Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (11): 1672-1679.doi: 10.3969/j.issn.2095-4344.2014.11.006
Previous Articles Next Articles
Low-density extracorporeal shock wave and low-dose intermittent recombinant human parathyroid hormone 1-34 influence proliferation and differentiation of osteoblasts
Liu Chang-jian1, Wang Li2, Luo Zong-jian3
- 1 Department of Orthopedics, First Affiliated Hospital of Dalian Medical University, Dalian 116001, Liaoning Province, China; 2 Department of Radiology, Dalian Friendship Hospital, Dalian 116001, Liaoning Province, China ; 3 Department of Orthopedics, First Affiliated Hospital, Changchun University of Chinese Medicine, Changchun 130011, Jilin Province, China
-
Revised:2014-01-07Online:2014-03-12Published:2014-03-12 -
Contact:Luo Zong-jian, M.D., Associate professor, Department of Orthopedics, First Affiliated Hospital, Changchun University of Chinese Medicine, Changchun 130011, Jilin Province, China -
About author:刘长剑,男,1973年生,辽宁省大连市人,汉族,2008年吉林大学白求恩医学部毕业,博士,副教授。
CLC Number:
Cite this article
Liu Chang-jian, Wang Li, Luo Zong-jian. Low-density extracorporeal shock wave and low-dose intermittent recombinant human parathyroid hormone 1-34 influence proliferation and differentiation of osteoblasts[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(11): 1672-1679.
share this article
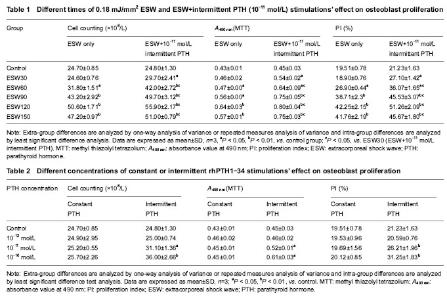
Detection of cell proliferation by cell counting with typan blue staining Different times of 0.18 mJ/mm2ESW stimulations’ effect on cell growth The cell count was promoted in turn as stimulating times of ESW ranging from 30 to 120, and the differences were significant (P < 0.05). But the cell number of the control and 30-time ESW stimulation group showed no difference (Table 1). Different patterns of rhPTH1-34 stimulations’ effect on cell growth Constant stimulations of 10-12,10-11 and 10-10 mol/L rhPTH1-34 had no effects on cell number. Intermittent stimulations of 10-11 and 10-10 mol/L rhPTH1-34 could promote cell number significantly compared with controls and constant stimulation groups (P < 0.05). Cell number of 10-10 mol/L rhPTH1-34 group was higher than that of 10-11 mol/L rhPTH1-34 group (P < 0.05; Table 2). Cooperation of 30-150 times of ESW and 10-11mol/L intermittent rhPTH1-34 stimulations’ effect on cell growth Cell number of 30-150 times ESW+10-11 mol/L rhPTH1-34 intermittent stimulation groups was respectively higher than that of only ESW stimulation groups (P < 0.05; Table 1). Cell number of 60-150 times of ESW+10-11 mol/L rhPTH1-34 intermittent stimulation groups were higher than that of only 10-11 mol/L rhPTH1-34 intermittent stimulation group (P < 0.05; Tables 1, 2). Detection of cell proliferation by MTT test Different times of 0.18 mJ/mm2 ESW stimulations’ effect on cell proliferation As stimulation time rose from 30 to 120 in turn, A490 nm value rose also. Except the differences between 30 times and control group, 90 and 150 times group, the differences of A490 nm among 30-150 times ESW stimulation groups were significant (P < 0.05; Table1). Different patterns of rhPTH1-34 stimulations’ effect on cell proliferation Constant stimulations of 10-12, 10-11 and 10-10 mol/L rhPTH1-34 had no effects on cell proliferation. Intermittent stimulations of 10-11 and 10-10 mol/L rhPTH1-34 could promote A490 nm value significantly as compared with controls and constant stimulation groups (P < 0.05). The A490 nm value of 10-10 mol/L intermittent rhPTH1-34 stimulation group was higher than that of 10-11 mol/L intermittent rhPTH1-34 stimulation group (P < 0.05; Table 2). Cooperation of 30-150 times of ESW and 10-11 mol/L intermittent rhPTH1-34 stimulations’ effect on cell proliferation A490 nm values of 30-150 times ESW+10-11 mol/L rhPTH1-34 intermittent stimulation groups were respectively higher than those of only ESW stimulation groups (P < 0.05; Table 3). A490 nm values of 60-150 times of ESW+10-11 mol/L rhPTH1-34 intermittent stimulation groups were higher than those of only 10-11 mol/L rhPTH1-34 intermittent stimulation group (P < 0.05; Tables 1, 2). "
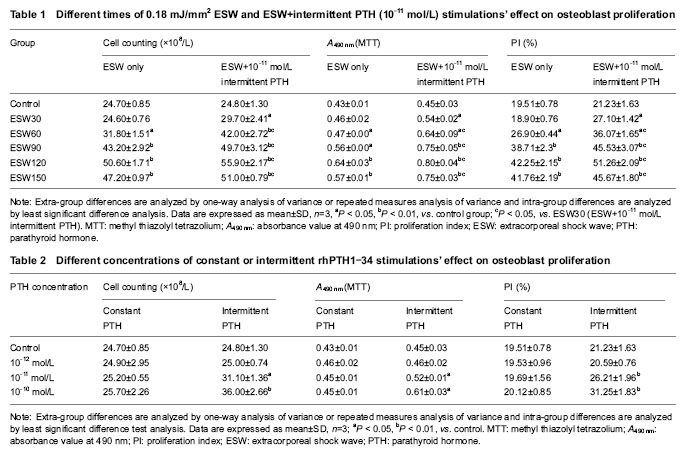

Detection of PI value by flow cytometry Different times of 0.18 mJ/mm2 ESW stimulations’ effect on PI As stimulation time rose from 30 to 120 in turn, PI value rose also. Except the differences between 30 times and control groups, and between 120 and 150 times groups, the differences of PI among 30-150 times ESW stimulation groups were significant (P < 0.05; Table 1). Different patterns of rhPTH1-34 stimulates’ effect on PI Constant stimulations of 10-12, 10-11 and 10-10 mol/L rhPTH1-34 had no effects on PI. Intermittent stimulations of 10-11 and 10-10 mol/L rhPTH1-34 could promote PI value significantly compared with controls and constant stimulation groups (P < 0.05). PI value of 10-10 mol/L rhPTH1-34 intermittent stimulation group was higher than that of 10-11 mol/L intermittent rhPTH1-34 stimulation group (P < 0.05; Table 3). Cooperation of 30-150 times of ESW and 10-11 mol/L pulse rhPTH1-34 stimulations’ effect on PI PI values of 30-150 times ESW+10-11 mol/L rhPTH1-34 intermittent stimulation groups were respectively higher than those of only ESW stimulation groups (P < 0.05; Table 1). PI values of 60-150 times ESW+10-11 mol/L rhPTH1-34 intermittent stimulation groups were higher than those of only 10-11 mol/L rhPTH1-34 intermittent stimulation group (P < 0.05; Tables 1, 2). Detection of osteoblast differentiation through ALP activity measurement Different times of 0.18 mJ/mm2 ESW stimulations’ effect on ALP activity As stimulation time rose from 30 to 120 in turn, ALP activity rose also. Except the differences between 30 times and control groups, and between 90 and 150 times groups, the differences of ALP activity among 30-150 times ESW stimulation groups were significant (P < 0.05; Table 3). "
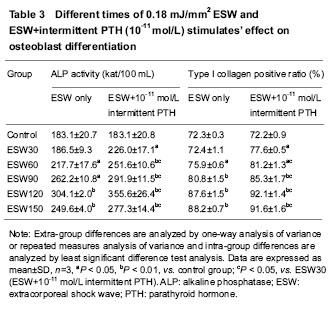
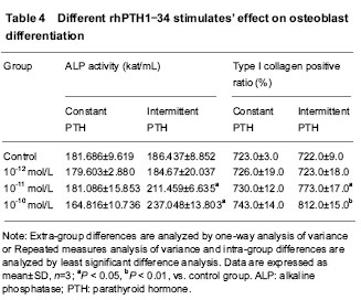
Different patterns of rhPTH1-34 stimulations’ effect on ALP activity Constant stimulations of 10-12, 10-11 and 10-10 mol/L rhPTH1-34 had no effects on ALP activity. Intermittent stimulations of 10-11 and 10-10 mol/L rhPTH1-34 could promote ALP activity significantly compared with controls and constant stimulation groups (P < 0.05). ALP activity of 10-10 mol/L rhPTH1-34 intermittent stimulation group was higher than that of 10-11 mol/L rhPTH1-34 intermittent stimulation group (P < 0.05; Table 4)."
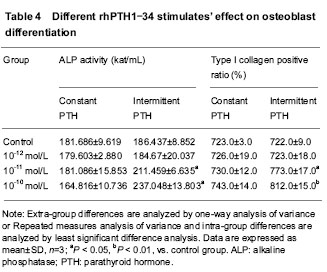

Cooperation of 30-150 times of ESW and 10-11 mol/L intermittent rhPTH1-34 stimulations’ effect on ALP activity ALP activities of 30-150 times ESW+10-11 mol/L rhPTH1-34 intermittent stimulation groups were respectively higher than those of only ESW stimulation groups (P < 0.05; Table 3). ALP activities of 60-150 times of ESW+10-11 mol/L rhPTH1-34 intermittent stimulation groups were higher than that of only 10-11 mol/L rhPTH1-34 stimulation group (P < 0.05; Tables 3, 4). Detection of type I collagen expression by immunocytochemistry Different times of 0.18 mJ/mm2 ESW stimulations’ effect on type I collagen expression As the stimulation time rose from 30 to 150 in turn, the ratio of type I collagen positive cells rose also. Except the differences between 30 times and control groups, and between 120 and 150 times groups, the differences of positive cell ratio among 30-150 times ESW stimulation groups were significant (P < 0.05; Table 3). Different patterns of rhPTH1-34 stimulates’ effect on type I collagen expression Constant stimulations of 10-12, 10-11 and 10-10 mol/L rhPTH1-34 had no effects on type I collagen positive cell ratio. Intermittent stimulations of 10-11 and 10-10 mol/L rhPTH1-34 could promote type I collagen positive cell ratio significantly compared with controls and constant stimulation groups (P < 0.05). Type I collagen positive cell ratio of 10-10 mol/L rhPTH1-34 intermittent stimulation group was higher than that of 10-11 mol/L rhPTH1-34 intermittent stimulation group (P < 0.05; Table 4). Cooperation of 30-150 times of ESW and 10-11 mol/L intermittent rhPTH1-34 stimulations’ effect on type I collagen expression Type I collagen positive cell ratios of 30-150 times ESW+10-11 mol/L rhPTH1-34 intermittent stimulation groups were respectively higher than those of only ESW stimulation groups (P < 0.05; Table3). Type I collagen positive cell ratios of 60-150 times of ESW+10-11 mol/L rhPTH1-34 intermittent stimulation groups were higher than that of only 10-11 mol/L rhPTH1-34 intermittent stimulation group (P < 0.05; Tables 3, 4)."

| [1] Saussine C. Extracorporeal shock wave lithotripsy. Prog Urol. 2013;23(14):1168-1171. [2] Kolk A, Auw Yang KG, Tamminga R, et al. Radial extracorporeal shock-wave therapy in patients with chronic rotator cuff tendinitis: a prospective randomised double-blind placebo-controlled multicentre trial. Bone Joint J. 2013;95-B(11):1521-1526. [3] Hausner T, Nógrádi A.The use of shock waves in peripheral nerve regeneration: new perspectives? Int Rev Neurobiol. 2013;109:85-98. [4] Tailly GG. Extracorporeal shock wave lithotripsy today. Indian J Urol. 2013;29(3):200-207.[5] Liu J, Zang YJ. Comparative study between three analgesic agents for the pain management during extracorporeal shock wave lithotripsy. Urol J. 2013;26; 10(3):942-945.[6] Mikami Y, Matsumoto T, Kano K, et al. Current status of drug therapies for osteoporosis and the search for stem cells adapted for bone regenerative medicine. Anat Sci Int. 2014;89(1):1-10.[7] Teotia PK, Hussein KE, Park KM, et al. Mouse adipose tissue-derived adult stem cells expressed osteogenic specific transcripts of osteocalcin and parathyroid hormone receptor during osteogenesis. Transplant Proc. 2013;45(8): 3102-3107. [8] Sheyn D, Cohn Yakubovich D, Kallai I, et al. PTH promotes allograft integration in a calvarial bone defect. Mol Pharm. 2013;10(12):4462-4471. [9] Smith BJ, Bu SY, Wang Y, et al. Comparative study of the bone metabolic response to dried plum supplementation and PTH treatment in adult, osteopenic ovariectomized rat. Bone. 2013;58C:151-159. [10] Zhong G, Pei FX, Li SF, et al. Experimental study on the proliferation and function of osteoblast cell induced by pBLAST49-mVEGF gene transfection. Sichuan Da Xue Xue Bao Yi Xue Ban. 2006;37(1):44-47.[11] Ejersted C, Andreassen TT, Nilsson MHL, et al. Human parathyroid hormone(1-34) increases bone formation and strength of cortical bone in aged rats. Europe J Endocrinol. 1994;130(2):201-207.[12] Xu J, Rong H, Ji H, et al. Effects of different dosages of parathyroid hormone-related protein 1-34 on the bone metabolism of the ovariectomized rat model of osteoporosis. Calcif Tissue Int. 2013;93(3):276-287.[13] Chen YJ, Kuo YR, Yang KD, et al. Activation of extracellular signal-regulated kinase (ERK) and p38 kinase in shock wave-promoted bone formation of segmental defect in rats. Bone. 2004;34(3):466-477.[14] Wang FS, Yang KD, Kuo YR, et al. Temporal and spatial expression of bone morphogenetic proteins in extracorporeal shock wave-promoted healing of segmental defect. Bone. 2003;32(4):387-396.[15] 15 Ishizuya T, Yokose S, Hori M, et al. Parathyroid hormone exerts disparate effects on osteoblast differentiation depending on exposure time in rat osteoblastic cells. J Clin Invest. 1997;99(12):2961-2970.[16] UzawaT, HoriM, Ejiri S, et al. Comparison of the effects of intermittent and continuous administration of human Parathyroid hormone (1-34) on rat bone. Bone. 1995;16(4): 477-484.[17] Wang YH, Qiu Y, Han XD, et al. Haploinsufficiency of endogenous parathyroid hormone-related peptide impairs bone fracture healing. Clin Exp Pharmacol Physiol. 2013; 40(11):715-723. [18] Khan MP, Mishra JS, Sharan K, et al. A novel flavonoid C-glucoside from Ulmus wallichiana preserves bone mineral density, microarchitecture and biomechanical properties in the presence of glucocorticoid by promoting osteoblast survival: a comparative study with human parathyroid hormone. Phytomedicine. 2013;20(14):1256-1266. [19] Hamedifar H, Salamat F, Saffarion M, et al. a novel approach for high level expression of soluble recombinant human parathyroid hormone (rhPTH 1-34) in Escherichia coli. Avicenna J Med Biotechnol. 2013;5(3):193-201.[20] Pereira Vasconcelos DF, Marques MR, Benatti BB, et al. Intermittent PTH administration improves periodontal healing in rats. J Periodontol. 2013.[21] Klein-Nulend J, Semeins CM, Burger EH. Metabolism in primary mouse osteoblastic Prostaglandin mediated modulation of transforming growth factor-β metabolism in primary mouse osteoblastic cells in vitro. J Cell Physiol. 1996;168:1-7.[22] Reich KM, Mcallister TN, Gudi S. Activation of G protein mediates flow-induced prostaglandin E2 production in osteoblast. Eradocranology. 1997;138:1014-1018.[23] Ajubi NE, Klein-Nulend J, Nijweide PJ, et al. Pulsatile fluid flow increases prostaglandin production by cultured chicken osteocytes--a cytoskeleton-dependent process. Biochem Biophys Res Commun. 1996;225:62-68.[24] Ueberle F, Rad AJ. Characterization of unfocused/weakly focused pressure pulse sources for extracorporeal pain therapy (“Radial Shock Wave Therapy” Sources). Biomed Tech (Berl). 2013. [25] Zhang X, Yan X, Wang C, et al. The dose-effect relationship in extracorporeal shock wave therapy: the optimal parameter for extracorporeal shock wave therapy. J Surg Res. 2014;186(1):484-492. [26] Moon SW, Kim JH, Jung MJ, et al. The effect of extracorporeal shock wave therapy on lower limb spasticity in subacute stroke patients. Ann Rehabil Med. 2013;37(4): 461-470. [27] Schmitz C, Császár NB, Rompe JD, et al. Treatment of chronic plantar fasciopathy with extracorporeal shock waves (review). J Orthop Surg Res. 2013;8(1):31. [28] Troncati F, Paci M, Myftari T, et al. Extracorporeal shock wave therapy reduces upper limb spasticity and improves motricity in patients with chronic hemiplegia: a case series. NeuroRehabilitation. 2013;33(3):399-405.[29] Qin L, Fok P, Lu H, et al. Low intensity pulsed ultrasound increases the matrix hardness of the healing tissues at bone-tendon insertion-a partial patellectomy model in rabbits. Clin Biomech (Bristol, Avon). 2006;21(4):387-394.[30] Vulpiani MC, Vetrano M, Conforti F, et al. Effects of extracorporeal shock wave therapy on fracture nonunions. Am J Orthop (Belle Mead NJ). 2012;41(9):E122-E127.[31] Fovargue DE, Mitran S, Smith NB, et al. Experimentally validated multiphysics computational model of focusing and shock wave formation in an electromagnetic lithotripter. J Acoust Soc Am. 2013;134(2):1598-609. [32] Speed C. A systematic review of shockwave therapies in soft tissue conditions: focusing on the evidence. Br J Sports Med. 2013. [33] Ha CH, Kim S, Chung J, et al. Extracorporeal shock wave stimulates expression of the angiogenic genes via mechanosensory complex in endothelial cells: mimetic effect of fluid shear stress in endothelial cells. Int J Cardiol. 2013;168(4):4168-4177. [34] Császár NB, Schmitz C. Extracorporeal shock wave therapy in musculoskeletal disorders. J Orthop Surg Res. 2013;8:22. [35] Suhr F, Delhasse Y, Bungartz G, et al. Cell biological effects of mechanical stimulations generated by focused extracorporeal shock wave applications on cultured human bone marrow stromal cells. Stem Cell Res. 2013;11(2): 951-964. [36] Yasuda H, Shima N, Nakagawa N, et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/ osteogenesis inhibitory factor and is identical to TRANCE/RANKL. Proc Nad Acad Sci USA. 1998;95: 3597-3602.[37] Saini V, Marengi DA, Barry KJ, et al. Parathyroid hormone (PTH)/PTH-related peptide type 1 receptor (PPR) signaling in osteocytes regulates anabolic and catabolic skeletal responses to PTH. J Biol Chem. 2013;288(28): 20122-20134. [38] Dossing DA, Radeff JM, Sanders J, et al. Parathyroid hormone stimulates translocation of protein kinase C isozymes in UMR-106 osteoblastic osteosarcoma cells. Bone. 2001;29(3):223-230.[39] Lee SJ, Kang JH, Kim JY, et al. Dose-related effect of extracorporeal shock wave therapy for plantar fasciitis. Ann Rehabil Med. 2013;37(3):379-388. [40] Raabe O, Shell K, Goessl A, et al. Effect of extracorporeal shock wave on proliferation and differentiation of equine adipose tissue-derived mesenchymal stem cells in vitro. Am J Stem Cells. 2013;2(1):62-73. [41] Rao DS, Parikh N, Palnitkar S, et al. The effect of endogenous parathyroid hormone in iliac bone structure and turnover in healthy postmenopausal women. Calcif Tissue Int. 2013;93(3):288-295.[42] Tamura Y, Kaji H. Parathyroid hormone and Wnt signaling. Clin Calcium. 2013;23(6):847-852. |
| [1] | Jiang Yong, Luo Yi, Ding Yongli, Zhou Yong, Min Li, Tang Fan, Zhang Wenli, Duan Hong, Tu Chongqi. Von Mises stress on the influence of pelvic stability by precise sacral resection and clinical validation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1318-1323. |
| [2] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [3] | Yang Weiqiang, Ding Tong, Yang Weike, Jiang Zhengang. Combined variable stress plate internal fixation affects changes of bone histiocyte function and bone mineral density at the fractured end of goat femur [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 890-894. |
| [4] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [5] | Zhang Guomei, Zhu Jun, Hu Yang, Jiao Hongwei. Stress of three-dimensional finite element models of E-MAX porcelain inlay [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 537-541. |
| [6] | Li Kun, Li Zhijun, Zhang Shaojie, Gao Shang, Sun Hao, Yang Xi, Wang Xing, Dai Lina . A 4-year-old child model of occipito-atlanto-axial joints established by finite element dynamic simulation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3773-3778. |
| [7] | Shu Qihang, Liao Yijia, Xue Jingbo, Yan Yiguo, Wang Cheng. Three-dimensional finite element analysis of a new three-dimensional printed porous fusion cage for cervical vertebra [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3810-3815. |
| [8] | Qin Wan’an, Cai Zhouyu, Wei Gejin, Lin Zhoudan. Finite element analysis of absorbable screws and ethibond sutures for the treatment of humerus shaft fractures caused by grenade throwing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3855-3859. |
| [9] | Zou Shouping, Lu Daoyun, Ye Li. Minimally invasive percutaneous pedicle screw technique for thoracolumbar fractures: biomechanical changes of the spine during 6-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3865-3869. |
| [10] | Zhang Qifu, Ma Yonghong, Wang Tao, Hu Yibo, Zhang Heling, Zong Qunchuan. Effects of anterior corpectomy and fusion versus posterior single open-door laminoplasty on cervical range of motion of patients with multilevel cervical spondylotic myelopathy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3870-3874. |
| [11] | Jie Ke, Deng Peng, Zeng Yirong. Application and comparison of four commonly used methods for patellar height measurement [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3875-3881. |
| [12] | Wang Ziao, Song Wenhui, Liu Changwen . Short-segment fixation of thoracolumbar burst fractures: method modification and strategies to reduce failure [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3902-3907. |
| [13] | Liu Jinwei, Chen Yunzhen, Wan Chunyou. Changes of osteogenic growth factors in the broken end of bone nonunion under stress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3619-3624. |
| [14] | Luo Anyu, Liu Hanlin, Xie Xiaofei, Huang Chen. Effect of antioxidant mixture on structural degeneration of an osteoarthritis rat model [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3625-3629. |
| [15] | Wang Zhen, Lin Haiqi, He Fei, Lin Wentao. Exercise activates skeletal muscle satellite cells: exercise prevention and treatment for age-related sarcopenia and muscle injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3752-3759. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||