Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (26): 4240-4245.doi: 10.3969/j.issn.2095-4344.2017.26.024
Previous Articles Next Articles
Effects of shear stress on cytoskeleton alignment, adhesion, migration and apoptosis of endothelial cells on micropatterned substrates
- Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing 100191, China
-
Received:2017-04-22Online:2017-09-18Published:2017-09-28 -
Contact:Gong Xiang-hui, M.D., Master’s supervisor, Lecturer, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing 100191, China -
About author:He Hong-ping, Studying for master’s degree, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing 100191, China -
Supported by:the National Natural Science Foundation of China, No. 11172031
CLC Number:
Cite this article
He Hong-ping, Zhao Xi-xi, Li Bin, Gong Xiang-hui.
share this article
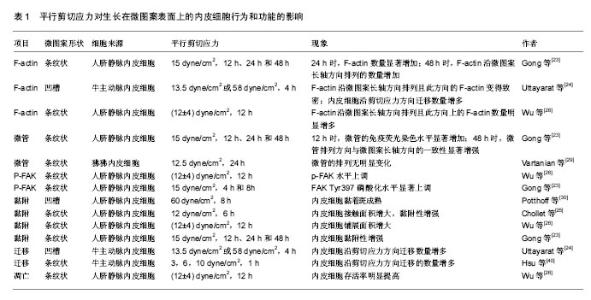
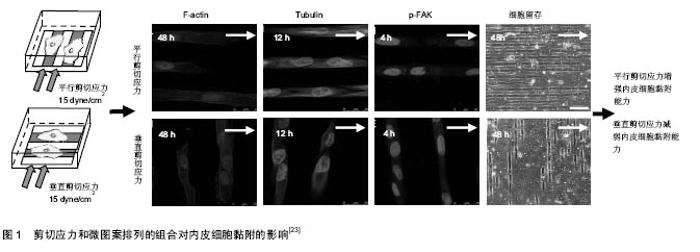
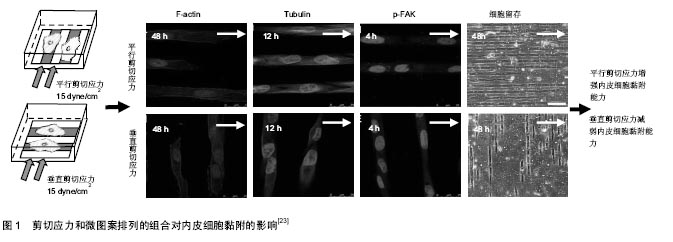
2.1 剪切应力方向对生长在微图案表面内皮细胞骨架排列的影响 细胞骨架主要由微丝、微管和中间纤维构成。微图案和剪切力对细胞骨架的影响的研究主要集中在微丝和微管方面。大量研究发现,剪切应力方向影响生长在微图案表面上的内皮细胞F-actin的排列(表1)。平行于微图案长轴的剪切应力促进内皮细胞的F-actin沿微图案长轴方向排列,进而促使内皮细胞沿微图案长轴方向排列。例如,Gong等[23]对生长在微图案表面上的内皮细胞施加平行剪切应力(15 dyne/cm2),发现在加载剪切应力24 h时F-actin应力纤维的数量显著增加;在加载剪切应力48 h时,F-actin沿微图案长轴方向排列的数量增加(图1)。Uttayarat等[24]发现对生长在微沟槽表面上的内皮细胞施加平行剪切应力(13.5 dyne/cm2或58 dyne/cm2)4 h后,F-actin沿微图案长轴方向排列且此方向的F-actin变得致密,内皮细胞沿剪切应力方向铺展。Chollet等[25]对生长在微图案表面上的内皮细胞施加平行剪切应力(12 dyne/cm2)6 h后,发现F-actin的水平及平均长度显著增加,并发现内皮细胞沿微图案长轴方向排列的数量增多。Wu等[26]对生长在微图案表面上的内皮细胞施加平行剪切应力[(12±4) dyne/cm2]12 h后,发现F-actin沿微图案长轴方向排列,并且此方向上的F-actin数量明显增多。Robotti等[27]对生长在微图案表面上内皮细胞施加平行剪切应力(14 dyne/cm2)16 h后,发现平行剪切应力促进内皮细胞沿微图案长轴方向的排列。Morgan等[28]对生长在微纳米尺寸的圆孔表面上内皮细胞施加平行剪切应力(20 dyne/cm2)24 h后,发现内皮细胞沿微图案长轴方向排列。"

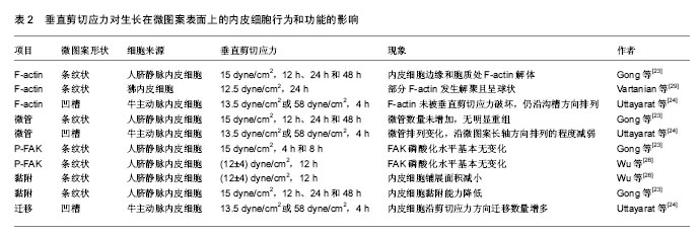
垂直于微图案长轴的剪切应力对内皮细胞F-actin的影响有不同的报道(表2)。Gong等[23]对生长在微图案表面上的内皮细胞施加垂直剪切应力(15 dyne/cm2) 24,48 h后,发现分布于内皮细胞边缘和细胞质处的F-actin纤维解体。剪切应力加载时间越长,F-actin的排列方向与微图案长轴方向的一致程度越弱(图1)。与Gong的结果类似,Vartanian等[29]对生长在微图案表面上的内皮细胞施加垂直于微图案长轴的12.5 dyne/cm2剪切应力24 h后,发现部分F-actin发生解聚且呈球状。但是Uttayarat等[24]对生长在微沟槽表面上的内皮细胞施加垂直剪切应力(13.5 dyne/cm2或58 dyne/cm2)4 h后,发现F-actin未被垂直剪切应力破坏,仍沿沟槽方向排列。 剪切应力作用对不同微图案上内皮细胞微管排列的影响研究较少。最近Gong等[23]研究发现剪切力方向影响微管的表达与排列:对生长在微图案表面上的内皮细胞分别施加平行剪切应力(15 dyne/cm2)12 h时,微管的免疫荧光染色水平显著增加,加载剪切应力48 h后,微管排列方向与微图案长轴方向的一致性显著增强;对内皮细胞加载垂直剪切应力后,微管数量未增加,未观察到明显的重组现象。Vartanian等[29]的研究结果与Gong等的结果不同:对生长在微图案表面上的内皮细胞施加 12.5 dyne/cm2平行剪切应力24 h后,发现微管的排列无明显变化;对内皮细胞加载垂直剪切应力后,微管发生排列发生变化,微管沿微图案长轴方向排列的程度减弱。尽管上述结果存在不一致之处,但仍可说明生物材料表面的拓扑结构与剪切应力方向共同影响微管的结构和排列。 2.2 剪切应力方向对生长在微图案表面上内皮细胞黏附的影响 研究发现剪切应力方向影响生长在微图案表面上内皮细胞的黏附能力。平行剪切应力增强内皮细胞的黏附能力。例如,Potthoff等[30]对生长在槽深0.1,0.4 μm微沟槽表面上的内皮细胞施加60 dyne/cm2平行剪切应力18 h后,发现平行剪切应力促进内皮细胞的黏着斑成熟,这一过程有利于内皮细胞的稳定黏附。Chollet等[25]对生长在微图案表面上的内皮细胞施加12 dyne/cm2平行剪切应力6 h后,发现内皮细胞的接触面积增大,黏附性增强。Wu等[26]对生长在微图案表面上的内皮细胞施加(12±4) dyne/cm2平行剪切应力12 h后,发现磷酸化黏着斑激酶(phosphorylated focal adhesion kinase,p-FAK)水平上调,内皮细胞的铺展面积增大。McCracken等[31]对生长在纳米孔阵列表面上的内皮细胞分别施加(1.7±0.3) dyne/cm2平行剪切应力和垂直剪切应力24 h后,发现平行剪切应力作用下的内皮细胞密度明显高于垂直剪切应力作用下的。Gong等[23]对生长在微图案上内皮细胞分别施加15 dyne/cm2平行剪切应力和垂直剪切应力48 h后,发现平行剪切应力作用下的内皮细胞存留量(73.92±10.61)%显著高于垂直剪切应力作用下的内皮细胞存留量(26.53±4.03)%。Wu等[26]对生长在微图案表面上的内皮细胞施加(12±4) dyne/cm2垂直剪切应力12 h后,发现内皮细胞铺展面积减小,最终导致内皮细胞不能耐受剪切应力而脱落。上述研究结果表明,生物材料表面的拓扑结构和剪切力方向的不同组合影响内皮细胞的黏附,这提示在血管材料设计时应充分考虑将微图案的方向与血流方向恰当组合,以增强内皮细胞的抗剪切能力。 细胞的黏附主要由黏着斑介导[32],黏着斑连接细胞外基质和细胞骨架蛋白actin[33],形成的跨膜结构参与调控细胞黏附和力信号的感知与传递。FAK是黏着斑的主要成分,FAK的磷酸化在调控细胞行为和功能中扮演着重要角色[34]。研究发现,体外培养的内皮细胞在受到剪切应力刺激后FAK在Tyr397位点的磷酸化水平升高[35],并激活了下游的Rho GTPase,Rho GTPase在调控细胞骨架重构、细胞黏附和细胞迁移中起关键作用[36-39]。近期的研究发现剪切应力与微图案的不同组合影响内皮细胞信号通路中FAK在Tyr397位点的磷酸化水平[23,26]。平行剪切应力上调生长在微图案表面上的内皮细胞FAK磷酸化水平,垂直剪切应力对生长在微图案表面上的内皮细胞FAK磷酸化无显著影响[23,26]。例如,Wu等[26]对生长在微图案表面上的内皮细胞施加分别施加(12±4) dyne/cm2平行剪切应力和垂直剪切应力12 h后,发现平行剪切应力上调FAK Tyr397的磷酸化水平;而垂直剪切应力作用下内皮细胞FAK Tyr397的磷酸化水平基本无变化。Gong等[23]对生长在微图案表面上的内皮细胞分别施加15 dyne/cm2平行剪切应力和垂直10 dyne/cm2)作用1 h后,发现内皮细胞沿剪切应力方向迁移的数量增多。 剪切应力大小对生长在微图案表面上内皮细胞的迁移也有影响。Uttayarat等[24]对生长在微沟槽表面上内皮细胞施加13.5,58 dyne/cm2剪切应力作用4 h后,结果显示58 dyne/cm2剪切应力作用下内皮细胞沿剪切应力方向迁移的数量显著高于13.5 dyne/cm2剪切应力下的迁移数量。Hsu等[40]对生长在微图案表面上的内皮细胞施加3 dyne/cm2垂直剪切应力作用1 h后,仅有29%内皮细胞沿剪切应力方向迁移;施加10 dyne/cm2垂直剪切应力作用1 h后,71%内皮细胞沿剪切应力方向迁移。 2.4 剪切应力方向对生长在微图案表面上内皮细胞凋亡的影响 细胞凋亡是一种受基因调控的主动的生理性细胞自杀行为,对内环境的稳定有着重要的作用[41]。内皮细胞凋亡的过程中往往伴随着FAK信号中断、黏着斑复合物的解体、F-actin的消失及actin重组成外周环等现象[42],AKT、eNOS、NF-kB等基因参与调控内皮细胞的凋亡[11]。目前已有研究发现剪切应力对生长在微图案表面上内皮细胞的凋亡有影响。例如,Wu等[26]发现对生长在微图案表面上内皮细胞施加(12±4) dyne/cm2平行剪切应力12 h后,内皮细胞在微图案上的铺展面积增大,信号通路中p-FAK水平上调,F-actin数量增加,内皮细胞存活率明显提高;而对生长在微图案表面上内皮细胞施加(12±4) dyne/cm2垂直剪切应力12 h后,未发现上述影响。Vartanian等[29]对生长在微图案表面上的内皮细胞施加12.5 dyne/cm2垂直剪切应力24 h后,发现部分F-actin解聚并呈球状,而球状actin是细胞凋亡的标志。通过分析不同角度的剪切应力对定向排列的内皮细胞产生的影响,Wang等[11]发现剪切应力的方向影响内皮细胞的抗动脉粥样硬化eNOS和AKT的磷酸化水平,当剪切应力方向与定向排列的内皮细胞长轴呈180°时,剪切应力对eNOS与AKT激活作用最强;而当剪切应力方向与定向排列的内皮细胞长轴呈90°时,剪切应力不影响eNOS与AKT的激活。剪切应力的方向也影响内皮细胞NF-κB的活性,当剪切应力方向与定向排列的内皮细胞长轴呈180°时,剪切应力对NF-κB的激活无影响;而当剪切应力方向与定向排列的内皮细胞长轴呈90°时,剪切应力对NF-κB激活作用最强。上述结果表明,剪切应力的方向对生长在微图案表面上内皮细胞的功能具有重要调控作用。 "

| [1]Writing Group Members,Mozaffarian D,Benjamin EJ,et al.Heart Disease and Stroke Statistics-2016 Update: A Report From the American Heart Association.Circulation.2016; 133(4):e38-360.[2]郭翔.表面微米拓扑结构的构建及对血小板和内皮细胞的影响[D].西南交通大学,2012.[3]Zilla P, Bezuidenhout D, Human P. Prosthetic vascular grafts: wrong models, wrong questions and no healing. Biomaterials, 2007;28(34): 5009-5027[4]Wang Y,Chen S,Pan Y,et al.Rapid in situ endothelialization of a small diameter vascular graft with catalytic nitric oxide generation and promoted endothelial cell adhesion.J Mater Chem B.2015;3(47): 9212-9222.[5]Chong DS,Turner LA,Gadegaard N,et al.Nanotopography and plasma treatment: redesigning the surface for vascular graft endothelialisation.Eur J Vasc Endovasc Surg. 2015; 49(3):335-343.[6]Bergmeister H,Strobl M,Grasl C,et al.Tissue engineering of vascular grafts.Eur Surg.2013;45(4): 187-193.[7]Meltem Aa GZ,Hans PW.Induction of EPC homing on biofunctionalized vascular grafts for rapid in vivo self-endothelialization — A review of current strategies Biotechnol Adv.2010;28(1):119-129.[8]Melchiorri AJ,Hibino N,Yi T,et al.Contrasting biofunctionalization strategies for the enhanced endothelialization of biodegradable vascular grafts. Biomacromolecules.2015;16(2):437-446.[9]Otsuka F,Finn AV,Yazdani SK,et al.The importance of the endothelium in atherothrombosis and coronary stenting.Nat Rev Cardiol.2012;9(8):439-453.[10]Ranjan A, Webster TJ. Increased endothelial cell adhesion and elongation on micron-patterned nano-rough poly (dimethylsiloxane) films. Nanotechnology.2009;20(30): 305102.[11]Gong X, Liu H, Ding X, et al. Physiological pulsatile flow culture conditions to generate functional endothelium on a sulfated silk fibroin nanofibrous scaffold. Biomaterials.2014; 35(17): 4782-4791.[12]Yazdani SK, Tillman BJ. The fate of an endothelium layer after preconditioning. J Vasc Surg.2010;51(1): 74-183.[13]Ohta S,Inasawa S,Yamaguchi Y.Alignment of vascular endothelial cells as a collective response to shear flow J Phys D Appl Phys.2015;48(24):245401.[14]Steward R Jr.,Tambe D,Hardin CC,et al.Fluid shear, intercellular stress, and endothelial cell alignment.Am J Physiol Cell Physiol.2015;308(8):C657-664.[15]Wang C,Baker BM,Chen CS,et al.Endothelial cell sensing of flow direction.Arterioscler Thromb Vasc Biol.2013;33(9): 2130-2136..[16]Li Y,Huang G,Zhang X,et al.Engineering cell alignment in vitro.Biotechnol Adv.2014;32(2): 347-365.[17]Dickinson LE,Rand DR,Tsao J,et al.Endothelial cell responses to micropillar substrates of varying dimensions and stiffness.J Biomed Mater Res A.2012;100A(6):1457-1466.[18]Huang NF,Lai ES,Ribeiro AJ,et al.Spatial patterning of endothelium modulates cell morphology, adhesiveness and transcriptional signature.Acta Biomater.2013;34(12):2928.[19]Boivin MC,Chevallier P,Hoesli CA,et al.Human saphenous vein endothelial cell adhesion and expansion on micropatterned polytetrafluoroethylene.J Biomed Mater Res A.2013;101(3):694-703.[20]Lin X,Helmke BP.Cell structure controls endothelial cell migration under fluid shear stress. Cell Mole Bioeng.2009; 2(2):231-243.[21]Lu J,Rao MP,Macdonald NC,et al.Improved endothelial cell adhesion and proliferation on patterned titanium surfaces with rationally designed, micrometer to nanometer features.Acta Biomater.2008;4(1): 192-201.[22]Liliensiek SJ,Wood JA,Yong J,et al.Modulation of human vascular endothelial cell behaviors by nanotopographic cues.Biomaterials.2010;31(20):5418-5426.[23]Gong X,Yao J,He H,et al.Combination of flow and micropattern alignment affecting flow-resistant endothelial cell adhesion.J Mech Behav Biomed.2017;74:11-20. [24]Uttayarat P,Chen M, Li M,et al.Microtopography and flow modulate the direction of endothelial cell migration.Am J Physiol-haert C.2008;294(2):H1027-H1035.[25]Chollet C,Bareille R,Remy M,et al.Impact of peptide micropatterning on endothelial cell actin remodeling for cell alignment under shear stress.Macromol Biosci. 2012;12(12): 1648-1659.[26]Wu CC,Li YS,Haga JH,et al.Directional shear flow and Rho activation prevent the endothelial cell apoptosis induced by micropatterned anisotropic geometry.Proc Natl Acad Sci U S A.2007;104(4): 1254-1259.[27]Robotti F,Franco D,Banninger L,et al.The influence of surface micro-structure on endothelialization under supraphysiological wall shear stress. Biomaterials, 2014, 35(30): 8479-8486.[28]Morgan JT,Wood JA,Shah NM,et al.Integration of basal topographic cues and apical shear stress in vascular endothelial cells.Biomaterials.2012;33(16):4126-4135.[29]Vartanian KB,Kirkpatrick SJ,Hanson SR,et al.Endothelial cell cytoskeletal alignment independent of fluid shear stress on micropatterned surfaces.Biochem Biophys Res Commun. 2008;371(4):787-792.[30]Potthoff E,Franco D,D'alessandro V,et al.Toward a rational design of surface textures promoting endothelialization.Nano Lett.2014;14(2):1069-1079.[31]Mccracken KE,Tran PL,You DJ,et al.Shear- vs. nanotopography-guided control of growth of endothelial cells on RGD-nanoparticle-nanowell arrays.J Biol Eng. 2013;7(1): 11.[32]Spatz JP,Geiger B.Molecular engineering of cellular environments: cell adhesion to nano-digital surfaces.Methods Cell Biol.2007;83(4):89-111.[33]张冠华,智发朝.黏着斑的结构、功能及在肿瘤转移中作用[J].现代消化及介入诊疗,2015,20(2):174-177.[34]Li Z,Lee H,Zhu C.Molecular mechanisms of mechanotransduction in integrin-mediated cell-matrix adhesion. Exp Cell Res.2016;349(1):85-94.[35]Li S,Butler P,Wang Y,et al.The role of the dynamics of focal adhesion kinase in the mechanotaxis of endothelial cells.Proc Natl Acad Sci U S A.2002;99(6):3546-3551.[36]Zeng L,Si X,Yu WP,et al.PTPαregulates integrin-stimulated FAK autophosphorylation and cytoskeletal rearrangement in cell spreading and migration.J Cell Biol.2003;160(1):137.[37]Mitra SK,Hanson DA,Schlaepfer DD.Focal adhesion kinase: in command and control of cell motility. Nat Rev Mol Cell Bio.2005;6(1):56-68.[38]Etiennemanneville S,Hall A.Rho GTPases in cell biology. Nature.2002;420(420):629-635.[39]Goldyn AM,Rioja BA,Spatz JP,et al.Force-induced cell polarisation is linked to RhoA-driven microtubule-independent focal-adhesion sliding.J Cell Sci.2009;122(20):3644-3651.[40]Hsu S,Thakar R,Liepmann D,et al.Effects of shear stress on endothelial cell haptotaxis on micropatterned surfaces. Biochem Biophys Res Commun.2005;337(1):401-409.[41]翟中和,王喜忠,丁明孝.细胞生物学[M].3版.北京:高等教育出版社,2007:442-443.[42]高清,吴波.细胞凋亡与细胞骨架的改变[J].医学研究生学报, 2008,21(8):877-880. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [3] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [4] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1045-1050. |
| [5] | Zhang Yujie, Yang Jiandong, Cai Jun, Zhu Shoulei, Tian Yuan. Mechanism by which allicin inhibits proliferation and promotes apoptosis of rat vascular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1080-1084. |
| [6] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [7] | Chen Shuo, Xiao Dongqin, Li Xingping, Ran Bin, Shi Feng, Zhang Chengdong, Deng Li, Huang Nanxiang, Liu Kang, Feng Gang, Duan Ke. Preparation and characterization of tantalum functional coating on titanium implant [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 546-552. |
| [8] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [9] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [10] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [11] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [12] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [13] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [14] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [15] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||