Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (4): 631-636.doi: 10.3969/j.issn.2095-4344.1948
Previous Articles Next Articles
Chitosan-based polysaccharide biomaterial for prevention and treatment of oral diseases
Liu Haiyan1,2, Hu Yang1, Wu Xiuping2, Pan Haobo1, Jing Xuan2
- 1Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences, Shenzhen 518055, Liaoning Province, China; 2School of Stomatology, Shanxi Medical University Stomatological Hospital, Taiyuan 030001, Shanxi Province, China
-
Received:2019-03-08Revised:2019-03-16Accepted:2019-05-23Online:2020-02-08Published:2020-01-07 -
Contact:Hu Yang, Associate investigator, Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences, Shenzhen 518055, Liaoning Province, China -
About author:Liu Haiyan, Master candidate, Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences, Shenzhen 518055, Liaoning Province, China; School of Stomatology, Shanxi Medical University Stomatological Hospital, Taiyuan 030001, Shanxi Province, China -
Supported by:the National Natural Science Foundation of China, No. 31570967, Nos. 31870956, 81672227
CLC Number:
Cite this article
Liu Haiyan, Hu Yang, Wu Xiuping, Pan Haobo, Jing Xuan. Chitosan-based polysaccharide biomaterial for prevention and treatment of oral diseases[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 631-636.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

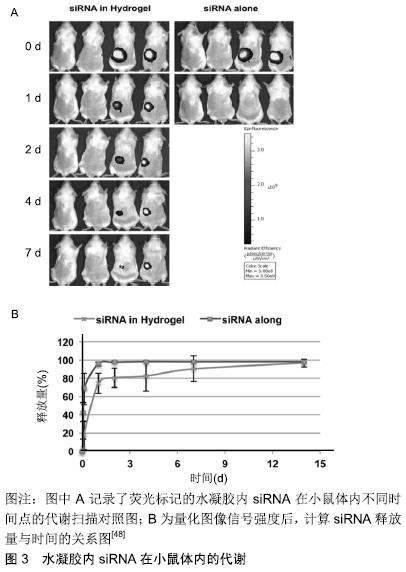
2.1 多糖材料对口腔疾病的预防 壳聚糖是一种包含共聚物葡糖胺和N-乙酰葡糖胺的多糖,具有良好的生物相容性和明显的抑菌性能[2],可作为龋病治疗的潜在药物。有研究证实,壳聚糖对中间普氏菌、牙龈卟啉单胞菌和聚核梭杆菌有良好的抗菌活性[19],且不同的分子质量可对抗口腔内不同的菌属。壳聚糖也可作为一种根管冲洗剂对抗粪肠球菌及白色念珠菌生物膜的细胞毒性,根据抑菌圈直径大小可见壳聚糖的抗菌活性与3%NaOCl和2%氯己定相当[20]。此外,在防龋牙膏中加入壳聚糖可提高含锡离子(Sn2+)牙膏的抗腐蚀性和抗磨性,这对于牙齿钙化差、不耐酸的患者是一个很好的选择[21]。此外,壳聚糖还有更为广泛的应用,如:含铜壳聚糖的杂合纳米颗粒对变形链球菌的抗菌活性与氯己定、氯化十六烷基吡啶相当。壳聚糖在与牙齿羟基磷灰石和细菌细胞壁相互作用下,可加强铜对牙齿表面的附着力及其抗菌斑生物膜作用[22]。与之类似,在壳聚糖内加入天然树脂蜂胶混合制成清漆,可在牙表面涂覆形成一层钝化膜,实现持续性释放,破坏体外微生物活性及预防牙菌斑生物膜形成,且效果较壳聚糖更强、敏感性更高[23]、范围更广[24]。这些多糖物质对菌斑的形成及感染性疾病的治疗和预后有很好的预防效果。 2.2 多糖材料对龋病的防治 在菌斑微生物作用下牙体硬组织缓慢丧失。新研究发现,在病变初期通过对天然聚阳离子多糖-壳聚糖进行磷酸化本体改性,在交联剂的作用下与Ⅰ型胶原进行化学组装,利用仿生唾液的矿化粒子进行磷灰石晶体在牙体原位的形成与组装,可实现再矿化[25]。将一种新的含釉原蛋白壳聚糖水凝胶通过釉原蛋白超分子组装后,与钙-磷(Ca-P)复合链进一步融合形成釉质晶体,可改善受侵蚀牙釉质的硬度和弹性模量,且该水凝胶的pH响应性和抗微生物性质可防止继发龋的发生[8]。 随着龋损范围的扩大,近髓的龋洞可使用磷酸钙和壳聚糖混合生物材料作盖髓剂,促进成牙本质细胞的生长,激发牙髓-牙本质复合体的再生潜力及第三期牙本质(修复性牙本质)的生长[9,26-27]。充填剂可使用壳聚糖改性的玻璃离子水门汀,利用壳聚糖与聚丙烯酸组成成网状结构降低玻璃离子水门汀组件中无机颗粒界面张力,从而在保留玻璃离子水门汀与牙体组织间化学结合的基础上对其机械性能进行提升,且牙体充填后可形成完美的封闭效果,降低了边缘微渗漏的风险[28]。但对于非化学充填材料需要建立疏水层(树脂的相互作用)和亲水性对应物(去矿物质牙质)之间的联系。Chit-MA70是一种酸蚀冲洗的黏结体系,具有亲疏水性,可与修复材料和脱矿的牙齿有机物之间发生物理或化学的相互作用。利用该黏结系统治疗后,在对牙齿进行修复体的热机械循环处理时其黏结性能不会发生改变。因此该系统可很好的应用于临床,减少在外界因素作用下的微渗漏或继发龋的发生[10]。总之,多糖材料的使用可遍及龋损发生的全过程,不论是早期预防、釉质再生还是龋损充填修复,均可发挥其良好的性能。 2.3 多糖材料对牙周病的防治 在机体防御能力下降时,定植于牙面的条件致病菌可选择性吸附于牙龈上皮,其胞外的脂多糖物质加之产生的酶元素如胶原酶、蛋白酶或透明质酸酶等可进行性破坏牙周组织,诱发牙周炎症。 2.3.1 多糖材料对牙周病早期的治疗 牙周炎症初期可通过局部冲洗上药控制炎症,但长期反复使用可引起潜在的不良反应,导致细菌耐药性和龈沟液稀释下抗生素浓度不足[29-31]。作为一种多孔药物递送载体,壳聚糖具有良好的生物相容性、低毒性和免疫刺激活性[25]。在热量和pH值刺激下,壳聚糖凝胶化,当药物到达指定位置后,壳聚糖可通过溶菌酶和壳聚糖酶在体内酶促降 解[32]。此外,壳聚糖载体可促进药物的释放、增加对牙本质的黏结[33],对特定对牙周炎初期的感染细菌发挥很好的抗菌性能,抑制疾病的进展,避免了到达指定位置药物浓度不足的缺陷,诱导促进组织的功能性修复。 2.3.2 多糖材料对牙周病进展期的治疗 随着病变的进展,结合上皮向根方迁移、牙周袋加深,暴露的无血管根面不适合细胞的生存,将人牙龈组织中的成纤维细胞在酯化苄基透明质酸支架上进行培养,可促进牙龈组织的增加和角化[34],但作用甚微。而利用富含抗氧化剂牛磺酸的壳聚糖薄膜,可极大刺激巨噬细胞和成纤维细胞活性,促进上皮组织的生长修复[35],以及牙周细胞的基因表达和成骨能力[36]。利用另一种多糖基物质如透明质酸聚合物对牙根表面进行处理,可促进成纤维细胞的黏附和增加生存力[37]。除此之外,临床上可通过翻瓣术对牙周炎症进行处理,但术后引起的感染、肿胀也不可避免。右派酚已被证明具有抗炎作用,可促进成纤维细胞增殖及伤口愈合处重新上皮化。右旋潘酚、尿囊素和壳聚糖的组合物可有效减少口腔外科手术介入后的并发症[38]。当牙周炎发展较为严重并伴随有牙槽骨吸收时,多糖物质也可很好的发挥其效能。 2.3.3 多糖材料对牙槽骨吸收的治疗 在炎症因子的刺激作用下,破骨细胞活性增强并发牙槽骨吸收及牙齿Ⅲ度松动时,多糖材料与骨形成间也建立了坚固的桥梁。作为成骨细胞的一种附加有效促分裂原,肾上腺髓质素刺激原代成骨细胞和成骨细胞样细胞系的增殖,也具有血管生成和抗菌作用[39-40]。壳聚糖微球通过乳液-离子交联形成肾上腺髓质素递送的方法。肾上腺髓质素的局部应用可能保留残余的牙槽嵴并加速牙槽骨重塑[41]。透明质酸具有骨诱导、抑菌和抗炎特性,可改善骨形成并加速感染牙槽窝的愈合[15]。此外,研究者发现新型壳聚糖- cPGA聚电解质复合水凝胶或乙酰化甘露聚糖处理过的海绵,作为拔牙创伤口敷料时利于拔牙后牙槽窝内新骨的形成[42],可促进骨愈合与增加骨密度[43]。硫酸钙因高度生物相容性、生物可吸收、材料成本低、成型性强,一直被作为骨骼填充剂和抗生素载体用于治疗骨骼和/或牙齿缺陷[44]。硫酸钙与壳聚糖聚合后具有较高的抗压强度和良好的促细胞活性[9]。而羟基磷灰石与壳聚糖的结合,不仅在机械强度上与骨组织类似,还可实现骨再 生[45]。由于外源性植体会引起异物反应,干细胞技术的应用可克服这一缺陷。磷酸锶对人脱落乳牙干细胞的成骨分化具有促进作用,虽壳聚糖对干细胞的膜内成骨和软骨形成有局限性,但含锶壳聚糖支架上的Ⅰ型胶原蛋白表达、碱性磷酸酶活性和钙沉积含量明显增加,且将细胞在3D支架中动态培养,最佳的流动速率促进细胞的成骨分化,极大促进了骨再生效果[46-47]。后期的研究发现,利用分子信号通路也可改善牙槽骨吸收。利用热敏壳聚糖水凝胶作为小干扰RNA储库和载体而用于RANK信号传导时,该水凝胶可延长siRNA释放,见图3,伴随着细胞内持续的基因沉默效应[48]。 "

2.3.4 多糖材料在美学方面的功效 功能性修复完成后,口腔临床医生还需解决患者的美观问题。牙周病患者,因后期牙齿松动不齐及牙龈退缩而形成黑三角,这对年轻女性患者来说是致命的一大缺陷。利用透明质酸填充剂做开放性牙龈充填物进行牙龈乳头重建[49],可产生很好的视觉美感。后期研究发现,透明质酸也可作再生制剂及用于局部控制牙周出血[50-51]。所以多糖聚合物可从分子、细胞及组织学等众多方面解决牙周炎发生发展过程中的不利影响,不仅满足了患者的功能要求,也实现了美学效果。 2.4 多糖材料在其他口腔疾患中的应用 在口腔医学领域中还有一类疾病较为常见,即阻生智齿。因为现代人类食物的精细化,使得颌骨较小,第三磨牙很难萌出,智牙拔除术是很好的解决方案。但因为阻生程度不同,对于重度阻生智齿的拔除将会引起一系列的术后反应,如出血、肿胀、炎症反应等。纤维素为目前药物(如抗炎杀菌药)薄膜包衣使用最多的材料,如乙基纤维素、羟丙基甲基纤维素、邻苯二甲酸羟丙基纤维素酯是目前研究较多的胃溶性药物包衣材料,但使用成本较高。与之相比,淀粉是一种易被微生物降解的碳水化合物,由单一类型的糖单元(D-葡萄糖)组成,易被人体降解吸收,成膜性好,通过改性可改善其阻隔性能和成膜性能[52],在指定位点发挥其生物效能[53]。这两者作为植物来源的2大物质,有较广泛的来源,也有很大的发展前途。透明质酸作为人体组织重要的一种碳水化合物,具有各种功能,包括:维护关节滑液的弹性黏度,控制组织水合作用和许多受体介导的细胞脱落作用,例如有丝分裂、迁移、肿瘤转移和炎症[54]。它的生物相容性和非免疫原性可协助伤口再生和皮肤填充[55-57]。与盐酸苯达胺(非类固醇类消炎镇痛药)喷雾剂相比,透明质酸喷雾剂可有效减少术后肿胀和张口受限[58]。此外,多糖类物质在口腔其他疾病治疗方面也有较多的应用,需要人们在后期不断探索中其在口腔众多领域的应用价值。 "

| [1] KOVACS CJ, FAUSTOFERRI RC, QUIVEY RG JR. RgpF Is Required for Maintenance of Stress Tolerance and Virulence in Streptococcus mutans.J Bacteriol.2017;199(24):1-16. [2] CHEN L, REN Z, ZHOU X, et al.Inhibition of Streptococcus Mutans Biofilm Formation, Extracellular Polysaccharide Production, and Virulence by an Oxazole Derivative. Appl Microbiol Biotechnol.2016; 100(2): 857-867. [3] XIAO J, HARA AT, KIM D, et al.Bio Fi Lm Three-Dimensional Architecture in Fl Uences in Situ PH Distribution Pattern on the Human Enamel Surface.Nat Publ Gr.2017;9(2):74-79. [4] MORADIAN-OLDAK J.Protein-Mediated Enamel Mineralization.Front Biosci.2012;17(7):1996. [5] PISTONE S, GOYCOOLEA FM, YOUNG A, et al.Formulation of polysaccharide- based nanoparticles for local administration into the oral cavity.Eur J Pharm Sci.2017;96:381-389. [6] KULKARNI DP, SABOO SS.Polymers Used in Floating Drug Delivery System : A Review.Eur J Pharm Med Res.2017;4(8):611-616. [7] HO YC, HUANG FM, CHANG YC.Cytotoxicity of Formaldehyde on Human Osteoblastic Cells Is Related to Intracellular Glutathione Levels.J Biomed Mater Res B Appl Biomater.2007;83(2):340-344. [8] COVARRUBIAS C, TREPIANA D, CORRAL C.Synthesis of hybrid copper-chitosan nanoparticles with antibacterial activity against cariogenic Streptococcus mutans.Dent Mater J.2018;37(3):379-384. [9] LOW A, MOHD YUSOF H, REZA F, et al.Gypsum-based biomaterials: Evaluation of physical and mechanical properties, cellular effects and its potential as a pulp liner.Dent Mater J. 2015;34(4):522-528. [10] DIOLOSÀ M, DONATI I, TURCO G, et al.Use of Methacrylate-Modified Chitosan to Increase the Durability of Dentine Bonding Systems. Biomacromolecules.2014;15(12):4606-4613. [11] 王凤川.载金属离子含羧基多糖基纳米材料的制备及其结构性能表征[D].上海:东华大学,2007. [12] 杨跃辉,杨希琴,丁平田.牙周用交联淀粉碘微球软膏初步药效学研究[J].中国临床药理学杂志,2011,27(3):206-209. [13] SOUZA FILHO MD, MEDEIROS JVR, VASCONCELOS DFP, et al. Orabase formulation with cashew gum polysaccharide decreases inflammatory and bone loss hallmarks in experimental periodontitis.Int J Biol Macromol.2018;107(Pt A):1093-1101. [14] CHEN S, YANG JY, ZHANG SY, et al.Effects of simvastatin gel on bone regeneration in alveolar defects in miniature pigs.Chin Medi J. 2012; 16(51):9501-9506. [15] KIM JJ, SONG HY, BEN AMARA H, et al.Hyaluronic Acid Improves Bone Formation in Extraction Sockets With Chronic Pathology:A Pilot Study in Dogs.J Periodontol.2016;87(7):790-795. [16] HENDRIKS J, RIESLE J, VAN BLITTERSWIJK CA.Co-culture in cartilage tissue engineering.J Tissue Eng Regen Med.2007;1(3):170-178. [17] VIJAN K.Mechanism of action and clinical effects of teeth whitening on enamel and dentin bonding for resin-based restorations.Asian Pacific J Health Sci.2018;5(1):142-145. [18] PARK S, KIM HH, YANG SB, et al.A Polysaccharide-Based Antibacterial Coating with Improved Durability for Clear Overlay Appliances.ACS Appl Mater Interfaces.2018;10(21):17714-17721. [19] JEON JG, ROSALEN PL, FALSETTA ML, et al.Natural products in caries research: current (limited) knowledge, challenges and future perspective.Caries Res.2011;45(3):243-263. [20] YADAV P, CHAUDHARY S, SAXENA RK, et al.Evaluation of Antimicrobial and Antifungal Efficacy of Chitosan as Endodontic Irrigant against Enterococcus Faecalis and Candida Albicans Biofilm Formed on Tooth Substrate.J Clin Exp Dent 2017;9(3):e361-e367. [21] SCHLUETER N, KLIMEK J, GANSS C.Effect of a Chitosan Additive to a Sn2+-Containing Toothpaste on Its Anti-Erosive/Anti-Abrasive Efficacy-a Controlled Randomised in Situ Trial.Clin Oral Investig. 2014;18(1):107-115. [22] COVARRUBIAS C, TREPIANA D, CORRAL C.Synthesis of hybrid copper-chitosan nanoparticles with antibacterial activity against cariogenic Streptococcus mutans.Dent Mater J.2018;37(3):379-384. [23] STROHMENGER L, BRAMBILLA E.The Use of Fluoride Varnishes in the Prevention of Dental Caries: A Short Review.Oral Dis. 2001;7(2):71-80. [24] FRANCA J R, DE LUCA MP, RIBEIRO TG, et al.Propolis--based chitosan varnish: drug delivery, controlled release and antimicrobial activity against oral pathogen bacteria.BMC Complement Altern Med. 2014;14:478. [25] 任迅,姚静,杜芹,等.通过自生长方式实现壳聚糖—胶原聚合物引导的牙体硬组织仿生再矿化[J].华西口腔医学杂志,2014,32(5):519-523. [26] ZHANG Y, WANG Y, SHI B, et al.A Platelet-Derived Growth Factor Releasing Chitosan/Coral Composite Scaffold for Periodontal Tissue Engineering.Biomaterials.2007;28(8):1515-1522. [27] PERINELLI DR, FAGIOLI L, CAMPANA R, et al.Chitosan-based nanosystems and their exploited antimicrobial activity.Eur J Pharm Sci. 2018;117:8-20. [28] LEE B, LEE C, WANG Y, et al.Controlled-release of tetracycline and lovastatin by poly(D,L-lactide-co-glycolide acid)-chitosan nanoparticles enhances periodontal regeneration in dogs.Int J Nanomedicine. 2016; 11:285-297. [29] HO YC, HUANG FM, CHANG YC.Cytotoxicity of Formaldehyde on Human Osteoblastic Cells Is Related to Intracellular Glutathione Levels.J Biomed Mater Res B Appl Biomater.2007;83(2):340-344. [30] SHRESTHA A, ZHILONG S, GEE NK, et al.Nanoparticulates for Antibiofilm Treatment and Effect of Aging on Its Antibacterial Activity.J Endod. 2010;36(6):1030-1035. [31] SILVA PV, GUEDES DFC, NAKADI FV, et al.Chitosan: A New Solution for Removal of Smear Layer after Root Canal Instrumentation.Int Endod J.2013;46(4):332-338. [32] TONETTI M, CUGINI MA, GOODSON JM.Zero-order delivery with periodontal placement of tetracycline-loaded ethylene vinyl acetate fibers.J Periodontal Res.1990;25(4):243-249. [33] PERCHYONOK V, GROBLER S, ZHANG S. IPNs from Cyclodextrin: Chitosan Antioxidants: Bonding, Bio-Adhesion, Antioxidant Capacity and Drug Release. J Funct Biomater. 2014;5(3):183-196. [34] PRATO GP, ROTUNDO R, MAGNANI C, et al.An autologous cell hyaluronic acid graft technique for gingival augmentation: a case series.J Periodontol.2003;74(2):262-267. [35] XU C, LEI C, MENG L, et al.Chitosan as a barrier membrane material in periodontal tissue regeneration.J Biomed Mater Res B Appl Biomater. 2012;100(5):1435-1443. [36] YAN XZ, VAN DEN BEUCKEN J, YUAN C, et al.Spheroid formation and stemness preservation of human periodontal ligament cells on chitosan films.Oral Dis.2018;24(6):1083-1092. [37] FRANÇA MCM, DA SILVA TM, SILVA GO,et al.Effect of ethylenediaminetetraacetic acid and hyaluronic acid on the viability and cytokine expression of periodontal ligament fibroblasts.Dent Traumatol. 2018. doi: 10.1111/edt.12404.[Epub ahead of print] [38] LOPEZ-LOPEZ J, JAN-PALLÍ E, LEZ-NAVARRO BG, et al.Efficacy of chlorhexidine, dexpanthenol, allantoin and chitosan gel in comparison with bicarbonate oral rinse in controlling post-interventional inflammation, pain and cicatrization in subjects undergoing dental surgery.Curr Med Res Opin. 2015;31(12):2179-2183. [39] BENYAHIA Z, DUSSAULT N, CAYOL M, et al. Stromal Fibroblasts Present in Breast Carcinomas Promote Tumor Growth and Angiogenesis through Adrenomedullin Secretion. Oncotarget. 2017; 8(9):15744-15762. [40] GRÖSCHL M, WENDLER O, TOPF HG, et al.Significance of Salivary Adrenomedullin in the Maintenance of Oral Health: Stimulation of Oral Cell Proliferation and Antibacterial Properties. Regu Pept.2009;154 (1-3):16-22. [41] WANG L, ZHENG L, LI C, et al.Adrenomedullin delivery in microsphere-scaffold composite for remodeling of the alveolar bone following tooth extraction: an experimental study in the rat.Biomed Eng Online. 2013; 12:99. [42] CHANG HH, WANG YL, CHIANG YC, et al.A novel chitosan-γPGA polyelectrolyte complex hydrogel promotes early new bone formation in the alveolar socket following tooth extraction.PLoS One. 2014;9(3): e92362. [43] PISTONE S, RYKKE M, SMISTAD G, et al.Polysaccharide-Coated Liposomal Formulations for Dental Targeting.Int J Pharm. 2017; 516(1-2):106-115. [44] HESARAKI S, MOZTARZADEH F, NEMATI R, et al.Preparation and characterization of calcium sulfate-biomimetic apatite nanocomposites for controlled release of antibiotics.J Biomed Mater Res B Appl Biomater.2009;91(2):651-661. [45] VACA-CORNEJO F,REYES H,JIMÉNEZ S,et al.Pilot Study Using a Chitosan-Hydroxyapatite Implant for Guided Alveolar Bone Growth in Patients with Chronic Periodontitis.J Funct Biomater.2017;8(3):29. [46] SU WT, WU PS, KO CS, et al.Osteogenic differentiation and mineralization of human exfoliated deciduous teeth stem cells on modified chitosan scaffold.Mater Sci Eng C Mater Biol Appl. 2014;41: 152-160. [47] FAREA M, HUSEIN A, HALIM AS, et al.Synergistic effects of chitosan scaffold and TGFβ1 on the proliferation and osteogenic differentiation of dental pulp stem cells derived from human exfoliated deciduous teeth.Arch Oral Biol.2014;59(12):1400-1411. [48] MA Z, YANG C, SONG W, et al.Chitosan Hydrogel as Sirna Vector for Prolonged Gene Silencing.J Nanobiotechnol.2014;12(1):23. [49] NAORUNGROJ S. Esthetic Reconstruction of Diastema with Adhesive Tooth-Colored Restorations and Hyaluronic Acid Fillers.Case Rep Dent. 2017;2017:5670582. [50] FALLACARA A, MANFREDINI S, DURINI E, et al.Hyaluronic Acid Fillers in Soft Tissue Regeneration.Facial Plast Surg.2017;33(1):87-96. [51] PI S, CHOI YJ, HWANG S, et al.Local Injection of Hyaluronic Acid Filler Improves Open Gingival Embrasure; Validation Through a Rat Model.J Periodontol.2017;88(11):1221-1230. [52] 纪趁趁.淀粉基材料复合改性及其在药用辅料方面的应用[D].贵阳:贵州大学,2017. [53] MIYAHARA T, NYAN M, SHIMODA A, et al.Exploitation of a Novel Polysaccharide Nanogel Cross-Linking Membrane for Guided Bone Regeneration(GBR).2012;2011:666-672. [54] GOCMEN G, GONUL O, OKTAY NS, et al.The Antioxidant and Anti-Inflammatory Efficiency of Hyaluronic Acid after Third Molar Extraction.J Cranio-Maxillofacial Surg.2015;43(7):1033-1037. [55] WANG Z, QIAN Y, LI L, et al.Evaluation of Emulsion Electrospun Polycaprolactone/Hyaluronan/Epidermal Growth Factor Nanofibrous Scaffolds for Wound Healing.J Biomater Appl.2016;30(6):686-698. [56] AYA KL, STERN R.Hyaluronan in Wound Healing: Rediscovering a Major Player.Wound Repair Regen. 2014;22(5):579-593. [57] TAMER TM, VALACHOVÁ K, HASSAN MA, et al. Chitosan/ Hyaluronan/ Edaravone Membranes for Anti-Inflammatory Wound Dressing: In Vitro and in Vivo Evaluation Studies.Mater Sci Eng C.2018;90(September 2017):227-235. [58] KORAY M, OFLUOGLU D, ONAL EA, et al. Efficacy of Hyaluronic Acid Spray on Swelling, Pain, and Trismus after Surgical Extraction of Impacted Mandibular Third Molars.Int J Oral Maxillofac Surg. 2014; 43(11):1399-1403. |
| [1] | Wang Feng, Zhou Liyu, Saijilafu, Qi Shibin, Ma Yanxia, Wei Shanwen. CaMKII-Smad1 promotes axonal regeneration of peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1064-1068. |
| [2] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [3] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [4] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [5] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [6] | Wang Yujiao, Liu Dan, Sun Song, Sun Yong. Biphasic calcium phosphate loaded with advanced platelet rich fibrin can promote the activity of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 504-509. |
| [7] | Liu Jiangfeng. Nano-hydroxyapatite/polyamide 66 composite filling combined with locking plate in the treatment of fibrous dysplasia of femoral bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 542-547. |
| [8] | Li Li, Ma Li. Immobilization of lactase on magnetic chitosan microspheres and its effect on enzymatic properties [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 576-581. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Li Shanshan, You Ran, Guo Xiaoxiao, Zhao Lu, Wang Yanling, Chen Xi. Advances in the mechanisms of optic nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3740-3745. |
| [11] | Li Xinping, Cui Qiuju, Zeng Shuguang, Ran Gaoying, Zhang Zhaoqiang, Liu Xianwen, Fang Wei, Xu Shuaimei. Effect of modification of β-tricalcium phosphate/chitosan hydrogel on growth and mineralization of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3493-3499. |
| [12] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| [13] | Lang Limin, He Sheng, Jiang Zengyu, Hu Yiyi, Zhang Zhixing, Liang Minqian. Application progress of conductive composite materials in the field of tissue engineering treatment of myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3584-3590. |
| [14] | Ma Qing, Shi Liyan, Huang Sixue, Zheng Zhangbowen, Zhang Aihua, Zhan Desong, Fu Jiale. Research status and prospect of zirconia ceramics in dental prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3597-3602. |
| [15] | Wen Zhijing, Gu Pengzhen, He Xijing, Li Jialiang, Wang Yibin, Wang Yiqun. Development of high molecular polymer polyetherketoneketone and its prospects in medical applications [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3603-3608. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||