Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (14): 2625-2632.doi: 10.3969/j.issn.2095-4344.2013.14.023
Previous Articles Next Articles
Glucocorticoids promote osteogenic differentiation of mesenchymal stem cells
Dong Ping, Xiao Ran
- Plastic Surgery Hospital, Chinese Academy of Medical Sciences (CAMS) and Peking Union Medical College (PUMC), Beijing 100144, China
-
Received:2012-08-29Revised:2012-10-08Online:2013-04-02Published:2013-04-02 -
Contact:Xiao Ran, M.D., Professor, Plastic Surgery Hospital, Chinese Academy of Medical Sciences (CAMS) and Peking Union Medical College (PUMC), Beijing 100144, China -
About author:Dong Ping★, Studying for master’s degree, Plastic Surgery Hospital, Chinese Academy of Medical Sciences (CAMS) and Peking Union Medical College (PUMC), Beijing 100144, China dpcyqwwj@163.com -
Supported by:the National Natural Science Foundation of China, No. 30871433, 30171305
CLC Number:
Cite this article
Dong Ping, Xiao Ran. Glucocorticoids promote osteogenic differentiation of mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(14): 2625-2632.
share this article
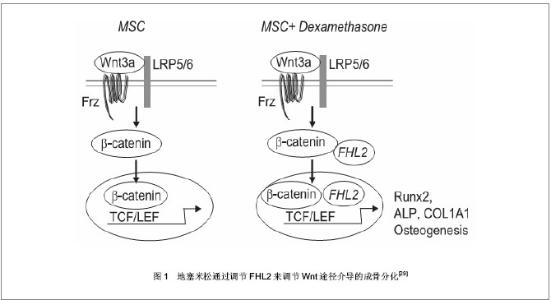
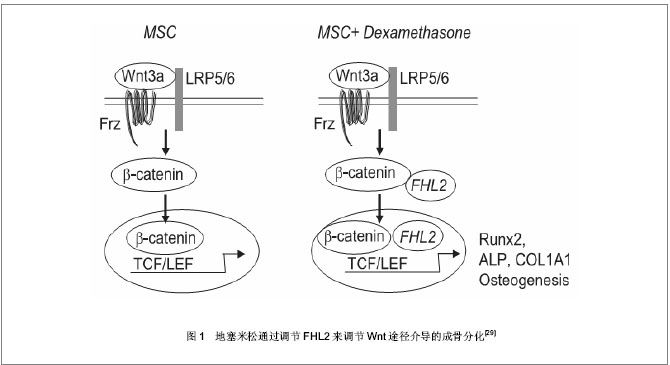
2.1 地塞米松促进间充质干细胞成骨分化 很多研究表明,地塞米松能够促进间充质干细胞表达成骨特异性的标志基因,其具体机制涉及众多基因以及信号通路[24-26]。 2.1.1 地塞米松调节Runx2表达促进间充质干细胞成骨分化 Runx2也称核心结合因子1(Cbfa1),是成骨过程中研究最广泛也是最重要的因子,该基因纯合缺失的小鼠缺乏有功能的成骨细胞。研究发现在Ⅰ型胶原(ColⅠ),骨钙素(OCN),骨涎蛋白(BSP)以及骨桥蛋白(OPN)等基因的启动子序列中都存在成骨特异性顺式元件(OSE)——PuCCPuCA,Runx2能够与OSE结合,从而反式激活这些成骨分化相关基因表达[27]。也有报道Runx2可以和rRNA基因启动子结合,抑制启动子活性,影响染色质组蛋白修饰,以控制细胞命运[28]。那么地塞米松作用于间充质干细胞时,是如何调控Runx2的呢?有人提出地塞米松是通过调节经典的Wnt途径来调控Runx2的表达。 Wnt蛋白可以激活LRP5/6受体,抑制糖原合成激酶3β的活性,使未磷酸化的β-catenin在核中堆积,增加TCF/LEF的转录活性和成骨基因的表达。地塞米松能够上调一种称为FHL2的LIM-domain蛋白表达,FHL2与β-catenin结合后,推动β-catenin向核转移,从而激活Runx2的表达和成骨分化[29],见图1。一旦部分间充质干细胞分化为成骨细胞后,地塞米松又可以刺激成骨细胞生成Wnt7b和Wnt10b,Wnt蛋白进一步激活间充质干细胞中经典的Wnt信号途径,上调Runx2表达促进成骨[30],形成协同刺激作用。"
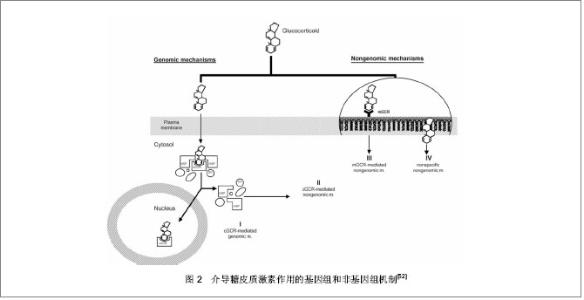
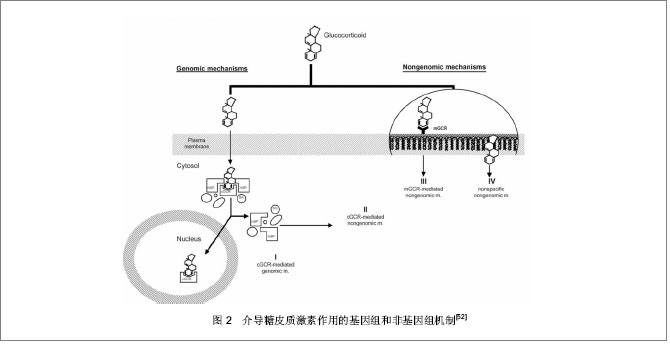
地塞米松还可能通过Wnt信号通路调节TAZ表达。TAZ也被称为β-catenin-like分子,是一种含PDZ结合基序的转录辅助激活子,在间充质干细胞分化过程中的作用类似于变阻器,抑制间充质干细胞向成脂方向分化,同时与Runx2相互作用并辅助激活Runx2促进间充质干细胞成骨分化。研究发现 10-8 mol/L地塞米松能够促进TAZ表达,Runx2表达也有显著提高[31]。 地塞米松也能通过刺激其他基因的表达来调控Runx2。有研究发现地塞米松作用于ROB-C26细胞过程中Dlx5(一种成骨转录因子)表达增强,随后Runx2表达也上升。Runx2,Dlx5,OCN在ROB-C26成骨分化过程中表现出地塞米松剂量和时间依赖性。Dlx5是Runx2和Osterix的上游调节子,说明地塞米松诱导Runx2的增强可能由Dlx5介导[32]。也有报道称地塞米松刺激鼠间充质干细胞成骨过程中会引起FGF18表达上调,FGF18的作用经由FGFR1/FGFR2可促进Runx2的表达进而促进成骨分化[33]。另外,地塞米松能促进间充质干细胞表达骨形态发生蛋白,Liu等[34]发现地塞米松处理间充质干细胞3 d引起细胞内骨形态发生蛋白2上升3至4倍,骨形态发生蛋白6提高20倍左右,可能是经骨形态发生蛋白信号途径促进Runx2表达,或经Msx2促进Osterix表达促进成骨发生[35]。 地塞米松不仅可以影响Runx2的表达量,还可以通过调节Runx2的翻译后修饰达到调节其活性的目的。体外研究报道称地塞米松通过糖皮质激素受体介导的途径上调MAPK磷酸酶1(MKP-1)水平,引起Runx2异构体Ⅱ125位丝氨酸磷酸化水平下降,而其对骨钙素,骨涎蛋白以及骨桥蛋白的转录活性上升,促进成骨发生[36]。 然而Runx2 C端Pro/Ser/Thr(PST)富含区的氨基酸残基磷酸化水平升高后其活性反而增强[37]。研究发现Runx2 PST富含区磷酸化水平受甲状旁腺激素相关蛋白受体(PTHR)调节,地塞米松能够上调间充质干细胞中甲状旁腺激素相关蛋白受体达20倍以上,从而激活胞内PKA途径,PKA与Runx2 PST结构域中PKA位点结合,使Runx2磷酸化水平升高而活性增强[38-39]。该过程可能在体内间充质干细胞成骨分化中有重要的影响,能够介导甲状腺激素及其相关蛋白刺激成骨的作用,参与骨发育。 2.1.2 地塞米松调节Noggin表达促进间充质干细胞成骨分化 Noggin属于可扩散蛋白家族的成员。过表达Noggin的转基因小鼠中发现,Noggin作为骨形态发生蛋白的拮抗剂能够与骨形态发生蛋白2、4和7结合从而抑制它们与其相应受体结合,对骨形态发生蛋白诱导的成骨发生抑制作用,影响小鼠骨发育[40]。后来发现,Noggin只抑制中胚层干细胞系C1向软骨分化,并不影响它向成骨分化[41]。最近研究表明,Noggin能够诱导人间充质干细胞向成骨方向分化,尽管诱导作用不显著。但若地塞米松和Noggin同时存在,间充质干细胞诱导后矿化程度比Noggin或地塞米松单独诱导作用都强,碱性磷酸酶活性也增加。同时,地塞米松能促进Noggin基因本身表达量增加12倍[42]。 2.1.3 地塞米松调节糖皮质激素诱导的亮氨酸拉链蛋白(GILZ)促进间充质干细胞成骨分化 亮氨酸拉链蛋白最初是在研究地塞米松作用基因时发现的,是一种新的介导糖皮质激素效应的蛋白,参与一系列生理过程如调节胸腺细胞的增殖与凋亡,抑制NF-κB的转录活性,促进肾上皮细胞钠离子转运等[43]。最近有研究显示亮氨酸拉链蛋白在介导抗炎症作用的同时具有促进成骨分化的能力。Zhang等[44]在间充质干细胞中过表达亮氨酸拉链蛋白,能诱导碱性磷酸酶活性增强以及矿化结节形成,促进其向成骨方向分化。这种促进作用一方面是提高成骨标志基因的表达,另一方面削弱了肿瘤坏死因子α对成骨的抑制作用。已经发现在地塞米松作用下,间充质干细胞中亮氨酸拉链蛋白表达会升高,因此该通路也可能参与地塞米松促进间充质干细胞成骨发生过程。 虽然地塞米松促进间充质干细胞成骨分化,但有人发现单纯的地塞米松作用于间充质干细胞却不能提高成骨细胞后期标志基因Osterix的表达[32]。这提示地塞米松在诱导过程中可能发挥启动以及协助促进效应,完全成熟成骨细胞的产生往往还需要其他诱导因子共同作用。尽管仍不知道Noggin 以及亮氨酸拉链蛋白促进成骨作用最终是否经由Runx2,但地塞米松的成骨诱导作用基本经由Runx2调控,并具有地塞米松剂量和时间依赖性。 2.2 地塞米松抑制间充质干细胞成骨分化 研究发现地塞米松并不总是促进间充质干细胞的成骨特异性基因的表达,地塞米松的成骨抑制作用与其在诱导液中的浓度有关。当用高浓度(10-7 mol/L)的地塞米松处理间充质前体细胞时,TAZ表达被抑制,进而Runx2的表达受到抑制[31];也有发现10-7 mol/L地塞米松处理时Runx2 mRNA稳定性降低[45],以及胰岛素样生长因子Ⅰ表达下降[46],甲状腺激素相关蛋白从间充质干细胞中释放受到抑制[39]。这都能够抑制间充质干细胞向成骨方向分化,引起骨量减少,也可能是糖皮质激素引起骨质疏松的原因。 Diefenderfer等[47]在研究中发现,尽管地塞米松(10-7 mol/L)处理间充质干细胞后矿化程度和碱性磷酸酶的活性增加了,但骨形态发生蛋白2的mRNA水平却下降了,并且当骨形态发生蛋白与地塞米松同时存在时,地塞米松抑制了外源骨形态发生蛋白对内源骨形态发生蛋白2的刺激作用及骨桥蛋白的表达,这在一定程度上影响了间充质干细胞成骨分化的速率。 地塞米松的成骨抑制作用可能还与间充质干细胞分化阶段有关。有人发现地塞米松(10-8 mol/L)促进有成骨分化倾向的C26细胞向成骨方向分化;然而在无成骨分化倾向的C26细胞中,地塞米松通过抑制Osterix异构体2表达,抑制C26成骨分化[48]。 2.3 糖皮质激素调控作用的介导 糖皮质激素可以调控很多基因,当然也不排除其中一些基因由于处于同一信号通路,糖皮质激素只调控了上游的某个基因而引起下游一系列基因表达改变。有研究报道糖皮质激素功能的发挥是由糖皮质激素受体来介导,干扰糖皮质激素受体能够抑制地塞米松诱导的人骨髓间充质干细胞成骨分化[49]。 糖皮质激素受体是一类激素配体依赖性的核受体,在人和动物发育中的组织及成熟组织中都有表达,包含3个结构域:激素结合域(GBD)、DNA结合域(DBD)和免疫原区(ID)。目前已知的糖皮质激素受体的介导方式有4种:①胞质糖皮质激素受体介导的经典基因组机制。糖皮质激素因其脂溶性特性穿过细胞膜与胞质中的糖皮质激素受体结合,形成活化的激素/受体复合物,然后以同源二聚体的形式转移至细胞核,结合到糖皮质激素应答元件(该元件所在位点可能与TATA box重合)转录激活靶基因;或以单体形式进入,与其他转录因子P65、AP1、NF-κB等结合,竞争性结合到靶基因激活位点抑制转录[50]。②胞质糖皮质激素受体介导的非基因组效应。和途径1一样,当糖皮质激素和其受体结合后,原来结合在糖皮质激素受体上的热激蛋白(HSP)、Src以及MAPK途径中的一些激酶分子从糖皮质激素受体上释放下来,参与细胞活动。如Src在成骨前体细胞中也有表达,它能够通过抑制Runx2转录因子YAP的活性进而抑制成骨分化[51]。③膜结合的糖皮质激素受体介导的特异性的非基因组糖皮质激素效应。④糖皮质激素非特异性地插入细胞膜[52],见图2。"
| [1] Blackwell GJ, Carnuccio R, Di Rosa M,et al. Macrocortin: a polypeptide causing the anti-phospholipase effect of glucocorticoids.Nature. 1980;287(5778):147-149.[2] Sapolsky RM, Pulsinelli WA.Glucocorticoids potentiate ischemic injury to neurons: therapeutic implications.Science. 1985;229(4720):1397-1400.[3] Scheinman RI, Cogswell PC, Lofquist AK,et al. Role of transcriptional activation of I kappa B alpha in mediation of immunosuppression by glucocorticoids.Science. 1995;270 (5234): 283-286.[4] Bamberger CM, Schulte HM, Chrousos GP. Molecular determinants of glucocorticoid receptor function and tissue sensitivity to glucocorticoids. Endocr Rev. 1996;17(3): 245-261.[5] Sapolsky RM, Romero LM, Munck AU. How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocr Rev. 2000;21(1):55-89.[6] Munck A, Guyre PM, Holbrook NJ.Physiological functions of glucocorticoids in stress and their relation to pharmacological actions.Endocr Rev. 1984;5(1):25-44.[7] Caplan AI.Mesenchymal stem cells.J Orthop Res. 1991;9(5): 641-650.[8] Oreffo RO, Cooper C, Mason C,et al. Mesenchymal stem cells: lineage, plasticity, and skeletal therapeutic potential. Stem Cell Rev. 2005;1(2):169-178.[9] Kern S, Eichler H, Stoeve J,et al. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue.Stem Cells. 2006;24(5):1294- 1301.[10] Pittenger MF, Mackay AM, Beck SC,et al. Multilineage potential of adult human mesenchymal stem cells.Science. 1999;284(5411):143-147.[11] Derfoul A, Perkins GL, Hall DJ,et al. Glucocorticoids promote chondrogenic differentiation of adult human mesenchymal stem cells by enhancing expression of cartilage extracellular matrix genes.Stem Cells. 2006;24(6):1487-1495.[12] D'Ippolito G, Schiller PC, Ricordi C,et al. Age-related osteogenic potential of mesenchymal stromal stem cells from human vertebral bone marrow.J Bone Miner Res. 1999;14(7): 1115-1122.[13] Dicker A, Le Blanc K, Aström G,et al. Functional studies of mesenchymal stem cells derived from adult human adipose tissue. Exp Cell Res. 2005;308(2):283-290.[14] Izadpanah R, Trygg C, Patel B,et al. Biologic properties of mesenchymal stem cells derived from bone marrow and adipose tissue.J Cell Biochem. 2006;99(5):1285-1297.[15] Zayzafoon M, Gathings WE, McDonald JM. Modeled microgravity inhibits osteogenic differentiation of human mesenchymal stem cells and increases adipogenesis. Endocrinology. 2004;145(5):2421-2432.[16] Aust L, Devlin B, Foster SJ,et al.Yield of human adipose-derived adult stem cells from liposuction aspirates. Cytotherapy. 2004;6(1):7-14.[17] Jackson L, Jones DR, Scotting P,et al. Adult mesenchymal stem cells: differentiation potential and therapeutic applications.J Postgrad Med. 2007;53(2):121-127.[18] Gustavo Duque, Douglas PK. Osteoporosis in older persons: Pathophysiology and Therapeutic Approach. US: Springer, 2009.[19] Yu Y, Wei N, Stanford C,et al. In vitro effects of RU486 on proliferation and differentiation capabilities of human bone marrow mesenchymal stromal cells. Steroids. 2012;77(1-2): 132-137.[20] Bouvard B, Audran M, Legrand E, et al. Ultrastructural characteristics of glucocorticoid-induced osteoporosis. Osteoporos Int. 2009;20(6):1089-1092.[21] Silbermann M, Maor G. Mechanisms of glucocorticoid- induced growth retardation: impairment of cartilage mineralization. Acta Anat (Basel). 1978;101(2):140-149.[22] Hong L, Wei N, Joshi V, et al. Effects of glucocorticoid receptor small interfering RNA delivered using poly lactic-co-glycolic acid microparticles on proliferation and differentiation capabilities of human mesenchymal stromal cells. Tissue Eng Part A. 2012;18(7-8):775-784.[23] 曹谊林,刘伟,崔磊,等.组织工程学理论与实践[M].上海:上海科学技术出版社, 2004: 163. [24] Hanada K, Dennis JE, Caplan AI.Stimulatory effects of basic fibroblast growth factor and bone morphogenetic protein-2 on osteogenic differentiation of rat bone marrow-derived mesenchymal stem cells. J Bone Miner Res. 1997;12(10): 1606-1614. [25] De Boer J, Wang HJ, Van Blitterswijk C. Effects of Wnt signaling on proliferation and differentiation of human mesenchymal stem cells.Tissue Eng. 2004;10(3-4):393-401.[26] Jadlowiec J, Koch H, Zhang X,et al. Phosphophoryn regulates the gene expression and differentiation of NIH3T3, MC3T3-E1, and human mesenchymal stem cells via the integrin/MAPK signaling pathway.J Biol Chem. 2004;279(51): 53323-53330.[27] Ducy P, Zhang R, Geoffroy V, et al. Osf2/Cbfa1: a transcriptional activator of osteoblast differentiation.Cell. 1997;89(5):747-754.[28] Young DW, Hassan MQ, Pratap J,et al. Mitotic occupancy and lineage-specific transcriptional control of rRNA genes by Runx2. Nature. 2007;445(7126):442-446.[29] Hamidouche Z, Haÿ E, Vaudin P, et al. FHL2 mediates dexamethasone-induced mesenchymal cell differentiation into osteoblasts by activating Wnt/beta-catenin signaling- dependent Runx2 expression. FASEB J. 2008;22(11): 3813-3822. [30] Zhou H, Mak W, Zheng Y, et al. Osteoblasts directly control lineage commitment of mesenchymal progenitor cells through Wnt signaling. J Biol Chem. 2008;283(4):1936-1945.[31] Hong JH, Hwang ES, McManus MT, et al. TAZ, a transcriptional modulator of mesenchymal stem cell differentiation.Science. 2005;309(5737):1074-1078.[32] Ito S, Suzuki N, Kato S,et al. Glucocorticoids induce the differentiation of a mesenchymal progenitor cell line, ROB-C26 into adipocytes and osteoblasts, but fail to induce terminal osteoblast differentiation. Bone. 2007;40(1):84-92.[33] Hamidouche Z, Fromigué O, Nuber U,et al. Autocrine fibroblast growth factor 18 mediates dexamethasone-induced osteogenic differentiation of murine mesenchymal stem cells. J Cell Physiol. 2010;224(2):509-515. [34] Liu Y, Titus L, Barghouthi M,et al. Glucocorticoid regulation of human BMP-6 transcription. Bone. 2004;35(3):673-681. [35] Liu T, Gao Y, Sakamoto K, et al. BMP-2 promotes differentiation of osteoblasts and chondroblasts in Runx2-deficient cell lines. J Cell Physiol. 2007;211(3):728- 735.[36] Phillips JE, Gersbach CA, Wojtowicz AM, et al. Glucocorticoid-induced osteogenesis is negatively regulated by Runx2/Cbfa1 serine phosphorylation. J Cell Sci. 2006; 119(Pt 3):581-591. [37] Xiao G, Jiang D, Thomas P,et al. MAPK pathways activate and phosphorylate the osteoblast-specific transcription factor, Cbfa1. J Biol Chem. 2000;275(6):4453-4459.[38] Susperregui AR, Viñals F, Ho PW, et al. BMP-2 regulation of PTHrP and osteoclastogenic factors during osteoblast differentiation of C2C12 cells.J Cell Physiol. 2008;216(1): 144-152.[39] Ahlström M, Pekkinen M, Lamberg-Allardt C. Dexamethasone downregulates the expression of parathyroid hormone-related protein (PTHrP) in mesenchymal stem cells. Steroids. 2009; 74(2):277-282.[40] Zimmerman LB, De Jesús-Escobar JM, Harland RM. The Spemann organizer signal noggin binds and inactivates bone morphogenetic protein 4. Cell. 1996;86(4):599-606.[41] Nifuji A, Kellermann O, Noda M. Noggin inhibits chondrogenic but not osteogenic differentiation in mesodermal stem cell line C1 and skeletal cells. Endocrinology. 2004;145(7): 3434-3442.[42] Rifas L. The role of noggin in human mesenchymal stem cell differentiation. J Cell Biochem. 2007;100(4):824-834.[43] Ayroldi E, Riccardi C. Glucocorticoid-induced leucine zipper (GILZ): a new important mediator of glucocorticoid action. FASEB J. 2009;23(11):3649-3658.[44] Zhang W, Yang N, Shi XM. Regulation of mesenchymal stem cell osteogenic differentiation by glucocorticoid-induced leucine zipper (GILZ). J Biol Chem. 2008;283(8):4723-4729.[45] Chang DJ, Ji C, Kim KK, et al. Reduction in transforming growth factor beta receptor I expression and transcription factor CBFa1 on bone cells by glucocorticoid. J Biol Chem. 1998;273(9):4892-4896.[46] Shao J, Xu X, Li G,et al. Inhibitory effects of pharmacological doses of dexamethasone on mineralization of mesenchymal progenitor cells in vitro. Pharmazie. 2009;64(10):674-679.[47] Diefenderfer DL, Osyczka AM, Garino JP,et al. Regulation of BMP-induced transcription in cultured human bone marrow stromal cells. J Bone Joint Surg Am. 2003;85-A Suppl 3: 19-28.[48] Nakashima K, Zhou X, Kunkel G, et al. The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell. 2002; 108(1):17-29. [49] Cárcamo-Orive I, Gaztelumendi A, Delgado J, et al. Regulation of human bone marrow stromal cell proliferation and differentiation capacity by glucocorticoid receptor and AP-1 crosstalk. J Bone Miner Res. 2010;25(10):2115-2125.[50] Beato M, Herrlich P, Schütz G. Steroid hormone receptors: many actors in search of a plot. Cell. 1995;83(6):851-857.[51] Murrills RJ, Fukayama S, Boschelli F, et al. Osteogenic effects of a potent Src-over-Abl-selective kinase inhibitor in the mouse. J Pharmacol Exp Ther. 2012;340(3):676-687.[52] Stahn C, Löwenberg M, Hommes DW, et al. Molecular mechanisms of glucocorticoid action and selective glucocorticoid receptor agonists. Mol Cell Endocrinol. 2007; 275(1-2):71-78.[53] Rauch A, Seitz S, Baschant U, et al. Glucocorticoids suppress bone formation by attenuating osteoblast differentiation via the monomeric glucocorticoid receptor. Cell Metab. 2010; 11(6):517-531.[54] Kuroda K, Venkatakrishnan R, Salker MS, et al. Induction of 11β-HSD 1 and activation of distinct mineralocorticoid receptor- and glucocorticoid receptor-dependent gene networks in decidualizing human endometrial stromal cells.Mol Endocrinol. 2013;27(2):192-202.[55] Lee SE, Kim JM, Jeong MK, et al.11β-hydroxysteroid dehydrogenase type 1 is expressed in human sebaceous glands and regulates glucocorticoid-induced lipid synthesis and toll-like receptor 2 expression in SZ95 sebocytes.Br J Dermatol. 2013;168(1):47-55.[56] Elvy Suhana MR, Farihah HS, Faizah O,et al. Effect of 11β-HSD1 dehydrogenase activity on bone histomorphometry of glucocorticoid-induced osteoporotic male Sprague-Dawley rats.Singapore Med J. 2011;52(11):786-793.[57] Garbrecht MR, Krozowski ZS, Snyder JM, et al. Reduction of glucocorticoid receptor ligand binding by the 11-beta hydroxysteroid dehydrogenase type 2 inhibitor, Thiram. Steroids. 2006;71(10):895-901.[58] Ge RS, Dong Q, Niu EM, et al. 11{beta}-Hydroxysteroid dehydrogenase 2 in rat leydig cells: its role in blunting glucocorticoid action at physiological levels of substrate.Endocrinology. 2005;146(6):2657-2664.[59] Cooper MS, Stewart PM. 11Beta-hydroxysteroid dehydrogenase type 1 and its role in the hypothalamus- pituitary-adrenal axis, metabolic syndrome, and inflammation. J Clin Endocrinol Metab. 2009;94(12): 4645-4654. [60] Hardy R, Cooper MS. Glucocorticoid-induced osteoporosis - a disorder of mesenchymal stromal cells. Front Endocrinol (Lausanne). 2011;2:24. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [3] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [4] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [5] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [6] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [7] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [8] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [9] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [10] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [11] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [12] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [13] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [14] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [15] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||