Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (28): 4452-4458.doi: 10.3969/j.issn.2095-4344.2289
Previous Articles Next Articles
Scaffolds for dental pulp regeneration and revascularization
Chen Leyi, Lü Xiaolin, Xu Wenan
Southern Medical University Nanfang Hospital/The First Clinical Medical College, Southern Medical University School of Stomatology, Guangzhou 510515, Guangdong Province, China
-
Received:2019-10-26Revised:2019-10-29Accepted:2019-12-19Online:2020-10-08Published:2020-08-31 -
Contact:Xu Wenan, MD, Associate professor, Master’s supervisor, Southern Medical University Nanfang Hospital/The First Clinical Medical College, Southern Medical University School of Stomatology, Guangzhou 510515, Guangdong Province, China -
About author:Chen Leyi, Southern Medical University Nanfang Hospital/The First Clinical Medical College, Southern Medical University School of Stomatology, Guangzhou 510515, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China, No. 81700956; 2018 Provincial-level College Students Innovation Training Program of Southern Medical University, No. 201812121134; Dean's Fund of Southern Medical University Nanfang Hospital, No. 2016B028
CLC Number:
Cite this article
Chen Leyi, Lü Xiaolin, Xu Wenan. Scaffolds for dental pulp regeneration and revascularization[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4452-4458.
share this article
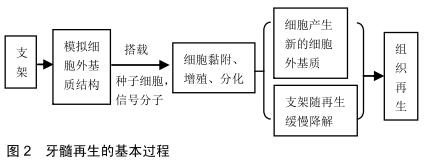
2.1 依据材料来源分类 牙髓再生组织工程支架材料依据材料来源可分为天然和人工合成2大类。天然来源支架材料主要包括了多糖类(藻酸盐、壳聚糖[3]、纤维素等[4])、细胞外基质来源类(脱细胞细胞外基质、透明质酸[5]、胶原[6]、明胶等[7])、蛋白多肽来源类(纤维蛋白[8]、丝蛋白[9]、自组装肽等)[10]。天然材料的机械性能较差,并且在制备过程中难以纯化,但是它们具有良好的亲水性和生物相容性,容易制备成水凝胶结构,使得天然材料在牙髓再生中受到了广泛关注。人工合成材料以聚合物(聚乳酸、聚乙酸醇、聚乳酸-羟基乙酸共聚物等[11])和生物陶瓷类2大类为主。合成材料来源广泛且容易大量制备,并且几乎不存在免疫原性问题,然而合成材料最大的困境是其生物相容性较差、有一定的细胞毒性,不能为细胞提供适宜的黏附和增殖环境[12]。 尽管可用于牙髓再生组织工程的支架材料种类繁多,目前最受关注和效果较好的支架材料并不多。文内将介绍血小板衍生物、脱细胞细胞外基质支架及自组装肽支架3种支架。 2.1.1 血小板衍生物 富血小板血浆由HARKE等[13]于1977年首次分离制得,其天然结构适合细胞生长,含有大量生长因子可促进细胞增殖分化和干细胞迁移,先已被广泛应用于临床实践和研究中。 研究表明改良富血小板血浆含有高浓度的血小板衍生因子AA和转化生长因子β1,能促进人类脱落乳牙干细胞的增殖并分化为成骨细胞,可作为临床牙齿重建治疗支架的备用选择[14]。此外,STAMBOLSKY等[15]发现在雌性比格犬年轻尖牙根尖周炎的治疗中,使用富血小板血浆作为支架的方案对比传统方法在组织学上表现出更高比率的根尖闭合,并能在牙髓腔内检测到高达87.5%的活性组织。但是多个实验报道均指出无论是血凝块或是富血小板血浆作为支架,都显著诱导再生牙髓腔壁增厚和根尖缩窄。ZHU等[16]利用免疫组织化学染色检测了牙髓拔除术后犬牙利用自体牙髓干细胞和富血小板血浆进行牙髓再生的情况,发现有多种支持再生组织为牙周韧带、牙本质和骨组织形成的分子标记。这说明富血小板血浆在牙髓再生过程中诱导生成的很可能并不是牙髓样组织,而是牙骨质或牙周组织。广泛的实验表明其具有强大的诱导成骨作用,故富血小板血浆在临床研究中多用于治疗大块骨缺损。 富血小板纤维蛋白是第2代血小板源性自体来源支架材料,相较富血小板血浆而言,其制备方法简单,无需加入凝血酶、抗凝剂和氯化钙等其他物质[17]。富血小板纤维蛋白还具有强大的纤维网络和优越的力学特 性[18],并能缓慢释放多种生长因子,有助于干细胞的迁移、增殖和分化[19-20],加速组织修复过程。 利用脂多糖模拟革兰阴性菌引发的牙髓炎,在脂多糖-牙髓干细胞-富血小板纤维蛋白共培养体系下发现,富血小板纤维蛋白可以使脂多糖刺激的牙髓干细胞表达炎症因子减少,减轻炎症反应[21]。富血小板纤维蛋白中含有大量白细胞,在对抗炎症反应和免疫调节中有重要作用。2013年KESWANI等[22]发表的临床报告中,利用自体富血小板纤维蛋白对一名右上颌中切牙外伤伴牙髓坏死的7岁男孩行牙髓血运重建术,12-15周后牙髓炎症状消失,影像学显示牙根发育和根尖闭合,并且牙髓活力测试与相邻侧切牙相似。随后许多研究者开展了相关研究和临床试验[23-24],肯定了富血小板纤维蛋白作为牙髓血运重建材料的可行性和安全性。CHEN等[23]将犬牙髓干细胞和富血小板纤维蛋白颗粒的细胞片装载在去髓消毒后的髓腔中植入裸鼠体内,8周后苏木精-伊红染色观察到明显的细胞外基质和毛细血管生成,而在单一牙髓干细胞组中没有观察到毛细血管的生成。在2/8和3/8含量富血小板纤维蛋白组中,检测到牙本质涎磷蛋白基因表达显著提升,这说明富血小板纤维蛋白能促进牙髓干细胞增殖分化为成牙本质细胞,并且有效促进血管再生。 除此之外,富血小板纤维蛋白具有更接近生理凝血过程和天然结构[18]、更少的免疫原性和交叉反应,让富血小板纤维蛋白成为较富血小板血浆更为热门的牙髓甚至口腔软硬组织的再生材料[25]。学者在各层次研究中也对两者在年轻恒牙中再生性能进行了比较,两者相较于传统的血凝块可促进根尖闭合和愈合,但富血小板纤维蛋白和富血小板血浆对比优势并不明显[26],有研究甚至显示富血小板血浆的疗效更好[27]。 2.1.2 脱细胞细胞外基质来源材料 细胞外基质是由动物细胞合成并分泌到胞外、分布在细胞表面或细胞之间的大分子,是细胞生长的天然支架。为尽可能模拟体内牙髓组织生长发育的环境,学者尝试通过对牙髓或牙髓内细胞培养物进行脱细胞处理,以获得利于血运重建或牙髓组织再生的支架。 目前牙髓再生脱细胞细胞外基质来源支架主要有2种来源,即牙髓组织来源或体外细胞分泌来源细胞外基质通过试剂洗脱细胞后获得细胞外基质骨架。实验证明人牙髓干细胞来源的细胞外基质可以在没有外源性生长因子加入的情况下,促进牙髓干细胞、牙周膜干细胞和人骨髓间充质干细胞表达牙源性标志蛋白和向成本牙本质细胞分化[28]。HU等[29]将猪牙髓脱细胞基质和人牙髓干细胞混合注入人第三磨牙牙根的1 mm牙片中,随后移植到裸鼠体内,2周后苏木精-伊红染色观察到牙髓样组织再生,并伴有高表达牙本质涎磷蛋白的成牙本质细胞样细胞再生。利用猪牙髓脱细胞基质对拔髓的比格犬进行治疗,8周后不仅观察到血管的形成,免疫组织化学染色还显示有犬源性牙本质涎蛋白阳性组织和CD31+细胞存在[30]。证明在没有加入外源性干细胞和细胞因子的情况下,外源性细胞外基质可能通过诱导干细胞迁移到再生部位,加快自体牙髓组织的再生,同时能进行自我降解。HUANG等[31]制备的人牙髓干细胞和上皮细胞双重来源细胞外基质支架对比单一来源支架,用于体外细胞培养中能明显提升牙髓干细胞和人骨髓间充质干细胞相关成血管因子的表达,并且在体实验后组织切片观察到血管生成,管内有大量红细胞通过,证明血管具有血流。 细胞外基质来源的支架无细胞毒性[32]、富含多种有利于血管再生的成分[29,32]、拥有利于干细胞定植和再生的结构,但如何改进其制备方法、减少有效成分和结构的丢失同时降低其免疫原性值得学者继续研究和探讨[30,33]。细胞外基质表现出的优异成血管能力,表明其作为潜在牙髓血运重建的支架材料的重大价值。不同来源细胞外基质对干细胞基因表达、分泌细胞因子的影响有所不同,这提示可以通过组合不同组织来源的细胞外基质设计一种最有利于牙髓再生和血管化的支架。 2.1.3 自组装肽 自组装肽由ZHANG等[34]首次发现并报道的生物离子自我组装短肽,主要有RAD16 (ARADADARARADADA)和EAK1(AEAEAKAKAEAEAKAK)两大类。因其具有含水量丰富、生物相容性好、能在体内酶的作用下逐渐降解、拥有与细胞外基质相似的3D结构等优点,并且具有纳米材料性质和水凝胶形态,使得自组装肽成为备受组织工程领域青睐的材料。在牙髓再生领域中,自组装肽促进干细胞增殖、分化[35]、组织矿化的能力也在体内外实验中得到肯定[36]。 商品化的自组装肽即PuramatrixTM,早在2005年已经进入市场,用于各种细胞培养和组织修复的研究中。实验证实PuramatrixTM支持牙髓干细胞[37]、脱落乳牙干细胞等的存活、增殖、成牙本质分化,同时因为该支架是液体,可以注射并适应不同形状的根管空间。在与另一商用支架人类重组胶原纤维Ⅰ的比较中,两者对脱落乳牙干细胞增殖、分化、体内成血管的能力不分伯仲(P > 0.05),但PuramatrixTM组中牙本质涎磷蛋白的高表达比人类重组胶原纤维Ⅰ提前了1周[38],这预示着商用自组装肽在牙髓再生方面具有广阔的临床应用前景。 随着自组装肽的发展,出现了不同结构或修饰方式的自组装肽三维生物支架,比如多结构域自组装肽能通过改变结构域来定制生物支架的性能。这种支架可以缓慢而平稳地释放生长因子,与牙髓干细胞共同移植到裸鼠皮下能明显观测到胶原、成牙本质样细胞、微血管的形成及成牙本质标志物牙本质涎蛋白的沉积[39]。尽管自组装肽目前在临床和实验中使用广泛,但其并没有组织特异性。而理想的支架可以通过其本身的特性诱导细胞分化的方向。通过在RADA16-I(Ac-(RADA)4-CONH2)的羧基端分别加上6种不同的成血管功能肽段获得的全新自组装肽,可以促进人脐静脉内皮细胞增殖和成管[40]。NGUYEN等[41]将SL基础(KSLSLSLSLSLSLK)的自组装肽水凝胶系统羧基端和细胞外基质磷酸化糖蛋白的活性结构之一Dentonin (TDLQERGDNDISPFSGDGQPFKD)相连接,用于实现诱导牙髓干细胞成牙本质分化的目的。自组装肽从简单寡肽到复杂多结构且多功能肽的演化,使天然来源的材料有了更丰富的人工特性。 2.2 材料的制备方式及形态 牙髓血运重建需要一定的干细胞能够稳定地增殖、分化、成熟、定型,最终再生为正常或近似正常的牙髓组织。为此,必须为干细胞提供满足其生长条件的环境。与此同时,除了细胞本身的生长外,牙髓血运重建更离不开细胞外基质相关成分的重建。又因为牙髓软组织由外部硬组织支撑,再生支架不需要太高的机械强度。随着硬度增加,干细胞的分化方向就越偏向于坚硬的骨质细胞,在最坚硬的支架中,间充质干细胞会分化为成骨细胞[42],反而使髓腔过度矿化,引起不必要的并发症。但支架一定的强度是必须的。所以目前大多使用半合成的聚合物作为牙髓血运重建的干细胞支架。 当前的思路基本上可概括为:制备人工合成或半合成材料模拟细胞外基质微环境,使相关干细胞能够在该微环境中附着、增殖、分化、成熟。不同的制备方法使得天然、合成或半合成的物质最终以不同的物理性质和形态呈现,也决定了其在牙髓再生和血运重建中的作用,见图2。 2.2.1 水凝胶 水凝胶是以物理(可逆的缠结链、氢键或疏水作用等)或化学(不可逆的共价结合)方式交联在一起的亲水聚合物网络,其可以溶胀但不溶于水中并具有3D网络结构,使其可以有效固定和释放活性物质和生物分子[43]。 水凝胶的本质特点是高含水量的聚合物,因此它比其他任何类型的聚合物生物材料更像天然软组织,能够表现出良好的生物相容性与对氧气、营养物质及其他水溶性物质的高度通透性,并能够很好地包裹、支持细胞生长[44]。绝大多数的水凝胶是可注射的,容易进入狭小的根管空间、适应多种根管结构[5,45]。 通过控制水凝胶的网络结构可以实现对水凝胶特性的设计,如水凝胶支架降解的特征、生物分子的扩散及细胞在网络中的迁移方式,达到重现科学家预设的再生过程[46]。而实现控制水凝胶结构特性的方法是:产生共价交联的水凝胶网络,并通过化学反应在高分子团的末端聚合官能化的大分子单体[44]。这些大分子单体能囊括多数组织工程中常用的材料,并能进行相互搭配。例如通过光聚合交联制备的聚乙二醇-纤维蛋白原水凝胶既兼顾了机械性质、维持足够的强度,又能恒定地保持纤维蛋白骨架,使牙髓干细胞能够在支架中生长、变成纺锤形态并且实现生物矿化[44,47]。触变性合成黏土-缺氧模拟剂水凝胶一方面利用合成黏土可吸附蛋白质的特性,又结合缺氧模拟剂模拟低氧对于间充质干细胞的趋化作用,理论上能使牙髓干细胞具有更高的活性和成血管能力[48]。 水凝胶本身可作为干细胞和细胞因子的优良载体,通过支架和其他物质的不同配比构建不同类型的再生单元。将牙髓干细胞与人脐静脉内皮细胞按3∶1、1∶1和1∶3比例混合后与PuramatrixTM共同注入牙根片段并植入裸鼠皮下,可以发现随着人脐静脉内皮细胞比例的提高,血管样组织生成增加并能促进牙髓干细胞的生长[37]。 水凝胶的流体性质与其可进行广泛修饰来改变自身生物相容性、细胞黏附性与空间结构的特点,决定了它在牙髓血运重建中的重要地位。现有的研究有力地证明了水凝胶及其衍生物在牙髓干细胞增殖[49]、分化[4-5]、迁移、定型[50]、血管生成等方面的优越性能。 2.2.2 纳米级材料 纳米材料是指尺度空间位于 100 nm内、合成后具有不同于原材料特性的材料。在组织工程中,适合制备纳米级支架的方法有:静电纺丝[51]、自我组装、热致相分离等[52]。 纳米材料可以在纳米级别模拟细胞外基质的3D骨架结构。相同质量的纳米材料具有更高的相对表面积,使细胞附着位点增多、附着能力增强[53]。另一方面,纳米材料的可塑性高,可植入狭小的根管空间,部分材料合成后可形成液体或类似水凝胶样的可注射物质。这两点特性能帮助材料适合复杂的根管结构,同时能相对携带更多的干细胞定植在牙髓腔内,有助于加速牙髓再生的进程。 纳米材料具有可控制孔裂隙,通过不同手段对支架进行改造[54],其能很好地与抗细菌、抗炎分子及生长因子结合,成为它们的运载体并且影响它们的释放方式[55]。目前在牙髓血运重建中很重要的一个步骤就是使用三联抗生素对坏死牙髓进行彻底的消毒灭菌,以确保牙髓腔内成为一个相对无菌的空间,以免炎症反应影响干细胞的增殖和分化。然而,抗生素对干细胞、内皮细胞的毒性及抑制血管相关生成因子表达的作用,成为再生牙髓血运重建的阻碍[56]。因此,一种基于纳米纤维的三联抗生素药物载体也应运而生[57],它不仅有与传统三联抗生素溶液相当的杀菌效果,还具有减轻抗生素细胞毒性、促进牙髓干细胞黏附增殖的能力[58]。纳米支架联合其他抑炎反应药物,如聚L-乳酸纳米纤维和降脂药辛伐他汀[59],能逆转脂多糖刺激作用导致的细胞外调节蛋白激酶1/2和Smad蛋白1磷酸化水平下调,促进牙髓干细胞的成牙本质分化,且体内实验表现出良好的血管生成能力。 分子大小的改变使得纳米材料相较于相当大体积材料获得了新的物理特性[60],有利于牙髓血运重建的进行。在缺氧条件下,聚左旋乳酸纳米纤维微球能增加血管内皮生长因子在牙髓干细胞中的表达,而在相同材料的普通微球中并无改变。组织学结果证明,这种缺氧诱导-纳米纤维微球在皮下植入和在体牙髓腔实验中均成功诱导成牙本质样细胞的形成,展现了强大的促成血管能力[61]。 "
|
[1] WIGLER R, KAUFMAN AY, LIN S, et al. Revascularization: a treatment for permanent teeth with necrotic pulp and incomplete root development.J Endod.2013;39(3):319-326.
[2] YANG J, YUAN G, CHEN Z. Pulp Regeneration: Current Approaches and Future Challenges. Front Physiol. 2016;7: 58.
[3] SOARES DG, ANOVAZZI G, BORDINI E, et al. Biological Analysis of Simvastatin-releasing Chitosan Scaffold as a Cell-free System for Pulp-dentin Regeneration. J Endod. 2018;44(6):971-976.
[4] TETI G, SALVATORE V, FOCAROLI S, et al. In vitro osteogenic and odontogenic differentiation of human dental pulp stem cells seeded on carboxymethyl cellulose-hydroxyapatite hybrid hydrogel. Front Physiol. 2015;6:297.
[5] CHREPA V, AUSTAH O, DIOGENES A. Evaluation of a Commercially Available Hyaluronic Acid Hydrogel (Restylane) as Injectable Scaffold for Dental Pulp Regeneration: An In Vitro Evaluation. J Endod. 2017;43(2):257-262.
[6] ZHANG L, YU Y, FENG K, et al. Templated dentin formation by dental pulp stem cells on banded collagen bundles nucleated on electrospun poly (4-vinyl pyridine) fibers in vitro. Acta Biomater. 2018;76:80-88.
[7] CHEN G, CHEN J, YANG B, et al. Combination of aligned PLGA/Gelatin electrospun sheets, native dental pulp extracellular matrix and treated dentin matrix as substrates for tooth root regeneration. Biomaterials. 2015;52:56-70.
[8] RUANGSAWASDI N, ZEHNDER M, WEBER FE. Fibrin gel improves tissue ingrowth and cell differentiation in human immature premolars implanted in rats. J Endod. 2014;40(2): 246-250.
[9] YANG JW, ZHANG YF, SUN ZY, et al. Dental pulp tissue engineering with bFGF-incorporated silk fibroin scaffolds. J Biomater Appl. 2015;30(2):221-229.
[10] 李龙飚,汪成林,叶玲.天然支架材料在牙髓组织工程再生中的研究进展[J].国际口腔医学杂志,2018,45(6):666-672.
[11] LI Y, HE L, PAN S, et al. Three-dimensional simulated microgravity culture improves the proliferation and odontogenic differentiation of dental pulp stem cell in PLGA scaffolds implanted in mice. Mol Med Rep. 2017;15(2): 873-878.
[12] MOUSSA DG, APARICIO C. Present and future of tissue engineering scaffolds for dentin-pulp complex regeneration. J Tissue Eng Regen Med. 2019;13(1):58-75.
[13] HARKE H, TANGER D, FURST-DENZER S, et al. Effect of a preoperative separation of platelets on the postoperative blood loss subsequent to extracorporeal circulation in open heart surgery (author's transl). Anaesthesist.1977;26(2): 64-71.
[14] WEN J, LI HT, LI SH, et al. Investigation of modified platelet-rich plasma (mPRP) in promoting the proliferation and differentiation of dental pulp stem cells from deciduous teeth. Braz J Med Biol Res. 2016;49(10):e5373.
[15] STAMBOLSKY C, RODRÍGUEZ-BENÍTEZ S, GUTIÉRREZ-PÉREZ JL, et al. Histologic characterization of regenerated tissues after pulp revascularization of immature dog teeth with apical periodontitis using tri-antibiotic paste and platelet-rich plasma. Arch Oral Biol. 2016;71:122-128.
[16] ZHU X, WANG Y, LIU Y, et al. Immunohistochemical and Histochemical Analysis of Newly Formed Tissues in Root Canal Space Transplanted with Dental Pulp Stem Cells Plus Platelet-rich Plasma. J Endod. 2014;40(10):1573-1578.
[17] DOHAN DM, CHOUKROUN J, DISS A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part I: technological concepts and evolution. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101(3):e37-e44.
[18] CASTRO AB, MESCHI N, TEMMERMAN A, et al. Regenerative potential of leucocyte- and platelet-rich fibrin. Part A: intra-bony defects, furcation defects and periodontal plastic surgery. A systematic review and meta-analysis. J Clin Periodontol. 2017;44(1):67-82.
[19] HUANG F, YANG S, ZHAO J, et al. Platelet-rich Fibrin Increases Proliferation and Differentiation of Human Dental Pulp Cells. J Endod. 2010;36(10):1628-1632.
[20] WOO S, KIM W, LIM H, et al. Combination of Mineral Trioxide Aggregate and Platelet-rich Fibrin Promotes the Odontoblastic Differentiation and Mineralization of Human Dental Pulp Cells via BMP/Smad Signaling Pathway. J Endod. 2016;42(1):82-88.
[21] KIM JDP, WOO SP, CHOI NDP, et al. Effect of Platelet-rich Fibrin on Odontoblastic Differentiation in Human Dental Pulp Cells Exposed to Lipopolysaccharide. J Endod. 2016;43(3): 433-438.
[22] KESWANI D, PANDEY RK. Revascularization of an immature tooth with a necrotic pulp using platelet-rich fibrin: a case report. Int Endod J. 2013;46(11):1096-1104.
[23] CHEN Y, ZHAO Y, ZHAO Y, et al. Potential dental pulp revascularization and odonto-/osteogenic capacity of a novel transplant combined with dental pulp stem cells and platelet-rich fibrin. Cell Tissue Res. 2015;361(2):439-455.
[24] BAKHTIAR H, ESMAEILI S, FAKHR TABATABAYI S, et al. Second-generation Platelet Concentrate (Platelet-rich Fibrin) as a Scaffold in Regenerative Endodontics: A Case Series. J Endod. 2017;43(3):401-408.
[25] 周延民,付丽.富血小板纤维蛋白在口腔软硬组织再生中的作用—回顾与展望[J].口腔医学, 2018,38(11):961-965.
[26] MURRAY PE. Platelet-Rich Plasma and Platelet-Rich Fibrin Can Induce Apical Closure More Frequently Than Blood-Clot Revascularization for the Regeneration of Immature Permanent Teeth: A Meta-Analysis of Clinical Efficacy. Front Bioeng Biotechnol. 2018;6:139.
[27] SHIVASHANKAR VY, JOHNS DA, MAROLI RK, et al. Comparison of the Effect of PRP, PRF and Induced Bleeding in the Revascularization of Teeth with Necrotic Pulp and Open Apex: A Triple Blind Randomized Clinical Trial. J Clin Diagn Res. 2017;11(6):C34-C39.
[28] RAVINDRAN S, HUANG C, GEORGE A. Extracellular matrix of dental pulp stem cells: applications in pulp tissue engineering using somatic MSCs. Front Physiol. 2014;4:395.
[29] HU L, GAO Z, XU J, et al. Decellularized Swine Dental Pulp as a Bioscaffold for Pulp Regeneration. Biomed Res Int. 2017; 2017:1-9.
[30] ALQAHTANI Q, ZAKY SH, PATIL A, et al. Decellularized Swine Dental Pulp Tissue for Regenerative Root Canal Therapy. J Dent Res. 2018;97(13):1460-1467.
[31] HUANG C, NARAYANAN R, WARSHAWSKY N, et al. Dual ECM Biomimetic Scaffolds for Dental Pulp Regenerative Applications. Front Physiol. 2018;9:495.
[32] MATOUG-ELWERFELLI M, DUGGAL MS, NAZZAL H, et al. A biocompatible decellularized pulp scaffold for regenerative endodontics. Int Endod J. 2018;51(6):663-673.
[33] SONG JS, TAKIMOTO K, JEON M, et al. Decellularized Human Dental Pulp as a Scaffold for Regenerative Endodontics. J Dent Res.2017;96(6):640-646.
[34] ZHANG S, HOLMES TC, DIPERSIO CM, et al. Self-complementary oligopeptide matrices support mammalian cell attachment. Biomaterials. 1995;16(18): 1385-1393.
[35] CAVALCANTI BN, ZEITLIN BD, NOR JE. A hydrogel scaffold that maintains viability and supports differentiation of dental pulp stem cells. Dent Mater. 2013;29(1):97-102.
[36] CHAN B, WONG RWK, RABIE B. In vivo production of mineralised tissue pieces for clinical use: a qualitative pilot study using human dental pulp cell. Int J Oral Maxillofac Surg. 2011;40(6):612-620.
[37] DISSANAYAKA WL, HARGREAVES KM, JIN L, et al. The Interplay of Dental Pulp Stem Cells and Endothelial Cells in an Injectable Peptide Hydrogel on Angiogenesis and Pulp RegenerationIn Vivo. Tissue Eng Part A. 2015;21(3-4): 550-563.
[38] ROSA V, ZHANG Z, GRANDE RHM, et al. Dental Pulp Tissue Engineering in Full-length Human Root Canals. J Dent Res. 2013;92(11):970-975.
[39] GALLER KM, HARTGERINK JD, CAVENDER AC, et al. A customized self-assembling peptide hydrogel for dental pulp tissue engineering. Tissue Eng Part A. 2012;18(1-2):176-184.
[40] HORII A. Screening of functionalized self-assembling peptide nanofiber scaffolds with angiogenic activity for endothelial cell growth. Progress in Natural Science: Materials International. 2011;21(2):111-116.
[41] NGUYEN PK, GAO W, PATEL SD, et al. Self-Assembly of a Dentinogenic Peptide Hydrogel. ACS Omega. 2018;3(6): 5980-5987.
[42] ENGLER AJ, SEN S, SWEENEY HL, et al. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126(4): 677-689.
[43] 夏鹍,张旗.水凝胶支架材料在牙髓再生中的研究进展[J].口腔医学,2019,39(7):651-654.
[44] ZHU J, MARCHANT RE. Design properties of hydrogel tissue-engineering scaffolds. Expert Rev Med Devices. 2014; 8(5):607-626.
[45] SILVA CR, BABO PS, GULINO M, et al. Injectable and tunable hyaluronic acid hydrogels releasing chemotactic and angiogenic growth factors for endodontic regeneration. Acta Biomater.2018;77:155-171.
[46] ZHANG YS, KHADEMHOSSEINI A. Advances in engineering hydrogels. Science. 2017;356(6337):f3627.
[47] LU Q, PANDYA M, RUFAIHAH AJ, et al. Modulation of Dental Pulp Stem Cell Odontogenesis in a Tunable PEG-Fibrinogen Hydrogel System. Stem Cells Int. 2015;2015:1-9.
[48] MÜLLER AS, ARTNER M, JANJIĆ K, et al. Synthetic Clay–based Hypoxia Mimetic Hydrogel for Pulp Regeneration: The Impact on Cell Activity and Release Kinetics Based on Dental Pulp–derived Cells In Vitro. J Endod. 2018;44(8): 1263-1269.
[49] PASQUI D, TORRICELLI P, CAGNA M, et al.Carboxymethyl cellulose—hydroxyapatite hybrid hydrogel as a composite material for bone tissue engineering applications. J Biomed Mater Res A. 2014;102(5):1568-1579.
[50] JONES TD, KEFI A, SUN S, et al. An Optimized Injectable Hydrogel Scaffold Supports Human Dental Pulp Stem Cell Viability and Spreading. Adv Med. 2016;2016:1-8.
[51] 张倩莉,袁重阳,王晓燕.静电纺纳米纤维支架在牙髓再生中的应用[J].口腔疾病防治,2018,26(11):738-742.
[52] CHIERUZZI M, PAGANO S, MORETTI S, et al.Nanomaterials for Tissue Engineering In Dentistry. Nanomaterials. 2016;6(7): 134.
[53] PRAVEENA C, MANNE P, KALLURI L, et al.Nanobiomaterials and Their Application in Prosthodontics//CHAUGHULE R S. Dental Applications of Nanotechnology.Cham: Springer International Publishing,2018:177-201.
[54] MOHAMMADIAN F, EATEMADI A. Drug loading and delivery using nanofibers scaffolds. Artif Cells Nanomed Biotechnol. 2017;45(5):881-888.
[55] KELLER L, OFFNER D, SCHWINTÉ P, et al. Active Nanomaterials to Meet the Challenge of Dental Pulp Regeneration. Materials. 2015;8(11):7461-7471.
[56] SAGHIRI MA, ASATOURIAN A,GARCIA-GODOY F, et al. Effect of biomaterials on angiogenesis during vital pulp therapy. Dent Mater J. 2016;35(5):701-709.
[57] BOTTINO MC, PANKAJAKSHAN D, NOR JE. Advanced Scaffolds for Dental Pulp and Periodontal Regeneration. Dent Clin North Am. 2017;61(4):689-711.
[58] PANKAJAKSHAN D, ALBUQUERQUE MTP, EVANS JD, et al. Triple Antibiotic Polymer Nanofibers for Intracanal Drug Delivery: Effects on Dual Species Biofilm and Cell Function.J Endod. 2016;42(10):1490-1495.
[59] SOARES DG, ZHANG Z, MOHAMED F, et al. Simvastatin and nanofibrous poly( l -lactic acid) scaffolds to promote the odontogenic potential of dental pulp cells in an inflammatory environment.Acta Biomater.2018;68:190-203.
[60] HASSAN S, PRAKASH G, BAL OZTURK A, et al. Evolution and clinical translation of drug delivery nanomaterials.Nano Today. 2017;15:91-106.
[61] KUANG R, ZHANG Z, JIN X, et al. Nanofibrous spongy microspheres for the delivery of hypoxia-primed human dental pulp stem cells to regenerate vascularized dental pulp. Acta Biomater. 2016;33:225-234. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Huang Maomao, Hu Yue, Wang Binchuan, Zhang Chi, Xie Yujie, Wang Jianxiong, Wang Li, Xu Fangyuan. Bibliometric and visual analysis of international literature addressing ischemic stroke rehabilitation in recent 10 years [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3725-3733. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||