Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (9): 1330-1335.doi: 10.3969/j.issn.2095-4344.1592
Previous Articles Next Articles
Identification of proliferation and differentiation capability of hippocampal neural stem/progenitor cells by adherent culture
Hu Yuting, Liu Fei, Liu Jing, Tao Xinrong
- School of Medicine, Anhui University of Science and Technology, Huainan 232001, Anhui Province, China
-
Revised:2018-11-15Online:2019-03-28Published:2019-03-28 -
Contact:Tao Xinrong, MD, Professor, School of Medicine, Anhui University of Science and Technology, Huainan 232001, Anhui Province, China -
About author:Hu Yuting, Master candidate, School of Medicine, Anhui University of Science and Technology, Huainan 232001, Anhui Province, China -
Supported by:the National Natural Science Foundation of China, No. 81471161 (to TXR); Anhui Province RC18-Discipline (Professional) Top Talents, No. gxbjZD16 (to TXR)
CLC Number:
Cite this article
Hu Yuting, Liu Fei, Liu Jing, Tao Xinrong. Identification of proliferation and differentiation capability of hippocampal neural stem/progenitor cells by adherent culture[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(9): 1330-1335.
share this article
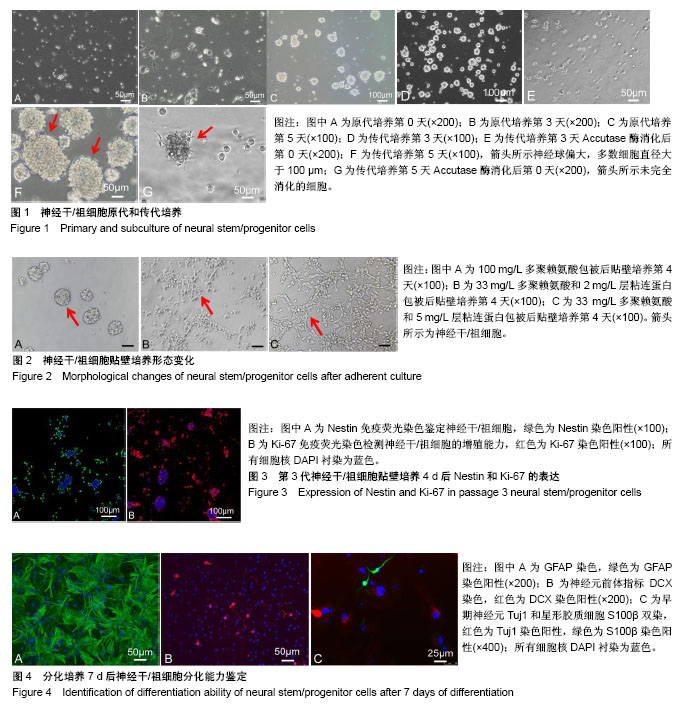
2.1 神经干/祖细胞原代和传代培养形态变化 从PBS悬液中收集原代细胞增加了提取细胞的数量,小鼠海马分离提取原代神经干/祖细胞当天,显微镜下可见细胞悬液中有单个细胞和细小组织碎片,单个细胞大部分呈现的是圆形,边缘清晰,透光性好,见图1A;继续培养3 d,悬液中依然有明显的细小组织碎片,漂浮着没有光泽的死细胞,培养皿底部有突起的贴壁细胞,部分单细胞存活聚集增殖形成小球,聚集的神经球稀疏且较松散,见图1B;培养至第5天,组织碎片仍然存在但所占比例较少,存活的神经干/祖细胞继续增殖形成大小不一的神经球,类似于桑葚状,细胞边缘有单个细胞凸起,边界清晰但不规则,透光性好,见图1C。 原代细胞培养5 d传代,消化后继续培养3 d,细胞增殖形成直径为50-80 μm大小不等的神经球,见图1D;培养3 d后形成的神经球,细胞消化时能够轻易地将细胞消化下来,见图1E;然而传代后的细胞培养至第5天,形成的神经球偏大,多数细胞直径大于100 μm,神经球中央部分细胞密集,见图1F;细胞消化时不易成功消化,仍然存在未完全消化的神经球状态,见图1G。 2.2 神经干/祖细胞贴壁培养形态变化 当只用100 mg/L多聚赖氨酸包被时,单细胞接种,培养4 d,神经干/祖细胞贴壁增殖形成边界清晰的克隆,见图2A,并且有部分神经球贴壁不牢固悬浮于培养液中;当用33 mg/L多聚赖氨酸和2 mg/L层粘连蛋白包被时,培养4 d,神经干/祖细胞增殖形成的克隆有一定程度的铺展,同时存在单个细胞贴壁且形态清晰,见图2B;当用33 mg/L多聚赖氨酸和5 mg/L层粘连蛋白包被时,培养4 d,神经干/祖细胞增殖形成的克隆完全铺展,细胞与细胞之间连接形成网状,见图2C。 2.3 神经干/祖细胞贴壁培养后Nestin的表达 用于鉴定的细胞为第3代细胞贴壁培养至第4天的神经干/祖细胞,免疫荧光法检测特异性标志蛋白Nestin的表达。结果显示,贴壁培养后细胞染色清楚,单个细胞的胞浆和细胞核清晰辨别,部分克隆球中央也成功被染色;Nestin阳性即胞浆绿色细胞所占比例约为82%,细胞核被DAPI衬染为蓝色,Nestin阳性表达率高,说明成功培养得到神经干/祖细胞,见图3A。 2.4 神经干/祖细胞贴壁培养后增殖指标Ki-67的表达 免疫细胞化学染色结果显示,部分神经干/祖细胞呈Ki-67阳性,经过ImageJ软件统计分析Ki-67阳性细胞占所有细胞比例约49%,见图3B。 2.5 神经干/祖细胞贴壁培养诱导分化及鉴定结果 第2代培养第3天的神经干/祖细胞消化成单细胞悬液,贴壁培养诱导分化7 d,免疫荧光染色结果显示,少部分细胞DCX表达阳性,呈红色,说明少部分神经干/祖细胞分化为神经元,见图4A;大部分细胞经过诱导分化后GFAP反应阳性,呈绿色,见图4B;同时早期神经元Tuj1和S100β双染,神经元和星形胶质细胞比例为1∶1.7,见图4C,进一步说明了培养的细胞为神经干/祖细胞,并且具有向不同神经细胞方向分化的能力。"
| [1] Qin W, Chen S, Yang S, et al. The Effect of Traditional Chinese Medicine on Neural Stem Cell Proliferation and Differentiation. Aging Dis. 2017;8(6):792-811.[2] Aimone JB, Li Y, Lee SW, et al. Regulation and function of adult neurogenesis: from genes to cognition. Physiol Rev. 2014;94(4):991-1026.[3] Altman J. Are new neurons formed in the brains of adult mammals. Science. 1962;135(3509):1127-1128.[4] Altman J, Das GD. Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J Comp Neurol. 1965;124(3):319-335.[5] Reynolds BA, Weiss S. Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science. 1992;255(5052):1707-1710.[6] Marei HE, Althani A, Rezk S,et al.Therapeutic potential of human olfactory bulb neural stem cells for spinal cord injury in rats. Spinal Cord. 2016;54(10):785-797.[7] Wu S, Sasaki A, Yoshimoto R,et al. Neural stem cells improve learning and memory in rats with Alzheimer’s disease. Pathobiology.2008; 75(3): 186-194.[8] Wu W, Chen X, Hu C, et al. Transplantation of neural stem cells expressing hypoxia-inducible factor-1α(HIF-1α) behavioral recovery in a rat stroke model. J Clin Neurosci. 2010; 17(1): 92-95.[9] Paul A, Chaker Z, Doetsch F. Hypothalamic regulation of regionally distinct adult neural stem cells and neurogenesis. Science. 2017;356(6345):1383-1386.[10] Bacigaluppi M, Pluchino S, Peruzzotti-Jametti L, et al. Delayed post-ischemic neuroprotection following systemic neural stem cell transplantation involves multiple mechanisms. Brain.2009;132(Pt8): 2239-2251.[11] Wang C, Lu CF, Peng J, et al.Roles of neural stem cells in the repair of peripheral nerve injury.Neural Regen Res. 2017; 12(12):2106-2112.[12] Hou B, Ma J, Guo X, et al. Exogenous Neural Stem Cells Transplantation as a Potential Therapy for Photothrombotic Ischemia Stroke in Kunming Mice Model. Mol Neurobiol. 2017; 54(2):1254-1262.[13] Ho SY, Ling TY, Lin HY, et al. SDF-1/CXCR4 Signaling Maintains Stemness Signature in Mouse Neural Stem/ Progenitor Cells. Stem Cells Int. 2017;2017:2493752.[14] Ziaee SM, Tabeshmehr P, Haider KH, et al. Optimization of time for neural stem cells transplantation for brain stroke in rats. Stem Cell Investig. 2017;4:29.[15] Telias M, Ben-Yosef D.Neural stem cell replacement: a possible therapy for neurodevelopmental disorders?Neural Regen Res. 2015;10(2):180-182.[16] Faigle R, Song H. Signaling mechanisms regulating adult neural stem cells and neurogenesis. Biochim Biophys Acta. 2013;1830(2):2435-2448.[17] Homem CC, Repic M, Knoblich JA. Proliferation control in neural stem and progenitor cells. Nat Rev Neurosci. 2015; 16(11):647-659.[18] Jessberger S, Toni N, Clemenson GD Jr, et al. Directed differentiation of hippocampal stem/progenitor cells in the adult brain. Nat Neurosci. 2008;11(8):888-893.[19] Aguirre A, Rubio ME, Gallo V. Notch and EGFR pathway interaction regulates neural stem cell number and self-renewal. Nature. 2010;467(7313):323-327.[20] Homayouni Moghadam F, Sadeghi-Zadeh M, Alizadeh-Shoorjestan B, et al. Isolation and Culture of Embryonic Mouse Neural Stem Cells. J Vis Exp. 2018;(141). doi: 10.3791/58874. [Epub ahead of print][21] Jadasz JJ, Tepe L, Beyer F, et al. Human mesenchymal factors induce rat hippocampal- and human neural stem cell dependent oligodendrogenesis. Glia. 2018;66(1):145-160.[22] Usui T, Sakurai M, Kawasaki H, et al. Establishment of a novel three-dimensional primary culture model for hippocampal neurogenesis. Physiol Rep. 2017;5(12): e13318.[23] Pachenari N, Kiani S, Javan M. Inhibition of glycogen synthase kinase 3 increased subventricular zone stem cells proliferation. Biomed Pharmacother. 2017;93:1074-1082.[24] Zhao L, Zhou C, Li L, et al. Acupuncture Improves Cerebral Microenvironment in Mice with Alzheimer's Disease Treated with Hippocampal Neural Stem Cells. Mol Neurobiol. 2017; 54(7):5120-5130.[25] Chen M, Puschmann TB, Marasek P, et al. Increased Neuronal Differentiation of Neural Progenitor Cells Derived from Phosphovimentin-Deficient Mice. Mol Neurobiol. 2018; 55(7):5478-5489.[26] Kumar V, Pandey A, Jahan S, et al. Differential responses of Trans-Resveratrol on proliferation of neural progenitor cells and aged rat hippocampal neurogenesis. Sci Rep. 2016;6: 28142.[27] Kim JY, Lee JH, Sun W. Isolation and Culture of Adult Neural Stem Cells from the Mouse Subcallosal Zone. J Vis Exp. 2016; (118). doi: 10.3791/54929.[28] Ludwig PE, Thankam FG, Patil AA, et al. Brain injury and neural stem cells. Neural Regen Res. 2018;13(1):7-18.[29] Gao X,Zhang J,Zhang J,et al.Identification of rat respiratory mucosa stem cells and comparison of the early neural differentiationpotential with the bone marrow mesenchymal stem cells in vitro.Cell Mol Neurobiol.2014,34(2):247-268.[30] Jiang DQ, Wei MD, Wang KW, et al. Nicotine contributes to the neural stem cells fate against toxicity of microglial-derived factors induced by Aβ via the Wnt/β-catenin pathway. Int J Neurosci. 2016;126(3):257-268.[31] Bergström T, Holmqvist K, Tararuk T, et al. Developmentally regulated collagen/integrin interactions confer adhesive properties to early postnatal neural stem cells. Biochim Biophys Acta. 2014;1840(8):2526-2532.[32] Zhang J, Li X, Zhang S, et al. Stromal cell derived factor-1α-laminin crosstalk induced neural stem cells migration and differentiation in vitro. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2017;29(1):57-60.[33] Mimura T, Yamagami S, Uchida S, et al. Isolation of adult progenitor cells with neuronal potential from rabbit corneal epithelial cells in serum- and feeder layer-free culture conditions. Mol Vis. 2010;16:1712-1719.[34] Park D, Xiang AP, Mao FF, et al. Nestin is required for the proper self-renewal of neural stem cells. Stem Cells. 2010; 28(12):2162-2171.[35] Ren M, Du C, Herrero Acero E, et al. A biofidelic 3D culture model to study the development of brain cellular systems. Sci Rep. 2016;6:24953.[36] Ahmed AI, Shtaya AB, Zaben MJ, et al. Endogenous GFAP-positive neural stem/progenitor cells in the postnatal mouse cortex are activated following traumatic brain injury. J Neurotrauma. 2012;29(5):828-842.[37] Choi HW, Hong YJ, Kim JS, et al. In vivo differentiation of induced pluripotent stem cells into neural stem cells by chimera formation. PLoS One. 2017;12(1):e0170735.[38] Petrik D, Latchney SE, Masiulis I, et al. Chromatin Remodeling Factor Brg1 Supports the Early Maintenance and Late Responsiveness of Nestin-Lineage Adult Neural Stem and Progenitor Cells. Stem Cells. 2015;33(12):3655-3665.[39] Radu BM, Dumitrescu DI, Mustaciosu CC, et al. Dual effect of methylglyoxal on the intracellular Ca2+ signaling and neurite outgrowth in mouse sensory neurons. Cell Mol Neurobiol. 2012;32(6):1047-1057.[40] Li RL, Zhang ZZ, Peng M, et al. Postoperative impairment of cognitive function in old mice: a possible role for neuroinflammation mediated by HMGB1, S100B, and RAGE. J Surg Res. 2013;185(2):815-824.[41] Sachewsky N, Leeder R, Xu W, et al. Primitive neural stem cells in the adult mammalian brain give rise to GFAP-expressing neural stem cells. Stem Cell Reports. 2014;2(6):810-824.[42] Huang F, Lan Y, Qin L, et al. Astragaloside IV Promotes Adult Neurogenesis in Hippocampal Dentate Gyrus of Mouse through CXCL1/CXCR2 Signaling. Molecules. 2018;23(9): E2178.[43] Raponi E, Agenes F, Delphin C, et al. S100B expression defines a state in which GFAP-expressing cells lose their neural stem cell potential and acquire a more mature developmental stage. Glia. 2007;55(2):165-177. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [3] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [4] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [5] | Zhao Xiang, Wei Cuilan, Zhang Yeting. Neurogenesis and neuroinflammation under exercise: alteration and regulation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 813-820. |
| [6] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [7] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [8] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [9] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [10] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [11] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [12] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [13] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [14] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [15] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||