Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (28): 4437-4444.doi: 10.3969/j.issn.2095-4344.2302
Previous Articles Next Articles
Application and significance of nanofibrous macroporous scaffold preparation technology for bone tissue engineering
Zhang Weizhong, Li Lei, He He, He Xin
School of Materials Science and Engineering, University of Shanghai for Science and Technology, Shanghai 200093, China
-
Received:2019-10-31Revised:2019-11-08Accepted:2019-12-19Online:2020-10-08Published:2020-08-29 -
About author:Zhang Weizhong, Master candidate, School of Materials Science and Engineering, University of Shanghai for Science and Technology, Shanghai 200093, China
CLC Number:
Cite this article
Zhang Weizhong, Li Lei, He He, He Xin. Application and significance of nanofibrous macroporous scaffold preparation technology for bone tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4437-4444.
share this article
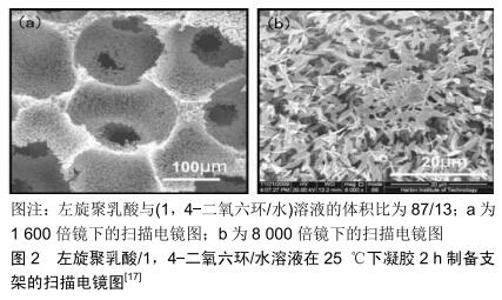
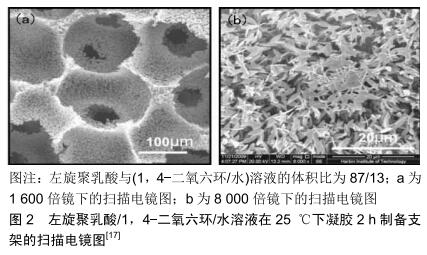
2 结果 Results 2.1 基于传统的热致相分离法 热致相分离是一种制备类似于天然细胞外基质的聚合物纳米纤维材料的方法。热致相分离首先是将聚合物与高沸点、低分子质量的液态或固态稀释剂混合,在高温时形成均相溶液,再将混合物溶液制成所需要的形状,降低温度使溶液发生相分离,然后用某些溶剂进行萃取除去稀释剂,最后进行冷冻干燥得到孔结构。郑雄飞等[17]在不结合其他致孔剂方法的条件下,基于单一的热致相分离方法制备出左旋聚乳酸纳米纤维大孔支架(图2),即将一定量的左旋聚乳酸高温溶解在一定比例的1,4-二氧六环和水的溶剂体系中,再倒入离心管中,然后进行热致相分离和凝胶一段时间,随后使用乙醇水溶液进行冷冻萃取除去1,4-二氧六环和水,最后在室温下干燥除去乙醇,冻干后得到含有100 μm以上的大孔纳米纤维左旋聚乳酸支架。CHEN等[18]使用了浊点热致相分离法在左旋聚乳酸/1,4-二氧六环/水三元体系中制备出超过300 μm的大孔纳米纤维左旋聚乳酸支架,然后再用壳聚糖溶液将左旋聚乳酸支架进行表面改性得到左旋聚乳酸/壳聚糖支架。相比于常规的热致相分离,支架的孔径会稍微小一点,但在孔壁上会形成血小板样的微观结构。最后用大鼠颅盖骨缺损模型来评估了支架在体内的成骨性,结果显示含有3%壳聚糖的复合支架显著增强了骨骼的再生。WANG等[19]使用此方法来制备具有80 μm大孔结构的纳米纤维支架-负载人骨形态发生蛋白2的聚乳酸-羟基乙酸共聚物微球/纳米纤维(BMP-2@MS/NF)复合支架,人骨形态发生蛋白2在复合支架的体外释放行为显示可持续释放超过5周。在这些载有人骨形态发生蛋白2复合支架上生长的小鼠原代成骨细胞具有更好的附着力和增殖能力。同样,碱性磷酸酶活性和茜素红S染色结果证明了这一点:载有人骨形态发生蛋白2复合支架显示成骨细胞的体外成骨分化增强。另外,来自大鼠颅盖缺损模型的体内成骨潜能评估组织学结果表明,这些负载人骨形态发生蛋白2的复合支架具有高骨诱导性。这些结果表明,载有人骨形态发生蛋白2的复合支架材料有可能被用作骨植入物,其成骨能力因人骨形态发生蛋白2的延长释放而得到增强。所以在使用单一的热致相分离法来制备纳米纤维大孔支架时,对热致相分离参数(如溶液浓度,溶剂体系,凝胶温度,凝胶时间等)、材料等要求比较高,并且产生的支架的孔隙也较小且不可控,连通性能也不好。 粒子沥滤法被广泛地运用于孔结构骨组织工程支架中。粒子沥滤法是通过将骨组织工程材料和致孔剂粒子进行混合,再利用二者不同的溶解性和挥发性把致孔剂粒子除去,最后致孔剂粒子的原有空间就变成了孔隙。所得到聚合物支架的孔尺寸取决于致孔剂粒子的尺寸。将热致相分离结合粒子沥滤法可制备出一定尺寸大孔结构的纤维支架。HAN等[20]使用此种复合方法制备由纳米羟基磷灰石颗粒增强的仿生纳米纤维大孔左旋聚乳酸支架,使用了300-450 μm的石蜡微球作为致孔剂来得到300-450 μm大孔的纳米纤维支架。LIU等[21]也使用了相同的方法和不同直径的石蜡微球(150-250,250-420,420-600 μm)制备出不同大孔结构的纳米纤维明胶支架。LIU等[22]也使用了相同的方法并且用糖作为致孔剂来制备出几百微米大孔的纳米纤维聚己内酯支架。SALERNO等[23]使用了200-400 μm和400-600 μm的明胶颗粒作为致孔剂来制备纳米纤维大孔支架。WEI等[24-25]和GUOTE等[26]均使用了糖球模板作为致孔剂来制备具有互连大孔和纳米纤维结构的左旋聚乳酸支架。CHEN等[27]使用了石蜡球来产生具有高度互连的球形大孔结构和纳米纤维结构支架。可以通过改变石蜡球的热处理时间来控制孔间连通性,并且可通过改变支架的孔隙率来控制力学性能。通过互连的大孔结构促进整个支架孔隙中的细胞接种,以及合成的胶原样基质,这些新型基质可能是骨组织工程的优秀支架。ZHANG等[28]首先将水溶性聚合物材料制成所需大孔结构的三维复制品,然后将聚合物溶液在模具中浇铸到致孔剂组件上,并进行热致相分离以形成纳米纤维基质,再用水浸出致孔剂材料,最终形成具有预先设计的大孔结构的合成纳米纤维细胞外基质。该技术有利于通过致孔剂粒子来控制大孔形状和尺寸,通过组装条件(热处理的时间和温度)控制孔间连通性,通过相分离和后处理参数控制孔壁形态,以及通过聚合物浓度和交联密度来控制力学性能。但是此方法很难将致孔剂除完,并且在除去致孔剂粒子后支架会在一定程度下产生萎缩,从而降低了支架的力学性能和孔与孔之间的连通性。 总之,热致相分离不需要复杂的设备来制备支架。通过使用致孔剂可以更好地控制热致相分离中支架的孔隙率,从而获得高于50 μm的孔径。通过溶剂相互作用方法产生的支架具有宏观构造结构,这不仅有助于细胞在纳米纤维和分化细胞的界面上生长,还有助于微观层面的组织形成。在相分离中,多孔支架不是以特定的、可再现的方式形成的,由于凝胶化、溶剂萃取等过程,制备过程耗时很长。使用有机溶剂作为溶解材料会引起人们对其形成支架中留下残留物的担忧。此外,当支架孔径低于所需孔径的1-20 μm时,将导致体内的支架与细胞整合缓慢。由热致相分离形成的支架在形成期间经历收缩,并且由于该材料具有不足的力学性能,因此通过该技术开发的支架大多数是失效的[29]。 "

2.2 基于传统的静电纺丝法 静电纺丝是一种专门制备纳米纤维材料的方法,能够模仿细胞外基质的结构。这些纳米纤维结构由于其高孔隙率和良好的表面积与体积比,以及较强的细胞黏附、迁移和增殖等特征而被广泛用于骨组织工程中。但是由于电纺纳米纤维非常长,缠结并且密集地堆积在表面上形成二维结构,因此电纺纳米纤维难以被用于制备骨组织工程支架的多孔三维结构。这是因为在静电纺丝的过程中,从喷丝头上喷出的射流存在不稳定鞭动,导致纤维随机地沉积到收集装置表面,最后就收集到排列非常紧密的二维纤维支架。 改变静电纺丝内部设备来制备不同孔径大小的纳米纤维大孔支架。CAI等[30]使用静电纺丝纱线组装技术制备了三维纳米纤维大孔左旋聚乳酸/聚己内酯复合支架,纤维长度在400-900 nm之间,孔大小在60-130 μm之间(图3)。在体外观察到,三维大孔纳米纤维支架有更多的细胞增殖和细胞向内生长,而且在人胚胎干细胞体外分化中明显检测到钙沉积。三维大孔纳米纤维支架修复骨缺损3,6周时,组织学和X射线显示在支架周围及支架内部均有精细的三维骨组织形成。这项研究表明三维大孔纳米纤维支架为人胚胎干细胞的生长提供了结构性支持并指导了骨的形成,表明该新策略成功地将电纺纤维用于骨组织工程。但是单一静电纺丝法制备的支架孔隙较小,力学性能偏低,具有不可控的三维结构。 "

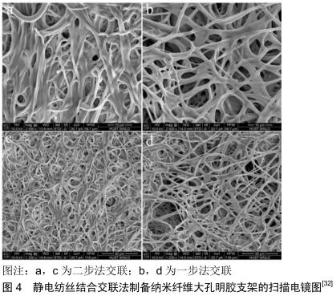
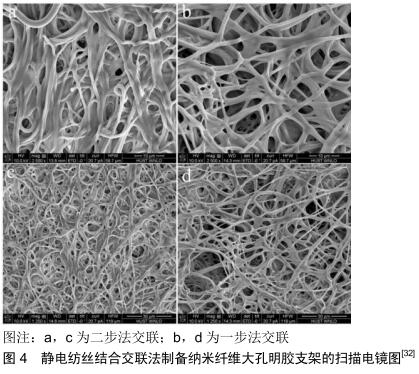
大多数由天然聚合物制造的纤维不稳定,因此需要交联以提高稳定性和力学性能。MAJIDI等[31]使用Ca2+交联结合静电纺丝来制备三维海藻酸钠/聚氧化乙烯水凝胶纳米纤维大孔支架。TENG等[32]使用戊二醛一步法交联结合静电纺丝制备了三维纳米纤维大孔明胶支架(图4)。一步法交联纤维的平均长度为(0.96±0.18) μm,短于两步法的交联纤维长度(1.62±0.43) μm;一步法交联的孔大小为(36.36±18.46) μm,大于二步法交联的孔径(13.15±4.13) μm,并且通过一步法交联制备的支架蓬松且多孔。在14 d内,两种纤维的降解率均未发现明显差异。在磷酸缓冲液溶液中浸泡14 d后,许多两步法交联的纤维融合在一起;相比之下,一步法交联纤维的形态和大孔结构没有明显变化(通常保持不变)。两步法和一步法交联明胶纤维的近似交联度分别为40%和54%。荧光显微镜和苏木精-伊红染色的结果表明,一步法交联的纤维支架中的MC3T3-E1亚克隆4细胞分布更加均匀和多样化。一步法交联的纤维增强了MC3T3-E1细胞的增殖和分化潜能。此外,一步法交联纤维对修复大鼠头骨的缺损是有益的。因此,在乙醇浴中通过戊二醛进行一步法交联是制备三维大孔纳米纤维支架最经济有效且简单的方法。该技术保留了明胶纤维的形态和结构,并增强了支架在体外和体内的生物学性能。KIM等[33]采用适当的加工条件交联以产生薄膜或大孔支架形式的生物玻璃-胶原纳米纤维复合材料,复合材料的平均纤维直径为320 nm,孔径在十到百微米之间。当浸泡在模拟体液中时,观察到生物玻璃纳米纤维均匀分布在基质中,从而在其表面上迅速形成骨状磷灰石矿物质,在体外表现出出色的生物活性。成骨细胞在生物玻璃-胶原纳米纤维复合材料上显示出良好的生长。特别地,纳米复合材料上细胞的碱性磷酸酶活性显著高于胶原蛋白上的碱性磷酸酶活性。所以这种新颖的生物玻璃-胶原蛋白纳米纤维复合材料在骨再生和组织工程应用中具有巨大潜力。这种方法只能加强支架的稳定性和力学性能,但是支架的孔隙还是很小,力学性能偏低且不能形成三维结构。 电纺纳米纤维支架的主要关注之一是二维(2D)阵列中密集堆积的纤维,这阻碍了它们在组织再生中的适用性。为了克服这个问题,JOSHI等[34]首次提出了静电纺丝结合原位气体发泡法来制备三维纳米纤维大孔聚己内酯/醋酸纤维素支架。将电纺纳米纤维毡浸入硼氢化钠溶液中,在相互连通的孔隙中充满硼氢化钠溶液,硼氢化钠溶液在毛细管力的驱动下水解生成氢气。原位生成的气体分子形成团簇,将导致孔隙成核的自由能降至最低,使纳米纤维重新组织成低密度、大孔、海绵状、多层的三维支架。使用此方法可以将密集堆积的二维电纺膜改性为低密度三维支架,但是导致支架的力学性能较差、孔径很小,三维结构也不可控制。 "
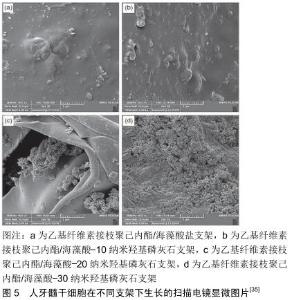
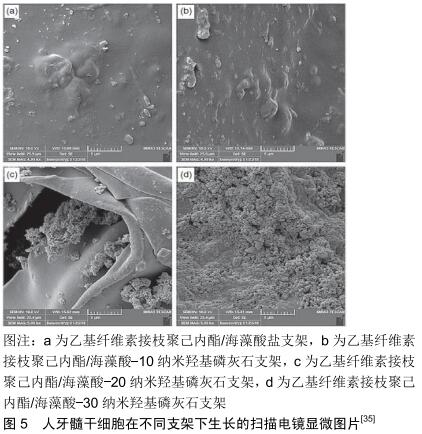
冷冻干燥也是可以用来制备孔结构的一种方法。HOKMABAD等[35]使用静电纺丝结合冷冻干燥法制备了掺杂纳米羟基磷灰石的乙基纤维素接枝聚己内酯/海藻酸盐纳米纤维大孔支架。乙基纤维素接枝聚己内酯/海藻酸盐、乙基纤维素接枝聚己内酯/海藻酸-10纳米羟基磷灰石、乙基纤维素接枝聚己内酯/海藻酸-20纳米羟基磷灰石和乙基纤维素接枝聚己内酯/海藻酸-30纳米羟基磷灰石的平均孔大小分别为(207.05±83.26),(183.19± 68.81),(175.29±68.04)和(160.4±57.14) μm。随着纳米羟基磷灰石含量的增加,导致乙基纤维素接枝聚己内酯/海藻酸盐支架膨胀,孔隙率和降解性能降低,但是观察到体外生物矿化和力学性能的增加。与原始乙基纤维素接枝聚己内酯/海藻酸盐支架相比,人牙髓干细胞在掺入纳米羟基磷灰石的支架上显示出更高的黏附、增殖和分化(图5)。但是此支架的力学性能还是偏低,孔径大小有待提升,无法形成三维结构。 "

静电纺丝支架被广泛用于骨组织工程,然而一个常见的问题是这些纤维支架的小孔径和紧密堆积的结构,导致细胞浸润性差,所以需要使用一种新技术来解决。KIM等[36-37]使用静电纺丝和10-200 μm的氯化钠作为致孔剂来制备大孔/纳米纤维玻璃酸/胶原复合支架(图6a),同时利用100-200 μm的氯化钠作为致孔剂来制备具有管状、大孔的纤维支架。软骨细胞黏附和增殖实验显示,随着胶原含量增加,细胞黏附和增殖能力增强,播种的软骨细胞保持成软骨形态的圆形特征。WANG等[38]同时使用静电纺丝和电雾化的聚氧化乙烯微粒作为致孔剂来制备纳米纤维大孔丝素蛋白支架(图6b)。小鼠3T3成纤维细胞体外培养实验显示,两种支架均可支持细胞附着、扩散和增殖。然而,静态培养条件下的体外细胞浸润只发生在大孔的静电纺丝支架上。HSU等[39]先使用静电纺丝制备介孔生物活性玻璃纳米纤维,再使用聚甲基丙烯酸甲酯微球作为大孔模板并利用静电液滴发生器来制备含有介孔生物活性玻璃纳米纤维的大孔微珠。含有介孔生物活性玻璃纳米纤维的大孔微珠平均孔大小为(125.91±27.42) μm,大于聚甲基丙烯酸甲酯微球的直径(80 μm)。GHOSH等[40]使用静电纺丝和250-425 μm氯化钠或100-250 μm的碳酸氢铵晶体作为致孔剂来制备不同孔大小的纳米纤维大孔聚己内酯支架。但是致孔剂很难除干净,即使除去了致孔剂,支架的力学性能也是很差,孔的连通性也不好,而且无法形成三维结构支架。 "


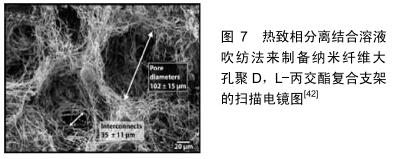
LEE等[41]使用静电纺丝结合超声法来制备纳米纤维结构的大孔左旋聚乳酸/明胶支架。他们发现超声处理后可以扩大支架的孔径、孔隙率、厚度。然而,支架的孔径、孔隙率、厚度的大小取决于超声的频率和处理时间。细胞实验显示在含或不含培养基的血清中,NIH3T3细胞黏附纳米纤维支架的数量明显高于非纤维支架。经过4 d的细胞培养发现,纳米纤维支架为细胞增殖提供了良好的环境。细胞与支架之间的相互作用发生在支架的孔壁上。纳米纤维与光滑结构的结合可用于控制表面积、蛋白质吸附和细胞相互作用。这种新型纳米纤维和大孔支架可为细胞黏附、增殖和分化提供更好的环境,以及用于研究细胞-支架的相互作用。与单一的静电纺丝相比,超声处理后可以得到更大孔径的支架,但是在使用超声的同时支架的力学性能将会变差且无法形成三维结构。 2.3 新型制备方法 溶液吹纺与静电纺丝最大的区别在于:溶液吹纺使用的是气体挤压式,而静电纺丝使用电场来挤出纳米纤维。MEDEIROS等[42]使用溶液吹纺和热致相分离方法来制备掺杂纳米生物活性玻璃的纳米纤维大孔聚D,L-丙交酯复合支架(图7)。通过在形成纤维的同时喷洒水滴,产生纤维和冰微球网络来生成大孔支架。由于存在冰球而具有相互连接的大孔,随后冷冻干燥形成多孔纳米纤维的三维支架。多喷嘴的使用使得冰微球能够原位喷射到纤维中,起到原位多孔化剂的作用,并且冷冻干燥可获得受控孔隙率的三维互连纳米纤维大孔支架。进一步的体内和体外实验显示,与暴露于模拟体液最初24 h内的光滑纤维相比,多孔纤维的可溶性二氧化硅离子释放更快,并且在84 h内羟基磷灰石的沉积是明显的。与单一的静电纺丝相比,此方法可以得到具有更大表面积(更多的开孔结构)和相互连通大孔结构的纳米纤维支架。 "
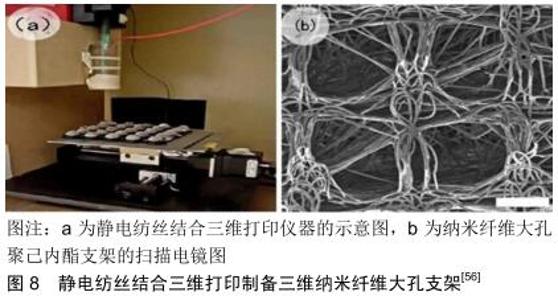
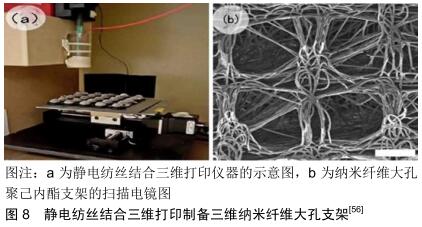
快速成型技术已被用于制作组织工程支架。该技术是一种相对先进的技术,用于制造具有完全互连多孔网络的支架[43]。快速成型技术通常以横截面的周期表示,然后将数据应用于实体自由形式制造机器以生成物理模型,通过从下到上构建各个层,每个新制造的层都堆积在前一层[44-45]。在制造过程中也可以使用生物制剂,例如生长因子和活细胞[46-48]。这些技术的优点是它们能够控制机械性能、基质结构(尺寸,形状,互连性,分支,几何和取向)、生物效应和支架的降解动力学[49-51]。快速成型技术包括不同类型的技术,例如三维印刷[52]、选择性激光烧结[53]、立体光刻[54]、熔融沉积建模等多种技 术[55]。由于三维打印技术具备速度快、精度高和可定制等优点,已逐渐被应用到骨组织工程领域。BROWN 等[56]使用静电纺丝结合三维打印技术制备了纳米纤维大孔聚己内酯支架(图8)。尽管允许聚合物按原样加工而不需要有毒溶剂,但基于熔融挤出直接写入骨组织工程的三维打印面临着静电纺丝的不同挑战:目前只能达到比静电纺丝大几个数量级的长丝直径;另一方面,直接熔融写入技术提供了对细丝放置的精确控制,以创建具有高度有序孔的三维结构,足够大的孔径(超过100 μm)以促进细胞浸润和血管形成。这种方法与单一的静电纺丝法相比可以得到结构孔隙可控的、具有良好孔道连通性、真正意义上的三维纳米纤维大孔支架。 除了以上方法之外,还有些研究者运用一些独特的方法来制备纳米纤维大孔支架,如激光切割、水凝胶涂层等。FULLER等[57]利用了静电纺丝和激光切割压缩技术来制备多层的纳米纤维大孔支架,其中通过激光切割引入30%圆形孔隙率,从而进一步降低了机械完整性。NIVEDHITHA等[58]将有大孔结构的凝胶涂在纤维支架上来制备微米纤维大孔支架,具有外覆盖物构建体上的蛋白质吸附显著降低,这可以延迟细胞与外层的黏附,并且构建体显示出良好的细胞附着和肌腱细胞的增殖。 "
|
[1] 曹谊林,刘伟,张文杰,等.组织工程研究进展[J].上海交通大学学报(医学版), 2012,32(9):1241-1250.
[2] CHUENJITKUNTAWORN B, OSATHANON T, NOWWAROTE N, et al. The efficacy of polycaprolactone/hydroxyapatite scaffold in combination with mesenchymal stem cells for bone tissue engineering. J Biomed Mater Res A.2016;104(1):264-271.
[3] 贺钧,李自良,谢志刚.骨替代材料的骨诱导性能研究进展[J].口腔疾病防治,2018,26(2):124-127.
[4] LANGER R, VACANTI JP. Tissue engineering. Sci. 1993;260 (5110):920-926.
[5] 曹谊林.组织工程学的研究进展[J].中国美容医学, 2005,14(2): 134-135.
[6] 张淦,程迅生.骨组织工程的研究进展[J].安徽医学, 2016,37(8): 1057-1061.
[7] DIECKMANN C, RENNER R, MILKOVA L, et al. Regenerative medicine in dermatology: biomaterials, tissue engineering, stem cells, gene transfer and beyond.Exp Dermatol.2010;19(8):697-706.
[8] MA PX. Scaffolds for tissue fabrication.Mater Today.2004;7(5): 30-40.
[9] MITCHELL GR, TOJEIRA A. Role of anisotropy in tissue engineering. Procedia Eng.2013;59(2):117-125.
[10] 黄霞,陈家昌,申长雨.骨组织工程支架材料研究进展[J].化工新型材料, 2010,38(9):65-68.
[11] KWON IK, MATSUDA T. Co-electrospun nanofiber fabrics of poly(L-lactide-co-epsilon-caprolactone) with type I collagen or heparin.Biomacromolecules.2005;6(4):2096-2105.
[12] BENIASH E, HARTGERINK JD, STORRIE H, et al. Self-assembling peptide amphiphile nanofiber matrices for cell entrapment.Acta Biomater.2005;1(4):387-397.
[13] HSU FY, WENG RC, LIN HM, et al. A biomimetic extracellular matrix composed of mesoporous bioactive glass as a bone graft material. Microporous Mesoporous Mater.2015;212:56-65.
[14] LIU Y, SUN Q, WANG S, et al. Preparation of SF/PLGA scaffold with microporous-nano/microfibrous architecture by thermal induced phase separation.Sci Adv Mater.2015;7(11):2380-2387.
[15] OH SH, PARK IK, KIM JM, et al. In vitro and in vivo characteristics of PCL scaffolds with pore size gradient fabricated by a centrifugation method.Biomaterials.2007;28(9):1664-1671.
[16] 吴林波,丁建东.组织工程三维多孔支架的制备方法和技术进展[J].功能高分子学报,2003,16(1):91-96.
[17] 郑雄飞,翟文杰.单一热致相分离法制备聚乳酸纳米纤维大孔支架[J].功能材料,2010,41 (S1):184-188.
[18] CHEN S, ZHAO X, DU C. Macroporous poly (L-lactic acid)/chitosan nanofibrous scaffolds through cloud point thermally induced phase separation for enhanced bone regeneration.Eur Polym J. 2018;109: 303-316.
[19] WANG W, MIAO Y, ZHOU X, et al. Local delivery of BMP-2 from poly(lactic-co-glycolic acid) microspheres incorporated into porous nanofibrous scaffold for bone tissue regeneration.J Biomed Nanotechno. 2017;13(11):1446-1456.
[20] HAN W, ZHAO J, TU M, et al. Preparation and characterization of nanohydroxyapatite strengthening nanofibrous poly(L-lactide) scaffold for bone tissue engineering.J Appl Polym Sci.2013;128(3): 1332-1338.
[21] LIU X, MA PX. Phase separation,pore structure, and properties of nanofibrous gelatin scaffolds.Biomaterials.2009;30(25): 4094-4103.
[22] LIU S, HE Z, XU G, et al. Fabrication of polycaprolactone nanofibrous scaffolds by facile phase separation approach.Mater Sci Eng C Mater Biol Appl.2014;44:201-208.
[23] SALERNO A, MAR FERNÁNDEZ-GUTIÉRREZ, BARRIO JSRD, et al. Macroporous and nanometre scale fibrous PLA and PLA-HA composite scaffolds fabricated by a bio safe strategy.RSC Adv. 2014; 4(106):61491-61502.
[24] WEI G, JIN Q, GIANNOBILE WV, et al. The enhancement of osteogenesis by nano-fibrous scaffolds incorporating rhBMP-7 nanospheres.Biomaterials.2007;28(12):2087-2096.
[25] WEI G, JIN Q, GIANNOBILE WV, et al. Nano-fibrous scaffold for controlled delivery of recombinant hµman PDGF-BB.J Control Release. 2006;112(1):103-110.
[26] GUPTE MJ, SWANSON WB, HU J, et al. Pore size directs bone marrow stromal cell fate and tissue regeneration in nanofibrous macroporous scaffolds by mediating vascularization.Acta Biomater.2018;82:1-11.
[27] CHEN VJ, MA PX. Nano-fibrous poly(L-lactic acid) scaffolds with interconnected spherical macropores.Biomaterials. 2004;25(11): 2065-2073.
[28] ZHANG R, MA PX. Synthetic nano-fibrillar extracellular matrices with predesigned macroporous architectures.J Biomed Mater Res. 2000; 52(2):430-438.
[29] SAKINA R, ALI M. An appraisal of the efficacy and effectiveness of nanoscaffolds developed by different techniques for bone tissue engineering applications: electrospinning a paradigm shift.Adv Polym Technol.2014;33(4).DOI:10.1002/adv.21429
[30] CAI YZ, ZHANG GR, WANG LL, et al. Novel biodegradable three-dimensional macroporous scaffold using aligned electrospun nanofibrous yarns for bone tissue engineering.J Biomed Mater Res A. 2012;100(5):1187-1194.
[31] MAJIDI SS, SLEMMING-ADAMSEN P, HANIF M, et al. Wet electrospun alginate/gelatin hydrogel nanofibers for 3D cell culture.Int J Biol Macromol.2018;118(PtB):1648-1654.
[32] TENG F, DING H, HUANG Y, et al. Fabrication of three- dimensional nanofibrous gelatin scaffolds using one-step crosslink technique.J Biomater Sci Polym Ed.2018;29(15):1859-1875.
[33] KIM HW, SONG JH, KIM HE. Bioactive glass nanofiber-collagen nanocomposite as a novel bone regeneration matrix.J Biomed Mater Res A.2010;79(3):698-705.
[34] JOSHI MK, PANT HR, TIWARI AP, et al. Multi-layered macroporous three-dimensional nanofibrous scaffold via a novel gas foaming technique. Chem Eng J.2015;275:79-88.
[35] HOKMABAD VR, DAVARAN S, AGHAZADEH M, et al. Fabrication and characterization of novel ethyl cellulose-grafted- poly(ɛ-caprolactone)/ alginate nanofibrous/ macroporous scaffolds incorporated with nano-hydroxyapatite for bone tissue engineering.J Biomater Appl. 2019;33(8):1128-1144.
[36] KIM TG, CHUNG HJ, PARK TG. Macroporous and nanofibrous hyaluronic acid/collagen hybrid scaffold fabricated by concurrent electrospinning and deposition/leaching of salt particles.Acta Biomater. 2008;4(6):1611-1619.
[37] KIM SH, KWON JH, CHUNG MS, et al. Fabrication of a new tubular fibrous PLCL scaffold for vascular tissue engineering.J Biomater Sci Polym Ed.2006;17(12):1359-1374.
[38] WANG K, XU M, ZHU M, et al. Creation of macropores in electrospun silk fibroin scaffolds using sacrificial PEO-microparticles to enhance cellular infiltration.J Biomed Mater Res A.2013;101(12):3474-3481.
[39] HSU FY, HSU HW, CHANG YH, et al. Macroporous microbeads containing apatite-modified mesoporous bioactive glass nanofibres for bone tissue engineering applications.Mater Sci Eng C Mater Biol Appl. 2018;89:346-354.
[40] GHOSH LD, JAIN A, SUNDERASAN NR, et al. Elucidating molecular events underlying topography mediated cardiomyogenesis of stem cells on 3D nanofibrous scaffolds. Mater Sci Eng C Mater Biol Appl. 2018;88:104-114.
[41] LEE JB, JEONG SI, BAE MS, et al. Poly(L-lactic acid) nanocylinders as nanofibrous structures for macroporous gelatin scaffolds.J Nanosci Nanotechnol.2011;11(7):6371-6376.
[42] MEDEIROS ELG, BRAZ AL, PORTO IJ, et al. Porous bioactive nanofibers via cryogenic solution blow spinning and their formation into 3D macroporous scaffolds.ACS Biomater Sci Eng. 2016;2(9): 1442-1449.
[43] WINDER J, BIBB R. Medical rapid prototyping technologies: state of the art and current Limitations for application in oral and maxillofacial surgery.J Oral Maxillofac Surg. 2005;63(7):1006-1015.
[44] LIN L, JU S, CEN L, et al. Fabrication of porous β-TCP scaffolds by combination of rapid prototyping and freeze drying technology. In 7th Asian-Pacific Conference on Medical and Biological Engineering. Springer, Berlin, Heidelberg.2008:88-91.
[45] WOODFIELD TB, GUGGENHEIM M, VON RECHENBERG B, et al. Rapid prototyping of anatomically shaped, tissue-engineered implants for restoring congruent articulating surfaces in small joints. Cell Prolif. 2010;42(4):485-497.
[46] YU D, LI Q, MU X, et al. Bone regeneration of critical calvarial defect in goat model by PLGA/TCP/rhBMP-2 scaffolds prepared by low-temperature rapid-prototyping technology.Int J Oral Maxillofac Surg. 2008;37(10):929-934.
[47] KHALIL S, SUN W. Bioprinting endothelial cells with alginate for 3D tissue constructs.J Biomech Eng.2009;131(11):111002.
[48] MIRONOV V, KASYANOV V, DRAKE C, et al. Organ printing: promises and challenges.Regen Med.2008;3(1):93-103.
[49] KAI H, WANG X, MADHUKAR KS, et al. Fabrication of a two-level tµmor bone repair biomaterial based on a rapid prototyping technique. Biofabrication.2009;1(2):025003.
[50] YANG S, LEONG KF, DU Z, et al. The design of scaffolds for use in tissue engineering. Part II.Rapid prototyping techniques.Tissue Eng. 2002;8(1):1-11.
[51] BAKER CT, BOCHAROV GA, PAUL CA, et al. Modelling and analysis of time-lags in some basic patterns of cell proliferation.J math biol. 1998;37(4):341-371.
[52] LAM CXF, MO XM, TEOH SH, et al. Scaffold development using 3D printing with a starch-based polymer.Mater Sci Eng C. 2002; 20(1-2): 49-56.
[53] PATTANAYAK DK,FUKUDA A,MATSUSHITA T,et al.Bioactive Ti metal analogous to hµman cancellous bone:fabrication by selective laser melting and chemical treatments.Acta Biomater. 2011;7(3):1398-1406.
[54] SUGAVANESWARAN M, ARµmAIKKANNU G. Analytical and experimental investigation on elastic modulus of reinforced additive manufactured structure.Mater Des(1980-2015).2015;66: 29-36.
[55] KALITA SJ, BOSE S, HOSICK HL, et al. Development of controlled porosity polymer-ceramic composite scaffolds via fused deposition modeling.Mater Sci Eng C. 2003;23(5):611-620.
[56] BROWN TD, EDIN F, DETTA N, et al. Melt electrospinning of poly(ε-caprolactone) scaffolds:phenomenological observations associated with collection and direct writing.Mater Sci Eng C. 2014; 45:698-708.
[57] FULLER KP, GASPAR D, DELGADO LM, et al. Development macro-porous electro-spun meshes with clinically relevant mechanical properties-A technical note.Biomed Mater.2019; 14(2):024103.
[58] NIVEDHITHA SUNDARAM M, DEEPTHI S, MONY U, et al. Chitosan hydrogel scaffold reinforced with twisted poly (l lactic acid) aligned microfibrous bundle to mimic tendon extracellular matrix.Int J Biol Macro.2019;122:37-44. |
| [1] | Li Wenjing, Li Haobo, Liu Congna, Cheng Dongmei, Chen Huizhen, Zhang Zhiyong. Comparison of different bioactive scaffolds in the treatment of regenerative pulp of young permanent teeth [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 499-503. |
| [2] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [3] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [4] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [5] | Liu Keke, Duan Xin, Ma Xiangrui, Zhang Yuntao. Effect of cinnamaldehyde on osteoblasts in high glucose environment with the electrospinning membrane as a carrier [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3500-3504. |
| [6] | Chen Song, He Yuanli, Xie Wenjia, Zhong Linna, Wang Jian. Advantages of calcium phosphate nanoparticles for drug delivery in bone tissue engineering research and application [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3565-3570. |
| [7] | Zhang Zhenhua, Liu Zichen, Yu Baoqing. Status and problems of polycaprolactone and its composite materials in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3571-3577. |
| [8] | Chen Jie, Liao Chengcheng, Zhao Hongbo, Zhao Wei, Chen Zhiwei, Wang Yan. Application of tissue engineering urethral stent and its preparation technology in urethral reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3591-3596. |
| [9] | Xu Hui, Kang Bingxin, Zhong Sheng, Gao Chenxin, Zhao Chi, Qiu Guowei, Sun Songtao, Xie Jun, Xiao Lianbo, Shi Qi. Pressing local acupoints plus adjustion of the knee joint in a sitting position for treating knee osteoarthritis: a randomized controlled trial [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 216-221. |
| [10] | Wang Liu, Song Dongzhe, Huang Dingming. Bone morphogenetic protein 9 regulates stem cell differentiation and bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3064-3070. |
| [11] | Xie Jian, Su Jiansheng. Advantages and characteristics of electrospun aligned nanofibers as scaffolds for tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2575-2581. |
| [12] | Li Yanle, Yue Xiaohua, Nie Zhen, Zhang Junwei, Li Zhaohui, Nie Weizhi, Jiang Hongjiang. Characteristics and application of bioabsorbable materials in orthopedics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2612-2617. |
| [13] | Han Ningning, Zuo Jinfu, Sun Miao, Tang Shengjian, Liu Fangjun. Application and progress of umbilical cord mesenchymal stem cells in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2079-2086. |
| [14] | Meng Maohua, Li Ying, Chen Xin, Cheng Lu, Dong Qiang. Effects and mechanisms of enamel matrix derivatives on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2108-2113. |
| [15] | Liu Zhigang, Guo Qinggong, Chen Jingtao. Effect of Capparis spinosa total alkaloid on proliferation and apoptosis of nucleus pulposus cells in an intervertebral disc degeneration rat model [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1699-1704. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||