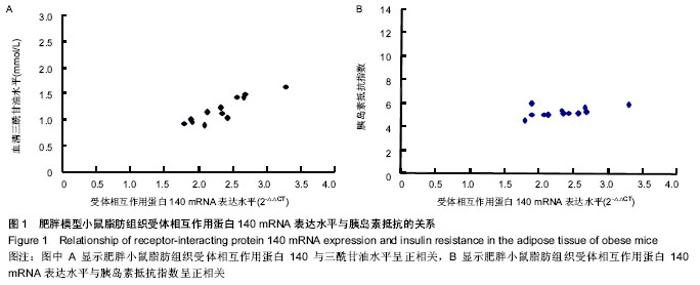
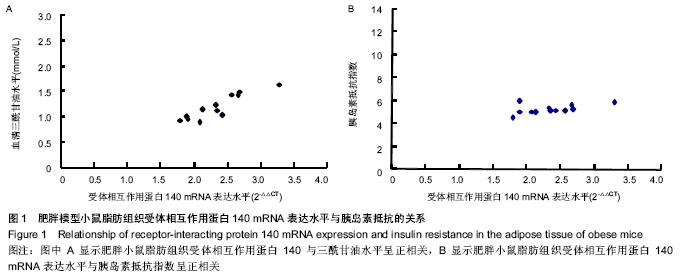
Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (18): 2892-2896.doi: 10.3969/j.issn.2095-4344.2014.18.018
Previous Articles Next Articles
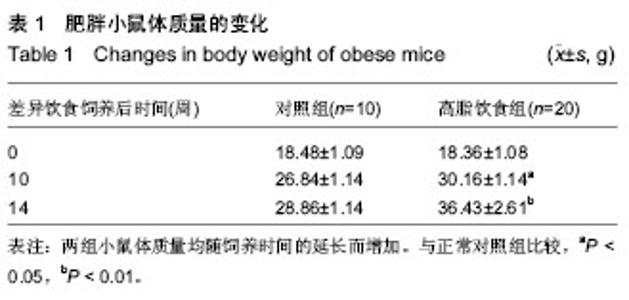
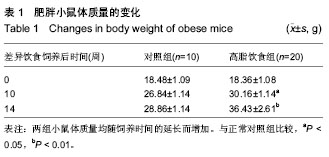
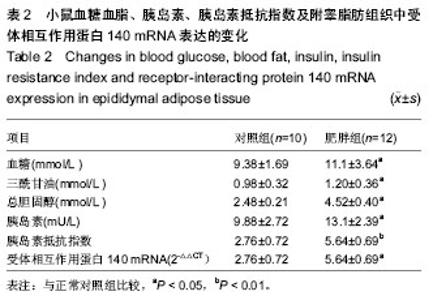
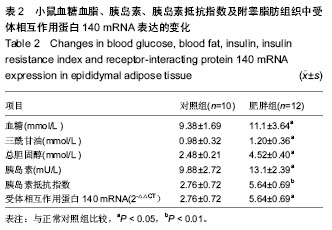
Receptor-interacting protein 140 mRNA expression in adipose tissue of a mouse model of high fat diet
Zhai Yan-pan, Wang Luan, Ma Rui-xin
- Department of Endocrinology, Affiliated Hospital of Qingdao University Medical College, Qingdao 266003, Shandong Province, China
-
Received:2014-04-08Online:2014-04-30Published:2014-04-30 -
Contact:Wang Luan, Master, Associate chief technician, Department of Endocrinology, Affiliated Hospital of Qingdao University Medical College, Qingdao 266003, Shandong Province, China Corresponding author: Ma Rui-xin, M.D., Chief physician, Professor, Department of Endocrinology, Affiliated Hospital of Qingdao University Medical College, Qingdao 266003, Shandong Province, China -
About author:Zhai Yan-pan, Studying for master’s degree, Department of Endocrinology, Affiliated Hospital of Qingdao University Medical College, Qingdao 266003, Shandong Province, China
CLC Number:
Cite this article
Zhai Yan-pan, Wang Luan, Ma Rui-xin. Receptor-interacting protein 140 mRNA expression in adipose tissue of a mouse model of high fat diet[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(18): 2892-2896.
share this article
| [1] Guyenet SJ, Schwartz MW. Clinical review: Regulation of food intake, energy balance, and body fat mass: implications for the pathogenesis and treatment of obesity. J Clin Endocrinol Metab. 2012;97(3):745-755. [2] Sethi JK, Vidal-Puig AJ. Targeting fat to prevent diabetes. Cell Metab. 2007;5(5):323-325. [3] Badman MK, Flier JS. The adipocyte as an active participant in energy balance and metabolism. Gastroenterology. 2007; 132(6):2103-2115. [4] Ahima RS, Lazar MA. Adipokines and the peripheral and neural control of energy balance. Mol Endocrinol. 2008; 22(5):1023-1031. [5] Francis GA, Fayard E, Picard F, et al. Nuclear receptors and the control of metabolism. Annu Rev Physiol. 2003;65: 261-311. [6] Desvergne B, Michalik L, Wahli W. Transcriptional regulation of metabolism. Physiol Rev. 2006;86(2):465-514. [7] Feige JN, Auwerx J. Transcriptional coregulators in the control of energy homeostasis. Trends Cell Biol. 2007;17(6): 292-301. [8] Herzog B, Hallberg M, Seth A, et al. The nuclear receptor cofactor, receptor-interacting protein 140, is required for the regulation of hepatic lipid and glucose metabolism by liver X receptor. Mol Endocrinol. 2007;21(11):2687-2697. [9] Rosell M, Jones MC, Parker MG. Role of nuclear receptor corepressor RIP140 in metabolic syndrome. Biochim Biophys Acta. 2011;1812(8):919-928. [10] Hallberg M, Morganstein DL, Kiskinis E, et al. A functional interaction between RIP140 and PGC-1alpha regulates the expression of the lipid droplet protein CIDEA. Mol Cell Biol. 2008;28(22):6785-6795. [11] Fritah A, Steel JH, Parker N, et al. Absence of RIP140 reveals a pathway regulating glut4-dependent glucose uptake in oxidative skeletal muscle through UCP1-mediated activation of AMPK. PLoS One. 2012;7(2):e32520. [12] Powelka AM, Seth A, Virbasius JV, et al. Suppression of oxidative metabolism and mitochondrial biogenesis by the transcriptional corepressor RIP140 in mouse adipocytes. J Clin Invest. 2006;116(1):125-136. [13] Ho PC, Lin YW, Tsui YC, et al. A negative regulatory pathway of GLUT4 trafficking in adipocyte: new function of RIP140 in the cytoplasm via AS160. Cell Metab. 2009;10(6):516-523. [14] Ho PC, Wei LN. Biological activities of receptor-interacting protein 140 in adipocytes and metabolic diseases. Curr Diabetes Rev. 2012;8(6):452-457. [15] Fritah A, Christian M, Parker MG. The metabolic coregulator RIP140: an update. Am J Physiol Endocrinol Metab. 2010; 299(3):E335-340. [16] O'Rahilly S. Human obesity and insulin resistance: lessons from experiments of nature. Novartis Found Symp. 2007;286: 13-23, 162-3, 196-203. [17] 陈蕾,贾伟平,项坤三,等.肥胖者胰岛素抵抗与总体脂、局部体脂关系的研究[J].中华内分泌代谢杂志,2001,17(5):276-279. [18] 中华人民共和国科学技术部.关于善待实验动物的指导性意见. 2006-09-30. [19] 杨爱君,崔雁,叶卉初,等.营养性肥胖动物模型的建立[J].临床和实验医学杂志,2005,4(3):156-157. [20] 石国富,张洪德,王丽娟,等. ELISA检测胰岛素的进一步研究[J].标记免疫分析与临床,1996(4):234. [21] Oliveira EP, Lima Md, Souza ML. Metabolic syndrome, its phenotypes, and insulin resistance by HOMA-IR. Arq Bras Endocrinol Metabol. 2007;51(9):1506-1515. [22] Duncan MH, Singh BM, Wise PH, et al. A simple measure of insulin resistance. Lancet. 1995;346(8967):120-121. [23] 张宏山,许明,张阳,等.小鼠心肌组织3种总RNA提取方法比较[J].临床心血管病杂志,2010,26(2):149-150. [24] 高海,李燕,周变华,等.动物脂肪代谢与肥胖基因调节研究进展[J].动物医学进展,2007,28(12):99-101. [25] Fortuño A, Rodríguez A, Gómez-Ambrosi J, et al. Adipose tissue as an endocrine organ: role of leptin and adiponectin in the pathogenesis of cardiovascular diseases. J Physiol Biochem. 2003;59(1):51-60. [26] Kahn SE, Hull RL, Utzschneider KM. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature. 2006;444(7121):840-846. [27] Calle EE. Obesity and cancer. BMJ. 2007;335(7630): 1107-1108. [28] Stefan N, Häring HU, Hu FB, et al. Metabolically healthy obesity: epidemiology, mechanisms, and clinical implications. Lancet Diabetes Endocrinol. 2013;1(2):152-162. [29] Cavaillès V, Dauvois S, L'Horset F, et al. Nuclear factor RIP140 modulates transcriptional activation by the estrogen receptor. EMBO J. 1995;14(15):3741-3751. [30] Lee CH, Chinpaisal C, Wei LN. Cloning and characterization of mouse RIP140, a corepressor for nuclear orphan receptor TR2. Mol Cell Biol. 1998;18(11):6745-6755. [31] Chen WH. Turning down the heat: the potential role of RIP140 in inflammation. Cell Mol Immunol. 2012;9(3):195-196. [32] Leonardsson G, Steel JH, Christian M, et al. Nuclear receptor corepressor RIP140 regulates fat accumulation. Proc Natl Acad Sci U S A. 2004;101(22):8437-8442. [33] Christian M, Kiskinis E, Debevec D, et al. RIP140-targeted repression of gene expression in adipocytes. Mol Cell Biol. 2005;25(21):9383-9391. [34] 查锡良.生物化学[M].7版.北京:人民卫生出版社,2008. [35] 李光伟,李春梅,孙淑湘,等.胰岛素抵抗--遗传和环境因素致高血压的共同途径?[J].中华内科杂志,2003,42(1):11-15. [36] 周鹏,陈南衡,王其民.高甘油三酯血症与胰岛素抵抗及糖代谢异常的关系[J].中华内科杂志,1998,37(7):447-450. [37] 窦梅,马爱国.胰岛素抵抗主要原因及机制的研究进展[J].国外医学卫生学分册,2009,36(3):175-179. [38] 李伶,杨刚毅.脂代谢紊乱和脂肪细胞因子与胰岛素抵抗[J].中国糖尿病杂志,2007,15(3):129-131. [39] Kamon J, Yamauchi T, Terauchi Y, et al. The mechanisms by which PPARgamma and adiponectin regulate glucose and lipid metabolism. Nihon Yakurigaku Zasshi. 2003;122(4): 294-300. [40] Yano W, Kubota N, Itoh S, et al. Molecular mechanism of moderate insulin resistance in adiponectin-knockout mice. Endocr J. 2008;55(3):515-522. [41] Yamauchi T, Kamon J, Minokoshi Y, et al. Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat Med. 2002; 8(11):1288-1295. [42] Gupta P, Ho PC, Huq MD, et al. PKCepsilon stimulated arginine methylation of RIP140 for its nuclear-cytoplasmic export in adipocyte differentiation. PLoS One. 2008;3(7): e2658. [43] Christian M, White R, Parker MG. Metabolic regulation by the nuclear receptor corepressor RIP140. Trends Endocrinol Metab. 2006;17(6):243-250. [44] White R, Morganstein D, Christian M, et al. Role of RIP140 in metabolic tissues: connections to disease. FEBS Lett. 2008; 582(1):39-45. |
| [1] | Chen Ziyang, Pu Rui, Deng Shuang, Yuan Lingyan. Regulatory effect of exosomes on exercise-mediated insulin resistance diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 4089-4094. |
| [2] | Liu Yulin, Li Guotai. Combined effects of hyperbaric oxygen, vibration training and astaxanthin on bone mineral density, glucose metabolism and oxidative stress in diabetic osteoporosis rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3117-3124. |
| [3] | Wang Chaoge, Weng Xiquan, Lin Baoxuan, Chen Lina, Xu Guoqin. Exercises under cold exposure change fat type and function in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3162-3167. |
| [4] | Huang Zhusong, Lin Yu, Chen Xiang, Lan Jinfu, Guan Yong, Gao Xi. Alcohol extract of Morinda officinalis improves lipid metabolism and bone metabolism in ovariectomized obese rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 205-210. |
| [5] | Li Zihan, Yin Wen, Sun Wei, Zhu Kun, Shen Di, Liu Yujia. An 8-week aerobic exercise promotes the function of voltage-dependent potassium channel in mesenteric vascular smooth muscle from obese rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2630-2635. |
| [6] | Zhang Lei, Yan Yu, Liu Yin, Xu Long, Yang Xinglei, Liu Yujia. An 8-week aerobic exercise improves obesity-induced myocardial fibrosis: role of nuclear factor-erythroid 2 p45-related factor 2 pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2650-2656. |
| [7] | Zhang Chuanhui, Li Jianjun, Yang Jun. Effects of dynamic pressure on the proliferation and mechanical properties of rabbit adipose mesenchymal stem cells transfected with insulin-like growth factor-1 [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 1982-1987. |
| [8] | Yang Luyao, Fu Pengyu, Tang Shuning, Zhu Rongxin, Gong Lijing . Change of Ghrelin-GHSR pathway in 4-week intermittent hypoxic exposure improving obesity in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1733-1739. |
| [9] | Cao Guolong, Tian Faming, Liu Jiayin. Lovastatin combined with insulin effects on fracture healing in rat models of bilateral ovariectomized type 2 diabetic mellitus [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(5): 673-681. |
| [10] | Ling Xuejian, Yang Qingchun, Zhang Yanding, Ma Gang. Effect of Noggin overexpression in hair follicle dermal sheath cells on the growth of skin and hair follicles [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5013-5017. |
| [11] |
Li Ying, Lin Wentao, Weng Xiquan.
Effects of different exercise intensities on visfatin level and glucose metabolism in type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(26): 4196-4200. |
| [12] | Jiang Han, Ding Jie, Chen Gen, Xie Qiang, Li Wei, Wang Zhibin, Zhu Lei. Hypoxia-mediated miRNAs affect glucose metabolism: new recognition of preventing and controlling glucose metabolism [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(2): 303-311. |
| [13] | Li Hong, Wu Shaohua, Qiu Mingxing, Deng Qingfu, Lei Guolin, Li Qinglong. Mechanism underlying basic fibroblast growth factor and insulin-like growth factor-1 effects on proliferation and apoptosis of spermatogonial stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(19): 3017-3022. |
| [14] | Liu Xing, Wei Xiaohan, Deng Jie, Li Zhongming. Resveratrol repairs skeletal muscle injury by up-regulating basic fibroblast growth factor and insulin-like growth factor 1 [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(14): 2184-2191. |
| [15] | Zhao Jianfeng, Geng Yu, Chen Qianbo, Yang Jinghui, Li Yan. Changes in insulin resistance and inflammatory factors in cataract patients with glaucoma after phacoemulsification and trabeculectomy: a self-controlled trial [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(11): 1750-1755. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||