation: past, present and future.Reproduction. 2018;156(1):F11-F27. [22]DONNEZ J, DOLMANS MM. The ovary: from conception to death. Fertil Steril.2017;108(4):594-595. [23]SILBER S. Ovarian tissue cryopreservation and transplantation: scientific implications.J Assist Reprod Genet. 2016;33(12):1595-1603. [24]DONNEZ J, DOLMANS MM, PELLICER A, et al. Restoration of ovarian activity and pregnancy after transplantation of cryopreserved ovarian tissue: a review of 60 cases of reimplantation.Fertil Steril 2013;99(6):1503-1513. [25]KONG HS, KIM EJ, YOUM HW, et al. Improvement in Ovarian Tissue Quality with Supplementation of Antifreeze Protein during Warming of Vitrified Mouse Ovarian Tissue. Yonsei Med J.2018; 59(2):331-336. [26]NATEGHI R, ALIZADEH A, JAFARI AHANGARI Y, et al: Ethylene Glycol and Dimethyl Sulfoxide Combination Reduces Cryoinjuries and Apoptotic Gene Expression in Vitrified Laying Hen Ovary.Biopreserv Biobank. 2017;15(6): 519-528. [27]BRITO DC, DOMINGUES SFS, RODRIGUES APR, et al. Vitrification of domestic cat (Felis catus) ovarian tissue: Effects of three different sugars.Cryobiology. 2018;83:97-99. [28]MEULI M, MEULI-SIMMEN C, FLAKE AW, et al. Premiere use of Integra artificial skin to close an extensive fetal skin defect during open in utero repair of myelomeningocele. Pediatr Surg Int.2013; 29(12): 1321-1326. [29]IBRAHIM SM, KAREEM OH, SAFFANAH KM, et al. Histological and mechanical evaluation of antifreeze peptide (Afp1m) cryopreserved skin grafts post transplantation in a rat model.Cryobiology.2018; 82: 27-36. [30]WILSON TC, WILSON JA, CRIM B, et al. The Use of Cryopreserved Human Skin Allograft for the Treatment of Wounds With Exposed Muscle, Tendon, and Bone.Wounds. 2016;28(4):119-125. [31]HOLZER PW, LEONARD DA, SHANMUGARAJAH K, et al: A Comparative Examination of the Clinical Outcome and Histological Appearance of Cryopreserved and Fresh Split-Thickness Skin Grafts.J Burn Care Res. 2017;38(1): e55-e61. [32]WOOD JM, SOLDIN M, SHAW TJ, et al. The biomechanical and histological sequelae of common skin banking methods.J Biomech. 2014;47(5):1215-1219. [33]NAALDIJK Y, JOHNSON AA, FRIEDRICH-STOCKIGT A, et al. Cryopreservation of dermal fibroblasts and keratinocytes in hydroxyethyl starch-based cryoprotectants.BMC Biotechnol. 2016; 16(1):85. [34]CORYDON C, HJORTDAL J, EHLERS N. Re-examination of organ-cultured, cryopreserved human corneal grafts after 27 years.Acta Ophthalmol.2009;87(2):173-175.
[35]BOURNE WM, NELSON LR. Human corneal studies with a vitrification solution containing dimethyl sulfoxide, formamide, and 1,2-propanediol. Cryobiology.1994;31(6):522-530. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [3] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [4] | Zhou Ying, Zhang Huan, Liao Song, Hu Fanqi, Yi Jing, Liu Yubin, Jin Jide. Immunomodulatory effects of deferoxamine and interferon gamma on human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1012-1019. |
| [5] | Mo Weibin, Huang Tianchang, Zeng Zhiwei, Yan Linbo. Effects of Pueraria lobata flavonoids on expressions of beta-catenin and glycogen synthase kinase 3beta in the brain of rats undergoing exhaustive exercise after long endurance exercise [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 736-741. |
| [6] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [7] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [8] | Baibujiafu·Yelisi, Renaguli·Maihemuti, Aizimaitijiang·Saiyiti, Wang Junxiang, Nijiati·Tuerxun. Stress analysis of maxillary central incisor crown implant restoration in different occlusal modes [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 567-572. |
| [9] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [10] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [11] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [12] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [13] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [14] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [15] | Maihemuti•Yakufu, Sun Qinqin, Chen Hongtao, Liu Xu, Yiliyaer•Abudusimu, Abudushalamu•Abudukelimu, Liu Jianjiang. Tension of 3D printed controllable tension band and skin regeneration of skin defect model rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 371-375. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||