Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (5): 773-779.doi: 10.3969/j.issn.2095-4344.1915
Previous Articles Next Articles
Effects of physical activity on hippocampal plasticity and cognition
Chen Yanlin1, Xu Lin2, Xu Shengjia3
- 1Department of Sports, Hohai University, Nanjing 210098, Jiangsu Province, China; 2Department of Exercise and Heath, Nanjing Sport Institute, Nanjing 210014, Jiangsu Province, China; 3the Research Center of Military Exercise Science, the Army Engineering University of PLA, Nanjing 211101, Jiangsu Province, China
-
Received:2019-04-19Revised:2019-04-29Accepted:2019-06-22Online:2020-02-18Published:2020-01-10 -
Contact:Xu Shengjia, Master, Lecturer, the Research Center of Military Exercise Science, the Army Engineering University of PLA, Nanjing 211101, Jiangsu Province, China -
About author:Chen Yanlin, Lecturer, Department of Sports, Hohai University, Nanjing 210098, Jiangsu Province, China -
Supported by:the Advanced Research Foundation of PLA University of Science and Technology, No. KYJYZLXY1902-36; the Science and Technology Innovation Project of Academic Degree Graduates in Jiangsu Province, No. SJKY19-1604
CLC Number:
Cite this article
Chen Yanlin, Xu Lin, Xu Shengjia. Effects of physical activity on hippocampal plasticity and cognition[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(5): 773-779.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
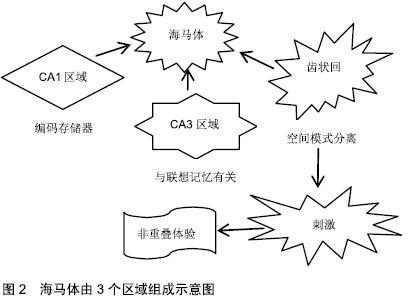
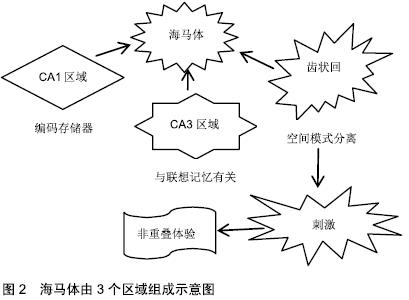
2.1 人类成人海马体的神经再生 初期成人脑中神经再生的研究表明,可在增殖细胞DNA合成期间使用胸苷类似物溴脱氧尿苷(BrdU),以证明海马体齿状回(dentate gyrus,DG)中细胞的增殖、分化和存活。在该研究中,齿状回的颗粒层和相邻的颗粒下区域(subgranular zone,SGZ)呈现标记细胞,表明存在新生神经元[4]。但这种现象仅局限于齿状回,而在人类海马体其他区域种并未发现类似的活性。 一些关注成人神经细胞计数的研究应用免疫组织化学方法鉴定增殖和未成熟/幼小神经元的标志物,如Ki67、双皮质素(DCX)或聚唾液酸化神经细胞黏附(PSA-NCAM),发现成人颗粒下区增殖活性与常规薄壁组织相似,且新生儿出生后第一年神经细胞数量很少,以此推测成年后齿状回中几乎没有神经再生的可能[5]。该结果否定了人类中颗粒下区再生的存在,且与应用相同内源标记的成人神经再生的阳性结果相矛盾[6-7]。在成人颗粒下区和齿状回颗粒层中已发现数千个神经祖细胞、双DCX和PSA-NCAM阳性的未成熟神经元。此外,上述研究的不一致性也可能来自方法本身。新神经元的溴脱氧尿苷染色可能不如DCX敏感,可能产生假阳性结果[7],若未能快速正确的固定保存,分子很可能发生快速变化,导致内源标记难以在成人脑组织中进行准确鉴别[4]。 2.1.1 神经源性活性的程度 当前,已经可以直接从成人海马体中分离出神经前体,继而在体外培养为成熟神经元[8]。但这种选择性提取缺乏定量分析,且细胞生成程度存疑[4,7]。一项研究则通过检测碳-14量化基因组DNA中的同位素浓度,发现即使少量的神经元前体也足以产生大量新生神经元[9],存在35%的海马体神经元在齿状回中不断地自我更新,更新率为1.75%/年[9]。这些比率证实,尽管可能是很小的成神经细胞群,但在整个生命周期中,可交换神经元的数量依旧很高(每天700个新神经元)[9]。相比之下,啮齿类动物的神经形成在哺乳动物中较为突出[10],在成年期仅表现出10%的齿状回神经元可再生率。 2.1.2 整个年龄段的神经再生变化 当前,需要分析人体神经元周转的动态变化,以及在整个年龄阶段中的变化,以全面了解海马体神经的发生发展[11]。普遍认为,与胎儿发育过程中观察到的神经细胞标记物数量相比,在产后早期神经细胞群数量减少[5]。 在此背景下,一些研究试图检测婴儿期后的神经母细胞,却未得到DCX阳性的结果,表明神经再生在成人海马体中停止[5]。但其他研究认为,成年期神经亦可再生,且DCX阳性细胞的数量和神经元周转率(来自海马体神经元的自我更新部分)表现为随基础值平行下降的趋势,可能与齿状回的神经原能力相关[9]。此外,尽管在齿状回中观察到静息神经祖细胞的总体水平下降,但在整个衰老期间存在稳定数量的双DCX阳性和PSA-NCAM阳性成神经细胞[7],表明早期祖细胞不会损害整个细胞谱系的增殖潜力[7],然而,该研究未能完善14岁以下人类海马样品情况的分析[7],因此无法对总年龄谱进行整体分析。因此,在整个年龄段齿状回中都存在神经再生的现象,表现为随年龄增长逐渐下降。 2.1.3 细胞数和海马体体积动力学 神经细胞数和神经元交换率可能与神经元计数和海马体体积的变化有关,新细胞的产生最终可能影响人类生命中各时间点的神经元数量和特定中枢神经系统区域的维度。但也有证据表明,随年龄增长,海马体中神经元会有所减少[12-13],目前已确定海马体各区受到的影响有所差异,而齿状回内主要为成神经细胞,该区域受神经元数量减少的影响最小[11]。 有研究认为,齿状回中成熟颗粒神经元的数量可能没有任何变化[7],在人类整个衰老过程中,神经再生是为了抵抗基础神经元的耗竭[7]。而其他研究发现,尽管具有神经原性潜力,但成年人类齿状回中呈现出颗粒细胞的净损失[9],且海马体(包括齿状回)所含神经元细胞的总体积减少,认为神经再生无法阻止细胞和体积的耗竭[13]。除上述假设外,由于神经再生现象,有研究认为海马体中神经元自我更新比例增加,不更新的细胞在无替换的情况下死亡[9]。此外,由于成年人新生细胞在成年期也会丢失,使得海马拥有更为特殊的细胞动力学性质。在新生神经元群体中,相比发育期间的细胞,成人新生脑细胞存活率更低[9],从而加深了海马体神经元调节的复杂性。 因此,海马体可被视为一种动态结构:最初在发育期间形成,之后表现为永久性地损失细胞,成年期间则由齿状回的颗粒状成神经细胞替代[11]。 2.2 身体活动可促进海马体齿状回的神经再生 海马体在新记忆获得方面非常重要[14],易受衰老和年龄相关的认知障碍的影响,如人类海马体积的退行性萎缩,而这一过程可通过运动得到改善[15-16]。研究表明,较高的体适能水平与较大的海马体体积相关,并且测试结果显示其记忆能力更好[17]。干预研究发现,每周3 d,维持3个月至1年的有氧训练可预防和/或逆转年龄相关海马体积下降的现象[15]。此外,研究认为运动引起的灰质体积增加发生在海马体、内嗅和前额叶皮质[15-16,18],而丘脑或尾状核部位没有变化[15]。 海马体由3个区域组成:CA1区域、CA3区域和齿状回,均与记忆功能相关。普遍认为CA1区域是编码存储器[19],CA3区域与联想记忆有关[20],齿状回则是对空间模式分离十分重要。齿状回非常独特,它可以在成人大脑中产生新的神经元,被认为在空间记忆和模式分离中起作用[21-22]。空间模式分离是将类似的传入信息或刺激转换为不同的非重叠体验的过程[21](见图2)。 "

位于齿状回内颗粒细胞层中的残余干细胞,在哺乳动物(包括人)中可能会继续增殖和分化[9]。干/祖细胞由不活跃的1型放射状胶质细胞组成:表达神经胶质纤维酸性蛋白、巢蛋白和Sox2;而2型细胞表达Sox-2,能够产生星形胶质细胞,以及快速增殖的神经元祖细胞,可在数周内分化为成熟的颗粒细胞[23]。其中一些内在因素如病理状况或运动等,可上调或下调新神经元的增殖、存活和整合。 运动可促进海马体齿状回的神经再生[22,24]。人体成像研究显示运动对海马体积的正面影响,被认为是神经发生的间接测量[22,24],对啮齿类动物的研究表明,依据遗传背景[25]、年龄[26]、跑轮类型[27]、使用的标记方法以及跑步距离的不同[28-29],运动诱导的新生神经元可增加两三倍。此类神经主要发生在齿状回背侧而不是腹侧[28],可能原因是,海马体的背侧对空间导航很重要[30],而海马体腹侧似乎对情绪变化更敏感[31]。新神经元又在齿状回介导的模式分离中发挥一定作 用[32],即区分相似信息的能力[33]。有学者研究表明,跑步可改善小鼠精细辨别能力[27],通过条件消除神经祖细胞中BAX依赖性程序性细胞凋亡来增强成体神经发生[33]。但最近研究未能重复上述结果,因此对这一假设的成立产生怀疑[34]。 运动可诱导新生神经元的出现,除促进神经元数量的增加,部分提高认知功能的效益外,对神经元的成熟、形态和连接性等特性的改变也十分重要。运动可缩短祖细胞的细胞周期[35],加速成体齿状回神经元成熟[36]。反转录病毒标记研究表明,跑步可促进成人的齿状颗粒细胞树突的外分子层中的脊形成[36]。此外,在反转录病毒注射后[30],在3周跑步者的再生齿状回神经元中,树突棘运动性、总树突长度、分支点、树突复杂性和线粒体密度均显著增加[36-37]。因此,可以认为运动诱导的神经元加速成熟沿时间轴变化,其中以背侧表现最快[38]。 同时跑步还会改变新神经元的整合通路,增加传入新生神经元和传入细胞突触的数量[28]。运动从内嗅皮质、乳头状核和内侧隔膜所募集的突触前输入相关信息[28],这些脑区对信息传递的内容和背景[39]、时空信息处理以及启动海马体θ节律来说非常重要[40]。运动降低了海马体内抑制性中间神经元、谷氨酸能苔藓细胞的神经支配与新生神经元的比例[28]。但新生神经元的抑制性突触传递并未受到影响,兴奋性突触传递仅小幅减少但频率不变。通过每个神经元的相对较少输入,实现相同的兴奋性和抑制性驱动,可促进新神经元完成更有效的整合[28]。此外,传入细胞与新生神经元的总体比例降低,可通过促进稀疏激活,使汇聚至单个神经元的输入减少,进而利于模式分离[28]。 2.3 海马体齿状回的神经再生与运动诱导的相关因素 2.3.1 调节神经递质 新生神经元的发育和整合需要多个神经递质的参与,其中谷氨酸和γ-氨基丁酸(GABA)是脑中最主要的兴奋性和抑制性神经递质,两者均参与调节新生神经元的整合和存活[41],而谷氨酸对运动诱导的齿状回突触可塑性的变化十分重要。跑步可通过降低长时增益效应的诱导阈值来提高齿状回的长期增益效应,这种突触可塑性被认为是体内和体外学习和记忆的细胞模型[42]。研究表明,跑步可提高谷氨酸受体亚单位NR2A、NR2B和谷氨酸受体5的齿状回基因表达[43],同时NR2B N-甲基-D-天冬氨酸(NMDA)受体亚基在新神经元中也高度表达[44],提示NR2B过表达可促进长期增益效应诱导[45]。一些研究表明,运动干预小鼠的脑切片中,个体新生神经元增加,且长期增益效应增强[46]。其他运动实验发现,弱θ图案刺激在对照组的齿状回中未发生长期增益效应,而在运动干预的大鼠中发现了持续性的长期增益效应[43]。此外,急性的自主运动和感觉刺激,可诱发海马体的θ活动模式,尤其是在CA1和齿状回区域[47]。因此,运动可通过抑制振荡,进而增强可塑性,最终完成网络学习[48]。 另外,跑步可调节抑制性神经递质。研究表明,一段时间耐力运动后,包括齿状回在内的海马体子区域中γ-氨基丁酸受体亚单位和GAD67的表达增加[49]。此外,运动增加了腹侧齿状回细胞外γ-氨基丁酸的释放和囊泡γ-氨基丁酸转运蛋白表达。对局部抑制的增强,可降低齿状回区即刻早期基因的表达[50],同时,抑制性神经递质的增加可减轻焦虑并改善认知功能。普遍认为,DG-CA3区域中抑制张力的降低与衰老相关的记忆缺陷有关[51]。又有研究证明,跑步可调节其他神经递质和神经调节剂,如单胺(多巴胺,5-羟色胺,去甲肾上腺素)和内源性大麻素等[52-53],均可促进海马体突触可塑性的变化,其中5-羟色胺系统在运动诱导的成人神经发生增加中占主导地位[54]。研究表明,5-羟色胺3受体亚基的消融消除了运动诱导的神经发生并弱化其抗抑郁作用[55]。 2.3.2 增加脑源性神经营养因子 神经营养因子在大脑可塑性调节中发挥重要作用,与运动相关且较为重要的生长因子是脑源性神经营养因子。在动物模型中,跑步可增加海马体中脑源性神经营养因子的表达水平[41],且与海马体可塑性、空间记忆和物体识别的改善相关[56]。短期和长期运动后,脑源性神经营养因子被上调,此外,运动诱导的乙酰胆碱,θ波和肾上腺素的增加也引发脑源性神经营养因子产生[57]。 在人类研究中发现,运动诱导的血清脑源性神经营养因子水平升高与海马体体积的变化有关[15]。而在衰老诱发的海马体体积下降现象中,可观察到血清脑源性神经营养因子水平降低[58]。通过其下游靶标,cAMP反应元件结合(CREB)蛋白、突触蛋白Ⅰ和突触素,同时增加其自身信使RNA(mRNA)及其受体酪氨酸激酶B(TrkB)含量,脑源性神经营养因子可增强突触可塑性[59]。研究发现,阻断大鼠海马体中脑源性神经营养因子可消除运动对认知功能和海马体可塑性的改善作用[60]。此外,神经祖细胞中TrkB受体的消融消除了对跑步的神经原性反应[61]。对海马体子区域的分析表明,运动增加脑源性神经营养因子的mRNA水平发生在齿状回区,而不是CA1区域[35]。 运动诱导的神经营养素水平变化可能是由几种神经细胞共同介导发生的[62]。星形胶质细胞与新生神经元树突的形成密切相关,可促进其发育[63]。星形胶质细胞能够合成脑源性神经营养因子,且含有TrkB受体[64],而跑步可增加海马体内星形胶质细胞数量及其体积,延长其发生发展过程[65],并提高TrkB表达水平[66],其他非神经元细胞也可能发挥类似作用。巨噬细胞移动抑制因子是一种通过运动上调的细胞因子,可诱导脑源性神经营养因子表达[67];小胶质细胞数量则会因跑步而下调[68],但海马体中小胶质细胞的消融损害了运动诱导的神经发生,这表明小胶质细胞产生的细胞因子可能参与神经发生的维持过程[69]。 表观遗传因子在神经营养因子的功能中同样发挥重要作用。脑源性神经营养因子基因(Val/Met多态性)的单核苷酸多态性在白种人中发生率为20%-30%[70],可降低活性依赖性脑源性神经营养因子分泌,并且与抑郁和焦虑相关疾病的易感性增加、记忆功能降低、以及抗抑郁药导致的神经可塑性受损有关[70]。此外,与Val/Val受试者相比,在携带Met位点人群中,运动对其海马体体积的影响减弱[71]。研究表明,在具有这种多态性的成年小鼠中,运动导致脑源性神经营养因子增加和神经发生减弱[72]。而运动诱导的脑源性神经营养因子增加,也可能通过表观遗传变化来介导[73]。研究表明,运动能够增加组蛋白H3乙酰化,有助于基因转录,特异性地上调脑源性神经营养因子基因表达[73]。另一方面,运动可下调海马体组蛋白去乙酰化酶(HDAC),从而上调脑源性神经营养因子表达[74]。这些结果表明,运动可能部分地通过脑源性神经营养因子,改善认知功能。 2.3.3 改善脑血管可塑性 运动可调节脑血管功能,提高灌注能力,输送氧气、营养素、神经营养因子和其他可能促进大脑功能的因素。一次性行走和跑步活动会增加动物脑内几个区域的脑血流量,包括海马体[75]、皮质和纹状体[76-77],但不影响嗅球或下丘脑区域[75-76],这表明运动对脑血流量具有区域特异性的控制作用。在大鼠开始移动后,其海马体内脑血流量水平快速增加,又很快恢复至基线水平,而较长时间的运动可导致运动后持续性高水平脑血流量(10-20 min)[78]。因此,运动的持续时间和强度可差异性地调节脑血流量表达水平。脑血流量的增加可影响神经元活动,而注射河豚毒素(TTX)、NMDA受体拮抗剂(MK-801)和一氧化氮合成酶抑制剂(L-NAME)又可抑制这一增加[75]。在人类研究中同样发现,运动会增加脑血流量,并削弱衰老相关脑血流量和脑血管反应性的下降[79]。 此外,在长期运动后,发现年轻人和老年人的海马体灌注发生变化[27,80];啮齿动物的脑血管可塑性得到改善,可能与运动诱导的脑区域(包括海马体、纹状体、小脑和皮质)血管生成的增加有关[81]。血管内皮生长因子可能是运动诱导海马体神经发生的必需品,它能够介导运动改善血管可塑性、成人神经再生,以及外周组织与脑之间的联系。血管内皮生长因子参与血管形成,并且可以延缓或减轻年龄增长相关的神经再生衰退[82]。在啮齿动物中,血管内皮生长因子的过表达,可促进海马体的血管增殖,伴随海马体神经发生的增加[83]。因此,身体活动、血管生成和神经发生之间存在一定的联系[84]。 2.3.4 重新连接神经元网络 就功能而言,身体活动可重新连接神经元网络从而影响海马体记忆,齿状回的新生神经元的增加可带来更多的益处,可能有助于维持棘突和突触的密度,并利于预防海马体衰老[85]。尽管成熟颗粒细胞仅占齿状回细胞总数的一小部分,但与发育中的颗粒细胞相比,它们具有增强的兴奋性,长期增益效应阈值降低和长期增益效应增强[46]。 此外,身体活动可改变成人新生神经元的突触可塑性和连接网络,增加内嗅皮质的神经支配、空间记忆与生成θ节律的重要区域,例如尾侧内嗅皮质、内侧隔、上乳头内侧和内侧乳头核[28]。此外,动物模型和人类初步结果的证据表明,身体活动诱导海马回路中θ节律的同步神经元放电[86];身体活动还可修饰侧向内嗅皮层中突触传递的重要基因表达[87],内嗅皮质是一种皮质区域,神经选择性控制成人新生颗粒细胞[88],而自发性跑步运动可增加侧内嗅皮质到成人新生神经元短期突触的可塑性。因此,身体活动能够协调神经系统的活动,有利于记忆的形成,可增强成人新生神经元与现有海马-内嗅通路的整合[89]。 "

| [1] VOSS MW, SOTO C, YOO S, et al.Exercise and Hippocampal Memory Systems.Trends Cogn Sci. 2019;23(4):318-333. [2] BAPTISTA P, ANDRADE JP. Adult Hippocampal Neurogenesis: Regulation and Possible Functional and Clinical Correlates.Front Neuroanat. 2018;12:44. [3] COOPER C, MOON HY, VAN PRAAG H. On the Run for Hippocampal Plasticity.Cold Spring.Harb Perspect Med.2018;8(4). [4] ERIKSSON PS, PERFILIEVA E, BJÖRK-ERIKSSON T, et al. Neurogenesis in the adult human hippocampus. Nat Med.1998;4(11): 1313-1317. [5] AUGUSTE KI, CHANG EF, GUTIERREZ AJ, et al. Human hippocampal neurogenesis drops sharply in children to undetectable levels in adults. Nature.2018;555(7696):377-381. [6] KNOTH R, SINGEC I, DITTER M, et al. Murine features of neurogenesis in the human hippocampus across the lifespan from 0 to 100 years.PLoS One.2010;5(1):e8809. [7] BOLDRINI M, FULMORE CA, TARTT AN, et al. Human hippocampal neurogenesis persists throughout aging. Cell Stem Cell.2018;22(4): 589-599. [8] ROY NS, WANG S, JIANG L, et al. In vitro neurogenesis by progenitor cells isolated from the adult human hippocampus.Nat Med.2000;6(3): 271-277. [9] SPALDING KL, BERGMANN O, ALKASS K, et al. Dynamics of hippocampal neurogenesis in adult humans. Cell.2013;153(6): 1219-1227. [10] AMREIN I, ISLER K, Lipp HP.Comparing adult hippocampal neurogenesis in mammalian species and orders: influence of chronological age and life history stage.Eur J Neurosci.2011;34(6): 978-987. [11] BERGMANN O, SPALDING KL, FRISÉN J. Adult Neurogenesis in Humans. Cold Spring Harb Perspect Biol. 2015;7(7):a018994. [12] WEST MJ, COLEMAN PD, FLOOD DG, et al.Differences in the pattern of hippocampal neuronal loss in normal ageing and Alzheimer’s disease. Lancet.1994;344(8925):769-772. [13] SIMIĆ G, KOSTOVIĆ I, WINBLAD B, et al.Volume and number of neurons of the human hippocampal formation in normal aging and Alzheimer’s disease.J Comp Neurol.1997;379(4):482-494. [14] RIEDEL G, MICHEAU J, LAM AG, et al. Reversible neural inactivation reveals hippocampal participation in several memory processes.Nat Neurosci.1999;2(10):898-905. [15] ERICKSON KI, VOSS MW, PRAKASH RS, et al. Exercise training increases size of hippocampus and improves memory. Proc Natl Acad Sci U S A.2011;108(7):3017-3022. [16] ERICKSON KI, LECKIE RL, WEINSTEIN AM.Physical activity, fitness, and gray matter volume. Neurobiol Aging. 2014;35 Suppl 2:S20-28. [17] VOSS MW, VIVAR C, KRAMER AF, et al.Bridging animal and human models of exercise-induced brain plasticity. Bridging animal and human models of exercise-induced brain plasticity.Trends Cogn Sci. 2013; 17(10):525-544. [18] WHITEMAN AS, YOUNG DE, BUDSON AE, et al. Entorhinal volume, aerobic fitness, and recognition memory in healthy young adults: A voxel-based morphometry study.Neuroimage.2016;126:229-238. [19] NAKAZAWA K, MCHUGH TJ, WILSON MA, et al. NMDA receptors, place cells and hippocampal spatial memory. Nat Rev Neurosci.2004; 5(5): 361-372. [20] NAKAZAWA K, SUN LD, QUIRK MC, et al. Hippocampal CA3 NMDA receptors are crucial for memory acquisition of one-time experience. Neuron.2003;38(2):305-315. [21] KESNER RP, ROLLS ET.A computational theory of hippocampal function, and tests of the theory: New developments.Neurosci Biobehav Rev. 2015;48:92-147. [22] BOLIJN S, LUCASSEN PJ. How the Body Talks to the Brain; Peripheral Mediators of Physical Activity-Induced Proliferation in the Adult Hippocampus.Brain Plasticity.2015;1(1):5-27. [23] BONAGUIDI MA, WHEELER MA, SHAPIRO JS, et al.In vivo clonal analysis reveals self-renewing and multipotent adult neural stem cell characteristics.Cell.2011;145(7):1142-1155. [24] PATTEN AR, YAU SY, FONTAINE CJ, et al.The benefits of exercise on structural and functional plasticity in the rodent hippocampus of different disease models.Brain Plast.2015;1(1):97-127. [25] GRÉGOIRE CA, BONENFANT D, LE NGUYEN A, et al.Untangling the influences ofvoluntary running, environmental complexity, social housing and stress on adult hippocampal neurogenesis.PLoS One. 2014;9(1): e86237. [26] VAN PRAAG H, SHUBERT T, ZHAO C, et al.Exercise enhances learning and hippocampal neurogenesis in aged mice.J Neurosci. 2005;25(38): 8680-8685. [27] CREER DJ, ROMBERG C, SAKSIDA LM, et al.Running enhances spatial pattern separation in mice. Proc Natl Acad Sci U S A.2010; 107(5): 2367-2372. [28] VIVAR C, PETERSON BD, VAN PRAAG H, et al. Running rewires the neuronal network of adult-born dentate granule cells.Neuroimage. 2016;131:29-41. [29] CLARK PJ, KOHMAN RA, MILLER DS, et al.Genetic influences on exercise-induced adult hippocampal neurogenesis across 12 divergent mouse strains.Genes Brain Behav.2011;10(3):345-353. [30] MOSER MB, MOSER EI, FORREST E, et al.Spatial learning with a minislab in the dorsal hippocampus. Proc Natl Acad Sci U S A.1995; 92(21): 9697-9701. [31] KJELSTRUP KG, TUVNES FA, STEFFENACH HA, et al. Reduced fear expression after lesions of the ventral hippocampus.Proc Natl Acad Sci U S A.2002;99(16):10825-10830. [32] SAHAY A, SCOBIE KN, HILL AS, et al. Increasing adult hippocampal neurogenesis is sufficient to improve pattern separation. Nature.2011; 472(7344):466-470. [33] AIMONE JB, DENG W, GAGE FH.Resolving new memories: A critical look at the dentate gyrus, adult neurogenesis, and pattern separation. Neuron.2011;70(4):589-596. [34] KODALI M, MEGAHED T, MISHRA V, et al.Voluntary running exercise-mediated enhanced neurogenesis does not obliterate retrograde spatial memory.J Neurosci.2016;36(31):8112-8122. [35] FARMER J, ZHAO X, VAN PRAAG H, et al. Effects of voluntary exercise on synaptic plasticity and gene expression in the dentate gyrus of adult male Sprague-Dawley rats in vivo.Neuroscience. 2004;124(1):71-79. [36] ZHAO C, JOU J, WOLFF LJ, et al.Spine morphogenesis in newborn granule cells is differentially regulated in the outer and middle molecular layers.J Comp Neurol.2014;522(12):2756-2766. [37] STEIB K, SCHÄFFNER I, JAGASIA R, et al.Mitochondria modify exercise-induced development of stem cell-derived neurons in the adultbrain.J Neurosci.2014;34(19):6624-6633. [38] PIATTI VC, DAVIES-SALA MG, ESPÓSITO MS, et al.The timing for neuronal maturation in the adult hippocampus is modulated by local network activity.J Neurosci.2011;31(21):7715-7728. [39] KNIERIM JJ. From the GPS to HM:Place cells, grid cells, and memory. Hippocampus.2015;25(6):719-725. [40] DILLINGHAM CM, FRIZZATI A, NELSON AJ, et al. How do mammillary body inputs contribute to anterior thalamic function? Neurosci Biobehav Rev.2015;54:108-119. [41] GE S, GOH EL,SAILOR KA, et al.GABA regulates synaptic integration of newly generated neurons in the adult brain.Nature.2006;439(7076): 589-593. [42] BLISS TV, COLLINGRIDGE GL. A synaptic model of memory: Long-term potentiation in the hippocampus. Nature.1993;361(6407):31-39. [43] FARMER J, ZHAO X, VAN PRAAG H, et al. Effects of voluntary exercise on synaptic plasticity and gene expression in the dentate gyrus of adult male Sprague–Dawley rats in vivo.Neuroscience.2004;124(1):71-79. [44] KHEIRBEK MA, TANNENHOLZ L, HEN R.NR2B-dependent plasticity of adult-born granule cells is necessary for context discrimination.J Neurosci.2012;32(25):8696-8702. [45] TANG YP, WANG H, FENG R, et al. Differential effects of enrichment on learning and memory function in NR2B transgenic mice. Neuropharmacology.2001;41(6):779-790. [46] SCHMIDT-HIEBER C, JONAS P, BISCHOFBERGER J. Enhanced synaptic plasticity in newly generated granule cells of the adult hippocampus. Nature.2004;429(6988):184-187. [47] BLAND BH, ODDIE SD. Theta band oscillation and synchrony in the hippocampal formation and associated structures:the case for its role in sensorimotor integration.Behav Brain Res.2001;127(1-2):119-136. [48] ABRAHAM WC, MASON-PARKER SE, BEAR MF, et al. Heterosynaptic metaplasticity in the hippocampus in vivo: A BCM-like modifiable threshold for LTP.Proc Natl Acad Sci U S A. 2001;98(19): 10924-10929. [49] HILL LE, DROSTE SK, NUTT DJ, et al. Voluntary exercise alters GABAA receptor subunit and glutamic acid decarboxylase-67 gene expression in the rat forebrain. J Psychopharmacol. 2010;24(5): 745-756. [50] SCHOENFELD TJ, RADA P, PIERUZZINI PR, et al.Physical exercise prevents stress-induced activation of granule neurons and enhances local inhibitory mechanisms in the dentate gyrus.J Neurosci. 2013; 33(18): 7770-7777. [51] BAKKER A, KRAUSS GL, ALBERT MS, et al. Reduction of hippocampal hyperactivity improves cognition in amnestic mild cognitive impairment. Neuron.2012;74(3):467-474. [52] DISHMAN RK. Brain monoamines, exercise, and behavioral stress: Animal models. Med Sci Sports Exerc.1997;29(1):63-74. [53] TANTIMONACO M, CECI R, SABATINI S, et al. Physical activity and the endocannabinoid system: An overview.Cell Mol Life Sci.2014; 71(14): 2681-2698. [54] KLEMPIN F, BEIS D, MOSIENKO V, et al. Serotonin is required for exercise-induced adult hippocampal neurogenesis.J Neurosci.2013; 33(19):8270-8275. [55] KONDO M, NAKAMURA Y, ISHIDA Y, et al. The 5-HT3 receptor is essential for exercise-induced hippocampal neurogenesis and antidepressant effects.Mol Psychiatry.2015;20(11):1428-1437. [56] CASSILHAS RC, LEE KS, FERNANDES J, et al. Spatial memory is improved by aerobic and resistance exercise through divergent molecular mechanisms.Neuroscience.2012;202:309-317. [57] BABAEI P, DAMIRCHI A, MEHDIPOOR M, et al.Long term habitual exercise is associated with lower resting level of serum BDNF. Neurosci Lett. 2014;566:304-308. [58] ERICKSON KI, PRAKASH RS, VOSS MW, et al.Brain-derived neurotrophic factor is associated with age-related decline in hippocampal volume.J Neurosci.2010;30(15):5368-5375. [59] VAYNMAN SS, YING Z, YIN D, et al. Exercise differentially regulates synaptic proteins associated to the function of BDNF.Brain Res.2006; 1070(1):124-130. [60] VAYNMAN S, YING Z, GOMEZ-PINILLA F. Hippocampal BDNF mediates the efficacy of exercise on synaptic plasticity and cognition. Eur J Neurosci.2004;20(10):2580-2590. [61] LI Y, LUIKART BW, BIRNBAUM S, et al. TrkB regulates hippocampal neurogenesis and governs sensitivity to anti-depressive treatment. Neuron.2008;59(3):399-412. [62] VIVAR C, POTTER MC, CHOI J, et al.Monosynaptic inputs to new neurons in the dentate gyrus. Nat Commun. 2012;3:1107. [63] SULTAN S, LI L, MOSS J, et al.Synaptic integration of adult-born hippocampal neurons is locally controlled by astrocytes.Neuron.2015; 88(5):957-972. [64] MIKLIC S, JURIC DM, CARMAN-KRZAN M. Differences in the regulation of BDNF and NGF synthesis in cultured neonatal rat astrocytes.Int J Dev Neurosci.2004;22(3):119-130. [65] SAUR L, BAPTISTA PP, DE SENNA PN, et al. Physical exercise increases GFAP expression and induces morphological changes in hippocampal astrocytes.Brain Struct Funct.2014;219(1):293-302. [66] FAHIMI A, BAKTIR MA, MOGHADAM S, et al. Physical exercise induces structural alterations in the hippocampal astrocytes: Exploring the role of BDNF-TrkB signaling.Brain Struct Funct. 2017;222(4): 1797-1808. [67] MOON HY, KIM SH, YANG YR, et al. Macrophage migration inhibitory factor mediates the antidepressant actions of voluntary exercise.Proc Natl Acad Sci U S A.2012;109(32):13094-13099. [68] GEBARA E, SULTAN S, KOCHER-BRAISSANT J, et al. Adult hippocampal neurogenesis inversely correlates with microglia in conditions of voluntary running and aging.Front Neurosci.2013;7:145. [69] SPEISMAN RB, KUMAR A, RANI A, et al.Daily exercise improves memory, stimulates hippocampal neurogenesis and modulates immune and neuroimmune cytokines in aging rats.Brain Behav Immun. 2013;28: 25-43. [70] EGAN MF, KOJIMA M, CALLICOTT JH, et al. The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function.Cell.2003;112(2):257-269. [71] BROWN BM, BOURGEAT P, PEIFFER JJ, et al. Influence of BDNF Val66Met on the relationship between physical activity and brain volume. Neurology.2014;83(15):1345-1352. [72] IERACI A, MADAIO AI, MALLEI A, et al.Brain-derived neurotrophic factor Val66Met human polymorphism impairs the beneficial exercise- induced neurobiological changes in mice.Neuropsychopharmacology. 2016;41(13):3070-3079. [73] GOMEZ-PINILLA F, ZHUANG Y, FENG J, et al.Exercise impacts brain-derived neurotrophic factor plasticity by engaging mechanisms of epigenetic regulation.Eur J Neurosci.2011;33(3):383-390. [74] SLEIMAN SF, HENRY J, AL-HADDAD R, et al.Exercise promotes the expression of brain derived neurotrophic factor (BDNF) through the action of the ketone body β-hydroxybutyrate. Elife.2016;5. pii: e15092. [75] NISHIJIMA T, OKAMOTO M, MATSUI T, et al.Hippocampal functional hyperemia mediated by NMDA receptor/NO signaling in rats during mild exercise.J Appl Physiol (1985).2012;112(1):197-203. [76] DELP MD, ARMSTRONG RB, GODFREY DA, et al. Exercise increases blood flow to locomotor, vestibular, cardiorespiratory and visual regions of the brain in miniature swine.J Physiol. 2001;533 (Pt 3):849-859. [77] Osborne PG. Hippocampal and striatal blood flow during behavior in rats: Chronic laser Doppler flowmetry study.Physiol Behav.1997; 61(4):485-492. [78] NISHIJIMA T, SOYA H.Evidence of functional hyperemia in the rat hippocampus during mild treadmill running. Neurosci Res.2006; 54(3):186-191. [79] MURRELL CJ, COTTER JD, THOMAS KN, et al.Cerebral blood flow and cerebrovascular reactivity at rest and during sub-maximal exercise: Effect of age and 12-week exercise training.Age (Dordr). 2013;35(3): 905-920. [80] PEREIRA AC, HUDDLESTON DE, BRICKMAN AM, et al. An in vivo correlate of exercise-induced neurogenesis in the adult dentate gyrus. Proc Natl Acad Sci U S A.2007;104(13):5638-5643. [81] VIBOOLVORAKUL S, PATUMRAJ S.Exercise training could improve age-related changes in cerebral blood flow and capillary vascularity through the upregulation of VEGF and eNOS.Biomed Res Int. 2014; 2014:230791. [82] LICHT T, ROTHE G, KREISEL T, et al. VEGF preconditioning leads to stem cell remodeling and attenuates age-related decay of adult hippocampal neurogenesis.Proc Natl Acad Sci U S A. 2016;113(48): E7828-E7836. [83] LICHT T, GOSHEN I, AVITAL A, et al. Reversible modulations of neuronal plasticity by VEGF.Proc Natl Acad Sci U S A.2011;108(12): 5081-5086. [84] FABEL K, FABEL K, TAM B, et al.VEGF is necessary for exercise-induced adult hippocampal neurogenesis.Eur J Neurosci. 2003;18(10): 2803-2812. [85] LISTER JP, BARNES CA.Neurobiological changes in the hippocampus during normative aging. Arch Neurol.2009; 66(7): 829-833. [86] RENDEIRO C, RHODES JS.A new perspective of the hippocampus in the origin of exercise-brain interactions.Brain Struct Funct.2018;223(6): 2527-2545. [87] GUERRIERI D, VAN PRAAG H. Exercise-mimetic AICAR transiently benefits brain function. Oncotarget. 2015;6(21):18293-18313. [88] WOODS NI, VAAGA CE, CHATZI C, et al. Preferential targeting of lateral entorhinal inputs onto newly integrated granule cells.J Neurosci. 2018; 38(26):5843-5853. [89] LEAL SL, YASSA MA.Integrating new findings and examining clinical applications of pattern separation.Nat Neurosci.2018;21(2):163-173. |
| [1] | Luo Lin, Song Naiqing, Huang Jin, Zou Xiaodong. Review and prospect of international research on preschool children’s movement development assessment: a CiteSpace-based visual analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1270-1276. |
| [2] | Shu Wenbo, Chen Mengchi, Li Hua, Huang Liqian, Huang Binbin, Zhang Wenhai, Wu Yachen, Wang Zefeng, Li Qiaoli, Liu Peng. Correlation between body fat distribution and characteristics of daily physical activity in college students [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1277-1283. |
| [3] | Zhao Xiang, Wei Cuilan, Zhang Yeting. Neurogenesis and neuroinflammation under exercise: alteration and regulation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 813-820. |
| [4] | Fan Jin, Zeng Luyao, Zhong Dongling, Li Yuxi, Tian Yanping, Huang Yijie, Jin Rongjiang. Development of functional near-infrared spectroscopy in recent 10 years: a visual analysis using CiteSpace [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3711-3717. |
| [5] | Su Mingzhu, Ma Yuewen. Radial extracorporeal shock wave therapy regulates the proliferation and differentiation of neural stem cells in the hippocampus via Notch1/Hes1 pathway after cerebral ischemia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3009-3015. |
| [6] | Dai Yaling, Chen Lewen, He Xiaojun, Lin Huawei, Jia Weiwei, Chen Lidian, Tao Jing, Liu Weilin. Construction of miR-146b overexpression lentiviral vector and the effect on the proliferation of hippocampal neural stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3024-3030. |
| [7] | Tian Ting, Li Xiaoguang. Problems and challenges in regeneration and repair of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3039-3048. |
| [8] | Zhang Shuang, Tan Rui, Wang Chunxiao, Wu Fengyu, Guo Hongyu. MicroRNAs for assessing the motion control of human skeletal muscles [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2755-2760. |
| [9] | Tian Lu, Liu Bin. Effects of physical activity and exercise on metabolic syndrome [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(2): 296-302. |
| [10] | Li Huijun, Wei Cuilan, Li Chuikun, Zhang Yeting. Sports and cognition: a visual analysis using Citespace software [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(14): 2277-2283. |
| [11] | Guo Xiaozheng, Wang Xing. Improvement of cognitive function in older adults based on near-infrared spectroscopy [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(11): 1790-1796. |
| [12] | Sun Nai, Wang Dali, Zhang Jun, Cao Jinming, Qiu Fucheng, Liu Huimiao, Li Dong, Gu Ping. Effects of cytokine-induced neutrophil chemoattractant 3 on the survival and proliferation of neural stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(1): 118-123. |
| [13] | Zhang Lin1, Liu Jinjie1, Zhao Yan2, Liu Yi1, Lin Jianwen1. N-butylphthalide affects cognitive function of APP/PS1 transgenic mice (Alzheimer’s disease model) [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(在线): 1-6. |
| [14] | Hu Yuting, Liu Fei, Liu Jing, Tao Xinrong. Identification of proliferation and differentiation capability of hippocampal neural stem/progenitor cells by adherent culture [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(9): 1330-1335. |
| [15] | Chen Lingling, Yang Zekun, Sun Jianjun, Zhang Cun. Motion compatibility recognition of walk-aid robot based on multi-scale permutation entropy [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(34): 5473-5478. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||