Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (39): 6359-6363.doi: 10.3969/j.issn.2095-4344.2014.39.022
Previous Articles Next Articles
Application of collagen and silk fibroin scaffolds in spinal cord tissue engineering
Zhu Xiang1, Chen Xu-yi2, Liu Ying-fu3, Xing Ran1, Tu Yue2
- 1Tianjin University of Traditional Chinese Medicine, Tianjin 300193, China; 2Department of Brain, Affiliated Hospital of Logistics College of CAPF, Tianjin 300162, China; 3Department of Clinical Medicine, Logistics College of CAPF, Tianjin 300309, China
-
Online:2014-09-17Published:2014-09-17 -
Contact:Tu Yue, Professor, Master’s supervisor, Department of Brain, Affiliated Hospital of Logistics College of CAPF, Tianjin 300162, China -
About author:Zhu Xiang, Studying for master’s degree, Tianjin University of Traditional Chinese Medicine, Tianjin 300193, China -
Supported by:the Science and Technology Support Program of Tianjin, No. 14ZCZDGX00500; the National Natural Science Foundation of China, No. 11102235; the Natural Science Foundation of Tianjin, No. 12JCZDJC24100; the Scientific Fund of Tianjin Health Bureau, No. 2013KZ134; the Seed Fund of the Affiliated Hospital of Logistics College of CAPF, No. FYM201432
CLC Number:
Cite this article
Zhu Xiang, Chen Xu-yi, Liu Ying-fu, Xing Ran, Tu Yue. Application of collagen and silk fibroin scaffolds in spinal cord tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(39): 6359-6363.
share this article
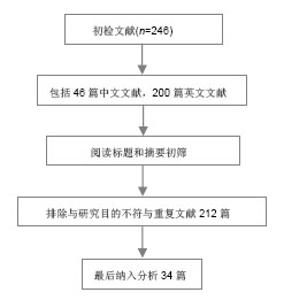
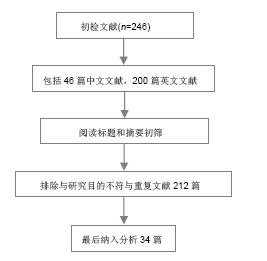
2.1 胶原蛋白 2.1.1 胶原蛋白的结构 胶原蛋白是细胞外基质的一种结构蛋白,不同于一般蛋白质的双螺旋结构,由3条多肽链组成,每条多肽链都左旋形成左手螺旋结构,再以氢键相互咬合组成右手超螺旋结构。胶原的螺旋区段是由左旋a链相互缠绕而成,最大的特征是氨基酸呈现(Gly-X-Y)n 周期性排列,其中X、Y位置为羟脯氨酸(Hyp)和脯氨酸(PrO),这两种是胶原特有的氨基酸[4]。3条多肽链中的α-肽链借范德化力、氢键及共价交联以平行、右手螺旋形式 文献检索流程图: 缠绕成3股螺旋结构,使胶原蛋白具有很高的拉伸强度[5]。胶原蛋白含有细胞的特殊黏附位点Arg-Gly-Asp(RGD)序列,使其具有良好的生物相容性,但单一的胶原蛋白构建三维多孔网状支架结构的能力较差,结构不稳定。胶原蛋白具有的整合结合位点,能与细胞表面的整合素识别并结合,通过生长因子(如神经营养蛋白)进一步增强对神经元的再生[6]。 2.1.2 胶原蛋白的特性 胶原蛋白是一种在组织工程中广泛使用的生物材料,具有低抗原性,优良的生物相容性和生物降解性,本身及其降解产物在人体内不会引起炎症反应,也避免了植入后再次手术取出对身体造成的伤害[7]。Gerburg等[8]对Ⅰ/Ⅲ型胶原复合导管的生物相容性进行了研究,结果表明术后5-7 d导管与宿主得到很好结合。由胶原蛋白制成的生物支架材料,其本身的半通透性有利于营养物质从机体中进入到支架;胶原蛋白支架材料有利于桥接脊髓损伤后轴突的再生,引导轴突的方向性生长[9]。胶原蛋白支架还可以与抗体、神经营养因子等结合,在大鼠脊髓损伤模型中作为药物缓释系统的载体,支持损伤处神经的连接,促进神经的再生和功能的恢复[10-11]。贾骏等[12]研究表明聚乳酸-羟基乙酸共聚物的纳米纤维支架经胶原改性后,改善了所述细胞的相容性。胶原凝胶、功能化胶原凝胶均具有良好的细胞相容性,神经生长因子活化的胶原蛋白能更好地促进大鼠脊髓神经功能的恢复[13]。但胶原蛋白在体内存在降解较快和力学性能差等缺点,诱导组织再生的胶原蛋白在修复较大的脊髓缺损时很容易降解,不能很好地充当脊髓再生的支架;要满足胶原蛋白在脊髓组织工程中应用的要求,除了对其本身进行改性和加工外,还需要寻找互补的天然高分子材料。 2.1.3 胶原蛋白在脊髓组织工程中的应用 已有研究证实,以胶原蛋白为支架材料植入脊髓损伤部位,神经细胞能够存活生长并促进轴突生长[14]。神经营养因子3对胶原蛋白结合域功能的重组,使其溶解到Ⅰ型胶原蛋白支架上,这些支架随后被植入到大鼠横断脊髓部位而起到作用,改善了脊髓损伤后的大鼠运动功能[15]。Yoshii等[16]在大鼠横断脊髓中植入了胶原蛋白支架,使脊髓功能得到了明显恢复。Liu等[17]认为表皮生长因子受体的抗体与功能化胶原蛋白支架的结合,对脊髓损伤后神经分化和修复有积极的促进作用,在这个过程中,胶原蛋白支架提供了一个可以引入额外刺激以改善脊髓损伤后恢复的平台。Liu等[18]将胶原蛋白制成的导管植入到动物脊髓半横断的腔隙,伤后9个月在脊髓损伤处发现神经纤维通过胶原蛋白导管从头侧生长到尾侧。吴立志等[19]将胶原蛋白与许旺细胞共移植到成年大鼠脊髓完全横断损伤处,3个月后观察发现动物后肢的自主运动功能有了显著恢复;通过免疫荧光发现神经元的轴突能在脊髓内穿越损伤区再生。Giannetti等[20]将结合胶原的羟化异丁酸多孔海绵支架植入到脊髓半切损伤的SD大鼠损伤处,2-4个月后的观察显示损伤处的纤维胶质瘢痕较少,有大量轴突进入多孔胶原海绵生长并贯穿整个支架。Han等[21]将抑制神经生长的中和性抗体表皮生长因子受体与神经保护蛋白脑源性神经营养因子(brain derived neurophic factor,BDNF)联合负载到胶原蛋白支架上,成功地促进了脊髓横断损伤后的再生和神经功能恢复。 2.2 丝素蛋白 2.2.1 丝素蛋白的结构 丝素蛋白是从蚕丝中提取的天然高分子纤维蛋白,主要由丝氨酸、丙氨酸、酪氨酸、乙氨酸和脯氨酸等组成,一条L链和一条H链通过S-S键结合,由于丝素蛋白主要成分中的氨基酸有氨基,易形成具有β折叠结晶结构的氢键,所以不溶于水。但在碱性溶液中,β折叠结晶结构被破坏后分解,降低了力学强度。丝素蛋白具有良好的透湿性和透气性,对水和氧的通透性较好,可被蛋白酶降解成被机体吸收的氨基酸或寡肽。蚕丝具有良好的柔韧性,在材料本身的性能上与神经相接近[22]。 2.2.2 丝素蛋白的特性 丝素蛋白与细胞良好的生物相容性是其作为组织工程支架材料最基本的条件,丝素蛋白材料能够支持骨髓间充质干细胞、表皮细胞、成纤维细胞、神经细胞、嗅鞘细胞、血管内皮细胞等多种细胞的黏附、生长、发育、增殖、分化等[23]。Yang等[24]将冷冻干燥法制成的丝素蛋白支架植入到兔的皮下,6个月后质量降解到初始质量的65.7%,表明丝素蛋白支架材料能被生物降解和吸收且降解速率较慢;又联合静电纺丝技术制备出丝素蛋白神经导管修复SD大鼠的坐骨神经缺损,发现6个月后大鼠的坐骨神经损伤处愈合良好,证明了丝素蛋白生物材料能促进轴突的延伸和神经的再生。汤欣等[25]将海马神经元与丝素蛋白在体外共培养一段时间后,通过免疫细胞化学染色显示神经元突起生长,说明了中枢神经系统的神经组织细胞可以在丝素纤维表面生长、迁移。Dal Pra等[26]将丝素蛋白支架植入到大鼠皮下,6个月后观察支架周围的新生组织无明显的T细胞免疫反应,也没有纤维化生成。Rebecca等[27]也将丝素蛋白支架植入到大鼠的皮下,观察1年,发现只引起轻微的免疫反应,大多数丝素蛋白支架在2-6个月内完全降解。这些都证明了丝素蛋白具有良好的生物相容性和生物降解性。Meinel等[28]发现了丝素蛋白的免疫原性较低,不会引起明显的免疫应答和炎症反应,有利于细胞的黏附、分化和组织的形成。丝素蛋白材料能被降解和吸收,降解速率与丝素蛋白支架的结构和形貌有密切关系。一般来说,支架材料的孔隙越小孔隙越均匀,材料的机械性能越好,降解速率越慢[29]。有研究发现不同的冻干温度会影响到丝素蛋白空隙的大小和孔隙率,而高浓度的丝素蛋白溶液容易形成较小的孔隙[30]。与其他天然纤维蛋白相比,丝素蛋白生物力学性能较好,在体内降解较慢,降解产物与周围组织生物相容性好,并对组织有营养和修复作用。 2.2.3 丝素蛋白在脊髓组织工程中的应用 目前,国内外的研究已初步证明了丝素蛋白在神经修复方面的优势。丝素蛋白支架具备了作为脊髓组织工程支架的基本条件,使脊髓的可塑性增强。Fitch等[31]研究发现丝素蛋白支架可以对脊髓的断段起到桥接作用,支架移植后可以通过促进抑制分子的表达进而抑制瘢痕的形成,为细胞和分子的迁移提供了支持。王新宏[32]将丝素蛋白支架移植到SD大鼠脊髓全横断处,通过观察脊髓损伤后胶质瘢痕的形成和功能改善情况,发现丝素蛋白移植组较空白对照组形成的瘢痕组织少,并且移植组的BBB评分较高,研究结果表明丝素蛋白支架对减少胶质瘢痕和促进轴突的再生有一定作用。Shen等[33]将嗅鞘细胞与丝素蛋白纤维支架共培养,观察细胞的生长及迁移状况,发现丝素蛋白纤维支架与嗅鞘细胞具有良好的生物相容性,支架对嗅鞘细胞的生长和迁移起到支持和引导作用。因此,丝素蛋白材料也可以成为修复脊髓损伤的生物支架材料。 2.3 胶原/丝素蛋白在脊髓组织工程中的应用 将胶原蛋白和丝素蛋白用静电共纺的方法制备成复合体,可以在保持良好生物相容性的基础上,提高材料的物理性能。丝素蛋白的降解速度较缓慢,可以为细胞提供支持,以匹配细胞的生长速度,胶原蛋白与丝素蛋白相结合,改善胶原蛋白在干态下的物理性能的同时还可以改善膜的抗水性能,降低其热水溶失率[34]。Kim等[29]将胶原蛋白和丝素蛋白复合后,材料的粗糙度和表面形态发生变化,与单一材料相比,复合材料粗糙的表面使其与酶溶液的接触面积增加,加快支架材料的降解,改良了单一丝素蛋白支架材料降解缓慢的问题。吴刚等[22]将静电纺丝法制备的聚乳酸-羟基乙酸共聚物-胶原-丝素三维多孔支架与单纯的聚乳酸-羟基乙酸共聚物支架相比,发现聚乳酸-羟基乙酸共聚物-胶原-丝素三维多孔支架孔隙率和直径更适合许旺细胞的生长,细胞的相容性好,是一个良好的神经组织工程载体。随着组织工程学的不断发展,新材料、新工艺的不断出现,以胶原和丝素蛋白两种生物材料相结合,用目前最新的快速成型三维打印技术(Three-dimensional printing,3DP)控制脊髓支架导管的复杂外形及内部微细结构,对支架的结构进行优化,或许能在脊髓损伤修复方面获得突破,使脊髓损伤的修复出现新的希望。 "
| [1]Zhong Y,Bellamkonda RV.Biomaterials for the central nervous system.J R Soc Interface.2008;5(26):957-975.
[2]Edwards SL,Mitchell W,Matthews JB,et al. Design of nonwoven scaffold structures for tissue engineering of the anterior cruciate ligament.AUTEX Res J.2004;4 (2):86-94.
[3]Pakulska MM,Ballios BG,Shoichet MS.Injectable hydrogels for central nervous system therapy.Biomed Mater. 2012;7(2): 024101.
[4]Fan J,Xiao Z,Zhang H,et al.Linear Ordered Collagen Scaffolds Loaded with Collagen-Binding Neurotrophin-3 Promote Axonal Regeneration and Partial Functional Recovery after Complete Spinal Cord Transection.J Neurotrauma. 2010;27(9):1671-1683.
[5]Bae I,Osatomi K,Yoshida A,et al. Biochemical properties of acid-soluble collagens extracted from the skins of underutilised fishes.Food Chem.2008;108:49-54.
[6]Wallace DG,Rosenblatt J. Collagen gel systems for sustained delivery and tissue engieeering.Adv Drug Deliv Rev. 2003; 55(12):1631-1649.
[7]王培伟,陈宗刚,王红声,等.静电纺壳聚糖/胶原蛋白复合纳米纤维的细胞相容性[J].中国组织工程研究与临床康复, 2008,12(1): 5-9.
[8]Gerburg K,Felix S,Gerald W,et al.Bio-compatibility of type I/III collagen matrix for peripheral nerve reconstruction. Biomaterials.2003;24:2779-2787.
[9]Lin H,Chen B,Wang B, et al.Novel nerve guidance material prepared from bovine aponeurosis.J Biomed Mater Res. 2006; 79:591-598.
[10]Cao J,Sun C,Zhao H,et al.The use of laminin modified linear ordered collagen scaffolds loaded with laminin-binding ciliary neurotrophic factor for sciatic nerve regeneration in rats. Biomaterials. 2011;32(16):3939-3948.
[11]Han Q,Jin W,Xiao Z,et al.The promotion of neural regeneration in an extreme rat spinal cord injury model using a collagen scaffold containing a collagen binding neuroprotective protein and an EGFR neutralizing antibody. Biomaterials.2010;31:9212-9220.
[12]贾骏,段螈螈,陈亚芍,等.胶原改性PLGA电纺纤维的制备及其细胞相容性研究[J].临床口腔医学杂志,2007,23(6):323-325.
[13]Liang WB,Han QQ,Jin W, et al.The promotion of neurological recovery in the rat spinal cord crushed injury model by collagen-binding BDNF. Biomaterials.2010;31: 8634-8641.
[14]Lin PW,Wu CH,Chert CH,et al.Characterization of cortical neuron outgrowth in two and three-dimensional culture systems.J Biomed Mater Res B Appl Biomater.2005; 75(1): 146-157.
[15]White DJ,Puranen S,Johnson MS,et al.The collagen receptor subfamily of the integrins.Int J Biochem Cell Biol.2004;36(8): 1405-1410.
[16]Yoshii S,Oka M,Shima M,et al. Restoration of function after spinal cord transecti on using a collagen bridge.J Biomed Mater Res.2004;70(4):569-575.
[17]Liu S,Said G,Tadie M.Regrowth of the rostral spinal axons into the caudal ventral roots through a collagen tube implanted into hemisected adult rat spinal cord.Neurosurgery.2001; 49(1): 143-150.
[18]Liu T,Teng WK,Chan BP,et al.Photochemical crosslinked electrospun collagen nanofibers: Synthesis, characterization and neural stem cell interactions.J Biomed Mater Res A. 2010; 95(1):276-282.
[19]吴立志,曾园山,李海标,等.胶原或明胶吸附施万细胞移植促进全横断脊髓损伤修复的研究[J].解剖学报,2003,34(3):289-293.
[20]Giannetti S,Lauretti L,Fernandez E,et al.Acrylic hydrogel implan after spinal cord lesion in the adult rat.Neurol Res. 2001;23(4):405-409.
[21]Han Q,Sun W,Lin H,et al.Linear ordered collagen scaffolds loaded with collagen-binding brain derived neurotrophic factor improve the recovery of spinal cord injury in rats.Tissue Eng Part A.2009;15:2927-2935.
[22]吴刚,董长超,王光林,等.PLGA-丝素-胶原纳米三维多孔支架材料的制备及细胞相容性研究[J].中国修复重建外科杂志,2009, 23(8): 1007-1011.
[23]Zhang F,Zuo B,Zhang HX.Studies of electrospun regenerated SF/TSF nanofibers. Polymer.2009;50:279-285.
[24]Yang YM,Zhao YH.Degradation behaviors of nerve guidance conduits made up of silk fibroin in vitro and in vivo.Poly Degra Stabil.2009;94:2213-2220.
[25]汤欣,杨宇民,丁斐,等.丝素纤维与海马神经元的生物相容性细胞培养研究[J].交通医学,2006,20(5):491-493.
[26]Dal Pra I,Freddi G,Minic J,et al.De novo engineering of reticular connective tissue in vivoby silk fibroin nonwoven materials.Biomaterials.2005;26(14): 1987-1999.
[27]Rebecca LH,Antle K.In vitro degradation of silk fibroin. Biomaterials.2005; 26:3385-3393.
[28]Meinel L,Hofmann S,Karageorgiou V.The inflammatory responses to silk films in vitro and in vivo.Biomaterials.2005; 26(2):147-155.
[29]Kim UJ,Park J,Kim HJ,et al.Three-dimensional aqueous-derived biomaterial scaffolds from silk fibroin. Biomaterials.2005;26(15):2775-2785.
[30]Mandal BB,Kundu SC.Non-bioengineered silk fibroin protein 3D scaffolds for potential biotechnological and tissue engineering applications.Macromol Biosci.2008;8(9):807-818.
[31]Fitch MT,Silver J.CNS injury, glial scars, and inflammation: Inhibitory extracellular matrices and regeneration failure.Exp Neurol.2008;209(2):294-301.
[32]王新宏.丝素蛋白支架对大鼠脊髓损伤后胶质瘢痕形成和功能改善的影响[D].苏州大学,2012.
[33]Shen Y,Qian Y,Zhang H,et al.Guidance of olfactory ensheathing cell growth and migration on electrospun silk fibroin scaffolds.Cell Transplant. 2010;19(2):147-157.
[34]Levin B,Redmond SL,Rajkhowa R,et al.Utilising silk fibroin membranes as scaffolds for the growth of tympanic membrane keratinocytes, and application to myringoplasty surgery.J Laryngol Otol.2013;127 Suppl 1:S13-20. |
| [1] | Shen Jinbo, Zhang Lin. Micro-injury of the Achilles tendon caused by acute exhaustive exercise in rats: ultrastructural changes and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1190-1195. |
| [2] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [3] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [4] | Liu Liu, Zhou Qingzhu, Gong Zhuo, Liu Boyan, Yang Bin, Zhao Xian. Characteristics and manufacturing techniques of collagen/inorganic materials for constructing tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 607-613. |
| [5] | Xu Xiaoming, Chen Yan, Song Qian, Yuan Lu, Gu Jiaming, Zhang Lijuan, Geng Jie, Dong Jian. Human placenta derived mesenchymal stem cell gel promotes the healing of radiation skin damage in SD rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3976-3980. |
| [6] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [7] | Chen Lei, Zheng Rui, Jie Yongsheng, Qi Hui, Sun Lei, Shu Xiong. In vitro evaluation of adipose-derived stromal vascular fraction combined with osteochondral integrated scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3487-3492. |
| [8] | Yang Li, Li Xueli, Song Jinghui, Yu Huiqian, Wang Weixia. Effect of cryptotanshinone on hypertrophic scar of rabbit ear and its related mechanism [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3150-3155. |
| [9] | Liu Zhendong, Wang Rui, Li Xiaolei, Wang Jingcheng. Review of interferon alpha-2b inhibiting scar formation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 317-321. |
| [10] | Chen Siyu, Li Yannan, Xie Liying, Liu Siqi, Fan Yurong, Fang Changxing, Zhang Xin, Quan Jiayu, Zuo Lin. Thermosensitive chitosan-collagen composite hydrogel loaded with basic fibroblast growth factor retards ventricular remodeling after myocardial infarction in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2472-2478. |
| [11] | Chen Liang, Meng Shu, Cheng Guoping, Ding Yi . Effects of fish scale collagen membrane on adhesion, proliferation and osteogenic differentiation of rat bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2494-2499. |
| [12] | Zhang Hui, Wang Shaohua, Wang Qian, Wang Hui, Gan Hongquan, Cui Yishuang, Li Qijia, Wang Zhiqiang. Porous tantalum coated with RGD polypeptide can activate the integrin/focal adhesion kinase signaling pathway of MG63 cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2535-2540. |
| [13] | Li Shao, Liang Yongkang, Gao Yi, Peng Qing. Establishment and functional in vitro characteristics of three-dimensional collagen HepaRG microsphere [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2541-2547. |
| [14] | Chen Zhenyu, Zhang Xiaoning, Luo Yuxin, Liang Jianwei, Yan Chi. Evaluation of silk fibroin/curcumin composite film for promoting wound healing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2554-2561. |
| [15] | Liu Yunyi, Wang Bo, Wang Lin. Effects of post-exercise gastrocnemius needling on Achilles tendon degeneration in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(14): 2211-2218. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||