Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (28): 4550-4555.doi: 10.3969/j.issn.2095-4344.2017.28.020
Previous Articles Next Articles
Application and progress of mandibular reconstruction
Ma Cheng, Huang Xuan-ping
- Department of Oral and Maxillofacial Surgery, Stomatological Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China
-
Revised:2017-08-07Online:2017-10-08Published:2017-11-10 -
Contact:Huang Xuan-ping, Master’s supervisor, Professor, Department of Oral and Maxillofacial Surgery, Stomatological Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
About author:Ma Cheng, Studying for master’s degree, Department of Oral and Maxillofacial Surgery, Stomatological Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:the Key Research & Development of Guangxi Zhuang Autonomous Region, No. 1598012-18; the Science and Technology Research of Universities in Guangxi Zhuang Autonomous Region, No. ZD2014035; the Medical and Health Appropriate Technology Development and Promotion Project of Guangxi Zhuang Autonomous Region, No. S201687
CLC Number:
Cite this article
Ma Cheng, Huang Xuan-ping. Application and progress of mandibular reconstruction[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(28): 4550-4555.
share this article
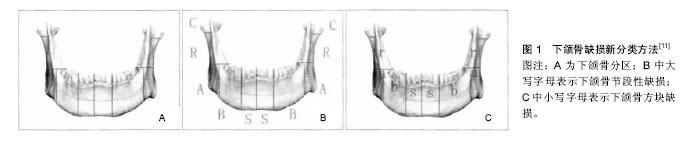
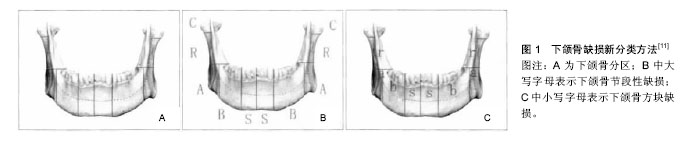
2.1 下颌骨缺损分类 下颌骨形态及功能的多样性决定了其缺损的复杂性,修复重建难度及预后存在差异性。合理的下颌骨缺损分类对下颌骨修复重建和临床治疗起到重要作用。其分类主要根据下颌骨的功能分区和咀嚼肌肉附着等进行划分。目前国内外对于下颌骨缺损的分类方法尚未统一[6],分类方法包括传统的David分类,Urken分类,蔡志刚分类等及新分类法(图1)[7-10]。Brown等[10]通过对下颌骨的功能区、肌肉附着、以及生物力学等因素进行考虑提出了一种新的分类方法,将下颌骨缺损分为8类。Ⅰ类:不超过下颌尖牙不包括髁突在内的一侧下颌骨缺损;Ⅰc类:不超过下颌尖牙包括髁突在内的一侧下颌骨缺损;Ⅱ类:超过下颌尖牙不包括髁突的一侧下颌骨缺损;IIc类:超过下颌尖牙包括髁突的一侧下颌骨缺损;Ⅲ类:包括双侧尖牙不涉及下颌角的下颌骨缺损;Ⅳ类:包括双侧尖牙涉及下颌角的下颌骨缺损;Ⅳc类:包括双侧尖牙设计髁突的下颌骨缺损。这种分类方法比以往分类更能全面地反映下颌骨缺损的复杂性,为下颌骨切除术后重建效果提供一个更科学和更有效的评估标准,对指导临床修复具有重要意义。"

2.2 下颌骨缺损重建的原则与时机 下颌骨缺损重建不仅需要恢复下颌骨的连续性与面部外形,还要恢复患者的咀嚼、吞咽、言语等基本生理功能,包括牙槽骨的高度、牙弓形态与宽度、周围咀嚼肌附着,以及感觉功能恢复和咬合功能重建等。成功植骨的前提主要包括[12]:①受区组织健康;②移植骨与宿主骨广泛接触;③术区血供良好;④移植骨固定持久可靠。对于下颌骨缺损修复重建的时机,尤其是对恶性肿瘤患者,传统观点认为应当延期修复,以便检测肿瘤复发。如何在保证肿瘤最大程度根治的前提下,充分利用整复手段恢复患者下颌骨的外形与功能是手术考虑的重要因素。随着颌面整复技术与显微外科技术的发展,特别是血管化游离组织瓣移植的广泛应用,外科手术不但能够保证肿瘤的广泛切除,而且还能恢复患者的面部外形与功能。与延期修复相比,即刻修复具有更多优点:①有利于保护重要组织与器官,减少术后并发症的发生;②有利于患者尽早解除心里障碍;③有利于早期恢复患者的面部外形与基本生理功能。因此,越来越多的学者认为即刻修复应当作为下颌骨的缺损重建的首选[13]。 2.3 下颌骨缺损重建的方法 目前下颌骨缺损重建的常用方法主要包括非血管化骨移植、带蒂骨肌皮瓣移植,成型钛板重建以及血管化游离骨皮瓣移植等。非血管化游离骨移植虽然手术过程简单,耗费时间少,但移植成活依赖于受植区的血供,易发生感染,导致愈合时间长,且不能修复软组织的缺损[14]。带蒂骨肌皮瓣可同时提供软组织修复肌肉、口腔黏膜和皮肤缺损,缺陷是在不影响自身血供的情况下难以塑形,易造成修复后的下颌骨外形欠佳[15]。游离腓骨瓣移植是目前应用最为广泛的修复方法[16-17]:游离腓骨瓣长度适宜,最长可达25 cm,骨量充足,血供来源于骨膜和骨内腓动脉,因此可对其进行多次截骨满足各种形状的三维立体塑形需求,且腓骨皮质骨较厚,更利于种植体的植入。血管直径为1.5-3.0 mm,为颈部血管的吻合提供了有利的条件。 近年来随着计算机技术与影像学的快速发展,快速成型技术、计算机辅助设计与制造(CAD/CAM)技术与骨移植、个体化钛支架植入相结合修复下颌骨缺损已经进入临床治疗阶段,且取得了理想的效果[18]。其优点是可以在术前模拟各种手术方案,进行数字化导板、重建钛板、钛螺钉固定位置的设计,优化修复体的生物力学,简化手术过程,缩短了手术时间[19-20]。快速成型技术在下颌骨缺损修复上的基本流程:首先利用MRI或螺旋CT对患者下颌骨进行扫描从而获得数字化数据,为保证仿真生物模型的精确度,目前选择的扫描层距为1.0 mm或者小于1.0 mm[21]。再利用逆向工程镜像还原技术模拟出患者缺损部位的下颌骨三维图像,然后以STL数据格式输出,控制快速成型机完成下颌骨仿真模型的制作,临床医生即可根据图像进行手术部位、重建钛板、固位和稳定等方面的设计,直至满意为止[22]。Foley等[23]在利用快速成型技术分别对8例下颌骨缺损患者进行修复的报告中,术前进行手术模拟,确定截骨范围,设计并制造了引导装装置,术后通过与术前模拟数据进行对比,分析发现利用快速成型技术制作手术导板修复下颌骨缺损是一种精确的修复方法。张新凤等[24]使用快速成型技术结合腓骨皮瓣修复80例下颌骨缺损的患者,术前在计算机软件上模拟病变切除及腓骨移植修复重建的手术操作,通过快速成型技术打印出修复后的实体模型及指导截骨和移植骨塑形的数字化导板,并根据模型进行重建钛板的预弯,术中根据术前制作的手术导板进行固位与塑形,术后患者均获得满意面容。将实际手术与模拟手术截除的下颌骨各个部位的长度进行对比,发现快速成型技术在血管化腓骨移植修复下颌骨缺损方面可获得较高的精度(表1)。"

| [1] Josep RP, Alejandra PG, Asteria AC, et al. Three-dimensional planning in craniomaxillofacial surgery. Ann Maxillofac Surg. 2016; 6(2): 281-286.[2] 丁姗姗,禹怡君,刘超,等.超顺磁性支架材料修复兔下颌骨缺损的实验研究[J].医学研究生学报,2017,30(3):251-256.[3] 罗婷苑,张翔,张森林.3D打印技术在髂骨移植修复下颌骨缺损中的应用研究[J].临床口腔医学杂志,2017,33(4):232-234.[4] 张治平,黄伟,黄雁翔.预构扩张耳前窄蒂皮瓣修复大面积面部软组织缺损[J].广东医学,2017,8(8):1236-1239.[5] 朱文赫, 钟秀宏, 张巍,等.鹿茸多肽-胶原蛋白/壳聚糖复合材料对家兔下颌骨缺损愈合的促进作用及其机制[J].吉林大学学报(医学版),2017,43(3):527-531.[6] 李静远,廖贵清.下颌骨缺损分类进展[J].中华口腔医学研究杂志(电子版),2017, 11(2):115-118.[7] David DJ, Tan E, Katsaros, et al. Mandibular reconstruction with vascularized iliac crest:a 10-year experience. Plast Reconstr Surg.1988;82(5):792-803.[8] Urken ML, Weinberg H, Vickery C, et al. Oromandibular reconstruction using microvascular composite free flaps.Arch Otolaryngol Head Neck Surg.1991; 17(7):733-744.[9] 蔡志刚,张杰,张建国,等.下颌骨缺损的修复与重建[J].中国耳鼻咽喉头颈外科,2004,11(5);285-287.[10] Brown JS, Barry C, Ho M, et al. A new classification for mandibular defects after oncological resection. Lancet Oncology. 2016;17(1):e23-e30.[11] 廖贵清,苏宇雄.下颌骨缺损分类[J].广东牙病防治,2008,16(2): 57-59.[12] Shnayder Y, Lin D, Desai SC, et al. Reconstruction of the lateral mandibular defect: a review and treatment algorithm. JAMA Facial Plast Surg. 2015;17(5):367-373.[13] Brown JS, Lowe D, Kanatas A, et al. Mandibular reconstruction with vascularised bone flaps: a systematic review over 25 years. Microsurgery.2017;55(2):113-126.[14] 谢富强,孙健.非血管化髂骨移植重建下颌骨缺损的临床分析[J].华西口腔医学杂志,2012,30(4):411-413.[15] 王玉龙,韩龙,毛驰.下颌骨缺损的修复重建[J].口腔颌面修复学杂志,2011, 12(2):126-128.[16] Kim JE, Broyles JM, Sacks JM. Mandible reconstruction. Eplasty. 2013; 13(2):ic19.[17] Ann K, Konstantine M, George L, et al. Reconstruction of mandibular defects with autogenous bone and decellularized bovine bone grafts with freeze-dried bone marrow stem cell paracrine factors. Oncol Lett. 2017;13(3):1811-1818.[18] Ciocca L, Mazzoni S, Fantini M, et al. CAD/CAM guided secondary mandibular reconstruction of a discontinuity defect after ablative cancer surgery. J Craniomaxillofac Surg. 2012; 40(8):e511-515.[19] Kumta S, Kumta M, Jain L, et al. A novel 3D template for mandible and maxilla reconstruction: rapid prototyping using stereolithography. Indian J Plast Surg. 2015; 48(3):263-273.[20] Azuma M, Yanagawa T, Ishibashi-Kanno N, et al. Mandibular reconstruction using plates prebent to fit rapid prototyping 3-dimensional printing models ameliorates contour deformity. Head Face Med. 2014;10:45.[21] L Chan HH, Siewerdsen JH, Vescan A, et al. 3D rapid prototyping for otolaryngology-head and neck surgery: applications in image-guidance, surgical simulation and patient-specific modeling. Plos One. 2015; 10(9):e0136370.[22] Rodby KA, Turin S, Jacobs RJ, et al. Advances in oncologic head and neck reconstruction: systematic review and future considerations of virtual surgical planning and computer aided design/computer aided modeling. J Plast Reconstr Aesthet Surg. 2014; 67(9):1171-1185.[23] Foley BD, Thayer WP, Honeybrook A, et al. Mandibular reconstruction using computer-aided design and computer-aided manufacturing: an analysis of surgical results. J Oral Maxillofac Surg. 2013;71(2):e111.[24] 张新凤,董智伟,鲍海宏,等.3D打印技术在提高80例应用血管化腓骨移植修复下颌骨缺损精确度方面的临床探讨[J].中国现代医学杂志,2016,26(10):51-55.[25] Wong R, Tideman H, Merkx M. The modular endoprosthesis for mandibular body replacement: finite element analysis of endoprosthesis reconstruction of the mandible. J Craniomaxillofac Surg. 2012;40(8):487-497.[26] 贾暮云,蒋济金,初晓艺,等.下颌骨缺损重建板修复术后十年回顾性分析[J].中华口腔医学杂志,2016,51(7):401-404.[27] Liu YF, Xu LW, Zhu HY, et al. Technical procedures for template-guided surgery for mandibular reconstruction based on digital design and manufacturing. BioMedical Engineering OnLine. 2014;13(1):63. [28] Tepper OM, Sorice S, Hershman GN, et al. Use of virtual 3-dimensional surgery in post-traumatic craniomaxillofacial reconstruction. J Maxillofac Oral Surg. 2011; 69(3):733-741.[29] Jacobsen C, Kruse A, Lübbers HT, et al. Is mandibular reconstruction using vascularized fibula flaps and dental implants a reasonable treatment? Clin Implant Dent Relat Res. 2014;16(3):419-428.[30] Chambrone L, Mandia J, Shibli JA, et al. Dental implants installed in irradiated jaws: a systematic review. J Dent Res. 2013;92(12 Suppl):119S-130S.[31] Gbara A, Darwich K, Li L, et al. Long-term results of jaw reconstruction with microsurgical fibula grafts and dental implants. J Maxillofac Oral Surg. 2007; 65(5):1005.[32] Boyd TG, Huber KM, Verbist DE, et al. Removal of exposed titanium reconstruction plate after mandibular reconstruction with a free fibula osteocutaneous flap with large surgical pin cutters: a case report and literature review. Eplasty. 2012; 12:e42.[33] Cotic J, Jamsek J, Kuhar M, et al. Implant-prosthetic rehabilitation after radiation treatment in head and neck cancer patients: a case-series report of outcome. Radiol Oncol. 2017;51(1):94-100.[34] Bartaire E, Mouawad F, Mallet Y, et al. Morphologic assessment of mandibular reconstruction by free fibula flap and donor-site functional impairment in a series of 23 patients. Ann Surg Oncol.2012;129(5):230-237.[35] Sakakibara A, Hashikawa K, Yokoo S, et al. Risk factors and surgical refinements of postresective mandibular reconstruction: a retrospective study. Plast Surg Int. 2014; 2014: 893746.[36] Markey J, Knott PD, Fritz MA, et al. Recent advances in head and neck free tissue transfer. Curr Opin Otolaryngol Head Neck Surg. 2015;23(4):297.[37] 张斌.口腔鳞状细胞癌的表观遗传学研究现状和进展[J].口腔颌面外科杂志,2016, 26(2):77-82.[38] Rao LP, Shukla M, Sharma V, et al. Mandibular conservation in oral cancer. Surg Oncol. 2012;21(2):109. [39] Saracoglu B, Atay A, Korkmaz C, et al. Quality of life assessment of implant-retained overdentures and fixed metal-acrylic resin prostheses in patients with marginal mandibulectomy. J Prosthet Dent. 2017.[40] 邱宇,林李嵩,施斌,等.2种下颌骨切除方式对口腔鳞癌预后的影响[J].中国口腔颌面外科杂志,2016,14(2):149-153.[41] Zhou L, Wang P, Han H, et al. Prototyped grafting plate for reconstruction of mandibular defects. J Craniomaxillofac Surg. 2014;42(8):1723-1729.[42] 吴纪楠,龚振宇,陈觉尧,等.基于快速成型技术制作个体化下颌人工骨的临床应用[J].中华口腔医学研究杂志(电子版), 2011,5(2): 31-34.[43] Yagihara K, Okabe S, Ishii J, et al. Mandibular reconstruction using a poly(L-lactide) mesh combined with autogenous particulate cancellous bone and marrow: a prospective clinical study. Int J Oral Maxillofac Surg. 2013;42(8):962-969.[44] Chenard KE, Teven CM, He TC, et al. Bone morphogenetic proteins in craniofacial surgery: current techniques, clinical experiences, and the future of personalized stem cell therapy. Biomed Res Int. 2012;2012(8):601549.[45] Hao ZC, Song ZH, Huang J, et al. Scaffold microenvironment for stem cell based bone tissue engineering. Biomater Sci. 2017;5:1382-1392. [46] Matsuo A, Chiba H, Takahashi H, et al. Clinical application of a custom-made bioresorbable raw particulate hydroxyapatite/poly-L-lactide mesh tray for mandibular reconstruction. Odontology. 2010;98(1):85.[47] Lee MK, DeConde AS, Lee M, et al. Biomimetic scaffolds facilitate healing of critical-sized segmental mandibular defects. Am J Otolaryngol. 2015;36(1):1-6.[48] Sándor GK, Tuovinen VJ, Wolff J, et al. Adipose stem cell tissue-engineered construct used to treat large anterior mandibular defect: a case report and review of the clinical application of good manufacturing practice-level adipose stem cells for bone regeneration. Br J Oral Maxillofac Surg. 2013;71(5):938-950. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [3] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [4] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [5] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [6] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [7] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [8] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [9] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [10] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [11] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [12] | Guan Jian, Jia Yanfei, Zhang Baoxin , Zhao Guozhong. Application of 4D bioprinting in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 446-455. |
| [13] | Zeng Yuwei, Huang Chuang, Wei Jianguo, Duan Dongming, Wang Le. Tracing transplanted bone marrow mesenchymal stem cells in rat calvarial defect by bioluminescence imaging [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 3968-3973. |
| [14] | He Qi, Xu Faya, Li Xiyan, Han Lei, Xiao Yanbing, Tu Jiao. Preparation and sterilization method of human acellular amniotic membrane scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4028-4033. |
| [15] | Wang Kang, Zhi Xiaodong, Wang Wei. Effect and mechanism of human amniotic epithelial cells on nerve injury repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4046-4051. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||