Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (44): 7777-7784.doi: 10.3969/j.issn.2095-4344.2013.44.020
Previous Articles Next Articles
CT manifestations of invasive pulmonary aspergillosis after solid organ transplantation
Dong Xin, Li Hai-bin
- Department of Radiology, the 309th Hospital of PLA, Beijing 100091, China
-
Online:2013-10-29Published:2013-10-31 -
Contact:Dong Xin, Chief physician, Department of Radiology, the 309th Hospital of PLA, Beijing 100091, China dongxin1963@sina.com -
About author:Dong Xin, Chief physician, Department of Radiology, the 309th Hospital of PLA, Beijing 100091, China dongxin1963@sina.com
CLC Number:
Cite this article
Dong Xin, Li Hai-bin. CT manifestations of invasive pulmonary aspergillosis after solid organ transplantation[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(44): 7777-7784.
share this article
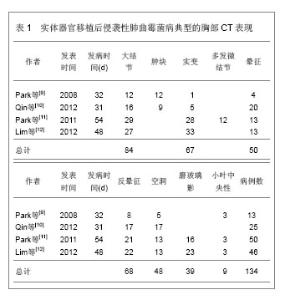
2.1 纳入文献基本情况 初检得到107篇文献,全部为英文。阅读标题和摘要进行初筛,排除因研究目的与此文无关11篇,内容重复性的研究41篇,共保存55篇文献做进一步的分析。其中重点研究实体器官移植后侵袭性肺曲霉菌病CT表现的文献26篇。文献全部为临床研究,其中3篇研究对象来源于同一单位同一时间范围的病例,排除2篇,保留1篇,其余27篇作为对照。 文献[1-8]研究了侵袭性肺曲霉菌病的临床和影像表现,文献[9-12]研究了实体器官移植后侵袭性肺曲霉菌病CT表现,文献[14-18]研究了CT晕征的诊断意义,文献[22-30]研究了CT反晕征的诊断意义,文献[31-33]研究了CT半月征的诊断意义,文献[34-39]研究了实变、磨玻璃影等其他征象的诊断意义,文献[49-55]研究了CT血管造影的诊断意义。 2.2 结果描述 2.2.1 侵袭性肺曲霉菌病的易感因素和诊断 侵袭性肺曲霉菌病的发病率逐年增高。尸检报告侵袭性肺曲霉菌病占全部真菌感染的60%。在中性白细胞减少症者,死亡率超过50%,而在造血干细胞移植接受者,死亡率高达90%[7-8]。肺曲霉病的易感因素包括,长期嗜中性白血球减少症(< 500细胞/mm3,>10 d)、移植(风险最高的是肺移植和造血干细胞移植)、长期(> 3周)和大剂量的皮质激素治疗、血液系统恶性肿瘤、化疗、AIDS晚期和慢性肉芽肿病[5]。 侵袭性肺曲霉菌病诊断依然严峻。在严重免疫妥协患者,早期诊断侵袭性肺曲霉菌病是困难的。诊断的金标准是通过胸腔镜或开胸肺活检获得的肺组织病理检查[5]。痰标本中分离出曲菌的意义取决于宿主的免疫状态。在免疫功能正常者,它几乎总是代表定植,无临床意义。然而,免疫妥协患者痰标本中分离出曲菌高度预示侵袭性肺曲霉菌病,反之,阴性的痰标本不能排侵袭性肺曲霉菌病。在已证实侵袭性肺曲霉菌病,血培养罕有阳性结果[5-6]。 在疾病的早期阶段,病变多为非特异性改变,胸片用处不大。常见的表现包括圆形密度,代表肺梗死的以胸膜为基底的楔形肺浸润和空洞。胸腔积液少见。CT,尤其是薄层CT,是非常有用的检查方法[7]。常规使用薄层CT导致更早地发现病变,改善治疗结果。它还有助于进一步引导发现病原体,如支气管镜和开胸肺活检[7]。侵袭性肺曲霉菌病典型的胸部CT表现包括结节、肿块、实变以及磨玻璃影,见表1。"
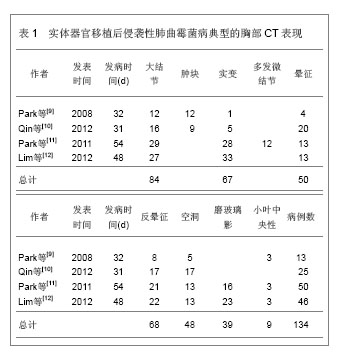

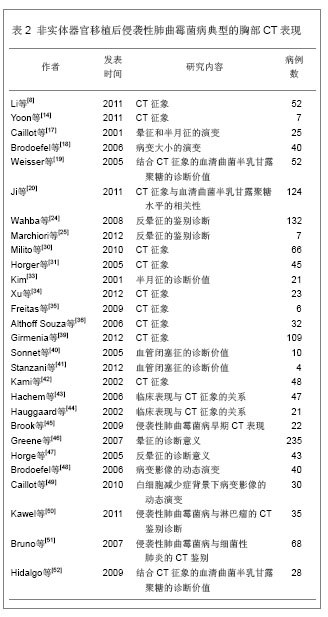
2.2.2 结节和肿块 结节和肿块是侵袭性肺曲霉菌病最常见的CT表现,结节是指一个局灶性,圆形的密度增高影,至少是中等程度边缘清楚,直径≤ 3 cm,大结节通常是指 > 1 cm的结节。肿块是指表现为直径> 3 cm的致密影(不管其轮廓、边缘或密度特征)。Greene[13]报告235例侵袭性肺曲霉菌病,发现94%的患者至少存在一个结节;Park等[9]报告13例实体器官移植后侵袭性肺曲霉菌病,12例表现为结节和肿块;Qin等[10]报告25例肝移植后侵袭性肺曲霉菌病,64%的病例表现为结节,36%的病例表现为肿块;Lim等[12]报告46例实体器官移植后侵袭性肺曲霉菌病,其中59%的病例表现为结节;Park等[11]报告50例实体器官移植后侵袭性肺曲霉菌病,56%的病例表现为大结节,26%表现为微结节(< 1 cm)。 结节或肿块周围通常出现磨玻璃密度区,称CT晕征[14-16]。病理学检查发现结节代表局灶性梗死,而晕征代表肺泡出血。结节中央坏死可以出现空洞。最初作为诊断侵袭性肺曲霉菌病的特异征象。然而随后发现这个征象既不敏感也不特异。晕征还可见于转移瘤、支气管泡癌、隐源性间质性肺炎和嗜酸细胞性肺炎或其他真菌感染[5]。晕征通常为侵袭性肺曲霉菌病的早期表现,随着疾病的进展,晕征倾向于逐渐消失[14-16]。Caillot等[17]的报告显示侵袭性肺曲霉菌病在发病的0,3,7,14 d晕征的发生率分别为 96%、68%、22%和19%,他们的结论是晕征是侵袭性肺曲霉菌病最早的放射学表现,但是短暂的,在感染发生3 d后,其发生率快速下降。Brodoefel等[18]的报告显示侵袭性肺曲霉菌病在发病的1,4,8,16 d,晕征的发生率分别为88%,63%,37%和18%,Lim等[12]报告晕征在发病的2周减少1半,在4周内完全消失。所以在如此狭窄的时间窗内,尽早对高危患者实施CT检查,以探测晕征的存在是非常重要的。早期系统抗真菌治疗可能影响晕征的发生率。尽管存在争议,基于CT早期征象的抗真菌治疗与基于曲菌半乳甘露聚糖试验的抗真菌治疗可能更可靠,因为曲菌半乳甘露聚糖不大可能在肺部出现病灶前就呈阳性[19-20]。尽管普遍认为在一定的临床背景下,在早期侵袭性肺曲霉菌病的患者经常可以观察到晕征。并非所有的作者均报告同样高的发生率。事实上,报告的晕征发生率差异很大,范围从25%-95%[9-15-17,21],这些差异是否与观察者之间定义的不同还是与CT扫描时间窗选择的不同有关尚不清楚。 与晕征的表现相反,在高分辨胸部CT肺窗上观察,还可见病灶中心密度低呈磨玻璃状,周围是新月形或环形高密度,厚度至少2 mm。称反晕征[22-23]。这种表现是由于中心为低密度由肺泡间隔浸润和细胞碎片,周围环形或新月形高密度是肺泡管机化性肺炎或致密、均匀肺泡间细胞浸润所导致的致密气腔实变。Wahba等[24]报告189例侵袭性真菌性肺炎,37例侵袭性毛霉菌性肺炎中7例(19%)可见反晕征;132例侵袭性肺曲霉菌病仅1例(< 1%)可见反晕征。似乎侵袭性肺曲霉菌病不常表现为反晕征。但是随后Marchiori等[25]的报告发现在79例各类表现为反晕征的病例中,有7例(8.86%)是侵袭性肺曲霉菌病。而最近的两个实体器官移植后侵袭性肺曲霉菌病的报告,反晕征发生率分别高达68%和47.83%[11,12]。反晕征亦代表肺梗死的表现,随时间可演变为空洞。除真菌感染以外,反晕征最常见于隐源性机化性肺炎,还见于肺结核、肺梗死以及结节病等[26-29]。Marchiori等[25]讨论了侵袭性肺曲霉菌病反晕征,Lim等[10]的报告描述为梗死性实变的形态学特征,发现内部呈网状结构,外部实变环厚度> 1 cm并有胸腔积液,强烈地提示侵袭性真菌性肺炎。 尽管大部分文献都强调晕征的诊断意义,但实际在严重免疫妥协的患者,发现肺内直径> 1 cm的结节,无论是否并有晕征,更可能代表侵袭性肺曲霉菌病,而不是其他非真菌感染[30]。这些侵袭性肺曲霉菌病患者,在病变的不同阶段,还可见结节和肿块内的空洞以及空气半月征[31-32]。特别是空气半月征是一个较晚的侵袭性肺曲霉菌病征象。为一个半月形空气积聚,半月形空气影将空洞壁与洞内肿块分开[33]。该征象通常被认为是曲菌移植到已有的空洞内或在血管侵袭性曲菌病中梗死的肺收缩的结 果[10,12,17-18,31,32]。Caillot等[17]的报告显示侵袭性肺曲霉菌病在发病的3,7,14 d,空气半月征的发生率分别为8%、28%和63%,气腔实变的发生率分别为31%、50%和18%。因此,空气半月征对侵袭性肺曲霉菌病诊断非常有用,但对于指导治疗的意义不大,因为此时患者已经渡过了中性粒细胞减少阶段。空气半月征同样也见于其他疾病,包括结核病、Wegener 肉芽肿、空洞内出血和肺癌[26-27,28,31-32]。Qin等[10]报告25例肝移植后侵袭性肺曲霉菌病,20%的病例表现为空气半月征。Lim等[12]报告46例实体器官移植后侵袭性肺曲霉菌病,仅1例(2%)表现为空气半月征。Park等[11]报告50例实体器官移植后侵袭性肺曲霉菌病,仅2例(4%)表现为空气半月征。空气半月征发生率如此之大,最大的可能是选择的病例处于不同的病变阶段。 2.2.3 实变 实变也是侵袭性肺曲霉菌病常见的CT表现,通常位于近胸膜的边缘,边界可以清楚或模糊,典型的呈基底紧贴胸膜的三角形高密度影。这种实变代表曲霉菌引起出血性肺梗死, 中央为坏死的肺组织(反晕征), 周围的晕圈代表坏死周围出血区(晕征)。邻近胸膜面实变病灶可引起胸膜凹陷, 但其程度较肺癌为重并伴有邻近胸膜增厚[22-24]。另外报告逐渐增多的是非血管侵袭性模式的实变,CT表现为单侧或双侧肺段或肺叶性实变, 病变内有时见支气管充气征。在病理上为渗出或化脓性肺炎。病变初期边缘模糊, 进入亚急性期, 病灶边缘趋于清楚, 较大病变常伴有纤维索条影。肺部病灶吸收较一般肺部炎症较慢时, CT 征象呈浸润状态是其特征之一。免疫功能抑制者, 肺泡实变可更为广泛。CT 征象缺乏特异, 主要与非特异性肺炎鉴别[34-36]。Park等[9]报告13例实体器官移植后侵袭性肺曲霉菌病,1例表现为实变。Qin等[10]报告25例肝移植后侵袭性肺曲霉菌病,20%的病例表现为实变,Lim等[12]报告46例实体器官移植后侵袭性肺曲霉菌病,72%的病例表现为肿块和实变,其中48%可见梗死性实变(反晕征)。Park等[11]报告50例实体器官移植后侵袭性肺曲霉菌病,72%的病例表现为肿块和实变,其中42%可见梗死性实变(反晕征)。然而,Xu等[34]回顾性分析了23例非血行感染的侵袭性肺曲霉菌病的早期CT表现,发现12例(52%)表现为实变,大部分实变沿支气管走行和胸膜下发布,此外,磨玻璃影外,其他征象少见。他们的结论是非血行感染的侵袭性肺曲霉菌病的早期CT表现与急性支气管炎近似,Althoff Souza等[36]比较了32例免疫妥协宿主肺侵袭性肺曲霉菌病和22例肺念珠菌感染的CT表现,结果显示肺侵袭性肺曲霉菌病84%(27/32)的病例表现为实变,肺念珠菌感染50%(11/22)的病例表现为实变,二者比较差异有显著性意义(P=0.01)。提示实变可能更多地发生在肺侵袭性肺曲霉菌病。 2.2.4 磨玻璃影 磨玻璃影是指CT图像上表现为肺密度轻度增加,但仍可显示血管影和支气管的区域,此征象常为早期肺部疾患的表现,缺乏特异性,可见于各种炎症、水肿、纤维化及肿瘤等病变,这种特征表现是由气腔不完全充盈、间质轻度增厚、肺泡部分萎陷、正常呼气末或肺血容量增加造成,由于其密度浅淡,仍可见血管影,可与肺实变鉴别[37-38]。Park等[9]报告13例实体器官移植后侵袭性肺曲霉菌病,无1例表现为磨玻璃影。Lim等[10]报告46例实体器官移植后侵袭性肺曲霉菌病,50%的病例表现为磨玻璃影。Park等[11]报告50例实体器官移植后肺侵袭性肺曲霉菌病,31%的病例表现为磨玻璃影。Xu等[34]回顾性分析了23例非血行感染的侵袭性肺曲霉菌病的早期CT表现,发现14例(61%)表现为磨玻璃影,其中双侧10例,单侧4例。主要累及两肺中野和下野。此外,除气腔实变外,其他征象少见。提示在非血行感染的侵袭性肺曲霉菌病,磨玻璃影可能是其早期CT表现。Freitas等[35]报告6例血液肿瘤和骨髓干细胞移植后白细胞减少症以及免疫妥协宿主的肺侵袭性肺曲霉菌病,发现最常见的CT表现是结节(5/6)和实变(2/6),亦有3个病例表现为磨玻璃影,然而,这种磨玻璃影均无一例外地伴随着结节和实变。他们的结论是在肺侵袭性肺曲霉菌病出现磨玻璃影,均为伴随征象,无特异性诊断意义。Althoff Souza等[36]比较了32例免疫妥协宿主肺侵袭性肺曲霉菌病和22例肺念珠菌感染的CT表现,结果显示肺侵袭性肺曲霉菌病44%(14/32)的病例表现为磨玻璃影,肺念珠菌感染64%(14/22)的病例表现为磨玻璃影,二者比较无显著性意义(P > 0.1)。提示磨玻璃影可以发生在不同的真菌性肺炎。 2.2.5 其他征象 实体器官移植后侵袭性肺曲霉菌病其他征象包括,支气管周围浸润、树芽征、胸腔积液等,这些征象既不常见,也不特异[4]。 2.2.6 预测出血 Horger等[31]的45例回顾性研究显示,没有早期HRCT征像(结节,实变,支气管周围浸润)能够预测患者的结果或进展为肺出血。然而,发生存在大的空洞病变或实变靠近较大的肺血管容易导致肺出血。 2.2.7 实体器官移植后与白细胞减少症侵袭性肺曲霉菌病的放射学模式 侵袭性肺曲霉菌病放射学模式被分为血管侵袭型或气道侵袭型[39],前者应满足以下3个表现中至少2个:①晕征。②梗死形实变。③内部低密度、空洞或空气新月征。后者应满足以下3个表现中至少2个:①小气道病变。②支气管周围实变。③支气管扩张。当CT表现同时满足血管侵袭型或气道侵袭型时,被认为是组合型。当CT表现不能满足上述的任何标准时,则称不确定型。简化以上模式为两种,血管侵袭性和非血管侵袭性模式。前者被定义为血管侵袭型或组合型,而后者被定义为气道侵袭型或不确定型。Park等[11]报告50例实体器官移植后无白细胞减少症和60例白细胞减少症的侵袭性肺曲霉菌病,发现前者实变或肿块和晕征的发生率低于后者(分别为56%,26%和78%,55%)。作者认为实体器官移植后与白细胞减少症侵袭性肺曲霉菌病的放射学模式有所不同,前者倾向于非血管侵袭性模式,而后者更倾向于血管侵袭性模式。然而,同期有关实体器官移植后侵袭性肺曲霉菌病的文献并未进一步证实这一特点[9,10,12]。 2.2.8 侵袭性肺曲霉菌病征象与患者预后的关 Lim等[12]报告46例实体器官移植后证实或高度可能的侵袭性肺曲霉菌病。发现生存者的实变或肿块发生率明显低于死亡者(62%∶93%)。生存者比死亡者更常见出现空洞(43%∶13%),并且明显为较小的空洞(7.5 cm3∶19 cm3)。随访CT显示生存者的晕征在4个星期内迅速吸收,其实变、梗死性实变及其内部低密度区的范围随着时间推移逐渐减小,在2周内减小一半大小。大结节持续存在7 d(84%),随后缓慢吸收。他们的结论是实变或肿块是实体器官移植接受者侵袭性肺曲霉菌病最常见的CT表现。无实变或肿块和小腔空洞存在可能与更好预后相关。治疗后每个模式吸收的时间各不相同。Park等[11]报告50例实体器官移植后侵袭性肺曲霉菌病,多变量分析显示大结节、多发梗死性实变、肾脏替代治疗和持久性的血清曲菌半乳甘露聚糖阳性是90 d的死亡率独立的预测因子。而代表出血的晕征与90 d的死亡率无关。 2.2.9 CT肺动脉造影诊断侵袭性肺曲霉菌病的价值 Sonnet等[40]用增强CT肺动脉造影研究诊断侵袭性真菌性肺炎,主要是侵袭性肺曲霉菌病和侵袭性毛霉菌肺炎。他们定义肺部病变边缘的肺动脉中断而无血管内和血管周围病变的征象未血管闭塞征。在CT肺血管造影上,5例组织病理学侵袭性真菌性肺炎,4例被发现存在血管闭塞征。这其中只有2例病变存在CT晕征。表明血管闭塞征在诊断侵袭性真菌性肺炎优于晕征。在另外9例病变,CT肺血管造影显示未闭塞的肺部血管而除外了侵袭性真菌性肺炎的诊断。这9个例最终诊断为瘢痕、血肿、淋巴瘤、非典型分枝杆菌感染和弥漫性肺泡损伤。最近Stanzani 等[41]的研究进一步证实了CT肺血管造影在诊断侵袭性真菌性肺炎的优势。他们的21例侵袭性真菌性肺炎中,19例(90%)表现为肺血管闭塞征阳性;15例非侵袭性真菌性肺炎中,仅1例表现为肺血管闭塞征阳性,为金黄色葡萄球菌脓毒栓塞。CT肺血管造影肺血管闭塞征诊断侵袭性真菌性肺炎的敏感性和特异性分别是83%和93%。优于晕征的敏感性和特异性。与Sonnet等[40]的研究结果相似,敏感性和特异性分别是80%和100%。 既往的回顾性研究肺侵袭性肺曲霉菌病主要为免疫妥协的白细胞减少症、骨髓干细胞移植后宿主,见表2。"
| [1]Küpeli E, Eyübo?lu FÖ, Haberal M.Pulmonary infections in transplant recipients. Curr Opin Pulm Med.2012;18(3): 202-212. [2]Kotloff RM, Ahya VN, Crawford SW.Pulmonary complications of solid organ and hematopoietic stem cell transplantation. Am J Respir Crit Care Med. 2004; 170(1):22-48. [3]Batista MV, Costa SF, Shikanai-Yasuda MA, et al. Current treatment options for invasive aspergillosis.Drugs Today (Barc). 2013;49(3):213-226. [4]Beigelman-Aubry C, Godet C, Caumes E. Lung infections: The radiologist's perspective.Diagn Interv Imaging. 2012; 93(6):431-440. [5]Kousha M, Tadi R, Soubani AO.Pulmonary aspergillosis: a clinical review.Eur Respir Rev. 2011;20(121):156-1574. [6]Wichmann D, Kluge S.Invasive pulmonary aspergillosis in the ICU: an emerging disease? Intensive Care Med. 2013 Jan 22. [7]Smith JA, Kauffman CA.Pulmonary fungal infections. Respirology. 2012;17(6):913-926. [8]Li XS, Zhu HX, Fan HX, et al.Pulmonary fungal infections after bone marrow transplantation: the value of high-resolution computed tomography in predicting their etiology.Chin Med J (Engl). 2011;124(20):3249-3254. [9]Park YS, Seo JB, Lee YK, et al.Radiological and clinical findings of pulmonary aspergillosis following solid organ transplant. Clin Radiol. 2008;63(6):673-680. [10]Qin J, Fang Y, Dong Y, et al.Radiological and clinical findings of 25 patients with invasive pulmonary aspergillosis: retrospective analysis of 2150 liver transplantation cases.Br J Radiol. 2012;85(1016):e429-435. [11]Park SY, Lim C, Lee SO, et al.Computed tomography findings in invasive pulmonary aspergillosis in non-neutropenic transplant recipients and neutropenic patients, and their prognostic value. J Infect. 2011;63(6):447-456. [12]Lim C, Seo JB, Park SY, et al. Analysis of initial and follo w-up CT findings in patients with invasive pulmonary aspergillosis after solid organ transplantation. Clin Radiol 2012;67(12): 1179-1186. [13]Greene R.The radiological spectrum of pulmonary aspergillosis. Med Mycol. 2005;43 Suppl 1:S147-154. [14]Yoon SH, Park CM, Goo JM. Pulmonary aspergillosis in immunocompetent patients without air-meniscus sign and underlying lung disease: CT findings and histopathologic features.Acta Radiol. 2011;52(7):756-761. [15]Parrón M, Torres I, Pardo M, et al. The halo sign in computed tomography images: differential diagnosis and correlation with pathology findings. Arch Bronconeumol. 2008;44(7): 386-392. [16]Lee YR, Choi YW, Lee KJ, et al.CT halo sign: the spectrum of pulmonary diseases. Br J Radiol. 2005;78(933):862- 865. [17]Caillot D, Couaillier JF, Bernard A, et al. Increasing volume and changing characteristics of invasive pulmonary aspergillosis on sequential thoracic computed tomography scans in patients with neutropenia. J Clin Oncol 2001;19(1): 253-259. [18]Brodoefel H, Vogel M, Hebart H, et al. Long-term CT follow-up in 40 non-HIV immunocompromised patients with invasive pulmonary aspergillosis: kinetics of CT morphology and correlation with clinical findings and outcome. AJR Am J Roentgenol 2006;187(2):404-413. [19]Weisser M, Rausch C, Droll A, et al. Galactomannan does not precede major signs on a pulmonary computerized tomographic scan suggestive of incasive aspergillosis in patients with hematological malignancies. Clin Infect Dis. 2005;41(8):1143-1149. [20]Ji Y, Xu LP, Liu DH, et al.Positive results of serum galactomannan assays and pulmonary computed tomography predict the higher response rate of empirical antifungal therapy in patients undergoing allogeneic hematopoietic stem cell transplantation.Biol Blood Marrow Transplant. 2011;17(5): 759-764. [21] Park SY, Kim SH, Choi SH ,et al.Clinical and radiological features of invasive pulmonary aspergillosis in transplant recipients and neutropenic patients. Transpl Infect Dis. 2010; 12(4):309-315. [22]Marchiori E, Zanetti G, Hochhegger B, et al.Reversed halo sign on computed tomography: state-of-the-art review. Lung. 2012;190(4):389-394. [23]Georgiadou SP, Sipsas NV, Marom EM, et al.The Diagnostic Value of Halo and Reversed Halo Signs for Invasive Mold Infections in Compromised Hosts Clin Infect Dis. 2011;52(9): 1144-1155. [24]Wahba H, Truong MT, Lei X, et al.Reversed halo sign in invasive pulmonary fungal infections. Clin Infect Dis. 2008; 46(11):1733-1737. [25]Marchiori E, Zanetti G, Escuissato DL, et al.Reversed halo sign: high-resolution CT scan findings in 79 patients. Chest. 2012;141(5):1260-1266. [26]Godoy MC, Viswanathan C, Marchiori E, et al. The reversed halo sign: update and differential diagnosis.Br J Radiol. 2012; 85(1017):1226-1235. [27]Marchiori E, Zanetti G, Hochhegger B, et al.Reversed halo sign in active pulmonary tuberculosis: criteria for differentiation from cryptogenic organizing pneumonia.Lung. 2012;190(4):389-394. [28]Cottin V, Cordier JF.Cryptogenic organizing pneumonia.Semin Respir Crit Care Med. 2012;33(5):462-475. [29]Marchiori E, Zanetti G, Irion KL,Reversed halo sign on computed tomography: state-of-the-art review.AJR Am J Roentgenol. 2011;197(6):1324-1327. [30]Milito MA, Kontoyiannis DP, Lewis RE, et al. Influence of host immunosuppression on CT findings in invasive pulmonary aspergillosis. 2010;48(6):817-823. [31]Horger M, Hebart H, Einsele H, et al.Initial CT manifestations of invasive pulmonary aspergillosis in 45 non-HIV immunocompromised patients: association with patient outcome?Eur J Radiol. 2005;55(3):437-44. [32]Fred HL, Gardiner CL.The air crescent sign: causes and characteristics. Tex Heart Inst J. 2009;36(3):264-265. [33].Tonelli AR, Khalife WT, Cao M. Spherules, hyphae, and air-crescent sign.Am J Med Sci. 2008;335(6):504-506. [34]Xu SC, Qiu LH, Liu WY, et al.Initial computed tomography findings of invasive pulmonary aspergillosis in non-hematological patients.Chin Med J (Engl). 2012; 125(17): 2979-2985. [35]Freitas DB, Piovesan AC, Szarf G, et al.Outbreak of invasive pulmonary aspergillosis among patients hospitalized in a bone marrow transplant ward: tomographic findings. J Bras Pneumol. 2009;35(9):931-936. [36]Althoff Souza C, Müller NL, Marchiori E, et al.Pulmonary invasive aspergillosis and candidiasis in immunocompromised patients: a comparative study of the high-resolution CT findings. J Thorac Imaging. 2006;21(3):184-189. [37]Battista G, Sassi C, Zompatori M, et al.Ground-glass opacity: interpretation of high resolution CT findings. Radiol Med. 2003; 106(5-6):425-444. [38]Diederich S. High resolution computed tomography of the lungs: ground glass opacity and its differential diagnosis. Radiologe. 2010;50(12):1141-1152. [39]Girmenia C, Guerrisi P, Frustaci AM, et al.New category of probable invasive pulmonary aspergillosis in haematological patients.Clin Microbiol Infect. 2012;18(10):990-996. [40] Sonnet S, Buitrago-Tellez CH, Tamm M, et al. Direct detection of angioinvasive pulmonary aspergillosis in immunosuppressed patients: preliminary results with high-resolution 16-MDCT angiography. AJR Am J Roentgenol. 2005;184(3):746-751. [41] Stanzani M, Battista G, Sassi C, et al.Computed tomographic pulmonary angiography for diagnosis of invasive mold diseases in patients with hematological malignancies. Clin Infect Dis. 2012;54(5):610-616. [42]Kami M, Kishi Y, Hamaki T. The value of the chest computed tomography halo sign in the diagnosis of invasive pulmonary aspergillosis. An autopsy-based retrospective study of 48 patients. Mycoses. 2002;45(8):287-294. [43]Hachem R, Sumoza D, Hanna H. Clinical and radiologic predictors of invasive pulmonary aspergillosis in cancer patients: should the European Organization for Research and Treatment of Cancer/Mycosis Study Group (EORTC/MSG) criteria be revised? Cancer. 2006;106(7):1581-1586. [44]Hauggaard A, Ellis M, Ekelund L. Early chest radiography and CT in the diagnosis, management and outcome of invasive pulmonary aspergillosis.Acta Radiol. 2002;43(3):292-298. [45]Brook O, Guralnik L, Hardak E. Radiological findings of early invasive pulmonary aspergillosis in immune-compromised patients.Hematol Oncol. 2009;27(2):102-106. [46]Greene RE, Schlamm HT, Oestmann JW. Imaging findings in acute invasive pulmonary aspergillosis: clinical significance of the halo sign. Clin Infect Dis. 2007;44(3):373-379. [47]Horger M, Einsele H, Schumacher U. Invasive pulmonary aspergillosis: frequency and meaning of the "hypodense sign" on unenhanced CT. Br J Radiol. 2005;78(932):697-703. [48]Brodoefel H, Vogel M, Hebart H. Long-term CT follow-up in 40 non-HIV immunocompromised patients with invasive pulmonary aspergillosis: kinetics of CT morphology and correlation with clinical findings and outcome. AJR Am J Roentgenol. 2006;187(2):404-413. [49]Caillot D, Latrabe V, Thiébaut A. Computer tomography in pulmonary invasive aspergillosis in hematological patients with neutropenia: an useful tool for diagnosis and assessment of outcome in clinical trials.Eur J Radiol. 2010;74(3): e172-175. [50]Kawel N, Schorer GM, Desbiolles. Discrimination between invasive pulmonary aspergillosis and pulmonary lymphoma using CT.Eur J Radiol. 2011;77(3):417-425. [51]Bruno C, Minniti S, Vassanelli A. Comparison of CT features of Aspergillus and bacterial pneumonia in severely neutropenic patients. J Thorac Imaging. 2007;22(2):160-165. [52]Hidalgo A, Parody R, Martino R. Correlation between high-resolution computed tomography and galactomannan antigenemia in adult hematologic patients at risk for invasive aspergillosis. Eur J Radiol. 2009;71(1):55-60. [53]Kwon JC, Kim SH, Park SH, et al. Prognosis of invasive pulmonary aspergillosis in patients with hematologic diseases in Korea.Tuberc Respir Dis (Seoul). 2012;72(3):284-292. [54]Kurosawa M, Yonezumi M, Hashino S, et al. Epidemiology and treatment outcome of invasive fungal infections in patients with hematological malignancies.Int J Hematol. 2012;96(6):748-757. [55]Herbrecht R, Roedlich MN. Earlier diagnosis of angioinvasive pulmonary mold disease: is computed tomography pulmonary angiography a new step? Clin Infect Dis. 2012;54(5):610-616. |
| [1] | Xuan Juanjuan, Bai Hongtai, Zhang Jixiang, Wang Yaoquan, Chen Guoyong, Wei Sidong. Role of regulatory T cell subsets in liver transplantation and progress in clinical application [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1143-1148. |
| [2] | Yang Xin, Jin Zhe, Feng Xu, Lu Bing. The current situation of knowledge and attitudes towards organ, eye tissue, body donation of residents in Shenyang [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 779-784. |
| [3] | Dong Liping, Luo Huaiqing, Yuan Heng, Long Juan, Xu Shaohui. Effect of aging on collateral vessel growth in rats with ischemic hind limbs [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3156-3161. |
| [4] | Zhang Shiyong, Wang Chengwei, Wang Xue, Yan Xiangli, Wang Jie. Application of digital subtraction angiography in dog hindlimb arteriography [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(5): 704-708. |
| [5] | Jia Ruiming, Li Haoxuan, Chen Yu, Huang Xiaoyong, Pu Xin, Shu Lixia. Preoperative CT angiography and intraoperative X-ray image registration algorithm for thoracic aortic endovascular repair [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(35): 5658-5663. |
| [6] | Cai Qiucheng, Fan Hongkai, Xiong Rihui, Jiang Yi. Intravenous administration of bone marrow mesenchymal stem cells protects liver function following fatty liver transplantation from donors after cardiac death [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(17): 2625-2629. |
| [7] | Jiang Shanshan, Wang Feng, Yu Limei. Immunomodulatory properties of mesenchymal stem cells and their application in organ transplantation [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(1): 103-109. |
| [8] | Liu Teng-fei, Zhou Jian-kang, Huang Tuan-jie, Xing Qu, Cheng Kang, Li Peng, Li Dong-peng, Yang Bo, Ma Shan-shan, Guan Fang-xia . MG53 protein protects against multiorgan ischemia/reperfusion injury: present and future [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(20): 3248-3254. |
| [9] | Wang Jiang, Liu Kai. Current development and clinical application of pancreatic stents [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(18): 2934-2639. |
| [10] | Li Ling, Li Ning, Ai Zi-ye, Yao Ya-jun, Wei Wan-hui, He Wei-yang, Wang Yan-feng, Ye Qi-fa. Intra-patient variability of tacrolimus concentration in transplant recipients: a prognostic predictor post transplantation [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(49): 7437-7422. |
| [11] | Lin Jing-xia, Su Fan, Luo Hong-shan. Transfusion of blood components in liver transplantation and abdominal multiple organ transplantation [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(33): 4957-4962. |
| [12] | Dong Jian-hui, Li Hai-bin, Sun Xu-yong, Qin Ke, Liao Ji-xiang, Li Mei-si, Huang Xiao-dan, Huang Chen, Huang Ying, Cao Song, Gao Zhao, Li Zhuang-jiang, Nie Feng, Yang Jian-jun. Clinical effects of renal transplantation with kidneys from donors dying of organophosphate poisoning [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(29): 4311-4318. |
| [13] | Zhang Wen-xin, Teng Sha, Peng Xiao, Lin Xiao-hong, Liu Hong-xia. Systematic review of the effects of education on medical students’ knowledge and attitudes toward organ donation and transplantation [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(5): 810-814. |
| [14] | Hu Xiao-yan, Wang Yan-feng, Ye Qi-fa, Chen Zhi-quan, Fan Xiao-li, Guo Yi, Li Ning. Effects of hypothermic machine perfusion versus static cold storage of kidney allografts on transplant outcomes: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(42): 6882-6888. |
| [15] | Wei Wen-jiang, Xiao Cheng-jiang, Li Li-heng, Xiao Ke-xi, Zhao Zhi-xiang, Xu Guang, Tang Ying-hong. Establishment of cerebral infarction models in beagle dogs by superselective catheterization via the vertebral basilar artery: cerebral arterial digital subtraction angiography manifestation [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(40): 6470-6474. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||