Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (50): 8184-8188.doi: 10.3969/j.issn.2095-4344.2014.50.026
Previous Articles Next Articles
Stem cell therapy for obstructive nephropathy: current situation and problems
Lv Chun-yan, Li Juan
- Department of Pathology, the Affiliated Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, Sichuan Province, China
-
Received:2014-11-12Online:2014-12-03Published:2014-12-03 -
About author:Lv Chun-yan, Master, Associate chief physician, Department of Pathology, the Affiliated Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, Sichuan Province, China -
Supported by:the Project of Sichuan Province Health Department, No. 110520; the Project of Chengdu Health Bureau, No. 2014004
CLC Number:
Cite this article
Lv Chun-yan, Li Juan. Stem cell therapy for obstructive nephropathy: current situation and problems[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(50): 8184-8188.
share this article
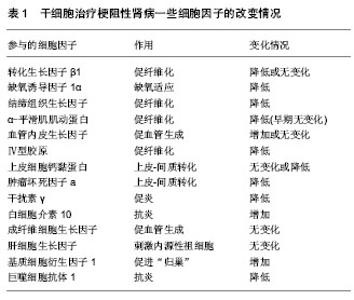
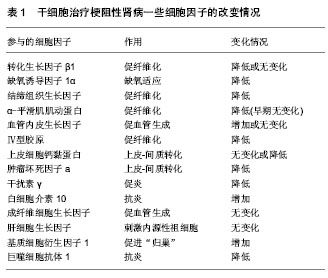
2.1 梗阻性肾病的动物模型研究 尿路梗阻性疾病是指尿路任何部位由于结构、功能等改变而导致尿流排出不畅的一组疾病。如果这种梗阻性疾病影响肾实质,导致肾脏结构(和)或功能的改变,就发展为梗阻性肾病[8],梗阻性肾病又分为完全性梗阻和部分性梗阻。在临床工作中,一些梗阻性肾病通过碎石、解除狭窄等方法得到治疗,但肾脏的损害并不能立即完全恢复,因此梗阻性肾损伤仍然存在。人们为了便于研究梗阻性肾病的病因、病机以及治疗等,结合临床情况,设计了以下3种动物模型:①单侧输尿管结扎造成完全梗阻性肾病的动物模型(complete unilateral ureteral occlusion,CUUO),简称单侧输尿管闭塞动物模型。该模型手术操作简单,在游离单侧输尿管后将其结扎便可,其结果稳定,是目前国际公认的动物模型[9-10],因此也被越来越多的研究者使用。②输尿管部分梗阻动物模型(partial unilateral ureteral obstruction,PUUO) [11]。该模型采用腰大肌掩埋法[12]、输尿管套管法、输尿管不全结扎法、输尿管内支架置入法等方法,使输尿管部分梗阻,该类方法较符合临床实际,但所选动物物种品系及模型建立的方法将对实验结果产生决定性影响,因此,目前尚缺乏国际公认的动物模型。③单侧输尿管梗阻再通模型(reversible unilateral ureteral obstruction ,RUUO)[13]。该模型在制作过程中,不离断输尿管,仅将输尿管夹闭。术后5-10 d再次手术,松解夹闭处,使输尿管再通。该模型更符合外科治疗实际,可更准确反映梗阻解除后的病理生理,为相应治疗方案的完善提供依据。但模型动物需经历二次手术,对动物影响更大。 2.2 干细胞类型 干细胞按来源不同可分为3类:胚胎干细胞、成体干细胞和诱导性多能干细胞。各种来源的干细胞各有其优缺点:①研究最早的是胚胎干细胞,但由于胚胎来源困难,体外保持其全能性条件复杂,安全性难以保证等原因,使其应用受限[14]。②成体干细胞是目前研究与应用最多最广的干细胞。成体干细胞是来源于成熟个体组织器官的一大类干细胞,其具有自我更新能力,但分化潜能窄,只能分化为相应或相邻组织器官组成的细胞,如造血干细胞、骨髓间充质干细胞[15]、神经干细胞及肾脏干细胞等[16]。来源于羊水的羊水干细胞也属于成体干细胞的范畴[17]。本文纳入23篇文章采用成体干细胞进行研究,其中对骨髓间充质干细胞的研究最多,其次有羊水干细胞、胚胎后肾间充质干细胞、脐带间充质干细胞及内皮祖细胞等[18-19]。用某种基因修饰干细胞,如携带肝细胞生长因子[20]、尿激酶型纤溶酶原激活基因等可增强干细胞的某种功能,以期更好地修复肾损伤。③诱导性多能干细胞是目前研究的最新方向[21]。诱导性多能干细胞是通过基因转染技术将某些转录因子导入动物或人的体细胞,使体细胞直接重构成为胚胎干细胞样的多潜能细胞。本文纳入4篇文章采用诱导多能干细胞进行研究,其体细胞来源有成纤维细胞[22]、单个核细胞等[23]。 2.3 干细胞移植方式及“归巢”率 目前干细胞治疗梗阻性肾病的移植方式主要有经静脉、动脉和局部移植[24]。Huang等[25]经大鼠尾静脉注射脐带间充质干细胞后14,21,28 d,取其心、肝、脾、肺、肾组织进行研究发现,干细胞主要分布于肺、脾、肾,在单侧输尿管闭塞大鼠肾的分布较假手术组肾更多,说明静脉注射干细胞是可行且安全的。干细胞有向创伤部位趋附的趋势,因此,经静脉移植是在大、小鼠等小型动物中最常使用的方式,该方式技术上也易于掌握。但经静脉移植的干细胞需经肺循环才能到达肾脏,在实施时必将损失一部分细胞。经肾动脉移植可减少肺循环损失的细胞量,但动脉穿刺操作难度大,容易造成不易控制的大出血[26]。肾包膜下直接注射可避免上述操作的缺点,但可造成局部炎症,且归巢时间较慢[27]。因此,在移植方式的选择上,可以根据不同的研究或治疗目的进行选择。 2.4 干细胞治疗梗阻性肾病的疗效及可能机制 2.4.1 移植的干细胞可定位于梗阻性肾 周盾等[28]将人脐血间充质干细胞静脉注入单侧输尿管闭塞大鼠,通过免疫组化检测肾间质HLA-1表达情况发现,HLA-1高表达于肾小球内、肾间质内微血管及大的血管内皮细胞以及部分损伤的小管部位。Bai等[29]将绿色荧光蛋白标记的间充质干细胞(luciferase reporter gene and green fluorescence protein genes,Luc-GFP-MSC)经左肾动脉注入单侧输尿管闭塞大鼠体内,用活体成像系统(in vivo imaging system,IVIS)进行追踪定位发现间充质干细胞移植3 d后荧光素酶在左肾出现并逐渐增加,7 d开始减弱,14 d消失。但程庆等[27]取肾组织经荧光显微镜观察发现,移植成纤维细胞诱导的多能干细胞8周后,肾小管、肾小球毛细血管及间质小动脉仍有丰富的荧光素蛋白细胞,这可能和检测方法不同有关。Yoo等[30]、李静[31]研究与上述结果一致。静脉注射和包膜下注射时,干细胞在肾间质出现时间延长。有人认为,迁移定位于肾间质的干细胞可以分化为肾脏实质细胞[32],从而修复改善肾功能[33]。因此,在体外将干细胞定向分化为“肾小管样细胞”,进一步移植到肾组织,以期治疗肾疾病成为目前干细胞治疗的一个研究热点。 2.4.2 干细胞治疗梗阻性肾可改善肾结构和功能 Ruan等[34]将成纤维细胞诱导的多能干细胞经肾动脉注入单侧输尿管闭塞兔子体内,8周后观察,干细胞治疗组动物血肌酐和尿素氮水平明显降低,肾小球滤过率较模型组增高,但仍低于正常组,说明干细胞可使肾功能得到改善,但不能完全恢复肾功能。而干细胞治疗组肾脏体积略增大或与正常肾脏大小相当,肾实质变薄,但质量与正常肾脏相当。马丹[35]也发现兔肾间质纤维化后,移植的自体骨髓间充质干细胞对血肌酐和尿素氮的升高有一定的抑制作用,促进了肾脏功能的恢复。 2.4.3 干细胞可减轻梗阻性肾间质纤维化程度 Sugandhi等[36]用干细胞治疗输尿管部分梗阻大鼠15 d后发现,干细胞治疗组肾皮质厚度明显高于未治疗组。组织学研究发现,干细胞治疗组肾间质纤维化程度、肾盂周纤维化程度、小管凋亡及肾小球硬化程度均较未治疗组轻。王代红等[37]也有类似发现。 2.4.4 干细胞可分泌、上调或下调一些细胞因子,发挥抗纤维化的作用 目前较多研究发现,干细胞归巢定位到肾间质后,可通过分泌、上调或下调一些细胞因子发挥抗纤维化的作用[37-46]。目前有研究报道的细胞因子见表1。由于细胞因子的抗纤维化作用,部分研究者将具有治疗肾纤维化的细胞因子基因转入干细胞,再将携带了这些基因的干细胞移植入单侧输尿管闭塞动物,以期增强干细胞抗纤维化的作用[47]。目前报道的有肝细胞生长因子[48]、尿激酶型纤溶酶原激活基因等[49]。"
| [1] 吕春燕,陈高莉,杨玲,等.大鼠脂肪干细胞分离培养及细胞表型的研究[J].海南医学,2014,25(9):1256-1259. [2] 周敬,潘兴华,庞荣清,等.干细胞移植治疗慢性肾脏疾病的可行性[J].中国组织工程研究与临床康复,2011,15(6):1107-1110. [3] 刘玉园,刘虹.干细胞在肾间质纤维化中的探究进展[J].中国中西医结合肾病杂志,2013,14(5):464-466. [4] 汪年松,姜珍珍.干细胞在肾脏疾病中的研究进展[J].中国中西医结合肾脏病杂志,2014,15(3):189-192. [5] 郭婷婷,黎丽茜,田虹.脂肪干细胞修复泌尿系统损伤:应用现状及问题[J].中国组织工程研究,2014,18(2):283-288. [6] 邢健生,白志明.MSCs治疗慢性肾脏病的新认知及进展[J].海南医学,2014,25(12):1794-1797. [7] Chou YH, Pan SY, Yang CH,et al.Stem cells and kidney regeneration.J Formos Med Assoc. 2014;113(4):201-209. [8] 邢健生,白志明.单侧输尿管梗阻模型引起肾损伤的新认知及进展[J].海南医学,2014,25(13):1964-1967. [9] 樊彩斌,周云,严向明,等.血管内皮生长因子对梗阻性肾病中肾小管周围毛细血管的调节作用[J].中国组织工程研究与临床康复, 2007,11(49):9821-9825. [10] 刘利峰,王砺.单侧输尿管梗阻对双侧肾脏病理改变的研究进展[J].医学综述,2012,18(17):2753-2755. [11] 蔡勇,白志明.输尿管梗阻动物模型的研究[J].局解手术学杂志, 2014,23(4):413-416. [12] 薛彦生,杨屹,王常林.SD大鼠可逆性不全梗阻肾病模型的建立和不同制作方法的比较[C].中华医学会第八次小儿外科学术会议论文集,2010:195-196. [13] Bai ZM, Deng XD, Li JD,et al.Arterially transplanted mesenchymal stem cells in a mouse reversible unilateral ureteral obstruction model: in vivo bioluminescence imaging and effects on renal fibrosis.Chin Med J (Engl). 2013;126(10): 1890-1894. [14] 杨建华,张富运,热吉普,等.胚胎干细胞移植对转化生长因子β1和髓鞘碱性蛋白影响[J].中国组织工程研究,2014,18(1): 112-118. [15] Ninichuk V, Anders HJ.Bone marrow-derived progenitor cells and renal fibrosis.Front Biosci. 2008;13:5163-5173. [16] Park HC, Yasuda K, Ratliff B,et al.Postobstructive regeneration of kidney is derailed when surge in renal stem cells during course of unilateral ureteral obstruction is halted.Am J Physiol Renal Physiol. 2010;298(2):F357-364. [17] Sun D, Bu L, Liu C,et al.Therapeutic effects of human amniotic fluid-derived stem cells on renal interstitial fibrosis in a murine model of unilateral ureteral obstruction.PLoS One. 2013;8(5):e65042. [18] 黄丹琳.人脐带间充质干细胞移植治疗大鼠梗阻性肾间质纤维化的实验研究[D].长沙:中南大学,2011:55-86. [19] 王代红.内皮祖细胞对单侧输尿管梗阻大鼠肾小管周毛细血管重建的实验研究[D].重庆:第三军医大学,2009:41-65. [20] 谢鸣部.肝细胞生长因子转染的骨髓间充质干细胞对肾间质纤维化的影响[D].福州:福建医科大学,2009:26-45. [21] 徐繁.兔肾间质纤维化模型的建立及诱导性自体干细胞移植治疗[D].昆明:昆明医学院,2010:28-29. [22] 徐繁,潘兴华,李潇,等.诱导性自体干细胞移植后间质纤维化肾的结构与功能变化[J].西南国防医药,2010,20(1):7-9. [23] 周敬.自体诱导性多能干细胞移植治疗慢性肾功能不全的研究[D].昆明:昆明医学院,2011:17-24. [24] 李金东.大鼠骨髓间充质干细胞三种移植途径在急性肾梗阻模型中的归巢研究[D].长沙:中南大学,2012:20-42. [25] Huang D, Yi Z, He X,et al.Distribution of infused umbilical cord mesenchymal stem cells in a rat model of renal interstitial fibrosis.Ren Fail. 2013;35(8):1146-1150. [26] 李东辉,刘振湘,李金东,等. 经肾动脉移植BMSC在小鼠单侧输尿管梗阻模型的建立[J].浙江临床医学,2014,16(2):211-214. [27] 程庆.大鼠骨髓间充质干细胞包膜下移植对梗阻肾病变影响的初步研究[D].长沙:中南大学,2011:23-25. [28] 周盾,孙洞箫,邱振宇.人脐血间充质干细胞静注对UUO大鼠肾间质纤维化程度的影响[J].山东医药,2012,52(22):9-11. [29] Bai ZM, Deng XD, Li JD,et al.Arterially transplanted mesenchymal stem cells in a mouse reversible unilateral ureteral obstruction model: in vivo bioluminescence imaging and effects on renal fibrosis.Chin Med J (Engl). 2013;126(10): 1890-1894. [30] Yoo JH, Park C, Jung DI,et al.In vivo cell tracking of canine allogenic mesenchymal stem cells administration via renal arterial catheterization and physiopathological effects on the kidney in two healthy dogs.J Vet Med Sci. 2011;73(2): 269-274. [31] 李静.肾小管间质纤维化模型中大鼠骨髓间充质干细胞在肾脏的靶向定位[D].成都:四川大学,2005:28-29. [32] 林金爱,高岩.骨髓间充质干细胞对大鼠肾小管损伤的保护作用[J].广东医学,2013,34(8):1172-1175. [33] 万建新,郭琦,潘阳彬,等.骨髓间充质干细胞修复损伤肾小管上皮细胞:可能与可行[J].中国组织工程研究与临床康复,2010, 14(32):5903-5907. [34] Ruan GP, Xu F, Li ZA,et al.Induced autologous stem cell transplantation for treatment of rabbit renal interstitial fibrosis.PLoS One. 2013;8(12):e83507. [35] 马丹.兔肾间质纤维化模型的建立及自体骨髓间充质干细胞移植治疗的实验研究[D].昆明医学院,2009:18-19. [36] Sugandhi N, Srinivas M, Agarwala S,et al.Effect of stem cells on renal recovery in rat model of partial unilateral upper ureteric obstruction.Pediatr Surg Int. 2014;30(2):233-238. [37] 王代红,李芙蓉,程悦,等.大鼠骨髓内皮祖细胞移植对单侧输尿管梗阻大鼠肾脏损伤的修复[J].重庆医学,2009,38(24): 3118- 3121. [38] Barreira AL, Takiya CM, Castiglione RC,et al.Bone marrow mononuclear cells attenuate interstitial fibrosis and stimulate the repair of tubular epithelial cells after unilateral ureteral obstruction.Cell Physiol Biochem. 2009;24(5-6):585-594. [39] Ma YY, Sun D, Li J,et al.Transplantation of endothelial progenitor cells alleviates renal interstitial fibrosis in a mouse model of unilateral ureteral obstruction.Life Sci. 2010;86 (21-22):798-807. [40] Liu X, Shen W, Yang Y,et al.Therapeutic implications of mesenchymal stem cells transfected with hepatocyte growth factor transplanted in rat kidney with unilateral ureteral obstruction.J Pediatr Surg. 2011;46(3):537-545. [41] Asanuma H, Vanderbrink BA, Campbell MT,et al.Arterially delivered mesenchymal stem cells prevent obstruction- induced renal fibrosis.J Surg Res. 2011;168(1):e51-59. [42] 罗燕萍.沙利度胺和人间充质干细胞对单侧输尿管梗阻大鼠肾间质纤维化的作用[D].镇江:江苏大学,2010:27-35. [43] 李佩倞.超声联合微泡促HGF修饰BMSCs靶向归巢抗肾纤维化的实验研究[D].重庆:第三军医大学,2013:92-96. [44] 张建江.骨髓干细胞动员对单侧输尿管梗阻大鼠肾间质微血管及纤维化的影响[D].长沙:中南大学,2006:53-72. [45] 孙洞箫,周盾,邱振宇.UC-MSCs对UUO大鼠肾间质ET-1、VEGF、肾小管周Ⅷ因子表达水平的影响[J].山东医药,2012, 52(10):28-29. [46] 王代红.内皮祖细胞对单侧输尿管梗阻大鼠肾小管周毛细血管重建的实验研究[D].重庆:第三军医大学,2009:50-56. [47] 陈昌金,袁惠,辜海英,等.多效生长因子基因转染小鼠脂肪来源干细胞的实验研究[J].中国修复重建外科杂志,2013,27(8): 897-901. [48] 沈伟.HGF基因修饰的骨髓间充质干细胞移植在输尿管梗阻大鼠肾脏中的分布[D].沈阳:中国医科大学,2009:10-13. [49] 毕凌云.胚胎后肾间充质细胞转染uPA基因治疗单侧输尿管梗阻大鼠的实验研究[D].长沙:中南大学,2008:64-104. [50] Liu YL, Wang YD, Zhuang F,et al.Immunosuppression effects of bone marrow mesenchymal stem cells on renal interstitial injury in rats with unilateral ureteral obstruction.Cell Immunol. 2012;276(1-2):144-152. [51] 庄峰,刘英莉,张薇,等. BMSCs移植对单侧输尿管结扎大鼠CD4+ CD25+Treg的影响[J].上海交通大学学报(医学版), 2011, 31(6): 739-743. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [14] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [15] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||