Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (2): 265-270.doi: 10.3969/j.issn.2095-4344.2014.02.017
Previous Articles Next Articles
Construction of a lentivirus vector for Trail gene in rats
Zhang Hai, Jiang Zheng-fang, Liu Yang, Zhang Bo, Wu Gui-qiang, Zeng Ling-yong
- Department of Neurosurgery, the Third Hospital of Mianyang, Mianyang 621000, Sichuan Province, China
-
Received:2013-11-18Online:2014-01-08Published:2014-01-08 -
Contact:Jiang Zheng-fang, Master’s supervisor, Chief physician, Department of Neurosurgery, the Third Hospital of Mianyang, Mianyang 621000, Sichuan Province, China -
About author:Zhang Hai, Master, Attending physician, Department of Neurosurgery, the Third Hospital of Mianyang, Mianyang 621000, Sichuan Province, China
CLC Number:
Cite this article
Zhang Hai, Jiang Zheng-fang, Liu Yang, Zhang Bo, Wu Gui-qiang, Zeng Ling-yong. Construction of a lentivirus vector for Trail gene in rats[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(2): 265-270.
share this article

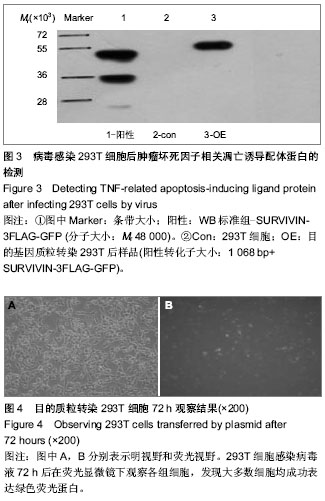
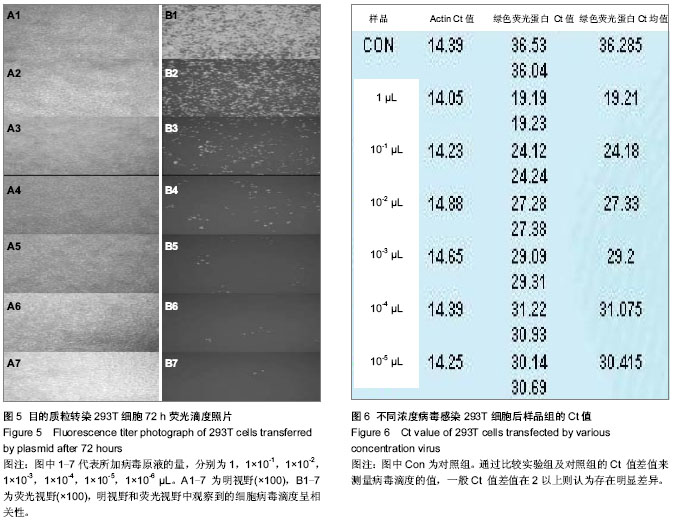
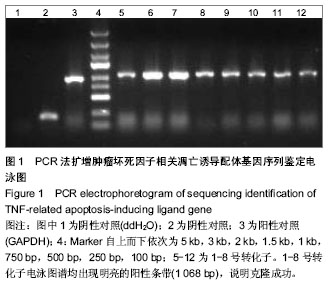
2.1 PCR鉴定重组克隆 阳性转化子大小为1 068 bp;阴性转化子大小为198 bp(图1)。 2.2 阳性克隆DNA测序结果 DNA测序鉴定的结果与实验要求的DNA序列基本一致(图2)。 2.3 Western Blot结果 观察到Mr 58 000附近处有特征条带,其大小和目的基因融合蛋白相吻合(图3)。 2.4 目的质粒转染后细胞荧光观察及病毒滴度的检测 293T细胞感染病毒液72 h后在荧光显微镜下观察各组细胞,发现大多数细胞均成功表达绿色荧光蛋白(图4)。根据图4中绿色荧光蛋白的表达情况,在加入1×10-6 μL病毒原液的孔中观察到存在2个带有荧光的细胞,说明该孔中至少有2个病毒感染了细胞,则该病毒的滴度就应该等于带有荧光的细胞数除以病毒原液量,就是2/(1×10-6)=2×106,单位为TU/μL,也就等于2×109 TU/mL(图5)。 2.5 Real-time PCR曲线图检测不同浓度病毒感染293T细胞后样品组的Ct值及表达量分析 在这次qPCR滴度检测中,10-4 μL组和对照组样品的Ct值相差5,且10-4 μL组和10-5 μL组样品间Ct值差异不大,故可以认为在10-4 μL组样品中存在病毒颗粒。在反转录后得到的20 μL cDNA 中只用了1 μL来检测滴度,所以在计算滴度时就应该乘以20。假定该组样品含有至少1个病毒颗粒,则病毒的滴度为:1/(1×10-5) ×20=2×106 TU/μL =2×109 TU/mL(图6)。"
| [1] Wirth T, Parker N, Ylä-Herttuala S. History of gene therapy. Gene. 2013; 525(2):162-169. [2] Boye SE, Boye SL, Lewin AS, et al. A comprehensive review of retinal gene therapy. Mol Ther. 2013;21(3):509-519.[3] Davanzo R, Zauli G, Monasta L, et al. Human colostrum and breast milk contain high levels of TNF-related apoptosis-inducing ligand (TRAIL). J Hum Lact. 2013; 29(1): 23-25.[4] Wajant H, Pfizenmaier K, Scheurich P. TNF-related apoptosis inducing ligand (TRAIL) and its receptors in tumor surveillance and cancer therapy. Apoptosis. 2002; 7(5): 449-459.[5] Gonzalvez F, Ashkenazi A. New insights into apoptosis signaling by Apo2L/TRAIL. Oncogene. 2010; 29(34): 4752- 4765. [6] Pietersen AM, van der Eb MM, Rademaker HJ, et al. Specific tumor-cell killing with adenovirus vectors containing the apoptin gene.Gene Ther. 1999; 6(5):882-892.[7] Shi J, Zheng D, Liu Y, et al. Overexpression of soluble TRAIL induces apoptosis in human lung adenocarcinoma and inhibits growth of tumorxenografts in nude mice. Cancer Res. 2005; 65(5):1687-1692.[8] Chen C, Akerstrom V, Baus J, et al. Comparative analysis of the transduction efficiency of five adeno associated virus serotypes and VSV-G pseudotype lentiviral vector in lung cancer cells. Virol J. 2013; 10:86.[9] Yan F, Zheng Y, Huang L. Adenovirus-mediated combined anti-angiogenic and pro-apoptotic gene therapy enhances antitumor efficacy in hepatocellular carcinoma. Oncol Lett. 2013; 5(1):348-354. [10] Zhang Y, Qu ZH, Cui M, et al. Combined endostatin and TRAIL gene transfer suppresses human hepatocellular carcinoma growth and angiogenesis in nude mice. Cancer Biol Ther. 2009; 8(5):466-473. [11] Sun W, Jiang Z, Xiang TX, et al. Anti-tumor effect of eukaryotic expressing plasmid containing soluble tumor necrotic factor-related apoptosis inducing ligand combined with human angiostatin Kringle (1 - 3) genes on human gastric cancer xenografts in nude mice. Zhonghua Yi Xue Za Zhi. 2009; 89(12):841-845.[12] Rasul A, Yu B, Zhong L, et al. Cytotoxic effect of evodiamine in SGC-7901 human gastric adenocarcinoma cells via simultaneous induction of apoptosis and autophagy. Oncol Rep. 2012; 27(5):1481-1487.[13] Zheng Y, Chen H, Zeng X, et al. Surface modification of TPGS-b-(PCL-ran-PGA) nanoparticles with polyethyleneimine as a co-delivery system ofTRAIL and endostatin for cervical cancer gene therapy. Nanoscale Res Lett. 2013; 8(1):161.[14] Qiu B, Ji M, Song X, et al. Co-delivery of docetaxel and endostatin by a biodegradable nanoparticle for the synergistic treatment of cervical cancer. Nanoscale Res Lett. 2012; 7(1): 666.[15] Moniri MR, Sun XY, Rayat J, et al. TRAIL-engineered pancreas-derived mesenchymal stem cells: characterization and cytotoxic effects on pancreatic cancer cells. Cancer Gene Ther. 2012; 19(9):652-658.[16] Mohr A, Albarenque SM, Deedigan L, et al. Targeting of XIAP combined with systemic mesenchymal stem cell-mediated delivery of sTRAIL ligand inhibits metastatic growth of pancreatic carcinoma cells. Stem Cells. 2010 ;28(11):2109- 120.[17] Xia P, Zhu J, Zhu G. Escherichia coli Nissle 1917 as safe vehicles for intestinal immune targeted therapy--a review. Wei Sheng Wu Xue Bao. 2013;53(6):538-544.[18] Cronin M, Stanton RM, Francis KP, et al. Bacterial vectors for imaging and cancer gene therapy: a review. Cancer Gene Ther. 2012;19(11):731-740.[19] Bhatia S, Menezes ME, Das SK, et al. Innovative approaches for enhancing cancer gene therapy. Discov Med. 2013; 15(84): 309-317.[20] Prinz-Hadad H, Mizrachi T, Irony-Tur-Sinai M, et al. Amelioration of autoimmune neuroinflammation by the fusion molecule Fn14•TRAIL. J Neuroinflammation. 2013;10:36. [21] Chiu TL, Wang MJ, Su CC. The treatment of glioblastoma multiforme through activation of microglia and TRAIL induced by rAAV2-mediated IL-12 in a syngeneic rat model. J Biomed Sci. 2012;19:45.[22] Balyasnikova IV, Ferguson SD, Han Y, et al. Therapeutic effect of neural stem cells expressing TRAIL and bortezomib in mice with glioma xenografts. Cancer Lett. 2011; 310(2): 148-159. [23] El-Aneed A. An overview of current delivery systems in cancer gene therapy. Journal of Controlled Release. 2004; 94(1):1-14.[24] Mancheño-Corvo P, Martín-Duque P. Viral gene therapy. Clin Transl Oncol. 2006;8(12):858-867.[25] Schabowsky RH, Sharma RK, Madireddi S, et al. ProtEx technology for the generation of novel therapeutic cancer vaccines. Exp Mol Pathol. 2009; 86(3):198-207. [26] Schweizer M, Merten OW. Large-scale production means for the manufacturing of lentiviral vectors. Curr Gene Ther. 2010; 10(6):474-486.[27] Segura MM, Mangion M, Gaillet B, et al. New developments in lentiviral vector design, production and purification. Expert Opin Biol Ther. 2013;13(7):987-1011.[28] Fu XR, Zhang XD, Zhang C, et al. Construction of a lentiviral vector carrying TRAIL gene and its infection efficiency to lymphoma cells in vitro. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2012;20(4):900-905. |
| [1] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [2] | Zhao Xiang, Wei Cuilan, Zhang Yeting. Neurogenesis and neuroinflammation under exercise: alteration and regulation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 813-820. |
| [3] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| [4] | Ma Zhijie, Li Jingyu, Cao Fang, Liu Rong, Zhao Dewei. Influencing factors and biological property of novel biomedical materials: porous silicon carbide coated with bioactive tantalum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 558-563. |
| [5] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [6] | Yang Junhui, Luo Jinli, Yuan Xiaoping. Effects of human growth hormone on proliferation and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3956-3961. |
| [7] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [8] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [9] | Li Xinping, Cui Qiuju, Zeng Shuguang, Ran Gaoying, Zhang Zhaoqiang, Liu Xianwen, Fang Wei, Xu Shuaimei. Effect of modification of β-tricalcium phosphate/chitosan hydrogel on growth and mineralization of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3493-3499. |
| [10] | Zhou Anqi, Tang Yufei, Wu Bingfeng, Xiang Lin. Designing of periosteum tissue engineering: combination of generality and individuality [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3551-3557. |
| [11] | Chen Song, He Yuanli, Xie Wenjia, Zhong Linna, Wang Jian. Advantages of calcium phosphate nanoparticles for drug delivery in bone tissue engineering research and application [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3565-3570. |
| [12] | Dai Min, Wang Shuai, Zhang Nini, Huang Guilin, Yu Limei, Hu Xiaohua, Yi Jie, Yao Li, Zhang Ligang. Biological characteristics of hypoxic preconditioned human amniotic mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3004-3008. |
| [13] | Li Xiangze, Bu Xianmin, Li Dongmei, Chi Yulei, Su Qiang, Jin Xintong, Zhao Jian, Zhang Gaotian, Wu Bin, Meng Chunyang . Stem cells, cytokines, hormones, neuropeptides and genes in traumatic brain trauma to promote fracture healing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3057-3063. |
| [14] | Ailimaierdan·Ainiwaer, Wang Ling, Gu Li, Dilidaer•Taxifulati, Wang Shan, Yin Hongbin. Effect of transforming growth factor-beta3 on the proliferation and osteogenic capability of osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2664-2669. |
| [15] | Huang Na, Liu Jiayue, Huang Yingjie, Wen Junmao, Wang Haibin, Zhang Qingwen, Zhou Chi . Bibliometric and visualized analysis of research on osteonecrosis of the femoral head from the Web of Science in the last 5 years [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2711-2718. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||