Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (12): 1946-1952.doi: 10.3969/j.issn.2095-4344.2017.12.024
Previous Articles Next Articles
Effects of mechanical factors on autophagy
Gui Jin-peng, Jiang Jing-yi, Zhang Ling-yu, Na Jing, Fan Yu-bo, Zheng Li-sha
- School of Biological Science and Medical Engineering, Beihang University, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, Beijing 100191, China
-
Received:2016-12-29Online:2017-04-28Published:2017-05-16 -
Contact:Zheng Li-sha, M.D., Associate professor, Master’s supervisor, School of Biological Science and Medical Engineering, Beihang University, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, Beijing 100191, China -
About author:Gui Jin-peng, Studying for master’s degree, School of Biological Science and Medical Engineering, Beihang University, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, Beijing 100191, China -
Supported by:the National Natural Science Foundation of China, No. 11572030
CLC Number:
Cite this article
Gui Jin-peng, Jiang Jing-yi, Zhang Ling-yu, Na Jing, Fan Yu-bo, Zheng Li-sha. Effects of mechanical factors on autophagy [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(12): 1946-1952.
share this article
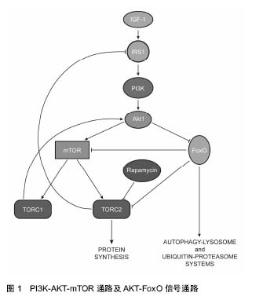
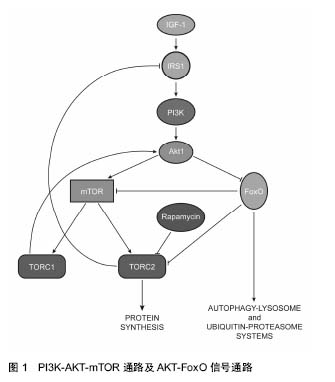
2.1 力学因素对心肌细胞自噬的影响 心脏是机体的供血器官,时刻处于强烈的血流压力环境中,心肌长时间处于压力超负荷或者血流容量超负荷条件下,就会出现左心室肥大、收缩异常、心律失常、能量代谢异常等异常现象,甚至会发展成心衰[12]。 Depre等[13]发现在压力超负荷导致左心室肥大的心肌组织中蛋白酶体的表达量和活性显著增强,而蛋白酶体活性与自噬密切相关。Zhu等[9]首次发现了自噬在压力超负荷诱导的心肌肥大中的作用,他们利用“自噬报告子”小鼠确认了压力超负荷会导致心肌细胞自噬活性持续性的增强,自噬的升高能够维持至少3周;Tannous[14]、Lin[15]以及其他的研究人员都发现了心肌细胞的自噬活性受到压力超负荷的促进[16-18]。为了进一步确认心肌细胞自噬、压力超负荷以及心肌细胞肥大的关系,Zhu等[9]利用动脉结扎使得敲除自噬相关基因Beclin-1的杂合子小鼠(Beclin-1+/-)心脏处于压力超负荷环境,发现敲除Beclin-1能够减弱压力超负荷促进的细胞自噬活性,减弱了心肌细胞对压力超负荷的病理性重建作用;而在小鼠体内过表达Beclin-1能起到相反的作用。Nakai等[10]的研究进一步确认了自噬在压力超负荷引起的心肌肥大中的作用,他们在成年小鼠体内特异性敲除自噬相关基因Atg5,小鼠出现了左心室扩张、收缩功能减弱等异常现象;敲除Atg5并在压力超负荷环境中处理2周后出现了心肌肥大等异常现象,说明了基础水平的细胞自噬是心肌细胞维持细胞稳态和心肌正常结构功能的一种机制,而自噬的上调是心肌应对血流压力超负荷的一种适应性反应。体外实验的结果也与体内实验一致,体外牵张能够诱导心肌细胞出现的肥大现象,是模拟压力超负荷环境下体内心肌细胞受力的常用方法。Hariharan等[19]对体外培养的原代大鼠心肌细胞加载36 h的应变为20%的牵张刺激,发现p62降解显著减少,说明自噬活性升高。Lin等[15]在体外对原代大鼠心肌细胞和COS7细胞施加48 h的应变为20%的牵张刺激,细胞出现肥大的同时,LC3b-Ⅱ表达增加,自噬体数量增多,自噬活性得到显著提高。 肥大的心肌细胞所处的压力超负荷环境恢复正常时,细胞可以逐渐回复正常形态,自噬在其中发挥重要作用[19]。Hariharan等[19]研究了细胞自噬在该过程中的作用,他们首先对小鼠进行动脉结扎1周使心肌细胞肥大,并去除主动脉结扎,发现心肌细胞肥大逐渐复原,同时,自噬相关的标志分子LC3b-Ⅱ表达升高,自噬活性极大的上调,而在体外实验中对心肌细胞进行机械牵张并去除牵张,也观察相同现象;基因敲除或沉默beclin-1能够抑制心肌细胞自噬活性升高,心肌肥大的恢复也受到抑制,说明细胞自噬在心肌肥大恢复中具有重要作用。 心肌细胞处于去应力负荷环境,心肌细胞的自噬也会受到影响。Cao等[20]研究了正常小鼠左心室的去应力负荷模型,发现去应力负荷后心肌细胞出现了萎缩,与此同时,细胞自噬相关的基因LC3b-Ⅱ和Beclin-1的表达上调,说明自噬也能被去应力负荷所促进,Baskin等[21]的研究也与之相符。而Kassiotis等[22]的研究却与之相反,他们比较了人的心肌在去应力负荷条件下自噬的变化情况,发现患者在佩戴左心室辅助装置前后心肌细胞中与自噬相关的基因Beclin-1、Atg5和LC3的表达显著下调,而20S蛋白酶体活性显著提高,显示去应力负荷使得心肌的自噬活性受到了抑制,他们认为在心衰患者体内心肌的自噬本身就处于一个较高水平,使用左心室辅助装置后的心肌细胞的能量需求减少,不需要维持高水平的自噬活性来提供细胞所需的能量,所以心肌细胞自噬水平出现降低。 力学因素对心肌细胞自噬的影响与蛋白酶体活性有关。压力超负荷使得心肌细胞中错误折叠的蛋白数量增多,这些蛋白在细胞核周围逐渐聚集,形成聚集体,从而被细胞自噬系统识别并降解。Tannous等[14]发现压力超负荷会使得左心室心肌细胞泛素化的蛋白累积、聚集体结构形成以及细胞自噬活性增加;在体外抑制心肌细胞蛋白酶体活性使泛素化蛋白积累,泛素化蛋白的积累能够引发细胞自噬;而抑制心肌细胞的自噬活性,能有效的增强聚集体结构的大小和数量。 PI3K-AKT-mTOR通路是调控细胞自噬的重要信号通路[23](图1),在AKT的下游,有一类转录因子——FoxO (forkhead box,class O)蛋白家族参与到心肌细胞自噬调控[24],能够促进自噬活性,通常情况下FoxO活性受到AKT的抑制,在心肌细胞受到刺激后,AKT对FoxOs的抑制被解除了[25],活化的FoxO1能够使得心肌细胞中特有的一种E3泛素连接酶atrogin-1/MAFbx的表达上调,促进泛素化活动和溶酶体活性,影响自噬途径,促进蛋白降解[26];而活化的FoxO3能够活化下游的Bcl-2家族成员Bnip3,抑制Bcl-2和Beclin-1的结合,促进自噬[27-28]。"
| [1] Ouyang L, Shi Z, Zhao S, et al. Programmed cell death pathways in cancer: a review of apoptosis, autophagy and programmed necrosis. Cell Prolif. 2012;45:487-498.[2] Periyasamy-Thandavan S, Jiang M, Schoenlein P, et al. Autophagy: molecular machinery, regulation, and implications for renal pathophysiology. Am J Physiol Renal Physiol. 2009; 297:F244-F256.[3] Reggiori F. Membrane origin for autophagy. Curr Top Dev Biol. 2006;74:1-30.[4] Klionsky DJ, Emr SD. Autophagy as a regulated pathway of cellular degradation. Science. 2000;290:1717-1721.[5] Fang Y, Tan J, Zhang Q. Signaling pathways and mechanisms of hypoxia-induced autophagy in the animal cells.Cell Biol Int.2015;39:891-898.[6] Meng XX, Yao M, Zhang XD, et al. ER stress-induced autophagy in melanoma. Clin Exp Pharmacol Physiol. 2015; 42: 811-816.[7] Lin WJ, Kuang HY. Oxidative stress induces autophagy in response to multiple noxious stimuli in retinal ganglion cells. Autophagy. 2014; 10: 1692-1701.[8] Liu J, Bi X, Chen T, et al. Shear stress regulates endothelial cell autophagy via redox regulation and Sirt1 expression. Cell Death Dis. 2015; 6: e1827.[9] Zhu HX, Tannous P, Johnstone JL, et al. Cardiac autophagy is a maladaptive response to hemodynamic stress. JCli Invest. 2007; 117: 1782-1793.[10] Nakai A, Yamaguchi O, Takeda T, et al. The role of autophagy in cardiomyocytes in the basal state and in response to hemodynamic stress. Nat Med. 2007; 13: 619-624.[11] Ma KG, Shao ZW, Yang SH, et al. Autophagy is activated in compression-induced cell degeneration and is mediated by reactive oxygen species in nucleus pulposus cells exposed to compression. Osteoarthr Cartilage. 2013; 21: 2030-2038.[12] Rothermel BA, Hill JA. Autophagy in load-induced heart disease. Circ Res. 2008;103:1363-1369.[13] Depre C, Wang Q, Yan L, et al. Activation of the cardiac proteasome during pressure overload promotes ventricular hypertrophy. Circulation. 2006;114:1821-1828.[14] Tannous P, Zhu HX, Nemchenko A, et al. Intracellular Protein Aggregation Is a Proximal Trigger of Cardiomyocyte Autophagy. Circ Res. 2008;117(24):3070-3078.[15] Lin L, Tang CY, Xu JF, et al. Mechanical Stress Triggers Cardiomyocyte Autophagy through Angiotensin II Type 1 Receptor-Mediated p38MAP Kinase Independently of Angiotensin II. Plos One. 2014;9(2):e89629.[16] Weng LQ, Zhang WB, Ye Y, et al. Aliskiren ameliorates pressure overload-induced heart hypertrophy and fibrosis in mice. Acta Pharmacol Sin. 2014;35:1005-1014.[17] Lin L, Liu XB, Xu JF, et al. High-density lipoprotein inhibits mechanical stress-induced cardiomyocyte autophagy and cardiac hypertrophy through angiotensin II type 1 receptor-mediated PI3K/Akt pathway. J Cell Mol Med. 2015; 19:1929-1938.[18] Fu L, Wei CC, Powell PC, et al. Volume overload induces autophagic degradation of procollagen in cardiac fibroblasts. J Mol Cell Cardiol. 2015;89:241-250.[19] Hariharan N, Ikeda Y, Hong C, et al. Autophagy Plays an Essential Role in Mediating Regression of Hypertrophy during Unloading of the Heart. Plos One. 2013;8(1):e51632.[20] Cao DJ, Jiang N, Blagg A, et al. Mechanical Unloading Activates FoxO3 to Trigger Bnip3-Dependent Cardiomyocyte Atrophy. J Am Heart Assoc. 2013;2(2):e000016.[21] Baskin KK, Rodriguez MR, Kansara S, et al. MAFbx/Atrogin-1 is required for atrophic remodeling of the unloaded heart. J Mol Cell Cardiol. 2014;72:168-176.[22] Kassiotis C, Ballal K, Wellnitz K, et al. Markers of Autophagy Are Downregulated in Failing Human Heart After Mechanical Unloading. Circulation. 2009; 120: S191-S197.[23] Hill JA. Autophagy in cardiac plasticity and disease. Pediatr Cardiol. 2011; 32: 282-289.[24] Hariharan N, Maejima Y, Nakae J, et al. Deacetylation of FoxO by Sirt1 Plays an Essential Role in Mediating Starvation-Induced Autophagy in Cardiac Myocytes. Circ Res. 2010; 107: 1470-1482.[25] Skurk C, Izumiya Y, Maatz H, et al. The FOXO3a transcription factor regulates cardiac myocyte size downstream of AKT signaling. J Biol Chem. 2005;280:20814-20823.[26] Sandri M, Sandri C, Gilbert A, et al. Foxo transcription factors induce the atrophy-related ubiquitin ligase atrogin-1 and cause skeletal muscle atrophy. Cell.2004;117: 399-412.[27] Mammucari C, Milan G, Romanello V, et al. FoxO3 controls autophagy in skeletal muscle in vivo. Cell Metab. 2007;6: 458-471.[28] Zhang HF, Bosch-Marce M, Shimoda LA, et al. Mitochondrial autophagy is an HIF-1-dependent adaptive metabolic response to hypoxia. J Biol Chem. 2008;283:10892-10903.[29] Porrello ER, D'amore A, Curl CL, et al. Angiotensin II type 2 receptor antagonizes angiotensin II type 1 receptor-mediated cardiomyocyte autophagy. Hypertension. 2009;53:1032-1040.[30] Yadav A, Vallabu S, Arora S, et al. ANG II promotes autophagy in podocytes. Am J Physiol Cell Physiol. 2010; 299: C488-C496.[31] Huang J, Sun W, Huang H, et al. miR-34a modulates angiotensin II-induced myocardial hypertrophy by direct inhibition of ATG9A expression and autophagic activity. PLoS One. 2014; 9(4): e94382.[32] Yue H, Li W, Desnoyer R, et al. Role of nuclear unphosphorylated STAT3 in angiotensin II type 1 receptor- induced cardiac hypertrophy. Cardiovasc Res. 2010; 85: 90-99.[33] Ramakrishnan P, Hecht BA, Pedersen DR, et al. Oxidant conditioning protects cartilage from mechanically induced damage. J Orthop Res. 2010;28:914-920.[34] Thomas CM, Fuller CJ, Whittles CE, et al. Chondrocyte death by apoptosis is associated with cartilage matrix degradation. Osteoarthr Cartilage. 2007;15:27-34.[35] Carames B, Taniguchi N, Seino D, et al. Mechanical Injury Suppresses Autophagy Regulators and Pharmacologic Activation of Autophagy Results in Chondroprotection. Arthritis Rheumus. 2012;64:1182-192.[36] Xu HG, Yu YF, Zheng Q, et al. Autophagy protects end plate chondrocytes from intermittent cyclic mechanical tension induced calcification. Bone. 2014;66:232-239.[37] Pan S. Molecular mechanisms responsible for the atheroprotective effects of laminar shear stress. Antioxid Redox Sign. 2009;11:1669-1682.[38] Guo FX, Li XH, Peng J, et al. Autophagy Regulates Vascular Endothelial Cell eNOS and ET-1 Expression Induced by Laminar Shear Stress in an Ex Vivo Perfused System. Ann Biomed Eng. 2014;42:1978-1988.[39] Ding Z, Liu S, Deng X, et al. Hemodynamic shear stress modulates endothelial cell autophagy: Role of LOX-1. Int J Cardiol. 2015;184:86-95.[40] Bharath LP, Mueller R, Li Y, et al. Impairment of autophagy in endothelial cells prevents shear-stress-induced increases in nitric oxide bioavailability. Can J Physiol Pharmacol. 2014; 92: 605-612.[41] Gumucio JP, Davis ME, Bradley JR, et al. Rotator cuff tear reduces muscle fiber specific force production and induces macrophage accumulation and autophagy. J Orthop Res. 2012;30:1963-1970.[42] Ning L, Xu Z, Furuya N, et al. Perlecan inhibits autophagy to maintain muscle homeostasis in mouse soleus muscle. Matrix Biology. 2015;48:26-35.[43] Ulbricht A, Eppler FJ, Tapia VE, et al. Cellular mechanotransduction relies on tension-induced and chaperone-assisted autophagy. Curr Biol. 2013;23:430-435.[44] Ulbricht A, Gehlert S, Leciejewski B, et al. Induction and adaptation of chaperone-assisted selective autophagy CASA in response to resistance exercise in human skeletal muscle. Autophagy. 2015; 11: 538-546.[45] Chen HC, Fong TH, Hsu PW, et al. Multifaceted effects of rapamycin on functional recovery after spinal cord injury in rats through autophagy promotion, anti-inflammation, and neuroprotection. J Surg Res. 2013;179: e203-e210.[46] Chen Z, Fu Q, Shen B, et al. Enhanced p62 expression triggers concomitant autophagy and apoptosis in a rat chronic spinal cord compression model. Mol Med Rep. 2014; 9: 2091-2096.[47] Wang ZY, Lin JH, Muharram A, et al. Beclin-1-mediated autophagy protects spinal cord neurons against mechanical injury-induced apoptosis. Apoptosis. 2014; 19: 933-945.[48] Park Y, Liu C, Luo T, et al. Chaperone-Mediated Autophagy after Traumatic Brain Injury. J Neurotrauma. 2015; 2: 1449-1457.[49] Lien SC, Chang SF, Lee PL, et al. Mechanical regulation of cancer cell apoptosis and autophagy: roles of bone morphogenetic protein receptor, Smad1/5, and p38 MAPK. Biochim Biophys Acta. 2013; 1833:3124-3133.[50] Chen H, Chen LY, Cheng BA, et al. Cyclic Mechanical Stretching Induces Autophagic Cell Death in Tenofibroblasts Through Activation of Prostaglandin E2 Production. Cell Physiol Biochem. 2015; 36:24-33.[51] Li D, Yan T, Xu Z, et al. Spironolactone Promotes Autophagy via Inhibiting PI3K/AKT/mTOR Pathway and Reduce Adhesive Damage in Podocytes under Mechanical Stress. Biosci Rep. 2016.[52] Porter KM, Jeyabalan N, Liton PB. MTOR-independent induction of autophagy in trabecular meshwork cells subjected to biaxial stretch. Bba-Mol Cell Res. 2014; 1843: 1054-1062. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [3] | Cui Xing, Sun Xiaoqi, Zheng Wei, Ma Dexin. Huangqin Decoction regulates autophagy to intervene with intestinal acute graft-versus-host disease in mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1057-1062. |
| [4] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [5] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [6] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [7] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [8] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [9] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [10] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [11] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [12] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [13] | Guan Jian, Jia Yanfei, Zhang Baoxin , Zhao Guozhong. Application of 4D bioprinting in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 446-455. |
| [14] | Sun Youqiang, Ma Chao, Liang Mengmeng, Xin Pengfei, Zhang Hua, Xiang Xiaobing. The pivotal role of autophagy in bone cells: bone-related cell activity and bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 276-282. |
| [15] | Liu Jiali, Suo Hairui, Yang Han, Wang Ling, Xu Mingen. Influence of lay-down angles on mechanical properties of three-dimensional printed polycaprolactone scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2612-2617. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||