Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (19): 3064-3070.doi: 10.3969/j.issn.2095-4344.3515
Previous Articles Next Articles
Bone morphogenetic protein 9 regulates stem cell differentiation and bone regeneration
Wang Liu, Song Dongzhe, Huang Dingming
- State Key Laboratory of Oral Diseases & National Clinical Research Center of Oral Disease & Department of Operative Dentistry and Endodontics, West China Hospital of Stomatology, Sichuan University, Chengdu 610000, Sichuan Province, China
-
Received:2020-06-17Revised:2020-06-20Accepted:2020-07-29Online:2021-07-09Published:2021-01-13 -
Contact:Huang Dingming, MD, Chief physician, Professor, State Key Laboratory of Oral Diseases & National Clinical Research Center of Oral Disease & Department of Operative Dentistry and Endodontics, West China Hospital of Stomatology, Sichuan University, Chengdu 610000, Sichuan Province, China -
About author:Wang Liu, Master, Physician, State Key Laboratory of Oral Diseases & National Clinical Research Center of Oral Disease & Department of Operative Dentistry and Endodontics, West China Hospital of Stomatology, Sichuan University, Chengdu 610000, Sichuan Province, China -
Supported by:the National Natural Science Foundation of China, No. 81970936 (to HDM); the National Natural Science Foundation of China, No. 81900996 (to SDZ)
CLC Number:
Cite this article
Wang Liu, Song Dongzhe, Huang Dingming. Bone morphogenetic protein 9 regulates stem cell differentiation and bone regeneration[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3064-3070.
share this article
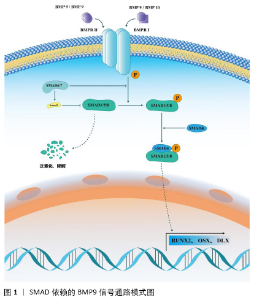
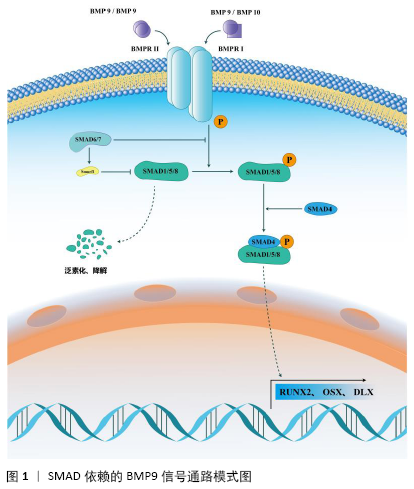
2.1 BMP9的结构及受体 BMP9又名转化生长因子2,最早在发育过程中的小鼠肝脏的基因组中发现,它在肝脏组织中的表达显著高于在其他组织中的表达[13]。在人类染色体中,BMP9定位于染色体10q11.22[13-14],由含有22个氨基酸的信号肽、33 kD的前结构域和12.5 kD的成熟蛋白质组成BMP9的前体形式。在丝氨酸蛋白内切酶的作用下,前体蛋白经过剪切、重构之后可以形成2种形式的成熟BMP9:①25 kD的短肽二聚体形式;②前结构域与成熟蛋白区域以非共价键的 方式结合在一起,形成大约100 kD的复杂成熟BMP9形式[15]。 BMP9在调控干细胞分化的过程中,主要是通过与两大类受体结合,激活下游的信号通路和级联反应,包括BMPⅠ型受体(type Ⅰ bone morphogenetic protein receptor,BMPRⅠ)和Ⅱ型受体(type Ⅱbone morphogenetic protein receptor,BMPRⅡ)。BMP9可以与BMPRⅠ中的受体间变性淋巴瘤激酶1(ALK1)和受体间变性淋巴瘤激酶2(ALK2)相结合,其中BMP9与间变性淋巴瘤激酶1有着较高的亲和性,与间变性淋巴瘤激酶2的亲和性则相对较弱[16-17]。对于BMPRⅡ型受体,BMP9则是可以它的多个亚型相结合,如ActRⅡ、ActRⅡB和BMPR2等[14,17-19]。这些受体大多都是跨膜蛋白受体,包含细胞膜外面的富含半胱氨酸的胞外结合区域、跨膜区域蛋白和胞浆内富含丝氨酸与苏氨酸的胞内区域。 2.2 BMP9调控干细胞成骨向分化的信号通路 BMP9的信号转导过程通常是由BMP9形成的自身同源二聚体聚合物特异性地结合到BMPRⅠ、BMPRⅡ异源二聚体复合物上,形成异源四聚体复合物,这个相互作用的过程促进了富含甘氨酸-丝氨酸基序的BMPRⅡ胞内区域的磷酸化,进而激活BMPRⅠ,从而激活下游的信号通路。既往研究报道,BMP9可以通过影响多条信号通路来实现对多种间充质干细胞成骨分化过程的调控作用[20-25],主要包括SMAD依赖的信号通路和非SMAD依赖的信号通路[6]。 2.2.1 SMAD依赖的BMP9信号通路 SMAD依赖的信号通路是BMP9调控干细胞成骨分化的最主要信号通路之一,见图1,其主要信号转导机制为:BMP9同源二聚体或BMP9/BMP10异源二聚体首先与BMPRⅡ相互作用,随后募集BMPRⅠ形成异源四聚体复合物,随后BMPRⅠ的胞内结构域启动SMAD1/5/8磷酸化。磷酸化的SMAD1/5/8和协同SMAD(co-SMAD,SMAD4)相结合成为SMAD1/5/8-SMAD4复合物,最终该复合物转运进入细胞核内,启动下游的成骨相关基因如RUNX2、DLX、OSX的转录[26]。 除了参与信号转导的SMAD1/5/8-SMAD4转录复合物,SMAD6和SMAD7也参与了SMAD依赖信号通路的信号转导。SMAD6/7又称抑制性SMAD(inhibitory SMAD,I-SMAD),在SMAD依赖信号通路中,过表达SMAD6/7可以抑制BMP9对干细胞成骨分化的调控作用,其主要分子机制有:①SMAD6/7复合物可以竞争性结合BMPR细胞内结构域的磷酸化位点,抑制SMAD1/5/8的磷酸化;②SMAD6/7复合物可以通过调控泛素连接酶Smurf1的活性,进而促进SMAD1/5/8的泛素化,随后加速SMAD1/5/8的降解,进而抑制SMAD依赖信号通路的转导过程。 "
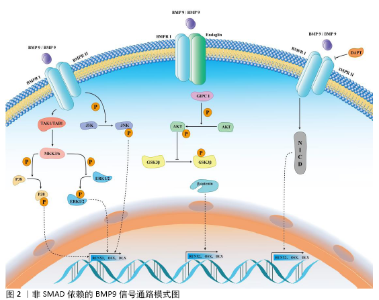
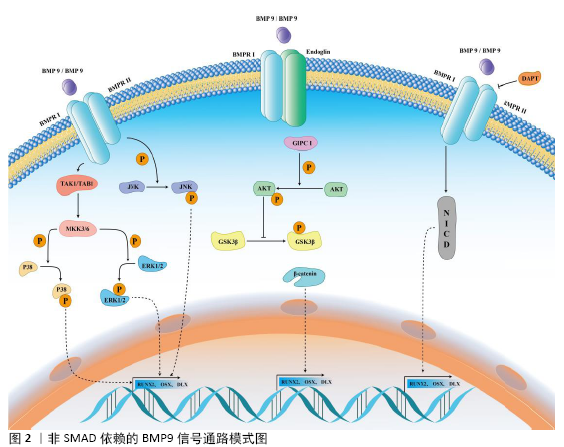
与BMP家族其他分子不同的是,BMP9不能被传统的BMP受体拮抗剂所抑制。例如:经典的BMP受体拮抗剂Noggin或BMP3可以显著抑制BMP2、BMP4和BMP7等BMP家族分子所介导的干细胞成骨分化过程,但他们对BMP9介导的干细胞成骨分化作用没有明显的影响,这提示BMP9可能不仅通过SMAD依赖信号通路来实现对干细胞成骨分化的调控作用,还可能通过其他信号通路来实现对干细胞的调控。 2.2.2 非SMAD依赖的BMP9信号通路 干细胞分化的调控网络错综复杂,多条信号通路可以参与调控BMP9介导的信号转导过程,这些信号通路统称非SMAD依赖的BMP9信号通路。它们与BMP9对干细胞成骨分化的调控作用相互影响、相互作用,主要包括MAPK、TGFβ、Wnt/β-catenin和Notch信号通路等,见图2。 (1)BMP9与MAPK信号通路:BMP9-MAPKs是一个重要的信号转导通路,其主要包含有细胞外信号相关激酶1/2 (extracellular signal-related kinases 1/2,ERK1/2)、ERK5、p38 MAPKs和Jun氨基末端激酶(Jun amino-terminal kinases,JNKs)。MAPK家族成员属于丝氨酸/苏氨酸蛋白激酶,在不同的物种间具有高度的保守性,在许多重要的生物学过程中发挥着重要的调控作用,包括成骨分化、基因表达、有丝分裂、代谢以及许多其他功能。当BMP9同源或异源二聚体作用于BMPR后,BMPRⅡ受体激活TAK1/TAB1复合物,进而激活MKK3/6,最终激活下游的p38或ERK1/2并使其磷酸化,随后发挥下游的信号转导功能[27-29]。 在MAPK信号通路中,转化生长因子β激活激酶1(transforming growth factor-β-activated kinase 1,TAK1)是目前研究较多的上游关键分子[29]。TAK1在成骨分化过程中起到了关键的调控作用,它可以参与p38 MAPK和BMP-SMAD的信号转导过程[30-32]。 在条件性敲除TAK1的小鼠中出现了颅骨组织发育缺陷、颅缝关闭不全等骨发育异常的表现[30-31]。p38选择性抑制剂或p38 siRNA均可以显著阻断BMP9介导的C3H10T1/2细胞成骨分化过程[33]。在条件性敲除BMPR的转基因小鼠模型中可以发现显著的骨发育障碍和磷酸化SMAD表达下调,磷酸化p38表达也显著下调[34]。BMP9介导的小鼠牙囊干细胞成骨向分化过程中磷酸化p38表达上调,而磷酸化ERK1/2表达下调。使用SB203580(p38抑制剂)和PD98059(ERK1/2抑制剂)后,小鼠牙囊干细胞的成骨分化能力分别下调和上调,提示p38、ERK1/2在BMP9介导的成骨分化过程中可能发挥不同的作用[35]。JNK MAPK信号通路在骨代谢与骨调控中的研究相对较少,研究发现,MKK4/7复合物被激活后,使JNK1/2/3激活,激活的JNK1/2/3促进JunB和ATF2基因的表达,进而促进骨组织的发育与形成[29]。在BMP9-MAPK信号通路中,BMP9可以上调JNK1/2/3复合体的磷酸化,选择性抑制JNK1/2/3的表达可以逆转BMP9介导的成骨分化,提示JNK MAPK信号轴也参与了这一调控过程[22]。p38、ERK1/2和JNK MAPK信号通路在骨组织发育与再生过程中发挥了重要的作用,它们在BMP9介导的成骨分化过程中也发挥了关键的调控作用。目前的研究主要集中于BMP9可以调控p38、ERK1/2和JNK等MAPKs的磷酸化,应用MAPKs抑制剂可以一定程度抑制BMP9介导的成骨分化过程,但是BMP9直接调控MAPKs信号通路关键蛋白激活的途径及其具体机制仍有待进一步的探究。 (2)BMP9与TGFβ信号通路:TGFβ信号通路是调控骨发育与骨代谢的经典信号通路。BMP9是TGFβ超家族的成员之一,介导二者的细胞表面受体、下游级联反应的关键分子均有所不同[26]。TGFβ主要包含TGFβ1、TGFβ2和TGFβ3三个成员,它们通过与细胞膜表面的TβRⅠ和TβRⅡ结合,激活并磷酸化SMAD2/3,然后募集SMAD4形成SMAD2/3-SMAD4复合物,转运进入到细胞核内对靶基因起到调控作用[36-37]。TGFβ也可以激活TAK1/TAB1复合物,进而活化MKK3/6复合物,随后激活p38和ERK1/2 MAPK信号通路,促进成骨分化关键分子RUNX2的磷酸化,激活成骨分化过程[26,36]。BMP9与TGFβ配体存在有相同的表面受体,如BMPRⅡ、ActRⅡ,提示BMP9与TGFβ之间可能存在相互作用或竞争性抑制[38-39]。 目前关于BMP9与TGFβ相互作用的研究相对较少,有待未来的进一步研究和阐明。 (3)BMP9与Wnt/β-catenin信号通路:Wnt/β-catenin信号通路是调节骨骼发生发育、骨组织代谢与维持骨组织平衡的关键信号通路[40]。Wnt配体与Frizzled和LRP5/6受体结合,磷酸化Dvl蛋白,进而募集GSK3β、Axin、APC、CK1形成GSK3β/Dvl/Axin/APC/CK1复合物,未被抑制的β-catenin进入细胞核内结合到TCF/LEF转录因子上,激活下游成骨分化相关基因的信号转导过程。在Wnt信号通路未被激活的情况下,游离的GSK3β/Axin/APC/CK1促进β-catenin磷酸化、泛素化和降解[40]。 研究发现与BMP2、BMP4相比,BMP9具有更强的促进成骨分化的潜能。BMP9可以促进Smad1/5、ERK和JNK的磷酸化,同时BMP9还可以通过调控Endoglin/GIPC1/Akt信号通路来促进GSK3β的磷酸化,稳定β-catenin的表达,提示BMP9也可以激活Wnt信号通路来调控干细胞的成骨分化过程[10]。BMP9可以通过增加β-catenin的表达与磷酸化,上调GSK3β的磷酸化来促进高磷酸盐状态下的血管平滑肌细胞的成骨分化过程[41]。近年来,BMP9与Wnt配体对干细胞成骨分化的共同调控作用也越来越受到关注。研究发现,BMP9可以促进C3H10T1/2细胞成骨分化,同时上调Wnt11的mRNA和蛋白表达,联合应用BMP9与Wnt11可以增强SMAD1/5/8与p38的磷酸化,进而促进成骨分化过程[42]。体内外研究发现,BMP9可以联合Wnt3a共同促进永生化小鼠根尖牙乳头干细胞的成骨向、成牙本质向分化[25]。BMP9信号通路与Wnt/β-catenin信号通路是调控干细胞成骨分化的信号通路,进一步阐明二者之间的相互作用关系及其具体机制,对于未来促进骨组织缺损的修复具有重要意义。 (4)BMP9与Notch信号通路:Notch信号通路分子是一个在发育上高度保守的蛋白家族,它们参与了胚胎发育、细胞命运决定、细胞增殖、细胞分化和细胞稳态等多个生物学过程。在人类基因组中,Notch信号通路包含5种Notch配体(JAG1、JAG2、DLL1、DLL3和DLL5)和4种受体(Notch1-4)[43]。在骨发育及骨代谢性疾病中,Notch受体或配体往往出现表达异常;既往研究也发现,Notch信号通路广泛参与到成骨、成软骨和破骨分化过程中[43]。Notch信号转导主要是通过细胞外受体配体的结合,随后Notch受体的细胞外结构域经过基质金属蛋白酶肿瘤坏死因子转换酶、γ-分泌酶复合物剪切后变成Notch细胞内结构域,进而介导细胞内的信号转导过程。 目前,关于BMP9与Notch信号通路的相互作用尚存争议,WANG等[44]研究发现沉默Notch1受体表达可以一定程度抑制BMP9介导的成骨分化作用。CAO等[45]研究发现过表达Notch信号通路中间关键分子Notch细胞内结构域对BMP9介导的成骨分化起到一定的抑制作用。WANG和CAO两个团队[44-45]均发现γ-分泌酶复合物抑制剂DAPT可以抑制BMP9介导的成骨分化作用。CAO等[45]研究发现,在小鼠胚胎成纤维细胞中,Notch1可以通过增加BMP9介导的SMAD1/5/8磷酸化来促进细胞的成骨分化,提示Notch信号通路与BMP9信号通路可能存在信号串化作用。BMP9与Notch信号通路之间的信号串化作用仍有许多尚未阐明的地方,未来的研究需要关注BMP9和Notch信号通路之间的相互影响作用及BMP9调控Notch信号通路关键分子表达的具体分子机制。 2.3 BMP9与骨组织工程技术 外伤、感染、炎症、肿瘤等多种疾病可以导致骨组织的破坏或缺损,治疗各种原因导致的骨相关疾病、修复骨缺损、恢复骨组织功能与美观是骨组织工程技术最需要解决的问题[46-49]。骨组织工程技术主要包括干细胞、细胞因子与支架材料3个组成部分。在过去20多年的研究中,有大量学者对BMP9复合多种生物支架材料或多种间充质干细胞对骨组织缺损、骨折不愈合、骨组织极限缺损及口腔颌面部骨组织重建与修复进行了初步的探究。 2.3.1 BMP9在口腔颌面部骨缺损修复中的应用 牙周病、根尖周病、外伤、口腔颌面部肿瘤等疾病导致的牙与骨组织缺损严重影响患者的生活质量。通过骨组织工程技术可以一定程度恢复患者的颌骨骨量与高度,为保存天然牙或为缺失牙区域的种植手术提供条件与保障。SAULACIC等[50]学者研究发现,应用重组BMP9蛋白(recombinant human BMP9 protein,rhBMP9)联合BioOss骨粉、BioGide骨膜对新西兰兔进行引导性骨再生术的效果要优于不使用rhBMP9,同时还发现用BioGide骨膜联合rhBMP9的修复效果要优于BioOss骨粉联合rhBMP9的修复效果。Nikola团队还发现联合脱蛋白牛骨粉进行颌骨位点保存术的骨修复潜能较强且优于 rhBMP2联合脱蛋白牛骨粉组,同时还发现高剂量和低剂量rhBMP9诱导骨修复的能力没有明显异[11]。DUMANIAN团队[51]使用PPCN-g材料搭载腺病毒BMP9感染后的小鼠牙囊细胞,可以在术后12周比较良好的修复颅骨极限缺损。TOSHIAKI 团队[52]对比使用搭载了相同剂量rhBMP9或rhBMP2 的可吸收胶原来修复新西兰兔颅骨极限缺损,术后2周时,rhBMP2组的骨缺损修复效果、形成新骨的体积与质量要优于rhBMP9组,在术后8周时,rhBMP9组则表现出更致密、更稳定的修复骨结构,可吸收胶原组则以结缔组织愈合为主。FUJIOKA-KOBAYASHI团队[12]研究发现,在新西兰兔颅骨缺损模型中,使用脱蛋白牛骨骨粉搭载不同质量(5,20 μg)的rhBMP2或rhBMP9,结果发现使用5 μg rhBMP9可以达到最好的骨缺损修复效果,而使用20 μg rhBMP2组与20 μg rhBMP9组骨缺损修复效果弱于5 μg rhBMP9组,5 μg rhBMP2组则不能完全修复缺损区域的骨组织。这一研究结果表明低剂量的rhBMP9可以用于修复极限骨缺损,但是其安全性仍有待进一步评估。 2.3.2 BMP9在骨关节疾病中的应用 外伤、感染、遗传性疾病可能导致截肢、肢端缺损等疾病,目前尚没有技术手段或治疗方式可以促进断端肢体的再生与修复。YU等[53]研究发现,在肢端缺损模型中,应用rhBMP2可以诱导小鼠肢体断端形成新的软骨骨化中心,但是却不能实现断端的修复与重建。随后该团队连续应用BMP2和BMP9处理小鼠肢体断端,结果发现包含有关节软骨与滑膜腔的滑膜关节样结构形成。该结构的形成依赖于蛋白多糖4基因的参与,若蛋白多糖4基因缺失,BMP9则不能实现对肢端缺损的修复作用[20]。尽管前述研究只是在实验室内完成,但其初步研究成果为未来实现肢端的自身修复提供了理论基础。骨关节炎是临床上常见的疾病,针对骨关节炎的治疗目的主要是消除病因与危险因素、减轻临床症状,而尚无有效办法可以逆转骨关节炎的进程。LIU等[54]研究发现,通过注射BMP9处理后的脂肪间充质干细胞可以促进膝骨关节炎软骨的修复,BMP9处理组Ⅱ型胶原与聚集蛋白聚糖的表达显著高于其他组,且该调控作用可能是通过Notch1/Jagged1信号通路来实现的,但该研究没有对炎症相关指标进行检测,也没有说明BMP9对骨关节炎的具体调控机制,仍有待进一步研究。WANG等[55]研究发现,在卵巢切除术后骨质疏松大鼠骨折模型中,应用腺病毒BMP9或绿色荧光蛋白腺病毒处理骨折区域,结果发现BMP9可以促进骨折区域的骨痂形成,提示BMP9有促进骨折区域修复的潜能。 目前,BMP9在骨组织工程中的应用目前仍在实验室研究阶段,重组蛋白和腺病毒是最常用的载体。小鼠、大鼠、新西兰兔和比格犬等颅骨、胫骨或牙槽骨缺损模型被用来评价BMP9在骨缺损修复中的作用,但是对于BMP9可能出现的毒副作用,目前鲜有团队进行相关的研究或者报道。因此,在未来的研究中,除了进一步确定BMP9促进骨缺损修复的最佳使用剂量或浓度外,对BMP9临床应用的安全性也需要进一步的评估。 "
| [1] LOPES D, MARTINS-CRUZ C, OLIVEIRA MB, et al. Bone physiology as inspiration for tissue regenerative therapies. Biomaterials. 2018;185: 240-275. [2] HO-SHUI-LING A, BOLANDER J, RUSTOM LE, et al. Bone regeneration strategies: Engineered scaffolds, bioactive molecules and stem cells current stage and future perspectives. Biomaterials. 2018;180:143-162. [3] BUNPETCH V, ZHANG ZY, ZHANG X, et al. Strategies for MSC expansion and MSC-based microtissue for bone regeneration. Biomaterials. 2019;196: 67-79. [4] HAUMER A, BOURGINE PE, OCCHETTA P, et al. Delivery of cellular factors to regulate bone healing. Adv Drug Deliv Rev. 2018;129:285-294. [5] URIST MR. Bone: formation by autoinduction. Science. 1965;150(3698): 893-899. [6] MOSTAFA S, PAKVASA M, COALSON E, et al. The wonders of BMP9: From mesenchymal stem cell differentiation, angiogenesis, neurogenesis, tumorigenesis, and metabolism to regenerative medicine. Genes Dis. 2019;6(3):201-223. [7] LI Y, LIU Z, TANG Y, et al. Schnurri-3 regulates BMP9-induced osteogenic differentiation and angiogenesis of human amniotic mesenchymal stem cells through Runx2 and VEGF. Cell Death Dis. 2020;11(1):72. [8] SONG D, ZHANG F, REID RR, et al. BMP9 induces osteogenesis and adipogenesis in the immortalized human cranial suture progenitors from the patent sutures of craniosynostosis patients. J Cell Mol Med. 2017;21(11): 2782-2795. [9] SONG D, HUANG S, ZHANG L, et al. Differential Responsiveness to BMP9 between Patent and Fused Suture Progenitor Cells from Craniosynostosis Patients. Plast Reconstr Surg. 2020;145(3):552e-562e. [10] EIRAKU N, CHIBA N, NAKAMURA T, et al. BMP9 directly induces rapid GSK3-β phosphorylation in a Wnt-independent manner through class I PI3K-Akt axis in osteoblasts. FASEB J. 2019;33(11):12124-12134. [11] SAULACIC N, SCHALLER B, MUÑOZ F, et al. Recombinant human BMP9 (RhBMP9) in comparison with rhBMP2 for ridge augmentation following tooth extraction: An experimental study in the Beagle dog. Clin Oral Implants Res. 2018;29(10):1050-1059. [12] FUJIOKA-KOBAYASHI M, ABD EL RAOUF M, SAULACIC N, et al. Superior bone-inducing potential of rhBMP9 compared to rhBMP2. J Biomed Mater Res A. 2018;106(6):1561-1574. [13] SONG JJ, CELESTE AJ, KONG FM, et al. Bone morphogenetic protein-9 binds to liver cells and stimulates proliferation. Endocrinology. 1995;136(10): 4293-4297. [14] BRAGDON B, MOSEYCHUK O, SALDANHA S, et al. Bone morphogenetic proteins: a critical review. Cell Signal. 2011;23(4):609-620. [15] MI LZ, BROWN CT, GAO Y, et al. Structure of bone morphogenetic protein 9 procomplex. Proc Natl Acad Sci U S A. 2015;112(12):3710-3715. [16] SCHARPFENECKER M, VAN DINTHER M, LIU Z, et al. BMP-9 signals via ALK1 and inhibits bFGF-induced endothelial cell proliferation and VEGF-stimulated angiogenesis. J Cell Sci. 2007;120(Pt 6):964-972. [17] SANCHEZ-DUFFHUES G, WILLIAMS E, GOUMANS MJ, et al. Bone morphogenetic protein receptors: Structure, function and targeting by selective small molecule kinase inhibitors. Bone. 2020;138:115472. [18] LUO J, TANG M, HUANG J, et al. TGFbeta/BMP type I receptors ALK1 and ALK2 are essential for BMP9-induced osteogenic signaling in mesenchymal stem cells. J Biol Chem. 2010;285(38):29588-29598. [19] KIM JH, PEACOCK MR, GEORGE SC, et al. BMP9 induces EphrinB2 expression in endothelial cells through an Alk1-BMPRII/ActRII-ID1/ID3-dependent pathway: implications for hereditary hemorrhagic telangiectasia type II. Angiogenesis. 2012;15(3):497-509. [20] YU L, DAWSON LA, YAN M, et al. BMP9 stimulates joint regeneration at digit amputation wounds in mice. Nat Commun. 2019;10(1):424. [21] ZHENG W, CHEN Q, ZHANG Y, et al. BMP9 promotes osteogenic differentiation of SMSCs by activating the JNK/Smad2/3 signaling pathway. J Cell Biochem. 2020;121(4):2851-2863. [22] WANG P, WANG Y, TANG W, et al. Bone Morphogenetic Protein-9 Enhances Osteogenic Differentiation of Human Periodontal Ligament Stem Cells via the JNK Pathway. PLoS One. 2017;12(1):e0169123. [23] TEVEN CM, ROSSI MT, SHENAQ DS, et al. Bone morphogenetic protein-9 effectively induces osteogenic differentiation of reversibly immortalized calvarial mesenchymal progenitor cells. Genes Dis. 2015;2(3):268-275. [24] WANG J, ZHANG H, ZHANG W, et al. Bone morphogenetic protein-9 effectively induces osteo/odontoblastic differentiation of the reversibly immortalized stem cells of dental apical papilla. Stem Cells Dev. 2014;23(12): 1405-1416. [25] ZHANG H, WANG J, DENG F, et al. Canonical Wnt signaling acts synergistically on BMP9-induced osteo/odontoblastic differentiation of stem cells of dental apical papilla (SCAPs). Biomaterials. 2015;39:145-154. [26] WU M, CHEN G, LI YP. TGF-β and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. Bone Res. 2016;4:16009. [27] RODRÍGUEZ-CARBALLO E, GÁMEZ B, VENTURA F. p38 MAPK Signaling in Osteoblast Differentiation. Front Cell Dev Biol. 2016;4:40. [28] THOUVEREY C, CAVERZASIO J. Focus on the p38 MAPK signaling pathway in bone development and maintenance. Bonekey Rep. 2015;4:711. [29] GREENBLATT MB, SHIM JH, GLIMCHER LH. Mitogen-activated protein kinase pathways in osteoblasts. Annu Rev Cell Dev Biol. 2013;29:63-79. [30] HOFFMANN A, PREOBRAZHENSKA O, WODARCZYK C, et al. Transforming growth factor-beta-activated kinase-1 (TAK1), a MAP3K, interacts with Smad proteins and interferes with osteogenesis in murine mesenchymal progenitors. J Biol Chem. 2005;280(29):27271-27283. [31] SHIM JH, GREENBLATT MB, XIE M, et al. TAK1 is an essential regulator of BMP signalling in cartilage. EMBO J. 2009;28(14):2028-2041. [32] GUNNELL LM, JONASON JH, LOISELLE AE, et al. TAK1 regulates cartilage and joint development via the MAPK and BMP signaling pathways. J Bone Miner Res. 2010;25(8):1784-1797. [33] XU DJ, ZHAO YZ, WANG J, et al. Smads, p38 and ERK1/2 are involved in BMP9-induced osteogenic differentiation of C3H10T1/2 mesenchymal stem cells. BMB Rep. 2012;45(4):247-252. [34] RIGUEUR D, BRUGGER S, ANBARCHIAN T, et al. The type I BMP receptor ACVR1/ALK2 is required for chondrogenesis during development. J Bone Miner Res. 2015;30(4):733-741. [35] LI C, YANG X, HE Y, et al. Bone morphogenetic protein-9 induces osteogenic differentiation of rat dental follicle stem cells in P38 and ERK1/2 MAPK dependent manner. Int J Med Sci. 2012;9(10):862-871. [36] DERYNCK R, BUDI EH. Specificity, versatility, and control of TGF-β family signaling. Sci Signal. 2019;12(570):eaav5183. [37] FENG XH, DERYNCK R. Specificity and versatility in tgf-beta signaling through Smads. Annu Rev Cell Dev Biol. 2005;21:659-693. [38] WU N, ZHAO Y, YIN Y, et al. Identification and analysis of type II TGF-β receptors in BMP-9-induced osteogenic differentiation of C3H10T1/2 mesenchymal stem cells. Acta Biochim Biophys Sin (Shanghai). 2010;42(10): 699-708. [39] LI RD, DENG ZL, HU N, et al. Biphasic effects of TGFβ1 on BMP9-induced osteogenic differentiation of mesenchymal stem cells. BMB Rep. 2012;45(9): 509-514. [40] OICHI T, OTSURU S, USAMI Y, et al. Wnt signaling in chondroprogenitors during long bone development and growth. Bone. 2020;137:115368. [41] HE F, WANG H, REN WY, et al. BMP9/COX-2 axial mediates high phosphate-induced calcification in vascular smooth muscle cells via Wnt/β-catenin pathway. J Cell Biochem. 2018;119(3):2851-2863. [42] ZHU JH, LIAO YP, LI FS, et al. Wnt11 promotes BMP9-induced osteogenic differentiation through BMPs/Smads and p38 MAPK in mesenchymal stem cells. J Cell Biochem. 2018;119(11):9462-9473. [43] ZIEBA JT, CHEN YT, LEE BH, et al. Notch Signaling in Skeletal Development, Homeostasis and Pathogenesis. Biomolecules. 2020;10(2):332. [44] WANG N, LIU W, TAN T, et al. Notch signaling negatively regulates BMP9-induced osteogenic differentiation of mesenchymal progenitor cells by inhibiting JunB expression. Oncotarget. 2017;8(65):109661-109674. [45] CAO J, WEI Y, LIAN J, et al. Notch signaling pathway promotes osteogenic differentiation of mesenchymal stem cells by enhancing BMP9/Smad signaling. Int J Mol Med. 2017;40(2):378-388. [46] CARREIRA AC, LOJUDICE FH, HALCSIK E, et al. Bone morphogenetic proteins: facts, challenges, and future perspectives. J Dent Res. 2014;93(4):335-345. [47] RAHMAN MS, AKHTAR N, JAMIL HM, et al. TGF-β/BMP signaling and other molecular events: regulation of osteoblastogenesis and bone formation. Bone Res. 2015;3:15005. [48] 汤显能,陈跃平,章晓云.骨与软骨组织工程中骨形态发生蛋白的特征与临床应用[J].中国组织工程研究,2019,23(4):591-596. [49] 朱正清,陈香润,贾方腾,等.骨形态发生蛋白9诱导成骨机制及其临床应用[J].中国组织工程研究,2019,23(33):5404-5412. [50] SAULACIC N, FUJIOKA-KOBAYASHI M, KOBAYASHI E, et al. Guided bone regeneration with recombinant human bone morphogenetic protein 9 loaded on either deproteinized bovine bone mineral or a collagen barrier membrane. Clin Implant Dent Relat Res. 2017;19(4):600-607. [51] DUMANIAN ZP, TOLLEMAR V, YE J, et al. Repair of critical sized cranial defects with BMP9-transduced calvarial cells delivered in a thermoresponsive scaffold. PLoS One. 2017;12(3):e0172327. [52] NAKAMURA T, SHIRAKATA Y, SHINOHARA Y, et al. Comparison of the effects of recombinant human bone morphogenetic protein-2 and -9 on bone formation in rat calvarial critical-size defects. Clin Oral Investig. 2017;21(9): 2671-2679. [53] YU L, HAN M, YAN M, et al. BMP2 induces segment-specific skeletal regeneration from digit and limb amputations by establishing a new endochondral ossification center. Dev Biol. 2012;372(2):263-273. [54] LIU X, DU M, WANG Y, et al. BMP9 overexpressing adipose-derived mesenchymal stem cells promote cartilage repair in osteoarthritis-affected knee joint via the Notch1/Jagged1 signaling pathway. Exp Ther Med. 2018; 16(6):4623-4631. [55] WANG X, HUANG J, HUANG F, et al. Bone morphogenetic protein 9 stimulates callus formation in osteoporotic rats during fracture healing. Mol Med Rep. 2017;15(5):2537-2545. [56] SONG Q, ZHONG L, CHEN C, et al. miR-21 synergizes with BMP9 in osteogenic differentiation by activating the BMP9/Smad signaling pathway in murine multilineage cells. Int J Mol Med. 2015;36(6):1497-1506. [57] LIAO J, YU X, HU X, et al. lncRNA H19 mediates BMP9-induced osteogenic differentiation of mesenchymal stem cells (MSCs) through Notch signaling. Oncotarget. 2017;8(32):53581-53601. [58] ZHANG Z, LIU J, ZENG Z, et al. lncRNA Rmst acts as an important mediator of BMP9-induced osteogenic differentiation of mesenchymal stem cells (MSCs) by antagonizing Notch-targeting microRNAs. Aging (Albany NY). 2019;11(24):12476-12496. [59] BEZ M, PELLED G, GAZIT D. BMP gene delivery for skeletal tissue regeneration. Bone. 2020;137:115449. [60] KIMELMAN-BLEICH N, PELLED G, ZILBERMAN Y, et al. Targeted gene-and-host progenitor cell therapy for nonunion bone fracture repair. Mol Ther. 2011;19(1):53-59. [61] FUJIOKA-KOBAYASHI M, SCHALLER B, ZHANG Y, et al. Recombinant human bone morphogenetic protein (rhBMP)9 induces osteoblast differentiation when combined with demineralized freeze-dried bone allografts (DFDBAs) or biphasic calcium phosphate (BCP). Clin Oral Investig. 2017;21(5): 1883-1893. [62] ZHAO C, ZENG Z, QAZVINI NT, et al. Thermoresponsive Citrate-Based Graphene Oxide Scaffold Enhances Bone Regeneration from BMP9-Stimulated Adipose-Derived Mesenchymal Stem Cells. ACS Biomater Sci Eng. 2018;4(8):2943-2955. [63] FUJIOKA-KOBAYASHI M, SAWADA K, KOBAYASHI E, et al. Recombinant Human Bone Morphogenetic Protein 9 (rhBMP9) Induced Osteoblastic Behavior on a Collagen Membrane Compared With rhBMP2. J Periodontol. 2016;87(6):e101-107. [64] ZHAO C, QAZVINI NT, SADATI M, et al. A pH-Triggered, Self-Assembled, and Bioprintable Hybrid Hydrogel Scaffold for Mesenchymal Stem Cell Based Bone Tissue Engineering. ACS Appl Mater Interfaces. 2019;11(9):8749-8762. [65] XIONG C, DAUBS MD, MONTGOMERY SR, et al. BMP-2 adverse reactions treated with human dose equivalent dexamethasone in a rodent model of soft-tissue inflammation. Spine (Phila Pa 1976). 2013;38(19):1640-1647. [66] BI W, LIU Y, GUO J, et al. All-trans retinoic-acid inhibits heterodimeric bone morphogenetic protein 2/7-stimulated osteoclastogenesis, and resorption activity. Cell Biosci. 2018;8:48. [67] GRGUREVIC L, CHRISTENSEN GL, SCHULZ TJ, et al. Bone morphogenetic proteins in inflammation, glucose homeostasis and adipose tissue energy metabolism. Cytokine Growth Factor Rev. 2016;27:105-118. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Yuan Jiawei, Zhang Haitao, Jie Ke, Cao Houran, Zeng Yirong. Underlying targets and mechanism of Taohong Siwu Decoction in prosthetic joint infection on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1428-1433. |
| [4] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [5] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [6] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [7] | Gu Xia, Zhao Min, Wang Pingyi, Li Yimei, Li Wenhua. Relationship between hypoxia inducible factor 1 alpha and hypoxia signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1284-1289. |
| [8] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [9] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [10] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [11] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [12] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [13] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [14] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [15] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||