Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (24): 3833-3838.doi: 10.3969/j.issn.2095-4344.2017.24.011
Previous Articles Next Articles
Effect of modified rabbit defensin 1 on peripheral nerve regeneration
Xu Chun-gui, Zhang Ji-sen, Xu Xin-zhong, Jing Jue-hua
- the Second Hospital of Anhui Medical University, Hefei 230041, Anhui Province, China
-
Revised:2017-07-01Online:2017-08-28Published:2017-08-30 -
Contact:Jing Jue-hua, M.D., Chief physician, Associate professor, Master’s supervisor, the Second Hospital of Anhui Medical University, Hefei 230041, Anhui Province, China -
About author:Xu Chun-gui, M.D., the Second Hospital of Anhui Medical University, Hefei 230041, Anhui Province, China -
Supported by:the Natural Science Foundation of Anhui Province, No. 1608085QH206; the Research Foundation of Anhui Medical University, No. 2015xkj017
CLC Number:
Cite this article
Xu Chun-gui, Zhang Ji-sen, Xu Xin-zhong, Jing Jue-hua . Effect of modified rabbit defensin 1 on peripheral nerve regeneration[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(24): 3833-3838.
share this article

2.1 实验动物数量分析 所有大鼠均进入结果分析,无脱失。 2.2 坐骨神经指数 神经营养因子组和改良兔防御素1组大鼠的坐骨神经指数大于生理盐水组(P < 0.05),但2组间差异无显著性意义(表1)。 2.3 神经传导速度 神经营养因子组和改良兔防御素1组大鼠的坐骨神经传导速度大于生理盐水组和改良兔防御素1组(P < 0.05;表1)。 2.4 有髓神经纤维数量及形态 神经营养因子组再生有髓神经纤维数较改良兔防御素1组多,而改良兔防御素1组有髓神经纤维数较生理盐水组多(P < 0.05)。对于有髓神经纤维直径以及髓鞘厚度,神经营养因子组>改良兔防御素1组>生理盐水组。而对于轴突直径以及G-ratio,神经营养因子组和改良兔防御素1组均大于生理盐水组(P < 0.05),而神经营养因子组和改良兔防御素1组轴突直径以及G-ratio差异无显著性意义(P > 0.05;表1,图5)。"
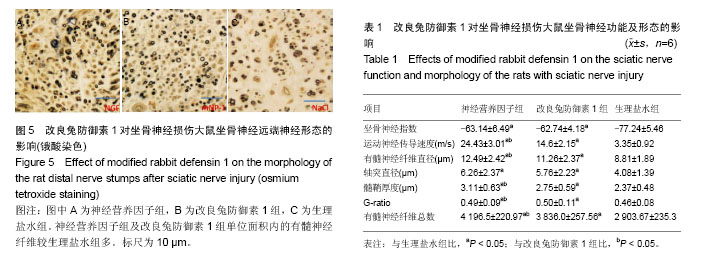
| [1] Guttmann E, Guttmann L, Medawar PB, et al. The rate of regeneration of nerve. Br Med Bull. 1943;19(7):14-44.[2] Al-Majed AA, Brushart TM, Gordon T. Electrical stimulation accelerates and increases expression of BDNF and trkB mRNA in regenerating rat femoral motoneurons. Eur J Neurosci. 2000;12(12):4381-4390.[3] Sulaiman OA, Gordon T. Role of chronic Schwann cell denervation in poor functional recovery after nerve injuries and experimental strategies to combat it. Neurosurgery. 2009;65(4 Suppl):A105-114.[4] 徐建广,顾玉东,李继峰.大鼠失神经支配骨骼肌及其运动终板退变观察[J].中华显微外科杂志,1999,22(3):215.[5] Adams L, Carlson BM, Henderson L, et al. Adaptation of nicotinic acetylcholine receptor, myogenin, and MRF4 gene expression to long-term muscle denervation. J Cell Biol. 1995;131(5):1341-1349.[6] Yang D, Biragyn A, Kwak LW, et al. Mammalian defensins in immunity: more than just microbicidal. Trends Immunol. 2002;23(6):291-296.[7] Verbanac D, Zanetti M, Romeo D. Chemotactic and protease-inhibiting activities of antibiotic peptide precursors. FEBS Lett. 1993;317(3):255-258.[8] Duits LA, Ravensbergen B, Rademaker M, et al. Expression of beta-defensin 1 and 2 mRNA by human monocytes, macrophages and dendritic cells. Immunology. 2002;106(4): 517-525.[9] Nozdrachev AD, Kolosova LI, Moiseeva AB, et al. The role of defensin NP-1 in restoring the functions of an injured nerve trunk. Neurosci Behav Physiol. 2006;36(3):313-315.[10] Lindsay RM. Nerve growth factors (NGF, BDNF) enhance axonal regeneration but are not required for survival of adult sensory neurons. J Neurosci. 1988;8(7):2394-2405.[11] Zhang P, Xue F, Kou Y, et al. The experimental study of absorbable chitin conduit for bridging peripheral nerve defect with nerve fasciculu in rats. Artif Cells Blood Substit Immobil Biotechnol. 2008;36(4):360-371. [12] Bhisitkul RB, Villa JE, Kocsis JD. Axonal GABA receptors are selectively present on normal and regenerated sensory fibers in rat peripheral nerve. Exp Brain Res. 1987;66(3):659-663.[13] Coronas V, Durand M, Chabot JG, et al. Acetylcholine induces neuritic outgrowth in rat primary olfactory bulb cultures. Neuroscience. 2000;98(2):213-219.[14] Udina E, Ceballos D, Gold BG, et al. FK506 enhances reinnervation by regeneration and by collateral sprouting of peripheral nerve fibers. Exp Neurol. 2003;183(1):220-231.[15] Sulaiman OA, Voda J, Gold BG, et al. FK506 increases peripheral nerve regeneration after chronic axotomy but not after chronic schwann cell denervation. Exp Neurol. 2002; 175(1):127-137.[16] Bai LL, Yin WB, Chen YH, et al. A new strategy to produce a defensin: stable production of mutated NP-1 in nitrate reductase-deficient Chlorella ellipsoidea. PLoS One. 2013; 8(1):e54966.[17] Ganz T, Selsted ME, Szklarek D, et al. Defensins. Natural peptide antibiotics of human neutrophils. J Clin Invest. 1985; 76(4):1427-1435.[18] García JR, Jaumann F, Schulz S, et al. Identification of a novel, multifunctional beta-defensin (human beta-defensin 3) with specific antimicrobial activity. Its interaction with plasma membranes of Xenopus oocytes and the induction of macrophage chemoattraction. Cell Tissue Res. 2001;306(2): 257-264.[19] García JR, Krause A, Schulz S, et al. Human beta-defensin 4: a novel inducible peptide with a specific salt-sensitive spectrum of antimicrobial activity. FASEB J. 2001;15(10):1819-1821.[20] McKerracher L, David S, Jackson DL, et al. Identification of myelin-associated glycoprotein as a major myelin-derived inhibitor of neurite growth. Neuron. 1994;13(4):805-811.[21] Fernandez-Valle C, Bunge RP, Bunge MB. Schwann cells degrade myelin and proliferate in the absence of macrophages: evidence from in vitro studies of Wallerian degeneration. J Neurocytol. 1995;24(9):667-679.[22] Rotshenker S. Microglia and macrophage activation and the regulation of complement-receptor-3 (CR3/MAC-1)-mediated myelin phagocytosis in injury and disease. J Mol Neurosci. 2003;21(1):65-72.[23] Chaly YV, Paleolog EM, Kolesnikova TS, et al. Neutrophil alpha-defensin human neutrophil peptide modulates cytokine production in human monocytes and adhesion molecule expression in endothelial cells. Eur Cytokine Netw. 2000; 11(2):257-266.[24] Kota S, Sabbah A, Chang TH, et al. Role of human beta-defensin-2 during tumor necrosis factor-alpha/NF-kappaB-mediated innate antiviral response against human respiratory syncytial virus. J Biol Chem. 2008;283(33):22417-22429. [25] Misuno NI, Kolesnikova TS, Lerer RI, et al. Effects of human defensin HNP-1 on the production of tumor necrosis factor-alpha by human blood monocytes in vitro. Biull Eksp Biol Med. 1992;113(5):524-527. [26] Leeyaphan C, Hau C, Takeoka S, et al. Immune response in human chromoblastomycosis and eumycetoma - focusing on human interleukin-17A, interferon-gamma, tumour necrosis factor-alpha, interleukin-1 beta and human beta-defensin-2. Mycoses. 2016. in press. [27] Huang FC. The differential effects of 1,25-dihydroxyvitamin D3 on Salmonella-induced interleukin-8 and human beta-defensin-2 in intestinal epithelial cells. Clin Exp Immunol. 2016;185(1): 98-106. [28] Tokunaga H, Saito S, Sakai K, et al. Halophilic beta-lactamase as a new solubility- and folding-enhancing tag protein: production of native human interleukin 1alpha and human neutrophil alpha-defensin. Appl Microbiol Biotechnol. 2010; 86(2):649-658.[29] Décanis N, Savignac K, Rouabhia M. Farnesol promotes epithelial cell defense against Candida albicans through Toll-like receptor 2 expression, interleukin-6 and human beta-defensin 2 production. Cytokine. 2009;45(2):132-140.[30] Jang BC, Lim KJ, Suh MH, et al. Dexamethasone suppresses interleukin-1beta-induced human beta-defensin 2 mRNA expression: involvement of p38 MAPK, JNK, MKP-1, and NF-kappaB transcriptional factor in A549 cells. FEMS Immunol Med Microbiol. 2007;51(1):171-184. [31] Jang BC, Lim KJ, Choi IH, et al. Triptolide suppresses interleukin-1beta-induced human beta-defensin-2 mRNA expression through inhibition of transcriptional activation of NF-kappaB in A549 cells. Int J Mol Med. 2007;19(5):757-763.[32] Derradjia A, Alanazi H, Park HJ, et al. α-tocopherol decreases interleukin-1β and -6 and increases human β-defensin-1 and -2 secretion in human gingival fibroblasts stimulated with Porphyromonas gingivalis lipopolysaccharide. J Periodontal Res. 2016;51(3):295-303.[33] Bogaczewicz J, Malinowska K, Sysa-Jedrzejowska A, et al. Medium-dose ultraviolet A1 phototherapy improves SCORAD index and increases mRNA expression of interleukin-4 without direct effect on human β defensin-1, interleukin-10, and interleukin-31. Int J Dermatol. 2016;55(7):e380-385.[34] Shimizu T, To M, Kamata Y, et al. Human β-defensin-2 and interleukin-1β expression in response to Porphyromonas gingivalis challenge in mice transplanted with periodontitic human gingiva. Microb Pathog. 2017;107:38-43.[35] Shamash S, Reichert F, Rotshenker S. The cytokine network of Wallerian degeneration: tumor necrosis factor-alpha, interleukin-1alpha, and interleukin-1beta. J Neurosci. 2002; 22(8):3052-3060. [36] Kiguchi N, Maeda T, Kobayashi Y, et al. Macrophage inflammatory protein-1alpha mediates the development of neuropathic pain following peripheral nerve injury through interleukin-1beta up-regulation. Pain. 2010;149(2):305-315.[37] Mirski R, Reichert F, Klar A, et al. Granulocyte macrophage colony stimulating factor (GM-CSF) activity is regulated by a GM-CSF binding molecule in Wallerian degeneration following injury to peripheral nerve axons. J Neuroimmunol. 2003;140(1-2):88-96. [38] Cheepudomwit T, Güzelsu E, Zhou C, et al. Comparison of cytokine expression profile during Wallerian degeneration of myelinated and unmyelinated peripheral axons. Neurosci Lett. 2008;430(3):230-235.[39] Katz Y, Nadiv O, Beer Y. Interleukin-17 enhances tumor necrosis factor alpha-induced synthesis of interleukins 1,6, and 8 in skin and synovial fibroblasts: a possible role as a "fine-tuning cytokine" in inflammation processes. Arthritis Rheum. 2001;44(9):2176-2184.[40] Perrin FE, Lacroix S, Avilés-Trigueros M, et al. Involvement of monocyte chemoattractant protein-1, macrophage inflammatory protein-1alpha and interleukin-1beta in Wallerian degeneration. Brain. 2005;128(Pt 4):854-866.[41] Ichinose M, Asai M, Imai K, et al. Enhancement of phagocytosis by corticostatin I (CSI) in cultured mouse peritoneal macrophages. Immunopharmacology. 1996; 35(2):103-109.[42] Ichinose M, Sawada M. A flow cytometric assay reveals a suppression of phagocytosis by rabbit defensin NP-3A in mouse peritoneal macrophages. Microbiol Immunol. 1995; 39(5):365-367.[43] Ichinose M, Asai M, Sawada M. Enhancement of phagocytosis by dynorphin A in mouse peritoneal macrophages. J Neuroimmunol. 1995;60(1-2):37-43.[44] Ichinose M, Asai M, Sawada M. beta-Endorphin enhances phagocytosis of latex particles in mouse peritoneal macrophages. Scand J Immunol. 1995;42(3):311-316.[45] Ichinose M, Hara N, Sawada M, et al. A flow cytometric assay reveals an enhancement of phagocytosis by platelet activating factor in murine peritoneal macrophages. Cell Immunol. 1994;156(2):508-518.[46] Ichinose M, Sawada M, Maeno T. Suppression of phagocytosis by adrenocorticotropic hormone in murine peritoneal macrophages. Immunol Lett. 1994;42(3):161-165. [47] Petrov V, Funderburg N, Weinberg A, et al. Human β defensin-3 induces chemokines from monocytes and macrophages: diminished activity in cells from HIV-infected persons. Immunology. 2013;140(4):413-420. [48] Barabas N, Röhrl J, Holler E, et al. Beta-defensins activate macrophages and synergize in pro-inflammatory cytokine expression induced by TLR ligands. Immunobiology. 2013; 218(7):1005-1011. [49] Ann SJ, Chung JH, Park BH, et al. PPARα agonists inhibit inflammatory activation of macrophages through upregulation of β-defensin 1. Atherosclerosis. 2015;240(2):389-397. [50] Terkawi MA, Takano R, Furukawa A, et al. Involvement of β-defensin 130 (DEFB130) in the macrophage microbicidal mechanisms for killing Plasmodium falciparum. Sci Rep. 2017;7:41772. [51] Lyu J, Bian T, Chen B, et al. β-defensin 3 modulates macrophage activation and orientation during acute inflammatory response to Porphyromonas gingivalis lipopolysaccharide. Cytokine. 2017;92:48-54. [52] Ganz T, Liu L, Valore EV, et al. Posttranslational processing and targeting of transgenic human defensin in murine granulocyte, macrophage, fibroblast, and pituitary adenoma cell lines. Blood. 1993;82(2):641-650.[53] Kawsar HI, Weinberg A, Hirsch SA, et al. Overexpression of human beta-defensin-3 in oral dysplasia: potential role in macrophage trafficking. Oral Oncol. 2009;45(8):696-702. [54] García JR, Jaumann F, Schulz S, et al. Identification of a novel, multifunctional beta-defensin (human beta-defensin 3) with specific antimicrobial activity. Its interaction with plasma membranes of Xenopus oocytes and the induction of macrophage chemoattraction. Cell Tissue Res. 2001;306(2): 257-264. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [3] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [4] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [5] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [6] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [7] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [8] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [9] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [10] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [11] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [12] | Guan Jian, Jia Yanfei, Zhang Baoxin , Zhao Guozhong. Application of 4D bioprinting in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 446-455. |
| [13] | Liu Jiali, Suo Hairui, Yang Han, Wang Ling, Xu Mingen. Influence of lay-down angles on mechanical properties of three-dimensional printed polycaprolactone scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2612-2617. |
| [14] | Huang Bo, Chen Mingxue, Peng Liqing, Luo Xujiang, Li Huo, Wang Hao, Tian Qinyu, Lu Xiaobo, Liu Shuyun, Guo Quanyi . Fabrication and biocompatibility of injectable gelatin-methacryloyl/cartilage-derived matrix particles composite hydrogel scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2600-2606. |
| [15] | Li Xuan, Sun Yimin, Li Longbiao, Wang Zhenming, Yang Jing, Wang Chenglin, Ye Ling. Manufacturing of nano-modified polycaprolactone microspheres and its biological effects in dental pulp cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1530-1536. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||