Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (23): 3740-3745.doi: 10.12307/2021.048
Previous Articles Next Articles
Advances in the mechanisms of optic nerve regeneration
Li Shanshan, You Ran, Guo Xiaoxiao, Zhao Lu, Wang Yanling, Chen Xi
- Department of Ophthalmology, Beijing Friendship Hospital, Capital Medical University, Beijing 100050, China
-
Received:2020-03-23Revised:2020-03-27Accepted:2020-07-04Online:2021-08-18Published:2021-02-24 -
Contact:Wang Yanling, MD, Chief physician, Department of Ophthalmology, Beijing Friendship Hospital, Capital Medical University, Beijing 100050, China Co-corresponding author: Chen Xi, MD, Physician, Department of Ophthalmology, Beijing Friendship Hospital, Capital Medical University, Beijing 100050, China -
About author:Li Shanshan, MD candidate, Department of Ophthalmology, Beijing Friendship Hospital, Capital Medical University, Beijing 100050, China -
Supported by:the National Natural Science Foundation of China (General Project), No. 81870686 (to WYL); Beijing Natural Science Foundation, No. 7184201 (to CX); Capital Health Development Research Special Fund, No. 2018-1-2021 (to WYL)
CLC Number:
Cite this article
Li Shanshan, You Ran, Guo Xiaoxiao, Zhao Lu, Wang Yanling, Chen Xi. Advances in the mechanisms of optic nerve regeneration[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3740-3745.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
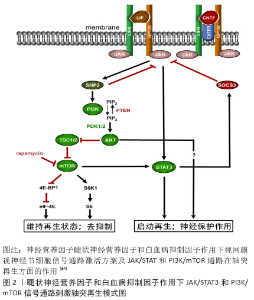
2.1 视网膜神经节细胞轴突再生 尽管成年哺乳动物视网膜神经节细胞轴突不可再生,但是如果将外周神经移植体缝合到视神经断端,部分视网膜神经节细胞可出现轴突再生;如果将外周神经移植物的远端植入上丘中,再生轴突可与上级神经元形成突触[4];如果将外周神经移植物的远端植入橄榄顶盖前核,可恢复瞳孔对光反射,表明光信号的传入和传出功能都得到了重建[5]。基于这些研究,虽然视网膜神经节细胞的轴突本身不可再生,但是可以通过外周神经再生,推测其可能的原因为中枢神经系统髓鞘对轴突再生的抑制作用。在1996年,BERRY等[6]对周围神经移植促进视网膜神经节细胞轴突再生提出了不同解释,认为可能原因为许旺细胞产生的营养因子。为验证这一假说,其团队将外周神经的自体片段植入眼球后方,成年大鼠视网膜神经节细胞视神经的轴突出现了再生。这一重要的结果首次提出了视神经并不是不可再生,并提供了确切的证据。 实现视神经再生的关键步骤包括促进视网膜神经节细胞存活、轴突再生、髓鞘再生,延伸投射到视觉中枢并建立具有功能的突触。当前促进视神经再生的治疗包括提供神经营养因子、炎症刺激、轴突生长抑制解除、细胞内信号传导修饰等。 2.1.1 神经营养性因子促进轴突再生 神经营养性因子是一系列在发育期间介导神经元细胞生长、分化和存活的可扩散的蛋白因子。经典的神经营养性因子可以大致分为神经营养因子、神经因子和其他生长因子。神经营养因子家族包括4种多肽,即脑源性神经营养因子、神经生长因子、神经营养因子3和神经营养因子4/5,这些多肽在结构和功能上相关,并通过特异性地与原肌球蛋白激酶受体A-C结合来介导它们的作用[7]。神经因子家族包括睫状神经营养因子和白血病抑制因子,这两者都通过包含糖蛋白130的信号转导受体和白血病抑制因子受体来介导其发挥作用[8]。其他生长因子包括肝细胞生长因子、成纤维细胞生长因子和神经胶质细胞分泌的营养因子等。 视网膜神经节细胞表达这些生长因子的受体,并且玻璃体腔内注射这些因子可在视神经受到损伤时促进视网膜神经节细胞的存活。然而,营养因子只是减缓,却不能避免视网膜神经节细胞的死亡[9]。脑源性神经营养因子是已被证实促进轴突切断的视网膜神经节细胞存活作用最明确的因子之一,玻璃体腔内注射脑源性神经营养因子可促进轴突切除的视网膜神经节细胞的存活[10];并且,脑源性神经营养因子注射到上丘中也减少了发育性视网膜神经节细胞的死亡[11];脑源性神经营养因子除可促进视网膜神经节细胞的存活,也可通过阻断轴突生长抑制因子而促进视网膜神经节细胞轴突生长[12]。而睫状神经营养因子刺激轴突再生的作用可能比脑源性神经营养因子更有效[13]。在大鼠青光眼模型中,玻璃体腔内注射脑源性神经营养因子未能显示保护作用,而注射睫状神经营养因子表现出显著的保护效果[14]。睫状神经营养因子的保护作用与cAMP水平,炎症刺激和STAT3途径的活化密切相关[15]。此外,肝细胞生长因子在体外和体内环境下,均可促进轴突再生和神经元存活[16]。也有研究表明,骨桥蛋白可显著增强αRGC对胰岛素样生长因子1或脑源性神经营养因子刺激的应答,并促进αRGC的存活和轴突再生[17]。骨桥蛋白是否对其他神经营养性因子具有相似的协同促进作用还有待进一步的研究。 2.1.2 炎症刺激视神经轴突再生 炎症刺激已被证实具有促进静息状态的视网膜神经节细胞转化为活化再生状态的作用[18]。研究显示,玻璃体腔移植外周神经的片段具有增强轴突切断的视网膜神经节细胞的再生反应[19]。最初,学者们认为促进再生的作用来自外周神经分泌的营养因子,但后续研究发现炎症刺激在其中也起到重要作用。浸润的炎症细胞(例如巨噬细胞和中性粒细胞)是其中起到调控作用的主体,并且为癌钙调蛋白(oncomodulin)来源[20]。癌钙调蛋白是来自小白蛋白家族的钙结合蛋白,由玻璃体和视网膜中活化的巨噬细胞和中性粒细胞分泌;此外,晶状体损伤或注射酵母聚糖等炎症反应也可招募炎症细胞并分泌高水平的癌钙调蛋白[21]。在炎性反应状态,癌钙调蛋白在轴突再生中起重要作用[21]。从巨噬细胞条件培养基中去除癌钙调蛋白则保护作用消失,其保护作用取决于升高的cAMP和甘露糖的存在[9]。而只有当cAMP升高或视网膜神经节细胞膜被穿透后,癌钙调蛋白才能与视网膜神经节细胞结合。据此,cAMP可能介导了癌钙调蛋白受体在视网膜神经节细胞上的易位,促进视网膜神经节细胞对癌钙调蛋白的应答[22]。因此,尽管癌钙调蛋白是否可发挥保护作用取决于cAMP和甘露糖的存在,但是甘露糖也仅在cAMP的存在下才能够发挥其作用。除此之外,神经因子也在损伤介导的炎症反应中发挥对轴突的保护作用。在CNTF-/-小鼠或CNTF-/-LIF-/-双敲除小鼠中,晶状体损伤对轴突生长的促进作用明显减弱,表明神经营养性因子是炎症诱导轴突再生所必需的[23]。在炎症反应情况下,促进轴突再生的关键因子已被确定为睫状神经营养因子、白血病抑制因子和白细胞介素6[24],而其中涉及的信号途径包括JAK/STAT3和PI3K/ATK/mTOR[25]。 2.1.3 视神经再生的内源性调控及基因治疗 在视觉投射的初始形成期间,在体内环境下视网膜神经节细胞的轴突可快速生长,在体外培养状态下视网膜神经节细胞受损的轴突仍具有再生能力;但是视网膜神经节细胞在出生后的早期阶段即丧失了轴突快速生长和再生的能力,这一阶段的视网膜神经节细胞朝向视网膜侧的树突生长增加,收到更多突触传入信号。体外实验证实,视网膜神经节细胞丧失轴突快速生长和再生的能力是由视网膜神经节细胞和无长突细胞的接触触发的[26]。这一结果显示,视网膜神经节细胞随着发育进程逐渐丧失的轴突再生能力和其减弱的轴突快速生长能力相关,而这一过程是由外界环境(与无长突细胞的接触)所触发,并不是由视网膜神经节细胞自身调控的。 在分子水平,视网膜神经节细胞的轴突再生可以通过增强或减弱细胞内特定的信号通路而得到部分恢复。哺乳动物无菌20样激酶3b(Mammalian sterile 20-like kinase 3b,Mst3b)属于嘌呤敏感蛋白激酶,为ERK通路的下游信号因子,可调节胚胎和成熟神经系统中的轴突生长[27]。抑制Mst3b可阻断癌钙调蛋白和其他营养因子如脑源性神经营养因子和神经生长因子对轴突的再生作用,而活化的Mst3b在没有其他营养因子的情况下仍可促进轴突再生[28]。此外,在条件B-RAF功能获得小鼠模型中,RAF-MEK信号的激活也可促进视神经损伤后的轴突再生[29]。 与神经营养因子相关的信号通路包括CaM激酶、MAP激酶、JAK/STAT和PI3K/mTOR等[30]。抑制CaM激酶可阻断癌钙调蛋白的保护作用,而抑制MAP激酶、JAK/STAT和PI3K/mTOR可阻断其他营养因子的保护作用(图2)。所以,恢复上述信号通路的表达可促进轴突持续再生。PTEN基因已被鉴定为肿瘤抑制基因,并且是PI3K/mTOR的负调节因子,而SOCS3基因可抑制JAK/STAT信号传导。PTEN的缺失可激活PI3K/mTOR通路并显著促使α-RGC的轴突再生[31];同时下调PTEN与SOCS3并配合使用睫状神经营养因子,可产生非常强大的促进轴突再生的作用。然而,几乎没有再生的轴突能够延伸进入视交叉[32]。此外,PTEN的缺失也可显著增强炎症反应对视神经再生的促进作用。下调PTEN同时组合使用cAMP类似物和酵母聚糖(可提高癌钙调蛋白和其它营养因子的水平),通过6周的时间一部分视网膜神经节细胞的轴突可再生形成完整视神经的长度并通过视交叉[15]。10-12周后,部分轴突可投射到视觉皮质的靶点位置[33]。 ROCK属于丝氨酸-苏氨酸激酶AGC(PKA/PKG/PKC)家族,主要通过作用于细胞骨架调节细胞的形状和运动。ROCK主要有2种同源异构体,ROCK1主要表达在肺、肝和肾,ROCK2主要分布在脑和心脏。ROCK为轴突延伸的负调节因子,PTEN则是其下游靶标之一。而ROCK通路可被髓鞘中表达的生长抑制剂(MAG、Nogo和少突胶质细胞髓鞘糖蛋白等)激活。ROCK抑制剂现被认为可能成为治疗青光眼的新型制剂[35]。 Krüppel样因子(Kru?ppel-like factor,Klf)是一系列锌指DNA结合蛋白,可调节神经系统的发育和再生[36]。其中,轴突生长能力随着发育成熟逐渐降低与Krüppel样因子4和Krüppel样因子9的表达增加有关。在细胞培养环境下,过表达这2个基因会抑制神经元的轴突生长[37]。在成熟小鼠中下调Krüppel样因子4的表达,可通过抑制JAK/STAT信号通路,促进视神经损伤后的再生[37]。此外,Krüppel样因子6和Krüppel样因子7表达下调也与轴突再生能力下降密切相关[37]。在斑马鱼,Krüppel样因子6和Krüppel样因子7表达下调会引起视神经的再生能力下降[38];在体外培养中,也有抑制轴突生长的能力。以上研究结果表明Krüppel样因子家族在成年哺乳动物中枢神经系统轴突再生能力的调控中起到重要作用。 cAMP可通过多种机制促进轴突再生[39-40]。cAMP使生长锥对生长抑制信号失去反应,并促进轴突再生蛋白的表达。细胞内cAMP水平的升高还增强损伤的视网膜神经节细胞对营养因子的应答。cAMP也可促使癌钙调蛋白结合于视网膜神经节细胞表面受体,并延长其结合时间[15]。 Mu?lle 细胞是视网膜放射状胶质细胞,研究发现Mu?lle 细胞通过一些条件可去分化为视网膜祖细胞来补充视网膜丢失的神经元,ZHOU等[41]发现Mu?lle 细胞内多聚嘧啶结合蛋白1(Ptbp1)下调后,分化为视网膜神经节细胞,从而改善了视网膜变性老鼠的视功能;Elsaeidi及其团队利用转基因老鼠,发现视网膜Mu?lle 细胞过表达Ascl1及Lin8a基因可补充视网膜内核层及丢失的神经元及视网膜神经节细胞[42]。重编程的Mu?lle 细胞可分化为视网膜神经节细胞,有望成为治疗视网膜神经节细胞丢失相关疾病的新方法。 2.2 中心视觉区域的重建和生理反应的恢复 在发育过程中,视网膜神经节细胞的轴突在趋化因子的引导下,延伸至视觉中枢相应的目标位置[43]。最近的研究表明,再生轴突能够正确地延伸至视觉中枢相应的靶点和恢复功能反应。正如前文所述,下调PTEN配合酵母聚糖与cAMP类似物可促使部分再生轴突延伸通过整个视神经,穿过视交叉并进入视束。在这种情况下,组织学证据显示在上级视觉通路存在一定程度的神经再支配,这些区域包括外侧膝状体背侧核、外侧膝状体腹侧核、上丘、视交叉上核等[33]。大多数轴突再生成功的小鼠能够重新建立昼夜节律,并且部分小鼠还可显示出对旋转黑白光栅的视动反应和悬崖回避,提示轴突再生后的小鼠视功能得到了部分恢复[33]。相比之下,通过病毒转染表达睫状神经营养因子可刺激少数视网膜神经节细胞轴突再生延伸至视交叉,但很少可通过视交叉,并且没有再生的轴突投射到视觉中枢[44]。同时下调PTEN和SOCS3的表达配合使用睫状神经营养因子,可促使大量轴突再生通过损伤区域,但是几乎没有轴突可以通过视交叉,即使通过视交叉的少量轴突延伸的轨迹也和正常视束不完全一致[41]。这些研究都没能得到下调PTEN/酵母聚糖/cAMP研究促使新生轴突正确投射至视觉中枢的结果。然而也有研究表明,当视神经损伤接近视交叉时,下调PTEN/SOCS3配合睫状神经营养因子可使轴突投射至上丘并恢复突触后反应[45]。过表达c-myc可显著增强下调PTEN/SOCS3组合睫状神经营养因子对轴突再生的促进作用,并促使大量轴突延伸进入视束,但其是否可投射至视觉中枢的正确位置还有待研究。 2.3 视网膜神经节细胞轴突再生存在的障碍 2.3.1 中枢神经系统髓鞘抑制因子 在中枢神经系统髓鞘中高表达的抑制轴突生长的髓鞘蛋白包括Nogo-A,MAG和少突胶质细胞髓鞘糖蛋白(oligodendrocyte-myelin GP,Omgp)。在中枢神经系统和外周神经系统中,这些髓鞘蛋白都有表达。但是中枢神经系统和外周神经系统轴突再生能力对于损伤的应答反应不同。在外周神经系统中,巨噬细胞和许旺细胞在损伤后可快速清除和下调髓鞘蛋白;在中枢神经系统中,受到损伤后少突胶质细胞可继续表达髓鞘蛋白,髓鞘蛋白的表达并没有下调,而髓鞘蛋白为中枢神经系统中轴突生长的最主要抑制剂之一。虽然这3种髓鞘蛋白分子结构不同,但是它们都结合于Nogo受体(Nogo receptor,NgR)和PirB受体[46]。使用病毒转染的方法,将NgR的显性阴性型(dominant negative form of NgR,d-nNgR)与Nogo、MAG和少突胶质细胞髓鞘糖蛋白结合,在视神经受到损伤后并没有显示出对轴突再生有明显的修复效果。然而,d-nNgR却明显增强了炎症反应对再生的影响[47]。NgR具有3种同源异构体,若将所有的同源异构体敲除则视神经可恢复部分再生能力;若同时存在眼内炎症,则再生作用还会增强[48]。 2.3.2 胶质瘢痕 在神经损伤部位形成的胶质瘢痕,并释放一系列轴突生长抑制因子,为轴突生长的另一个障碍[49]。损伤中心周围小胶质细胞出现活化和增殖,并募集巨噬细胞,与增殖的星形胶质细胞共同形成了胶质瘢痕。胶质瘢痕中抑制轴突生长的因子包括硫酸软骨素蛋白聚糖、信号蛋白3A、Slit-1和腱生蛋白-R等,其中最重要的是硫酸软骨素蛋白聚糖。瘢痕形成过程中各种细胞因子触发了硫酸软骨素蛋白聚糖的释放[50]。硫酸软骨素蛋白聚糖通过特定的NgR同源异构体[51],以及受体酪氨酸磷酸酶LAR和PTP-σ起到抑制轴突生长的作用[52],敲除PTP-σ基因可使视神经恢复部分再生能力[53]。 2.3.3 外源性抑制因子的共同信号通路 髓鞘蛋白的细胞内信号传导途径以及与胶质瘢痕相关抑制因子的共同通路为RhoA/ROCK途径(ras homolog gene A/rho-associated protein kinase,RhoA/ROCK)[54],其通过LIM激酶和丝切蛋白刺激导致肌动蛋白解聚,最终导致生长锥形成障碍[55]。细菌酶C3核糖基转移酶具有使RhoA失活的作用。向大鼠眼内注射细菌酶C3核糖基转移酶,或者通过基因操作使视网膜神经节细胞表达细菌酶C3核糖基转移酶,均可使视神经的轴突获得再生能力[56]。除此之外,RhoA失活还可明显增强炎症反应的效果,使再生的轴突几乎完全通过视神经损伤部位[56]。 2.3.4 轴突损伤引起视网膜神经节细胞的凋亡 凋亡的发生和进展取决于损伤部位与眼部的距离:损伤部位越靠近眼部,凋亡的发生和进展得越快。造成这一现象的原因可能为:若损伤部位距离眼部较远,神经营养因子可通过轴突逆行运输,暂时支持视网膜神经节细胞生存延长;损伤部位靠近眼部,神经营养因子的储库将快速耗尽[57]。另一种可能的解释为:在轴突损伤后,视网膜神经节细胞对细胞外神经毒性因子的摄取增加,例如钙蛋白酶通过逆行转运,触发细胞凋亡。凋亡被触发后又通过激活p53的下游因子进一步导致视网膜神经节细胞的死亡。这一过程伴随小胶质细胞的激活,进而也促进了视网膜神经节细胞的变性和死亡,形成恶性循环[34]。神经元中的内质网的自主应激反应也被认为存在于轴突切断诱导的视网膜神经节细胞死亡的过程中[58]。"

| [1] LAHA B, STAFFORD BK, HUBERMAN AD. Regenerating optic pathways from the eye to the brain. Science. 2017;356(6342):1031-1034. [2] Y CAJAL SR. Degeneration & regeneration of the nervous system: Hafner Pub. Co.1959. [3] BENOWITZ LI, HE Z, GOLDBERG JL. Reaching the brain: Advances in optic nerve regeneration. Exp Neurol. 2017;287(Pt 3):365-373. [4] AGUAYO AJ, RASMINSKY M, BRAY GM, et al. Degenerative and regenerative responses of injured neurons in the central nervous system of adult mammals. Philos Trans R Soc Lond B Biol Sci. 1991;331(1261):337-343. [5] WHITELEY SJ, SAUVE Y, AVILES-TRIGUEROS M, et al. Extent and duration of recovered pupillary light reflex following retinal ganglion cell axon regeneration through peripheral nerve grafts directed to the pretectum in adult rats. Exp Neurol. 1998;154(2):560-572. [6] BERRY M, CARLILE J, HUNTER A. Peripheral nerve explants grafted into the vitreous body of the eye promote the regeneration of retinal ganglion cell axons severed in the optic nerve. J Neurocytol. 1996;25(2):147-170. [7] HENDERSON CE. Role of neurotrophic factors in neuronal development. Curr Opin Neurobiol. 1996;6(1):64-70. [8] FISCHER D, LEIBINGER M. Promoting optic nerve regeneration. Prog Retin Eye Res. 2012;31(6):688-701. [9] BENOWITZ LI, YIN Y. Optic nerve regeneration. Arch Ophthalmol. 2010; 128(8):1059-1064. [10] MANSOUR-ROBAEY S, CLARKE DB, WANG YC, et al. Effects of ocular injury and administration of brain-derived neurotrophic factor on survival and regrowth of axotomized retinal ganglion cells. Proc Natl Acad Sci U S A. 1994;91(5):1632-1636. [11] GALINDO-ROMERO C, VALIENTE-SORIANO FJ, JIMENEZ-LOPEZ M, et al. Effect of brain-derived neurotrophic factor on mouse axotomized retinal ganglion cells and phagocytic microglia. Invest Ophthalmol Vis Sci. 2013;54(2):974-985. [12] MA YT, HSIEH T, FORBES ME, et al. BDNF injected into the superior colliculus reduces developmental retinal ganglion cell death. J Neurosci. 1998;18(6):2097-2107. [13] PERNET V, DI POLO A. Synergistic action of brain-derived neurotrophic factor and lens injury promotes retinal ganglion cell survival, but leads to optic nerve dystrophy in vivo. Brain. 2006;129(Pt 4):1014-1026. [14] MULLER A, HAUK TG, LEIBINGER M, et al. Exogenous CNTF stimulates axon regeneration of retinal ganglion cells partially via endogenous CNTF. Mol Cell Neurosci. 2009;41(2):233-246. [15] LAUGHTER MR, BARDILL JR, AMMAR DA,et al. Injectable Neurotrophic Factor Delivery System Supporting Retinal Ganglion Cell Survival and Regeneration Following Optic Nerve Crush. ACS Biomater Sci Eng. 2018;4(9): 3374-3383. [16] PEASE ME, ZACK DJ, BERLINICKE C, et al. Effect of CNTF on retinal ganglion cell survival in experimental glaucoma. Invest Ophthalmol Vis Sci. 2009; 50(5):2194-2200. [17] KURIMOTO T, YIN Y, OMURA K, et al. Long-distance axon regeneration in the mature optic nerve: contributions of oncomodulin, cAMP, and pten gene deletion. J Neurosci. 2010;30(46):15654-15663. [18] TONGES L, OSTENDORF T, LAMBALLE F,et al. Hepatocyte growth factor protects retinal ganglion cells by increasing neuronal survival and axonal regeneration in vitro and in vivo. J Neurochem. 2011;117(5):892-903. [19] DUAN X, QIAO M, BEI F, et al. Subtype-specific regeneration of retinal ganglion cells following axotomy: effects of osteopontin and mTOR signaling. Neuron. 2015;85(6):1244-1256. [20] LEIBINGER M, ANDREADAKI A, DIEKMANN H,et al. Neuronal STAT3 activation is essential for CNTF- and inflammatory stimulation-induced CNS axon regeneration. Cell Death Dis. 2013;4:e805. [21] LAU KC, SO KF, TAY D. Intravitreal transplantation of a segment of peripheral nerve enhances axonal regeneration of retinal ganglion cells following distal axotomy. Exp Neurol. 1994;128(2):211-215. [22] SINGH B, PLEMEL JR. Neutrophil contribution in facilitating optic nerve regeneration. J Neurosci. 2014;34(4):1081-1082. [23] YIN Y, CUI Q, GILBERT HY, et al. Oncomodulin links inflammation to optic nerve regeneration. Proc Natl Acad Sci U S A. 2009;106(46):19587-19592. [24] MEYER-FRANKE A, WILKINSON GA, KRUTTGEN A, et al. Depolarization and cAMP elevation rapidly recruit TrkB to the plasma membrane of CNS neurons. Neuron. 1998;21(4):681-693. [25] LEIBINGER M, MULLER A, ANDREADAKI A, et al. Neuroprotective and axon growth-promoting effects following inflammatory stimulation on mature retinal ganglion cells in mice depend on ciliary neurotrophic factor and leukemia inhibitory factor. J Neurosci. 2009;29(45):14334-14341. [26] LEIBINGER M, MULLER A, GOBRECHT P, et al. Interleukin-6 contributes to CNS axon regeneration upon inflammatory stimulation. Cell Death Dis. 2013;4:e609. [27] TAZAKI A, TANAKA EM, FEI JF. Salamander spinal cord regeneration: The ultimate positive control in vertebrate spinal cord regeneration. Dev Biol. 2017;432(1):63-71. [28] GOLDBERG JL, KLASSEN MP, HUA Y,et al. Amacrine-signaled loss of intrinsic axon growth ability by retinal ganglion cells. Science. 2002;296(5574): 1860-1864. [29] LORBER B, HOWE ML, BENOWITZ LI, et al. Mst3b, an Ste20-like kinase, regulates axon regeneration in mature CNS and PNS pathways. Nat Neurosci. 2009;12(11):1407-1414. [30] O’DONOVAN KJ, MA K, GUO H, et al. B-RAF kinase drives developmental axon growth and promotes axon regeneration in the injured mature CNS. J Exp Med. 2014;211(5):801-814. [31] GALVAO J, IWAO K, APARA A, et al. The Kruppel-Like Factor Gene Target Dusp14 Regulates Axon Growth and Regeneration. Invest Ophthalmol Vis Sci. 2018;59(7):2736-2747. [32] APARA A, GALVAO J, WANG Y,et al. KLF9 and JNK3 Interact to Suppress Axon Regeneration in the Adult CNS. J Neurosci. 2017;37(40):9632-9644. [33] YU B, GU X. Combination of biomaterial transplantation and genetic enhancement of intrinsic growth capacities to promote CNS axon regeneration after spinal cord injury. Front Med. 2018;13(2):131-137. [34] KOLE C, BROMMER B, NAKAYA N, et al. Activating Transcription Factor 3 (ATF3) Protects Retinal Ganglion Cells and Promotes Functional Preservation After Optic Nerve Crush. Invest Ophthalmol Vis Sci. 2020;61(2):31. [35] MAK HK, NG SH, REN T, et al. Impact of PTEN/SOCS3 deletion on amelioration of dendritic shrinkage of retinal ganglion cells after optic nerve injury. Exp Eye Res. 2020;192:107938. [36] BELIN S, NAWABI H, WANG C,et al. Injury-induced decline of intrinsic regenerative ability revealed by quantitative proteomics. Neuron. 2015; 86(4):1000-1014. [37] LUO X, SALGUEIRO Y, BECKERMAN SR, et al. Three-dimensional evaluation of retinal ganglion cell axon regeneration and pathfinding in whole mouse tissue after injury. Exp Neurol. 2013;247:653-662. [38] DE LIMA S, KORIYAMA Y, KURIMOTO T, et al. Full-length axon regeneration in the adult mouse optic nerve and partial recovery of simple visual behaviors. Proc Natl Acad Sci U S A. 2012;109(23):9149-9154. [39] WANG SK, CHANG RT. An emerging treatment option for glaucoma: Rho kinase inhibitors. Clin Ophthalmol. 2014;8:883-890. [40] TRAKHTENBERG EF, LI Y, FENG Q, et al. Zinc chelation and Klf9 knockdown cooperatively promote axon regeneration after optic nerve injury. Exp Neurol. 2018;300:22-29. [41] ZHOU H, SU J, HU X, et al. Glia-to-Neuron Conversion by CRISPR-CasRx Alleviates Symptoms of Neurological Disease in Mice. Cell. 2020;181(3): 590-603.e16. [42] MOORE DL, BLACKMORE MG, HU Y, et al. KLF family members regulate intrinsic axon regeneration ability. Science. 2009;326(5950):298-301. [43] VELDMAN MB, BEMBEN MA, THOMPSON RC, et al. Gene expression analysis of zebrafish retinal ganglion cells during optic nerve regeneration identifies KLF6a and KLF7a as important regulators of axon regeneration. Dev Biol. 2007;312(2):596-612. [44] GOULART CO, MENDONCA HR, OLIVEIRA JT, et al. Repulsive Environment Attenuation during Adult Mouse Optic Nerve Regeneration. Neural Plast. 2018;2018:5851914. [45] ELSAEIDI F, MACPHERSON P, MILLS EA, et al. Notch Suppression Collaborates with Ascl1 and Lin28 to Unleash a Regenerative Response in Fish Retina, But Not in Mice. J Neurosci. 2018;38(9):2246-2261. [46] OSTERHOUT JA, JOSTEN N, YAMADA J, et al. Cadherin-6 mediates axon-target matching in a non-image-forming visual circuit. Neuron. 2011;71(4): 632-639. [47] PERNET V, JOLY S, DALKARA D, et al. Long-distance axonal regeneration induced by CNTF gene transfer is impaired by axonal misguidance in the injured adult optic nerve. Neurobiol Dis. 2013;51:202-213. [48] LI S, HE Q, WANG H, et al. Injured adult retinal axons with Pten and Socs3 co-deletion reform active synapses with suprachiasmatic neurons. Neurobiol Dis. 2015;73:366-376. [49] ATWAL JK, PINKSTON-GOSSE J, SYKEN J, et al. PirB is a functional receptor for myelin inhibitors of axonal regeneration. Science. 2008;322(5903): 967-970. [50] FISCHER D, HE Z, BENOWITZ LI. Counteracting the Nogo receptor enhances optic nerve regeneration if retinal ganglion cells are in an active growth state. J Neurosci. 2004;24(7):1646-1651. [51] DICKENDESHER TL, BALDWIN KT, MIRONOVA YA, et al. NgR1 and NgR3 are receptors for chondroitin sulfate proteoglycans. Nat Neurosci. 2012; 15(5):703-712. [52] WANG X, HASAN O, ARZENO A, et al. Axonal regeneration induced by blockade of glial inhibitors coupled with activation of intrinsic neuronal growth pathways. Exp Neurol. 2012;237(1):55-69. [53] ORR MB, GENSEL JC. Spinal Cord Injury Scarring and Inflammation: Therapies Targeting Glial and Inflammatory Responses. Neurotherapeutics. 2018;15(3):541-553. [54] FITCH MT, DOLLER C, COMBS CK,et al. Cellular and molecular mechanisms of glial scarring and progressive cavitation: in vivo and in vitro analysis of inflammation-induced secondary injury after CNS trauma. J Neurosci. 1999;19(19):8182-8198. [55] TRAN AP, WARREN PM, SILVER J. The Biology of Regeneration Failure and Success After Spinal Cord Injury. Physiol Rev. 2018;98(2):881-917. [56] FISHER D, XING B, DILL J, et al. Leukocyte common antigen-related phosphatase is a functional receptor for chondroitin sulfate proteoglycan axon growth inhibitors. J Neurosci. 2011;31(40):14051-14066. [57] OHTAKE Y, WONG D, ABDUL-MUNEER PM,et al. Two PTP receptors mediate CSPG inhibition by convergent and divergent signaling pathways in neurons. Sci Rep. 2016;6:37152. [58] SCHWEIGREITER R, WALMSLEY AR, NIEDEROST B, et al. Versican V2 and the central inhibitory domain of Nogo-A inhibit neurite growth via p75NTR/NgR-independent pathways that converge at RhoA. Mol Cell Neurosci. 2004; 27(2):163-174. [59] LINGOR P, TONGES L, PIEPER N, et al. ROCK inhibition and CNTF interact on intrinsic signalling pathways and differentially regulate survival and regeneration in retinal ganglion cells. Brain. 2008;131(Pt 1):250-263. [60] FISCHER D, PETKOVA V, THANOS S, et al. Switching mature retinal ganglion cells to a robust growth state in vivo: gene expression and synergy with RhoA inactivation. J Neurosci. 2004;24(40):8726-48740. [61] CHITRANSHI N, DHEER Y, ABBASI M, et al. Glaucoma Pathogenesis and Neurotrophins: Focus on the Molecular and Genetic Basis for Therapeutic Prospects. Curr Neuropharmacol. 2018;16(7):1018-1035. [62] HU Y, PARK KK, YANG L, et al. Differential effects of unfolded protein response pathways on axon injury-induced death of retinal ganglion cells. Neuron. 2012;73(3):445-452. [63] HOWELL GR, SOTO I, LIBBY RT, et al. Intrinsic axonal degeneration pathways are critical for glaucomatous damage. Exp Neurol. 2013;246:54-61. |
| [1] | Xie Jingshu, Zhang Xianglin, Liu Jinlei, Wen Jing. Application of High Resolution reconstruction algorithm in precision CT scans of the middle and inner ears [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3614-3618. |
| [2] | Liang Meifu, Qu Shuhua. Optimal power load forecasting of the skeletal muscle based on back propagation neural network [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3641-3647. |
| [3] | Huang Maomao, Hu Yue, Wang Binchuan, Zhang Chi, Xie Yujie, Wang Jianxiong, Wang Li, Xu Fangyuan. Bibliometric and visual analysis of international literature addressing ischemic stroke rehabilitation in recent 10 years [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3725-3733. |
| [4] | Zhu Rui, Zeng Qing, Huang Guozhi. Ferroptosis and stroke [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3734-3739. |
| [5] | Liu Zhiwei, Xie Rui, Sun Kai, Li Kaiming, Wang Xiongwei, Zhan Jiawen, Zhu Liguo. Interpretation of diagnostic criteria for cervicogenic headache: challenges and understandings in diagnosis and differential diagnosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3746-3751. |
| [6] | Lin Haishan, Mieralimu Muertizha, Li Peng, Ma Chao, Wang Li. Correlation between skeletal muscle fiber characteristics and bone mineral density in postmenopausal women with hip fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3144-3149. |
| [7] | Zuo Xiuqin, Yin Sasa, Xie Huimin, Jia Zishan, Zhang Lining. Applicability and specifications of platelet-rich plasma in musculoskeletal repair [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3239-3245. |
| [8] | Bai Xiaotian, Huo Hongfeng. Biomechanical evaluation of foot and ankle function: constructing static and dynamic indexes of the foot [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2747-2754. |
| [9] | He Shaobo, Liu Jichao, Yang Lifeng, Ding Yongfeng, Li Wujian. Finite element analysis of transposition of posterior tibial tendon for foot drop [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(15): 2334-2340. |
| [10] | Xu Xiaopei, Lü Xin. Classification of intertrochanteric fractures with medial wall destruction in elderly patients and recovery of bony support and continuity of the cortical bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(14): 2259-2265. |
| [11] | Liu Lu, Zhang Nini, Dai Min, Huang Guilin. Pathological changes and functional reconstruction of radiation-induced salivary glands repaired by stem cells and biomaterials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2103-2107. |
| [12] | Zhang Yicen, Wang Peixin, Liu Zhicheng. Ultrasound-guided injection of hyaluronic acid and corticosteroid for treating plantar fasciitis: evaluation of pain, fascia thickness and ankle-foot function [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1670-1674. |
| [13] | Tian Yanping, Li Juan, Liu Xiaobo, Zhang Huiling, Shi Lihong, Jin Rongjiang. Knowledge network mapping of literature regarding platelet-rich plasma in recent 5 years: a visual analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1745-1752. |
| [14] | Xu Baoyun, Fu Weili. Posterior cruciate ligament injury: diagnosis, treatments and rehabilitation strategies [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1766-1772. |
| [15] | Long Qian, Guan Xiaoyan, Wang Qian, Hu Huan, Liu Jianguo. Transcriptome sequencing technology and its application in oral diseases, dental implants and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1791-1798. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||