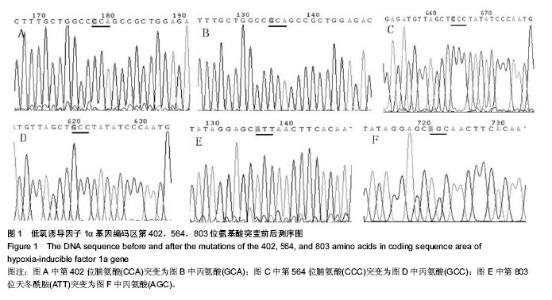
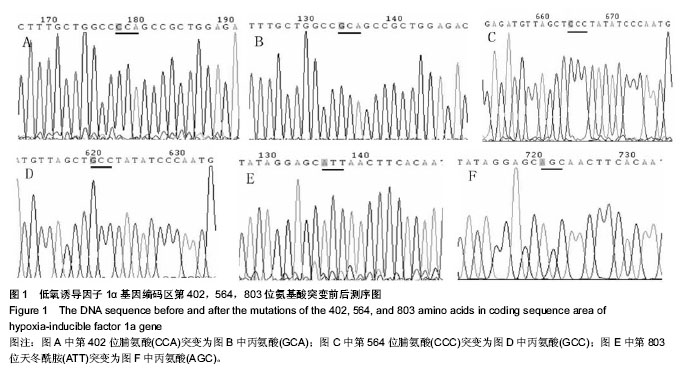
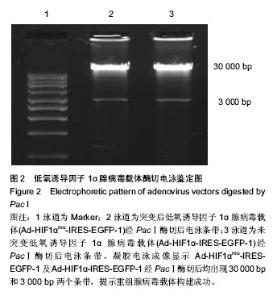
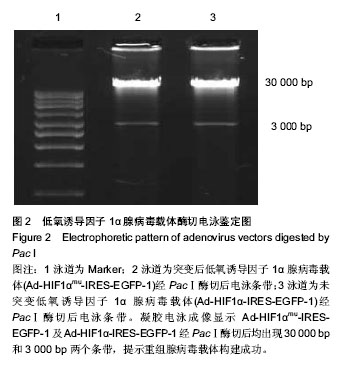
Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (19): 2953-2960.doi: 10.3969/j.issn.2095-4344.2014.19.001
In vitro transfection of triple-point mutants of hypoxia-inducible factor 1 alpha into bone marrow mesenchymal stem cells
Hui Chun-ying1, Xiao Hong-yan2, He Xin-ling1, Wang Wei3
- 1Department of Cardiocerebral Surgery, 2Department of Intervention Medicine, 3Department of Rehabilitation, First Affiliated Hospital of Liaoning Medical University, Jinzhou 121001, Liaoning Province, China
-
Revised:2014-02-26Online:2014-05-07Published:2014-05-07 -
Contact:Wang Wei, Master, Associate professor, Department of Rehabilitation, First Affiliated Hospital of Liaoning Medical University, Jinzhou 121001, Liaoning Province, China -
About author:Hui Chun-ying, Department of Cardiocerebral Surgery, First Affiliated Hospital of Liaoning Medical University, Jinzhou 121001, Liaoning Province, China -
Supported by:the Funded Project of the Social Development Department of Liaoning Science and Technology Bureau, No. 2012408002
CLC Number:
Cite this article
Hui Chun-ying, Xiao Hong-yan, He Xin-ling, Wang Wei. In vitro transfection of triple-point mutants of hypoxia-inducible factor 1 alpha into bone marrow mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(19): 2953-2960.
share this article
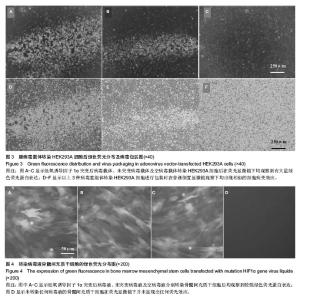
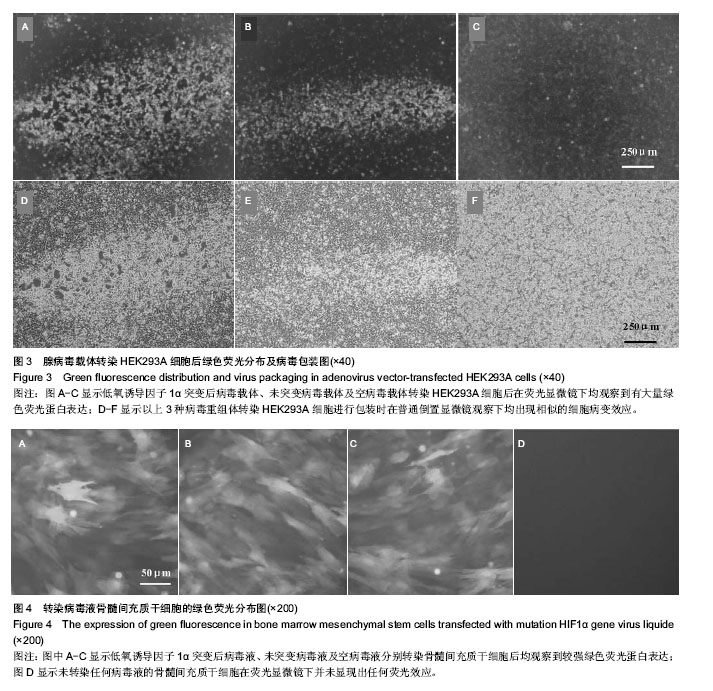
2.2 腺病毒包装结果 经3种病毒重组体转染的HEK293A细胞在荧光显微镜下均观察到有大量绿色荧光蛋白表达(图3A-C),同时在普通倒置显微镜下观察均出现相似的细胞病变效应:细胞触角回收、肿胀变圆,一部分细胞脱落,悬浮于视野中(图3D-F);病毒滴度检测结果依次为2.3×108 pfu/mL,2.6×108 pfu/mL和1.8×108 pfu/mL,符合后续转染实验要求。 2.3 病毒转染骨髓间充质干细胞表达结果 3种包装成功的病毒液在高倍数荧光显微镜下均观察到较强绿色荧光蛋白表达并且表达绿色荧光蛋白的大鼠骨髓间充质干细胞也较多,转染效率较高;而未转染任何病毒液的骨髓间充质干细胞在荧光显微镜下并未显现出任何荧光效应(图4)。"
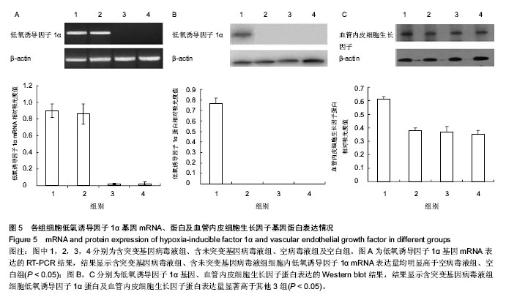
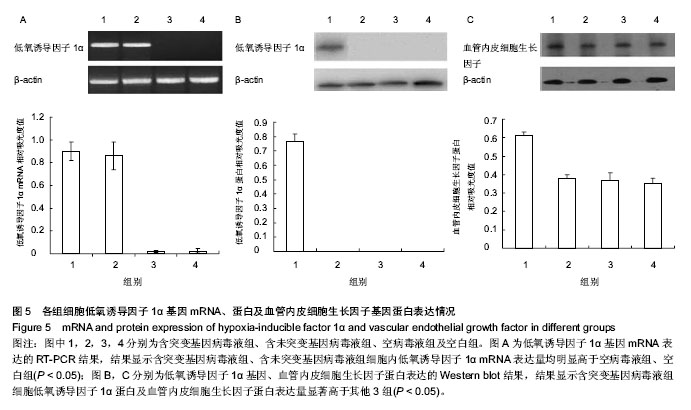
2.4 RT-PCR结果 含突变基因病毒液组、含未突变基因病毒液组细胞内低氧诱导因子1α mRNA表达相对吸光度值分别为0.85±0.06,0.83±0.07,两组差异无显著性意义(P > 0.05);空病毒液组、空白组低氧诱导因子1α mRNA的表达量非常低,几乎可以忽略,差异也无显著性意义(P > 0.05);而含突变基因病毒液组、含未突变基因病毒液组细胞内低氧诱导因子1α mRNA表达量均明显高于空病毒液组、空白组,差异有显著性意义(P < 0.05,图5A)。 2.5 Western blot结果 含突变基因病毒液组细胞低氧诱导因子1α蛋白及血管内皮细胞生长因子蛋白表达相对吸光度值分别为0.79±0.08和0.61±0.05,显著高于含未突变基因病毒液组、空病毒液组、空白组(P < 0.05);而含未突变基因病毒液组、空病毒液组、空白组3组差异均无显著性意义(P > 0.05,图5B,C)。结果显示,三点突变型低氧诱导因子1α不仅可以在常氧条件下高效表达,还能够促进其下游基因血管内皮细胞生长因子的高效表达。"
| [1] Kuniyoshi Y, Koja K, Miyagi K, et al. Prevention of postoperative paraplegia during thoracoabdominal aortic surgery. Ann Thorac Surg. 2003; 76(5): 1477-1484. [2] Dppolito G, Diabriras S, Howard GA, et al. Low oxygentension inhibits osteogenic differentiation and enhances stermness of human MIAMI cell. Bone. 2006; 39(3): 513-522. [3] Nishimura R, Okuda K. Hypoxia is important for establishing vascularization during corpus luteum formation in cattle. J Reprod Dev. 2010; 56(1): 110-116. [4] Giatromanolaki A, Fiska A, Pitsiava D, et al. Erythropoietin receptors in endometrial carcinoma as related to HIF1{alpha} and VEGF expression. In Vivo. 2009; 23(5): 699-703. [5] Seregin SS, Amalfitano A. Overcoming pre-existing adenovirus immunity by genetic engineering of adenovirus-based vectors. Expert Opin Biol Ther. 2009; 9(12): 1521-1531. [6] Fiorenzo P, Mongiardi MP, Dimitri D, et al. HIF1-positive and HIF1-negative glioblastoma cells compete in vitro but cooperate in tumor growth in vivo. Int J Oncol. 2010; 36(4): 785-791. [7] Wan C, Gilbert SR, Wang Y, et al. Activation of the hypoxia-inducible factor-1-alpha pathway accelerates. Proc Nat Acad Sci. 2008; 105(2): 686-691. [8] Giaccia A , Siim BG , Johnson RS. HIF-1 as a target for drug development. Nat Rev Drug Discov. 2003; 2(10): 803-811. [9] Khan TA , Sellke FW , Laham RJ. Gene therapy progress and prospects: therapeutic angiogenesis for limb and myocardial ischemia. Gene Ther. 2003; 10(4): 285-291. [10] Semenza GL. Evaluation of HIF-1 inhibitors as anticancer agents. Drug Discov Today. 2007; 12(19-20): 853-859. [11] Bruick RK , McKnight SL. A conserved family of prolyl- 4- hydroxylases that modify HIF. Science. 2001; 294(5545): 1337-1340. [12] Evans CE, Humphries J, Mattock K, et al. Hypoxia and upregulation of hypoxia- inducible factor 1{alpha} stimulate venous thrombus recanalization. Arterioscler Thromb Vasc Biol. 2010; 30(12): 2443-2451. [13] Lando D, Peet DJ, Whelan DA, et al. Asparagine hydroxylation of the HIF transactivation domain a hypoxic switch. Science. 2002; 295(5556): 858-861. [14] Zhang Z, Wang G, Li C, et al. Construction and characterization of a recombinant human adenovirus vector expressing bone morphogenetic protein 2. Exp Ther Med. 2013; 6(2):329-334. [15] Liu D, Hu L, Zhang Z, et al. Construction of human BMP2-IRES-HIF1αmu adenovirus expression vector and its expression in mesenchymal stem cells. Mol Med Rep. 2013; 7(2):659-663. [16] Emans PJ , Spaapen F , Surtel DA , et al. A novel in vivo model to study endochondral bone formation;HIF-1α activation and BMP expression. Bone。 2007; 40(2): 409-418. [17] Lim W, Cho J, Kwon HY, et al. Hypoxia-inducible factor 1 alpha activates and is inhibited by unoccupied estrogen receptor beta. FEBS Lett. 2009; 583(8): 1314-1318. [18] Thurston G, Suri C, Smith K, et al. Leakage-resistant blood vessels in mice transgenically overexpressing angiopoietin-1. Science. 1999; 286(5449):2511-2514. [19] Visconti RP, Richardson CD, Sato TN. Orchestration of angiogenesis and arteriovenous contribution by angiopoietins and vascular endothelial growth factor (VEGF). Proc Natl A cad Sci USA.2002; 99(12):8219-8224. [20] 朱国强,吴织芬,王勤涛,等. IGF-1对骨髓基质细胞分泌BMP-2和VEGF的影响[J].临床口腔医学杂志,2004, 20(4):215-217. [21] 郜勇,杨述华,杨操,等.人血管内皮细胞生长因子重组腺病毒载体的构建、鉴定及在骨髓间充质干细胞的表达[J].中华实验外科杂志,2006,23(2):138-140. [22] Fam NP, Verma S, Kutryk M, et al. Clinician guide to angiogenesis. Circulation. 2003; 108(21):2613-2618. [23] Bruick RK, McKnight SL. Building better vasculature. Genes Dev. 2001; 15(19): 2497-2502. [24] Semenza GL . HIF-1: mediator of physiological and pathophysiological responses to hypoxia. J Appl Physiol. 2000; 88(4):1474-1480. [25] Rebours V, Albuquerque M, Sauvanet A, et al. Hypoxia pathways and cellular stress activate pancreatic stellate cells: development of an organotypic culture model of thick slices of normal human pancreas. PLoS One. 2013; 8(9): e76229. [26] Cury V, Moretti AI, Assis L, et al. Low level laser therapy increases angiogenesis in a model of ischemic skin flap in rats mediated by VEGF, HIF-1α and MMP-2. J Photochem Photobiol B. 2013; 125: 164-170. [27] Pawlus MR, Hu CJ. Enhanceosomes as integrators of hypoxia inducible factor (HIF) and other transcription factors in the hypoxic transcriptional response. Cell Signal. 2013; 25(9): 1895-1903. [28] Ding G, Liu HD, Liang HX, et al. HIF1-regulated ATRIP expression is required for hypoxia induced ATR activation. FEBS Lett. 2013; 587(7): 930-935. [29] Tong Y, Li QG, Xing TY, et al. HIF1 regulates WSB-1 expression to promote hypoxia-induced chemoresistance in hepatocellular carcinoma cells. FEBS Lett. 2013; 587(16): 2530-2535. [30] Wu WJ, Zhang XK, Zheng XF, et al. SHH-dependent knockout of HIF-1 alpha accelerates the degenerative process in mouse intervertebral disc. Int J Immunopathol Pharmacol. 2013; 26(3): 601-609. [31] Huang C, Hales BF. Effects of exposure to a DNA damaging agent on the hypoxia inducible factors in organogenesis stage mouse limbs. PLoS One. 2012; 7(12): e51937. [32] Zimmermann SM, Würgler-Hauri CC, Wanner GA, et al. Echinomycin in the prevention of heterotopic ossification - an experimental antibiotic agent shows promising results in a murine model. Injury. 2013; 44(4):570-575. [33] Huang SM, Chen TS, Chiu CM, et al. GDNF increases cell motility in human colon cancer through VEGF-VEGFR1 interaction. Endocr Relat Cancer. 2013; 21(1): 73-84. [34] Steunou AL, Ducoux-Petit M, Lazar I, et al. Identification of the hypoxia-inducible factor 2α nuclear interactome in melanoma cells reveals master proteins involved in melanoma development. Mol Cell Proteomics. 2013; 12(3): 736-748. [35] Maher ER. HIF2 and endocrine neoplasia: an evolving story. Endocr Relat Cancer. 2013; 20(3): C5-7. [36] Bensellam M, Duvillié B, Rybachuk G, et al. Glucose-induced O? consumption activates hypoxia inducible factors 1 and 2 in rat insulin-secreting pancreatic beta-cells. PLoS One. 2012; 7(1): e29807. [37] Lange C, Caprara C, Tanimoto N, et al. Retina-specific activation of a sustained hypoxia-like response leads to severe retinal degeneration and loss of vision. Neurobiol Dis. 2011; 41(1):119-130. [38] Pawlus MR, Wang L, Ware K, et al. Upstream stimulatory factor 2 and hypoxia-inducible factor 2α (HIF2α) cooperatively activate HIF2 target genes during hypoxia. Mol Cell Biol. 2012; 32(22): 4595-4610. [39] Aragonés J, Elorza A, Acosta-Iborra B, et al. Myeloid hypoxia-inducible factors in inflammatory diseases. Crit Rev Immunol. 2011; 31(1): 1-13. [40] Simon F, Bockhorn M, Praha C, et al. Deregulation of HIF1-alpha and hypoxia- regulated pathways in hepatocellular carcinoma and corresponding non-malignant liver tissue--influence of a modulated host stroma on the prognosis of HCC. Langenbecks Arch Surg. 2010; 395(4): 395-405. [41] Wu XQ, Huang C, He X, et al. Feedback regulation of telomerase reverse transcriptase: new insight into the evolving field of telomerase in cancer. Cell Signal. 2013; 25(12): 2462-2468. [42] Coudyzer P, Lemoine P, Jordan BF, et al. Hypoxia is not required for human endometrial breakdown or repair in a xenograft model of menstruation. FASEB J. 2013; 27(9): 3711-3719. [43] Sena JA, Wang L, Hu CJ. BRG1 and BRM chromatin-remodeling complexes regulate the hypoxia response by acting as coactivators for a subset of hypoxia-inducible transcription factor target genes. Mol Cell Biol. 2013; 33(19): 3849-3863. [44] Shen G, Li X, Jia YF, et al. Hypoxia-regulated microRNAs in human cancer. Acta Pharmacol Sin. 2013; 34(3): 336-341. [45] Lin J, Qin X, Zhu Z, et al. FHL family members suppress vascular endothelial growth factor expression through blockade of dimerization of HIF1α and HIF1β. IUBMB Life. 2012; 64(11): 921-930. [46] Minervini G, Masiero A, Moro S, et al. In silico investigation of PHD-3 specific HIF1-α proline 567 hydroxylation: a new player in the VHL/HIF-1α interaction pathway? FEBS Lett. 2013; 587(18):2996-3001. [47] Green L, Cookson A, Bruce IN, et al. Identification of multiple, oxygen-stable HIF1 alpha isoforms, and augmented expression of adrenomedullin in rheumatoid arthritis. Clin Exp Rheumatol. 2013; 31(5):672-682. [48] Elson DA , Thurston G , Huang LE , et al. Induction of hypervascularity without leakage or inflammation in transgenic mice overexpressing hypoxia-inducible factor-1alpha. Genes Dev. 2001; 15(19): 2520-2532. [49] Li C, Liu D, Zhang Z, et al. Triple point-mutants of hypoxia-inducible factor-1α accelerate in vivo angiogenesis in bone defect regions. Cell Biochem Biophys. 2013; 67(2): 557-566. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [14] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [15] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||