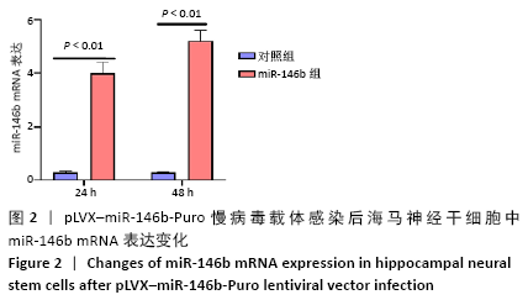
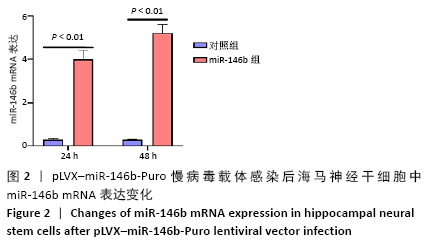
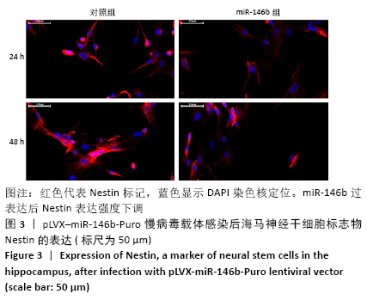
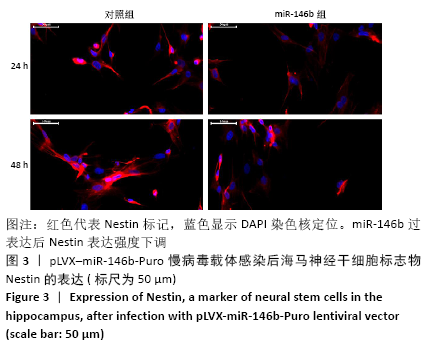
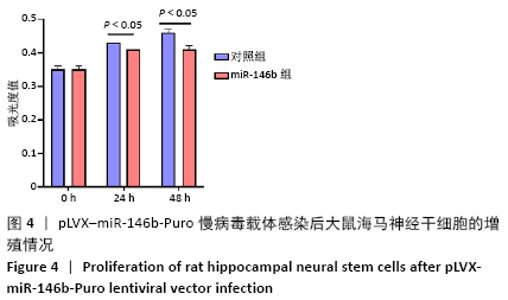
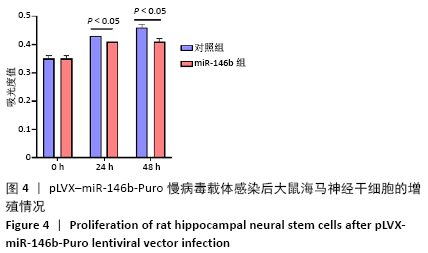
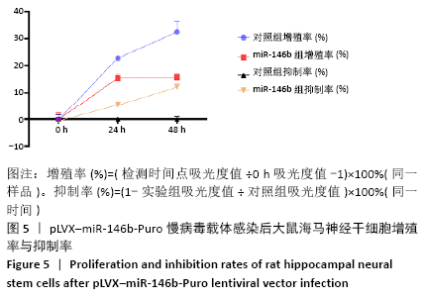
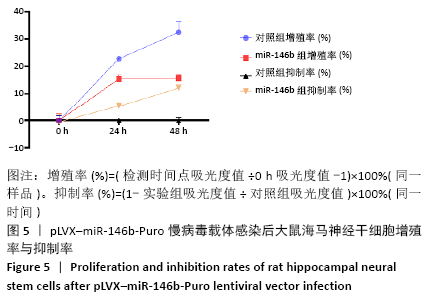
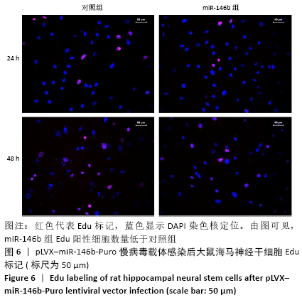
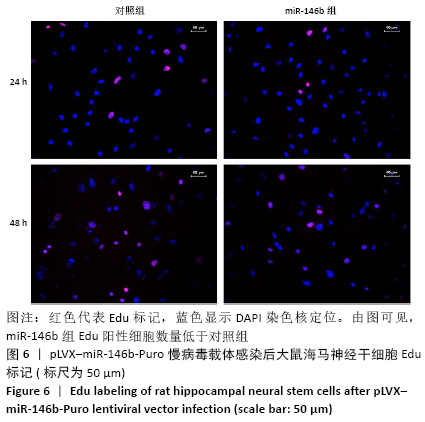
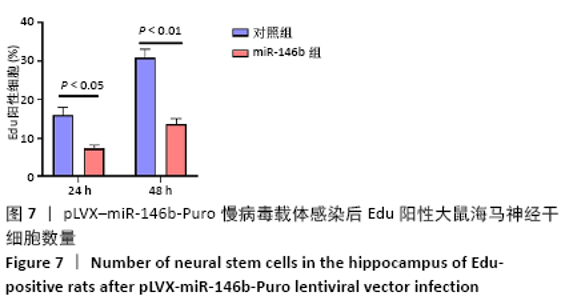
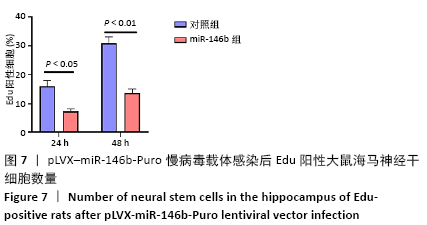
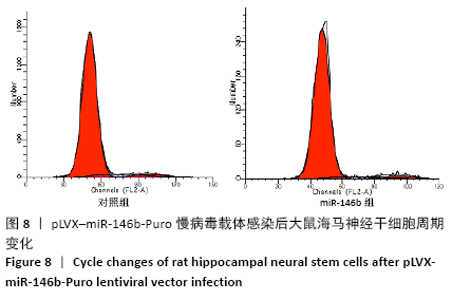
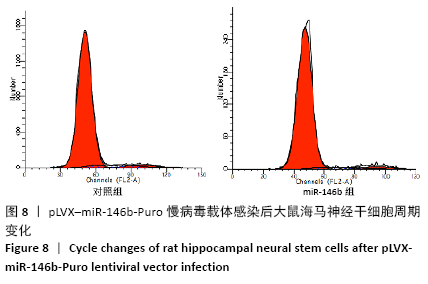
Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (19): 3024-3030.doi: 10.3969/j.issn.2095-4344.3539
Previous Articles Next Articles
Construction of miR-146b overexpression lentiviral vector and the effect on the proliferation of hippocampal neural stem cells
Dai Yaling1, Chen Lewen1, He Xiaojun1, Lin Huawei1, Jia Weiwei1, Chen Lidian1, 2, Tao Jing1, 2, Liu Weilin2
- 1College of Rehabilitation Medicine, 2The Academy of Rehabilitation Industry, Fujian University of Traditional Chinese Medicine, Fuzhou 350122, Fujian Province, China
-
Received:2020-07-07Revised:2020-07-13Accepted:2020-08-25Online:2021-07-09Published:2021-01-13 -
Contact:Liu Weilin, MD, Associate professor, The Academy of Rehabilitation Industry, Fujian University of Traditional Chinese Medicine, Fuzhou 350122, Fujian Province, China -
About author:Dai Yaling, Master, College of Rehabilitation Medicine, Fujian University of Traditional Chinese Medicine, Fuzhou 350122, Fujian Province, China -
Supported by:the Science and Technology Platform Construction Project of Fujian Science and Technology Department, No. 2018Y2002 (to CLD); the Natural Science Foundation of Fujian Province, No. 2016J01382 (to CLD)
CLC Number:
Cite this article
Dai Yaling, Chen Lewen, He Xiaojun, Lin Huawei, Jia Weiwei, Chen Lidian, Tao Jing, Liu Weilin. Construction of miR-146b overexpression lentiviral vector and the effect on the proliferation of hippocampal neural stem cells[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3024-3030.
share this article
| [1] TANG Y, YU P, CHENG L. Current progress in the derivation and therapeutic application of neural stem cells. Cell Death Dis. 2017;8(10): e3108. [2] DING WY, HUANG J, WANG H. Waking up quiescent neural stem cells: Molecular mechanisms and implications in neurodevelopmental disorders. PLoS Genet. 2020 ;16(4):e1008653. [3] SELVARAJ V, JIANG P, CHECHNEVA O, et al. Differentiating human stem cells into neurons and glial cells for neural repair. Front Biosci (Landmark Ed). 2012;17:65-89. [4] VOTTELER M, KLUGER PJ, WALLES H, et al. Stem cell microenvironments--unveiling the secret of how stem cell fate is defined. Macromol Biosci. 2010;10(11):1302-1315. [5] MORIZUR L, CHICHEPORTICHE A, GAUTHIER LR, et al. Distinct Molecular Signatures of Quiescent and Activated Adult Neural Stem Cells Reveal Specific Interactions with Their Microenvironment. Stem Cell Reports. 2018;11(2):565-577. [6] YANG H, ZHANG L, AN J, et al. MicroRNA-Mediated Reprogramming of Somatic Cells into Neural Stem Cells or Neurons. Mol Neurobiol. 2017;54(2):1587-1600. [7] HAO J, ZHANG S, ZHOU Y, et al. MicroRNA 483-3p suppresses the expression of DPC4/Smad4 in pancreatic cancer. FEBS Lett. 2011; 585(1):207-213. [8] XIA X, ZHANG K, CEN G, et al. MicroRNA-301a-3p promotes pancreatic cancer progression via negative regulation of SMAD4. Oncotarget. 2015;6(25):21046-21063. [9] ZHAO Y, JI S, WANG J, et al. mRNA-Seq and microRNA-Seq whole-transcriptome analyses of rhesus monkey embryonic stem cell neural differentiation revealed the potential regulators of rosette neural stem cells. DNA Res. 2014;21(5):541-554. [10] XIAO WZ, LU AQ, LIU XW, et al. Role of miRNA-146 in proliferation and differentiation of mouse neural stem cells. Biosci Rep. 2015;35(5): e00245. [11] 李强,张明伟,李建明,等. miR-146b促进诱导多能干细胞向神经元样细胞的分化[J].中国组织工程研究,2018,22(17):2711-2716. [12] KHAN-MALEK R, WANG Y. Statistical Analysis of Quantitative RT-PCR Results. Methods Mol Biol. 2017;1641:281-296. [13] WANG M, ZHOU A, AN T, et al. N-Hydroxyphthalimide exhibits antitumor activity by suppressing mTOR signaling pathway in BT-20 and LoVo cells. J Exp Clin Cancer Res. 2016;35:41. [14] MCKINNON KM. Flow Cytometry: An Overview. Curr Protoc Immunol. 2018;120:5.1.1-5.1.11. [15] STENUDD M, SABELSTRÖM H, FRISÉN J. Role of endogenous neural stem cells in spinal cord injury and repair. JAMA Neurol. 2015;72(2): 235-237. [16] D’AIUTO L, ZHI Y, KUMAR DAS D, et al. Large-scale generation of human iPSC-derived neural stem cells/early neural progenitor cells and their neuronal differentiation. Organogenesis. 2014;10(4):365-377. [17] GONÇALVES JT, SCHAFER ST, GAGE FH. Adult Neurogenesis in the Hippocampus: From Stem Cells to Behavior. Cell. 2016;167(4):897-914. [18] YAO J, MU Y, GAGE FH. Neural stem cells: mechanisms and modeling. Protein Cell. 2012;3(4):251-261. [19] URBÁN N, BLOMFIELD IM, GUILLEMOT F. Quiescence of Adult Mammalian Neural Stem Cells: A Highly Regulated Rest. Neuron. 2019; 104(5):834-848. [20] CLERVIUS H, BAIG M, MAHAVADI A, et al. Human neural stem cell transplants to address multiple pathologies associated with traumatic brain injury. Neural Regen Res. 2019;14(10):1699-1700. [21] SHI Y, ZHAO X, HSIEH J, et al. MicroRNA regulation of neural stem cells and neurogenesis. J Neurosci. 2010;30(45):14931-14936. [22] DELALOY C, LIU L, LEE JA, et al. MicroRNA-9 coordinates proliferation and migration of human embryonic stem cell-derived neural progenitors. Cell Stem Cell. 2010;6(4):323-335. [23] HAN J, DENLI AM, GAGE FH. The enemy within: intronic miR-26b represses its host gene, ctdsp2, to regulate neurogenesis. Genes Dev. 2012;26(1):6-10. [24] AMBASUDHAN R, TALANTOVA M, COLEMAN R, et al. Direct reprogramming of adult human fibroblasts to functional neurons under defined conditions. Cell Stem Cell. 2011;9(2):113-118. [25] YANG RS, WANG YH, DING C, et al. MiR-146 regulates the repair and regeneration of intervertebral nucleus pulposus cells via Notch1 pathway. Eur Rev Med Pharmacol Sci. 2019;23(11):4591-4598. [26] ZHANG N, LYU Y, PAN X, et al. miR‑146b‑5p promotes the neural conversion of pluripotent stem cells by targeting Smad4. Int J Mol Med. 2017;40(3):814-824. [27] TAGANOV KD, BOLDIN MP, CHANG KJ, et al. NF-kappaB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc Natl Acad Sci U S A. 2006; 103(33):12481-12486. [28] QUINN SR, O’NEILL LA. A trio of microRNAs that control Toll-like receptor signalling. Int Immunol. 2011;23(7):421-425. [29] SONKOLY E, STÅHLE M, PIVARCSI A. MicroRNAs and immunity: novel players in the regulation of normal immune function and inflammation. Semin Cancer Biol. 2008;18(2):131-140. [30] TESTA U, PELOSI E, CASTELLI G, et al. miR-146 and miR-155: Two Key Modulators of Immune Response and Tumor Development. Noncoding RNA. 2017;3(3):22. [31] LU LF, BOLDIN MP, CHAUDHRY A, et al. Function of miR-146a in controlling Treg cell-mediated regulation of Th1 responses. Cell. 2010; 142(6):914-929. [32] GHARWAN H, HIRATA RK, WANG P, et al. Transduction of human embryonic stem cells by foamy virus vectors. Mol Ther. 2007;15(10): 1827-1833. [33] GUO Q, ZHANG J, ZHENG Z, et al. Lentivirus-mediated microRNA-26a-modified neural stem cells improve brain injury in rats with cerebral palsy. J Cell Physiol. 2020;235(2):1274-1286. [34] SHI Z, ZHOU H, LU L, et al. MicroRNA-29a regulates neural stem cell neuronal differentiation by targeting PTEN. J Cell Biochem. 2018; 119(7):5813-5820. [35] JIA M, WANG X, ZHANG H, et al. MicroRNA-132 in the Adult Dentate Gyrus is Involved in Opioid Addiction Via Modifying the Differentiation of Neural Stem Cells. Neurosci Bull. 2019;35(3):486-496. [36] 黄国辉,杨西涛,陈奎,等. Porf-2基因过表达慢病毒载体的构建以及神经细胞的转染[J].上海交通大学学报(医学版), 2015,35(12): 1809-1813. [37] LENDAHL U, ZIMMERMAN LB, McKay RD. CNS stem cells express a new class of intermediate filament protein. Cell. 1990;60(4):585-595. [38] MEI J, BACHOO R, ZHANG CL. MicroRNA-146a inhibits glioma development by targeting Notch1. Mol Cell Biol. 2011;31(17): 3584-3592. [39] MITSUHASHI T, TAKAHASHI T. Proliferation and differentiation characteristics of neural stem cells during course of cerebral cortical histogenesis. Congenit Anom (Kyoto). 2016;56(1):6-11. [40] HONDA M, NAKASHIMA K, KATADA S. Epigenetic Regulation of Human Neural Stem Cell Differentiation. Results Probl Cell Differ. 2018;66: 125-136. [41] MENS MMJ, GHANBARI M. Cell Cycle Regulation of Stem Cells by MicroRNAs. Stem Cell Rev Rep. 2018;14(3):309-322. [42] DALTON S. Linking the Cell Cycle to Cell Fate Decisions. Trends Cell Biol. 2015;25(10):592-600. [43] BARTA T, DOLEZALOVA D, HOLUBCOVA Z, et al. Cell cycle regulation in human embryonic stem cells: links to adaptation to cell culture. Exp Biol Med (Maywood). 2013;238(3):271-275. [44] WANG Y, FAN H, ZHAO G, et al. miR-16 inhibits the proliferation and angiogenesis-regulating potential of mesenchymal stem cells in severe pre-eclampsia. FEBS J. 2012;279(24):4510-4524. [45] HE B, XU Z, CHEN J, et al. Upregulated microRNA-143 inhibits cell proliferation in human nasopharyngeal carcinoma. Oncol Lett. 2016;12(6):5023-5028. [46] HAMAM D, ALI D, KASSEM M, et al. microRNAs as regulators of adipogenic differentiation of mesenchymal stem cells. Stem Cells Dev. 2015;24(4):417-425. [47] SALOMONI P, CALEGARI F. Cell cycle control of mammalian neural stem cells: putting a speed limit on G1. Trends Cell Biol. 2010;20(5):233-243. [48] LENOS MG, TSANIKLIDOU SM. Calcium oscillations, G1 phase duration and neurogenesis timing. Trends Cell Biol. 2010;20(10):577,578. [49] VELAPPAN Y, SIGNORELLI S, CONSIDINE MJ. Cell cycle arrest in plants: what distinguishes quiescence, dormancy and differentiated G1? Ann Bot. 2017;120(4):495-509. [50] NYGREN JM, BRYDER D, JACOBSEN SE. Prolonged cell cycle transit is a defining and developmentally conserved hemopoietic stem cell property. J Immunol. 2006;177(1):201-208. [51] GAO SW, LIU F. Novel insights into cell cycle regulation of cell fate determination. J Zhejiang Univ Sci B. 2019;20(6):467-475. [52] LABBAYE C, TESTA U. The emerging role of MIR-146A in the control of hematopoiesis, immune function and cancer. J Hematol Oncol. 2012;5:13. [53] NAKASA T, MIYAKI S, OKUBO A, et al. Expression of microRNA-146 in rheumatoid arthritis synovial tissue. Arthritis Rheum. 2008;58(5): 1284-1292. [54] WANG X, YU Y. MiR-146b protect against sepsis induced mice myocardial injury through inhibition of Notch1. J Mol Histol. 2018; 49(4):411-417. [55] DE LA POMPA JL, WAKEHAM A, CORREIA KM, et al. Conservation of the Notch signalling pathway in mammalian neurogenesis. Development. 1997;124(6):1139-1148. [56] MIZUTANI T, TANIGUCHI Y, AOKI T, et al. Conservation of the biochemical mechanisms of signal transduction among mammalian Notch family members. Proc Natl Acad Sci U S A. 2001;98(16): 9026-9031. [57] HAI L, ZHANG C, LI T, et al. Notch1 is a prognostic factor that is distinctly activated in the classical and proneural subtype of glioblastoma and that promotes glioma cell survival via the NF-κB(p65) pathway. Cell Death Dis. 2018;9(2):158. |
| [1] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [2] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [3] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [4] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [5] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [6] | Zhao Xiang, Wei Cuilan, Zhang Yeting. Neurogenesis and neuroinflammation under exercise: alteration and regulation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 813-820. |
| [7] | Wang Yujiao, Liu Dan, Sun Song, Sun Yong. Biphasic calcium phosphate loaded with advanced platelet rich fibrin can promote the activity of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 504-509. |
| [8] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| [9] | Zhou Jihui, Yao Meng, Wang Yansong, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Influence of novel nanoscaffolds on biological behaviors of neural stem cells and the related gene expression [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 532-536. |
| [10] | Chen Yang, Huang Denggao, Gao Yuanhui, Wang Shunlan, Cao Hui, Zheng Linlin, He Haowei, Luo Siqin, Xiao Jingchuan, Zhang Yingai, Zhang Shufang. Low-intensity pulsed ultrasound promotes the proliferation and adhesion of human adipose-derived mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3949-3955. |
| [11] | Hao Xiaona, Zhang Yingjie, Li Yuyun, Xu Tao. Bone marrow mesenchymal stem cells overexpressing prolyl oligopeptidase on the repair of liver fibrosis in rat models [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3988-3993. |
| [12] | Zhou Wu, Wang Binping, Wang Yawen, Cheng Yanan, Huang Xieshan. Transforming growth factor beta combined with bone morphogenetic protein-2 induces the proliferation and differentiation of mouse MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3630-3635. |
| [13] | Chen Yutong, Li Chenchen, Liu Yang, Zheng Yaqin, Yang Xihua, An Meiwen. Establishment of an acute radioactive skin injury model in Wistar rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 237-241. |
| [14] | Yu Chengshuai, Du Gang, Pang Shenning, Lao Shan. Chemerin, a pro-inflammatory adipokine, regulates chondrocyte proliferation and metabolism by increasing production of nitric oxide [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 258-263. |
| [15] | Huang Zhusong, Lin Yu, Chen Xiang, Lan Jinfu, Guan Yong, Gao Xi. Alcohol extract of Morinda officinalis improves lipid metabolism and bone metabolism in ovariectomized obese rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 205-210. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||