Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (13): 2039-2046.doi: 10.3969/j.issn.2095-4344.2032
Previous Articles Next Articles
Regulatory effect of uncarboxylated osteocalcin on osteogenic and adipogenic differentiation of mouse bone marrow mesenchymal stem cells under high-glucose conditions
Liu Zhongsheng, Yang Jianhong
- Medical School, University of Chinese Academy of Sciences, Beijing 101400, China
-
Received:2019-07-11Revised:2019-07-13Accepted:2019-07-31Online:2020-05-08Published:2020-03-07 -
Contact:Yang Jianhong, PhD, Professor, Medical School, University of Chinese Academy of Sciences, Beijing 101400, China -
About author:Liu Zhongsheng, Medical School, University of Chinese Academy of Sciences, Beijing 101400, China -
Supported by:the Knowledge Innovation Program of Chinese Academy of Sciences, No. KSCX2-EW-J-29 and Y129015EA2; the College of Life Sciences of University of Chinese Academy of Sciences, No. KJRH2015-006
CLC Number:
Cite this article
Liu Zhongsheng, Yang Jianhong. Regulatory effect of uncarboxylated osteocalcin on osteogenic and adipogenic differentiation of mouse bone marrow mesenchymal stem cells under high-glucose conditions[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(13): 2039-2046.
share this article
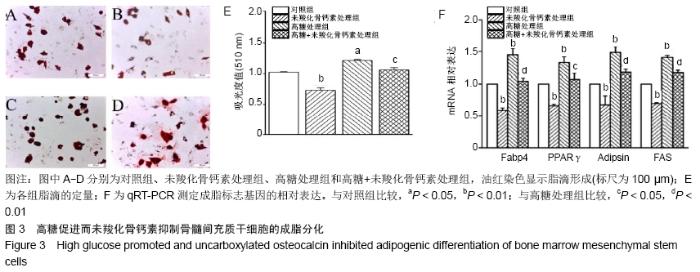
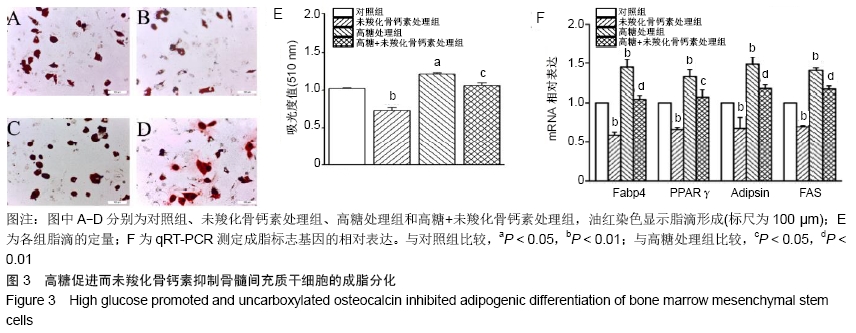
2.3 未羧化骨钙素抑制高糖条件下骨髓间充质干细胞的成脂分化 油红染色显示,与对照组相比,高糖促进骨髓间充质干细胞产生脂滴,经过未羧化骨钙素处理后,骨髓间充质干细胞产生脂滴的量显著减少。对油红染色进行定量分析,高糖处理组吸光度值明显高于对照组,差异有显著性意义(P < 0.05);未羧化骨钙素处理组吸光度值明显低于对照组,差异有非常显著性意义(P < 0.01);高糖+未羧化骨钙素处理组吸光度值明显低于高糖处理组,差异有显著性意义(P < 0.05),见图3A-E。 qRT-PCR检测结果显示,高糖处理组成脂分化标志基因的表达明显高于对照组(PFabp4 < 0.01;PPPARγ < 0.01;PAdipsin < 0.01;PFAS < 0.01);未羧化骨钙素处理组成脂分化基因的表达明显低于对照组(PFabp4 < 0.01;PPPARγ < 0.01;PAdipsin < 0.01;PFAS < 0.01)。高糖+未羧化骨钙素处理组成脂分化基因的表达明显低于高糖处理组(PFabp4 < 0.01;PPPARγ < 0.05;PAdipsin < 0.01;PFAS < 0.01),见图3F。 "
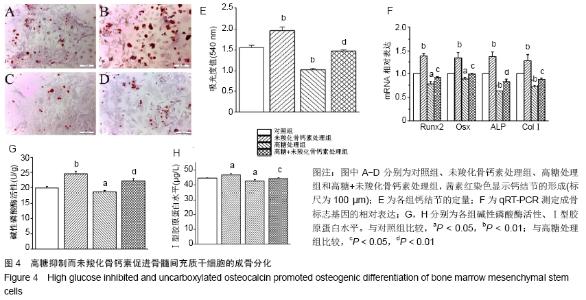
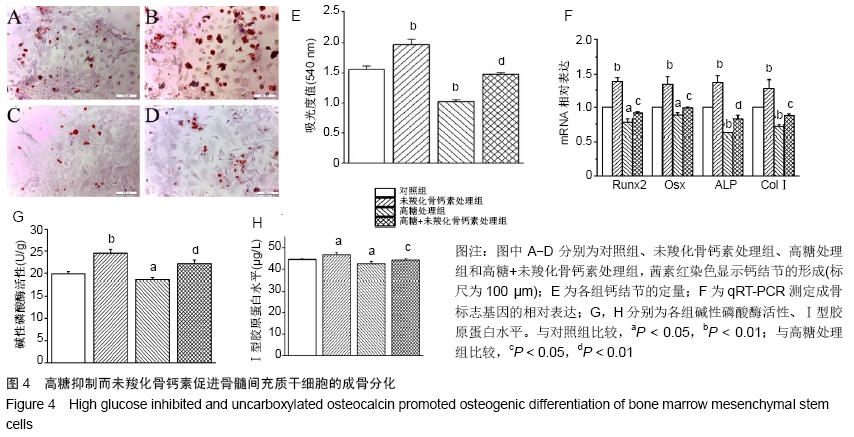
2.4 未羧化骨钙素促进高糖条件下骨髓间充质干细胞的成骨分化 茜素红染色显示,与对照组相比,高糖明显抑制骨髓间充质干细胞形成钙结节,而未羧化骨钙素促进骨髓间充质干细胞形成钙结节。对茜素红染色进行定量分析,高糖处理组吸光度值明显低于对照组,差异有显著性意义(P < 0.05);未羧化骨钙素处理组吸光度值明显高于对照组,差异有非常显著性意义(P < 0.01);高糖+未羧化骨钙素处理组吸光度值明显高于高糖处理组,差异有显著性意义(P < 0.05),见图4A-E。 qRT-PCR检测结果显示,高糖处理组成骨分化标志基因的表达明显低于对照组(PRunx2 < 0.05;POsx < 0.05; PALP < 0.01;PCOLⅠ < 0.01);未羧化骨钙素处理组成骨分化基因的表达明显高于对照组(PRunx2 < 0.01;POsx < 0.01;PALP < 0.01;PCOLⅠ < 0.01);高糖+未羧化骨钙素处理组成骨分化基因的表达明显高于高糖处理组(PRunx2 < 0.05;POsx < 0.05;PALP < 0.01;PCOLⅠ < 0.01),见图4F。 高糖处理组碱性磷酸酶活性以及Ⅰ型胶原蛋白水平明显低于对照组(P < 0.05);未羧化骨钙素处理组碱性磷酸酶活性以及Ⅰ型胶原蛋白水平明显高于对照组(碱性磷酸酶活性,P < 0.01;Ⅰ型胶原蛋白水平,P < 0.05);高糖+未羧化骨钙素处理组成骨分化基因的表达明显高于高糖处理组(碱性磷酸酶活性,P < 0.01;Ⅰ型胶原蛋白水平,P < 0.05),见图4G,H。 "
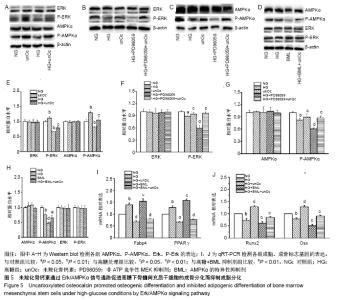
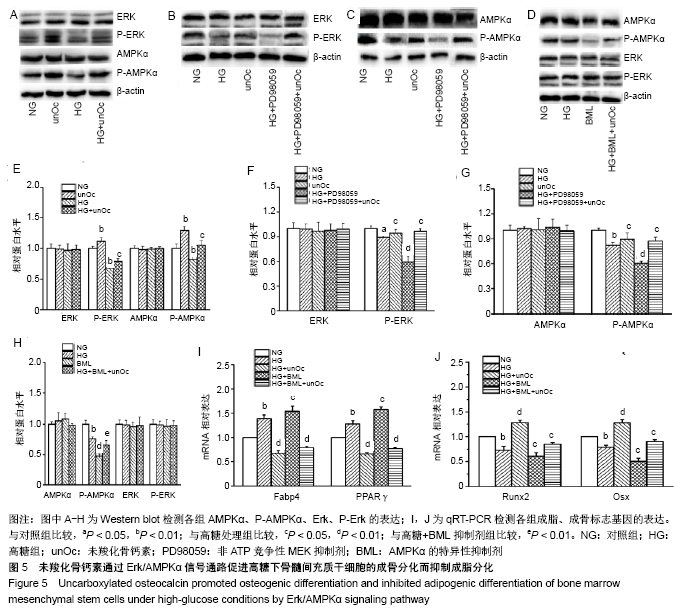
2.5 未羧化骨钙素通过Erk/AMPKα促进高糖条件下骨髓间充质干细胞的成骨分化而抑制成脂分化 Western blot表明,未羧化骨钙素可上调P-Erk(P < 0.01)和P-AMPKα (P < 0.01)的表达;P-Erk抑制剂PD98059使P-Erk、P-AMPKα下调(P < 0.01),这种作用被未羧化骨钙素所逆转(P < 0.05);当使用AMPKα抑制剂BML时,P-AMPKα下调(P < 0.01),这种作用被未羧化骨钙素所逆转(P < 0.01),而P-Erk水平未发生明显变化(P > 0.05)。上述结果说明P-AMPKα处于P-Erk下游,见图5A-H。 为验证未羧化骨钙素是否通过Erk/AMPKα通路调控骨髓间充质干细胞的分化,应用10 μmol/L BML预处理细胞1 h,未羧化骨钙素处理细胞7 d。qRT-PCR结果显示,抑制剂BML抑制AMPKα后,成脂分化基因表达上调,而成骨分化基因表达下调(高糖+BML抑制剂组与高糖处理组相比,PFabp4 < 0.01;PPPARγ < 0.01;PRunx2 < 0.01,POsx < 0.01)。然而,未羧化骨钙素逆转了抑制剂对分化的影响(高糖+BML抑制剂+未羧化骨钙素组与高糖+BML抑制剂组相比,PRunx2 < 0.01,POsx < 0.01),见图5I,J。上述结果说明,未羧化骨钙素是通过Erk/AMPKα通路抑制高糖条件下骨髓间充质干细胞向脂肪细胞分化而促进成骨分化。 "
| [1] YAMAGISHI S, NAKAMURA K, INOUE H. Possible participation of advanced glycation end products in the pathogenesis of osteoporosis in diabetic patients. Med Hypotheses. 2005;65(6):1013-1015. [2] HOUGH FS, PIERROZ DD, COOPER C, et al. MECHANISMS IN ENDOCRINOLOGY: Mechanisms and evaluation of bone fragility in type 1 diabetes mellitus. Eur J Endocrinol. 2016;174(4):R127-138. [3] 亢晨,张斌,孙瑶.骨代谢紊乱与糖尿病:有价值的防治并举[J].中国组织工程研究, 2014, 18(20): 3263-3266. [4] MARIA D, SAMSONRAJ RM, MUNMUN F, et al. Biological effects of melatonin on osteoblast/osteoclast cocultures, bone, and quality of life: Implications of a role for MT2 melatonin receptors, MEK1/2, and MEK5 in melatonin-mediated osteoblastogenesis. Journal of Pineal Research. 2018;64(3): e12465. [5] KHANDUKER S, AHMED R, KHONDKER F, et al. Electrolyte Disturbances in Patients with Diabetes Mellitus. Bangladesh J Med Biochem. 2017;10(1): 27-35. [6] DIMEGLIO LA, IMEL EA. Calcium and phosphate: hormonal regulation and metabolism. Basic and Applied Bone Biology. 2014:261-282. [7] PALERMO A, D'ONOFRIO L, BUZZETTI R, et al. Pathophysiology of Bone Fragility in Patients with Diabetes. Calcif Tissue Int. 2017;100(2): 22-132. [8] NAPOLI N, CHANDRAN M, PIERROZ DD, et al. Mechanisms of diabetes mellitus-induced bone fragility. Nat Rev Endocrinol. 2017; 13(4):208-219. [9] HARDOUIN P, MARIE PJ, ROSEN CJ. New insights into bone marrow adipocytes: Report from the First European Meeting on Bone Marrow Adiposity (BMA 2015). Bone. 2016;93:212-215. [10] LI J, ZHANG H, YANG C, et al. An overview of osteocalcin progress. J Bone Miner Metab. 2016;34(4):367-379. [11] OLDKNOW KJ, MACRAE VE, FARQUHARSON C. Endocrine role of bone: recent and emerging perspectives beyond osteocalcin. J Endocrinol. 2015;225(1):R1-19. [12] TANGSEEFA P, MARTIN SK, FITTER S, et al. Osteocalcin-dependent regulation of glucose metabolism and fertility: Skeletal implications for the development of insulin resistance. J Cell Physiol. 2018;233(5): 769-3783. [13] NG KW. Regulation of glucose metabolism and the skeleton. Clin Endocrinol (Oxf). 2011;75(2):147-155. [14] BONNEAU J, FERLAND G, KARELIS AD, et al. Association between osteocalcin gamma-carboxylation and insulin resistance in overweight and obese postmenopausal women. J Diabetes Complications. 2017; 31(6):1027-1034. [15] GUEDES JAC, ESTEVES JV, MORAIS MR, et al. Osteocalcin improves insulin resistance and inflammation in obese mice: Participation of white adipose tissue and bone. Bone. 2018;115:68-82. [16] LIU J, YANG J. Uncarboxylated osteocalcin inhibits high glucose-induced ROS production and stimulates osteoblastic differentiation by preventing the activation of PI3K/Akt in MC3T3-E1 cells. Int J Mol Med. 2016;37(1):173-181. [17] ZHANG Y, YANG JH. Activation of the PI3K/Akt pathway by oxidative stress mediates high glucose-induced increase of adipogenic differentiation in primary rat osteoblasts. J Cell Biochem. 2013;114(11): 595-2602. [18] AMBROSI TH, SCIALDONE A, GRAJA A, et al. Adipocyte Accumulation in the Bone Marrow during Obesity and Aging Impairs Stem Cell-Based Hematopoietic and Bone Regeneration. Cell Stem Cell. 2017;20(6):771-784. [19] MIRANDA C, GINER M, MONTOYA MJ, et al. Influence of high glucose and advanced glycation end-products (ages) levels in human osteoblast-like cells gene expression. BMC Musculoskelet Disord. 2016;17:377. [20] OKAZAKI R, INOUE D. Mechanism for the Development of Bone Disease in Diabetes: Abnormal Glucose Metabolism. Musculoskeletal Disease Associated with Diabetes Mellitus.2016:43-61. [21] LIANG Y, LIU Y, LAI W, et al. 1,25-Dihydroxy vitamin D3 treatment attenuates osteopenia, and improves bone muscle quality in Goto-Kakizaki type 2 diabetes model rats. Endocrine. 2019;64(1): 84-195. [22] WANG C, MENG H, WANG X, et al. Differentiation of Bone Marrow Mesenchymal Stem Cells in Osteoblasts and Adipocytes and its Role in Treatment of Osteoporosis. Med Sci Monit. 2016;22:226-233. [23] AKABERI S, LINDERGÅRD B, SIMONSEN O, et al. Impact of parathyroid hormone on bone density in long-term renal transplant patients with good graft function. Transplantation. 2006;82(6):749-752. [24] KANAZAWA I, TANAKA S, SUGIMOTO T. The Association Between Osteocalcin and Chronic Inflammation in Patients with Type 2 Diabetes Mellitus. Calcif Tissue Int. 2018;103(6):599-605. [25] KIM KM, LIM S, MOON JH, et al. Lower uncarboxylated osteocalcin and higher sclerostin levels are significantly associated with coronary artery disease. Bone. 2016;83:178-183. [26] XUAN Y, SUN LH, LIU DM, et al. Positive association between serum levels of bone resorption marker CTX and HbA1c in women with normal glucose tolerance. J Clin Endocrinol Metab. 2015;100(1): 74-281. [27] SHIN DW, KIM SN, LEE SM, et al. (-)-Catechin promotes adipocyte differentiation in human bone marrow mesenchymal stem cells through PPAR gamma transactivation. Biochem Pharmacol. 2009;77(1): 125-133. [28] SIMANN M, LE BLANC S, SCHNEIDER V, et al. Canonical FGFs Prevent Osteogenic Lineage Commitment and Differentiation of Human Bone Marrow Stromal Cells Via ERK1/2 Signaling. J Cell Biochem. 2017;118(2):263-275. [29] GU H, HUANG Z, YIN X, et al. Role of c-Jun N-terminal kinase in the osteogenic and adipogenic differentiation of human adipose-derived mesenchymal stem cells. Exp Cell Res. 2015;339(1):112-121. [30] ABDALLAH BM, ALZAHRANI AM, KASSEM M. Secreted Clusterin protein inhibits osteoblast differentiation of bone marrow mesenchymal stem cells by suppressing ERK1/2 signaling pathway. Bone. 2018;110: 221-229. [31] KIM H, SAKAMOTO K. (-)-Epigallocatechin gallate suppresses adipocyte differentiation through the MEK/ERK and PI3K/Akt pathways. Cell Biol Int. 2012;36(2):147-153. [32] WU W, YIN Y, XU K, et al. Knockdown of LGALS12 inhibits porcine adipocyte adipogenesis via PKA-Erk1/2 signaling pathway. Acta Biochim Biophys Sin (Shanghai). 2018;50(10):960-967. [33] WANG YG, QU XH, YANG Y, et al. AMPK promotes osteogenesis and inhibits adipogenesis through AMPK-Gfi1-OPN axis. Cell Signal. 2016;28(9):1270-1282. [34] ZENG Y, PU X, YANG J, et al. Preventive and Therapeutic Role of Functional Ingredients of Barley Grass for Chronic Diseases in Human Beings. Oxid Med Cell Longev. 2018;2018:3232080. [35] MA C, LI G, HE Y, et al. Pronuciferine and nuciferine inhibit lipogenesis in 3T3-L1 adipocytes by activating the AMPK signaling pathway. Life Sci. 2015;136:120-125. [36] LIU B, YANG T, LUO Y, et al. Oat β-glucan inhibits adipogenesis and hepatic steatosis in high fat diet-induced hyperlipidemic mice via AMPK signaling. Journal of Functional Foods. 2018;41:72-82. |
| [1] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [2] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [3] | Ye Haimin, Ding Linghua, Kong Weihao, Huang Zutai, Xiong Long. Role and mechanism of hierarchical microchanneled bone scaffolds in promoting osteogenesis and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 621-625. |
| [4] | Liu Yulin, Li Guotai. Combined effects of hyperbaric oxygen, vibration training and astaxanthin on bone mineral density, glucose metabolism and oxidative stress in diabetic osteoporosis rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3117-3124. |
| [5] | Li Zhen, Huang Yonghui, Sun Jifu, Sun Haitao. Role and mechanism of focal adhesion kinase in inducing osteogenic differentiation of mouse embryonic fibroblasts cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 165-171. |
| [6] | Guo Zhibin, Wu Chunfang, Liu Zihong, Zhang Yuying, Chi Bojing, Wang Bao, Ma Chao, Zhang Guobin, Tian Faming. Simvastatin stimulates osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 2963-2968. |
| [7] | Wang Liu, Song Dongzhe, Huang Dingming. Bone morphogenetic protein 9 regulates stem cell differentiation and bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3064-3070. |
| [8] | Chen Liang, Meng Shu, Cheng Guoping, Ding Yi . Effects of fish scale collagen membrane on adhesion, proliferation and osteogenic differentiation of rat bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2494-2499. |
| [9] | Liu Yan, Gao Xiang, Zhao Xiaoxia, Chen Qingyu, Gao Junwu. Effects of conditioned medium of human periodontal ligament stem cells on proliferation and osteogenic differentiation of inflammatory tissue-derived human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2005-2010. |
| [10] | Meng Maohua, Li Ying, Chen Xin, Cheng Lu, Dong Qiang. Effects and mechanisms of enamel matrix derivatives on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2108-2113. |
| [11] | Chen Junyi, Wang Ning, Chen Ximiao, Zhu Lunjing, Duan Jiangtao, Zhang Xianping, Bei Chaoyong. Osteogenic differentiation of bone marrow mesenchymal stem cells induced by demineralized bone matrix in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(1): 26-31. |
| [12] | Zhang Shengmin, Liu Chao. Research progress in osteogenic differentiation of adipose-derived stem cells induced by bioscaffold materials [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1107-1116. |
| [13] | Zhang Chao, Li Xingyong, Ma Guifu, Pu Xingyu, Luo Wenyuan. Hoxa9 silencing promotes tibial fracture healing by regulating osteogenic differentiation [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(35): 5600-5606. |
| [14] | Yu Xingge, Lin Kaili. Application of nanocomposite hydrogels in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5441-5446. |
| [15] | Liu Hao, Liu Jun, Rui Yongjun, Tang Fenglin, Lu Miao, Ding Tao. Co-transfection by Nell-1 and Noggin-shRNA promotes osteoblast differentiation of adipose derived mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4966-4970. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||