Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (18): 2865-2871.doi: 10.3969/j.issn.2095-4344.1733
Previous Articles Next Articles
Coaxial printed double crosslinked alginate/silk fibroin vascular network scaffold
- 1College of Life Information and Instrument Engineering, 2College of Automation, Hangzhou Dianzi University, Hangzhou 310018, Zhejiang Province, China; 3Zhejiang Provincial Key Lab of Medical Information and Three-Dimensional Bio-Printing, Hangzhou 310018, Zhejiang Province, China
-
Received:2019-02-12Online:2019-06-28Published:2019-06-28 -
Contact:Xu Mingen, PhD, Professor, College of Automation, Hangzhou Dianzi University, Hangzhou 310018, Zhejiang Province, China; Zhejiang Provincial Key Lab of Medical Information and Three-Dimensional Bio-Printing, Hangzhou 310018, Zhejiang Province, China -
About author:Li Ningning, Master candidate, College of Life Information and Instrument Engineering, Hangzhou Dianzi University, Hangzhou 310018, Zhejiang Province, China -
Supported by:the National Key Research & Development Program of China, No. 2018YFB1105600 (to WL); the National Natural Science Foundation of China, No. 61675059 (to WL)
CLC Number:
Cite this article
Li Ningning, Xu Mingen, Suo Hairui, Wang Ling. Coaxial printed double crosslinked alginate/silk fibroin vascular network scaffold[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(18): 2865-2871.
share this article
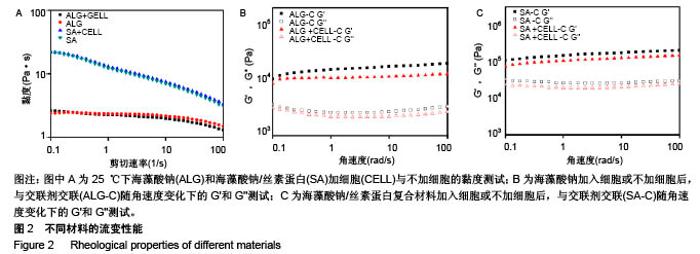
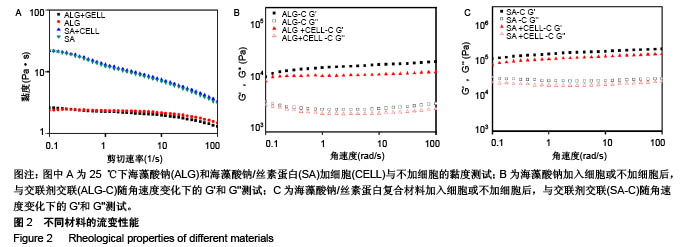
2.1 材料流变性能 3D细胞打印是将细胞与生物材料混合成的生物墨水打印成预先设计的3D结构,因此材料自身的流变性能及混合细胞后的流变性能对生物墨水的可打印性至关重要。图2A为材料的黏度曲线,发现对比单纯海藻酸钠材料,海藻酸钠/丝素蛋白复合材料具有明显的剪切变烯特性,更有利于从喷头中挤出,从而在挤出过程中减少对细胞的损伤;而加入细胞后2种材料的黏度并无明显变化,说明细胞的加入对打印参数影响不大。海藻酸钠/丝素蛋白复合材料的黏度范围为3.07-22 Pa•s,与脱细胞外基质的黏度(2.8-23.6 Pa•s)相似[27],有利于保持细胞活性。血管壁属于生物软组织,其最大的特点就是具有黏弹性。图2B,C中,有无细胞材料的黏弹性测量曲线显示出相似的趋势,将细胞与材料混合后,其储能模量(G')和损耗模量(G'')有轻微降低,这是因为与水凝胶相比,细胞可被认为是软颗粒,因此嵌入细胞会使水凝胶的网络结构发生轻微的变化[28]。在角速度为100 r/s的流变测试中,交联后海藻酸钠水凝胶支架的储存模量为19.7 kPa,损耗模量为 3.36 kPa;而交联后海藻酸钠/丝素蛋白水凝胶支架的储能模量为199.0 kPa,损耗模量为29.9 kPa,较交联后海藻酸钠水凝胶支架的测试结果有显著提高(P < 0.05)。 复合剪切模量由剪切储存模量(G')和剪切损耗模量(G'')决定,复合剪切模量的绝对值能够表示在动态条件下水凝胶的硬度。支架需要具有的机械性能,除适宜细胞生长外,也要保证三维结构在处理、体外培养过程中维持结构的稳定性[29]。相比单纯海藻酸钠水凝胶支架,复合水凝胶支架的储存和损耗模量都有所增加则,其复合剪切模量绝对值也相应增加,表明复合水凝胶的硬度大于纯海藻酸钠水凝胶;相对于纯海藻酸钠支架,复合水凝胶支架更足以承受内部和外部机械载荷的强度,来确保生物打印结构体的稳定性,这源于复合凝胶中钙离子螯合交联海藻酸钠和丝素蛋白形成的β折叠结构,形成的双交联网络。"

2.2 支架的打印及工艺参数优化 利用同轴喷头进行3D打印的工艺参数,包括材料的挤出速率、喷头的移动速度等。打印前,将同轴喷头固定在生物3D打印机的移动轴上,设置合适的注射泵流速和打印速度,生物墨水在针尖处和交联剂接触反应(图1b),形成具有一定机械强度的凝胶中空丝[30],打印机移动轴带动同轴喷头按照预先规划好的打印路径运动,实现3D结构的叠层制造。同轴喷头外管中的水凝胶溶液被交联,形成管壁,交联剂通过内管在生物墨水中流通形成中空通道。交联剂的离子完全渗透进水凝胶需要一定的时间,取决于交联剂离子浓度、海藻酸钠溶液浓度、管壁厚度等。因此,刚挤出的中空纤维内层先被交联,而外层未交联,利用未交联的外层实现丝之间的黏合,以保证支架结构的稳定性。 生物墨水与交联剂挤出速率不匹配时会导致材料堆积,使打印的管道出现内外壁不光滑的现象,如图3A所示;在出丝速度过快而喷头移动速度过慢时,会出现管道弯曲的现象,如图3B所示;当前2种情况同时存在时,打印结果如图3C所示,通道弯曲且内外壁都有材料堆积。经调试,设定生物墨水的挤出速率为7 μL/s、交联剂溶液挤出速率为5 μL/s时,通过测试单位时间丝的垂直挤出长度,得到出丝速度为11 mm/s,于是设定打印速度与出丝速度同为11 mm/s,与之匹配的打印层高设为0.65 mm,间距设为 2 mm。合适的层高可避免破坏已打印的结构,并能够维持通道结构的形状和完整性;而合适的打印间距能够保证支架具有高的孔隙率,利于氧气和营养物质的运输及代谢废物的排出。参数调试后打印出的2层通道如图3D所示,管壁光滑且孔隙均匀。通过图3E的4层通道图可看出,采用间隔层错位打印的方法,使同向的路径相互交错,达到了提高孔隙精度的目的。"


2.3 支架的OCT表征结果 OCT利用弱相干光干涉仪的基本原理,可非侵入性地对3D打印支架内部进行任意截面的断层扫描,比普通光学显微镜更能反映支架内部的真实情况[31-32]。如图4A支架上表面图所示,可清晰看到不同层凝胶丝交叉排列,血管结构为多层复合,中空的管道;图4B,C为不同角度的三维支架图,由图可观测到凝胶丝具有完整的中空结构,且各通道之间互相贯通,这种结构类似血管的中空通道,有利于营养物质和代谢废物的运输。由于3D打印的特性和交联剂F127的去除,打印出的海藻酸钠/丝素蛋白支架具有3个维度的孔隙结构:一为打印凝胶丝之间的孔隙结构;二为同轴打印出来的中空管道结构;三为管道壁上由于F127去除而导致的微孔结构。"

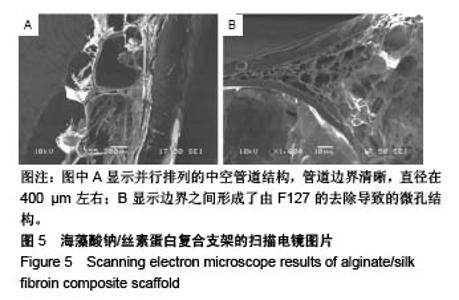
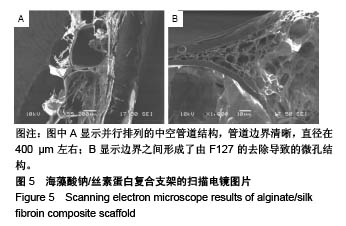
2.4 海藻酸钠/丝素蛋白支架的微观结构 支架内部的中空管道的扫描电镜图像如图5所示,图5A中可见并行排列的中空管道结构,管道边界清晰,直径在400 μm左右,且边界之间形成了由F127的去除导致的微孔结构,如图5B所示。因此,F127在管道结构成型过程中,不仅具有交联丝素蛋白、使结构更加稳定的作用,还具有制孔剂的作用。管壁的微孔结构增加了材料的比表面积,更有利于细胞的生长与黏附[33],在一定程度上解决了交联后海藻酸钠内部孔隙小影响细胞迁移生长的问题[34]。支架的中空管道结构及管壁的微孔结构可为毛细血管的长入提供更多的空间和通道,毛细血管是血液与周围组织进行物质交换的主要部位,它直接给细胞供血、供氧、供给能量及有关营养物质,同时还将带走对有害的代谢产物。多项研究表明,支架的粗糙度和微观孔隙能够提高细胞在材料上的黏附,促进细胞的增殖及功能表达[35-37]。"
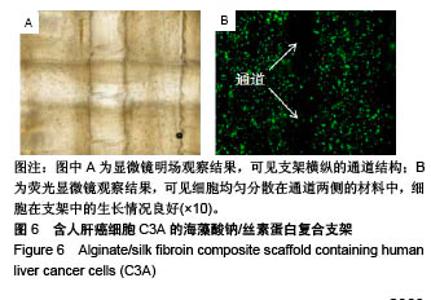
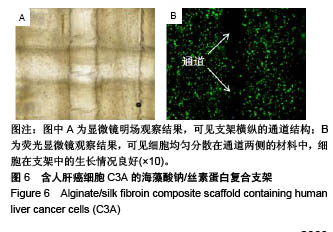
2.5 海藻酸钠/丝素蛋白支架的细胞活性 将海藻酸钠/丝素蛋白复合材料和人肝癌细胞C3A混合后,同轴打印出具有中空管道结构的含细胞支架,培养后的支架在明场条件下可清晰观测出支架横纵的通道结构,见图6A,纵向通道是支架的第1层,横向通道是支架的第2层。图6B为支架的荧光染色图片,可见细胞均匀分散在通道两侧的材料中,细胞在支架中的生长情况良好(绿色为活细胞,红色为死细胞),细胞存活率高于95%,说明海藻酸钠/丝素蛋白生物墨水的细胞相容性良好,且打印过程未对细胞造成明显损伤。对比Gao等[38]使用海藻酸钠获得的92%细胞存活率及Jia等[29]使用海藻酸钠/GelMA混合材料获得的75%细胞存活率,证明海藻酸钠/丝素蛋白生物墨水及其温和的交联方式和打印方法,使得细胞在管壁中有较佳的生存环境。虽然实验中使用人肝癌细胞C3A来测试所打印结构的生物学相关性,但所提出的生物打印系统可用于打印其他活细胞,例如内皮细胞和平滑肌细胞,用于中空管道的仿生发展。"
| [1]Battistella E,Varoni E,Cochis A,et al.Degradable polymers may improve dental practice J Appl Biomater Biomech. 2011;9(3):223-231.[2]Lee W,Debasitis JC, Lee VK,et al.Multi-layered culture of human skin fibroblasts and keratinocytes through three-dimensional freeform fabrication.Biomaterials. 2009;30(8):1587-1595.[3]Yang EK,Seo YK,Youn HH,et al.Tissue engineered artificial skin composed of dermis and epidermis. Artif Organs. 2000;24(1):7-17.[4]陈红.英国研究出3D打印人类眼角膜新材料[J].计算机与网络, 2018,44(12): 10.[5]Lee W,Lee V,Polio S,et al.On-demand three-dimensional freeform fabrication of multi-layered hydrogel scaffold with fluidic channels. Biotechnol Bioeng.2010;105(6):1178-1186.[6]Hribar KC,Soman P,Warner J,et al.Light-assisted direct-write of 3D functional biomaterials. Lab Chip.2013;14(2):268-275.[7]Yuichi N,Makoto N,Chizuka H,et al.Development of a three-dimensional bioprinter: construction of cell supporting structures using hydrogel and state-of-the-art inkjet technology.J Biomech Eng.2009;131(3):35001.[8]Christensen K,Xu C,Chai W,et al.Freeform inkjet printing of cellular structures with bifurcations.Biotechnol Bioeng.2015;112(5):1047-1055.[9]Xu C,Chai W,Huang Y,et al.Scaffold-free inkjet printing of three-dimensional zigzag cellular tubes. Biotechnol Bioeng. 2015; 109(12):3152-3160.[10]Ozbolat IT,Monika H.Current advances and future perspectives in extrusion-based bioprinting. Biomaterials.2016;76(37):321-343.[11]Zhang Y,Yu Y,Ozbolat IT.Direct Bioprinting of Vessel-Like Tubular Microfluidic Channels.J Nanotechnol Eng Med.2013;4(2):21001.[12]Massa S,Sakr MA,Seo J,et al. Bioprinted 3D vascularized tissue model for drug toxicity analysis. Biomicrofluidics.2017;11(4):44109.[13]Zhang Y,Yu Y,Chen H,et al.Characterization of printable cellular micro-fluidic channels for tissue engineering.Biofabrication.2013;5(2): 25004.[14]Luo Y,Lode A,Gelinsky M.Direct Plotting of Three‐Dimensional Hollow Fiber Scaffolds Based on Concentrated Alginate Pastes for Tissue Engineering. Adv Healthc Mater.2013;2(6):777-783.[15]Cheng Y,Zheng F,Lu J,et al.Bioinspired multicompartmental microfibers from microfluidics. Adv Mater.2014;26(30):5184-5190.[16]Gao Q,Liu Z,Lin Z,et al.3D Bioprinting of Vessel-like Structures with Multi-level Fluidic Channels.Acs Biomater Sci Eng.2017;3(3). DOI: 10.1021/acsbiomaterials.6b00643[17]Onoe H,Okitsu T,Itou A, et al. Metre-long cell-laden microfibres exhibit tissue morphologies and functions.Nat Mater.2013;12(6):584.[18]Rowley JA,Madlambayan G,Mooney DJ.Alginate hydrogels as synthetic extracellular matrix materials.Biomaterials.1999;20(1):45-53.[19]Wang S, Zhang Y, Yin G, et al. Electrospun polylactide/silk fibroin– gelatin composite tubular scaffolds for small‐diameter tissue engineering blood vessels.J Appl Polym Sci. 2010;113(4): 2675-2682.[20]Wang SD,Zhang YZ,Yin GB,et al.Fabrication of a composite vascular scaffold using electrospinning technology.MaterSci Eng C.2010;30(5): 670-676.[21]Rodriguez MJ,Brown J,Giordano J,et al.Silk based bioinks for soft tissue reconstruction using 3-dimensional (3D) printing with in vitro and in vivo assessments. Biomaterials 2017;117:105-115.[22]Chawla S,Midha S,Sharma A,et al.Silk‐Based Bioinks for 3D Bioprinting. Adv Healthc Mater. 2018;7(8):e1701204.[23]Unger RE,Wolf M,Peters K,et al.Growth of human cells on a non-woven silk fibroin net: a potential for use in tissue engineering.Biomaterials. 2004;25(6):1069-1075.[24]Liu H,Xu GW,Wang YF,et al.Composite scaffolds of nano-hydroxyapatite and silk fibroin enhance mesenchymal stem cell-based bone regeneration via the interleukin 1 alpha autocrine/paracrine signaling loop.Biomaterials. 2015;49:103-112.[25]Zhou J, Cao C, Ma X, et al.In vitro and in vivo degradation behavior of aqueous-derived electrospun silk fibroin scaffolds.Polym Degrad Stabil. 2010;95(9):1679-1685.[26]Kweon HY, Yeo JH, Lee KG, et al. Effects of poloxamer on the gelation of silk sericin. Macromol Rapid Commun. 2000;21(18):1302-1305[27]Kimsey RB,Spielman A.Motility of Lyme disease spirochetes in fluids as viscous as the extracellular matrix.J Infect Dis.1990;162(5):1205-1208.[28]Zhao Y,Li Y,Mao S,et al. The influence of printing parameters on cell survival rate and printability in microextrusion-based 3D cell printing technology.Biofabrication.2015;7(4):45002.[29]Jia W,Gungor-Ozkerim PS,Zhang YS,et al.Direct 3D bioprinting of perfusable vascular constructs using a blend bioink.Biomaterials. 2016; 106:58-68.[30]王秀娟,张坤生,任云霞,等.海藻酸钠凝胶特性的研究[J].食品工业科技,2008, 29(2):259-262.[31]Wang L,Xu M,Zhang L,et al. Automated quantitative assessment of three-dimensional bioprinted hydrogel scaffolds using optical coherence tomography.Biomed Opt Express.2016;7(3):894-910.[32]Wang L,Xu ME, Luo L,et al.Iterative feedback bio-printing-derived cell-laden hydrogel scaffolds with optimal geometrical fidelity and cellular controllability.Sci Rep.2018;8(1):2802.[33]罗会涛,赵婧,范兴平,等.不同类型多孔结构生物材料支架制备及其性能优化[J].中国材料进展, 2012,31(5):30-39.[34]Armstrong JP,Burke M, Carter BM, et al.3D Bioprinting Using a Templated Porous Bioink.Adv Healthc Mater. 2016;5(14):1724-1730.[35]Kantlehner M,Schaffner P,Finsinger D,et al.Surface Coating with Cyclic RGD Peptides Stimulates Osteoblast Adhesion and Proliferation as well as Bone Formation.Chembiochem.2015;1(2):107-114.[36]Ishaug-Riley SL,Crane-Kruger GM,Yaszemski MJ,et al. Three-dimensional culture of rat calvarial osteoblasts in porous biodegradable polymers.Biomaterials. 1998;19(15):1405.[37]Mikos AG,Lyman MD,Freed LE,et al.Wetting of poly(L-lactic acid) and poly(DL-lactic-co-glycolic acid) foams for tissue culture.Biomaterials. 1994;15(1):55-58.[38]Gao Q,He Y,Fu JZ,et al.Coaxial nozzle-assisted 3D bioprinting with built-in microchannels for nutrients delivery.Biomaterials. 2015;61: 203-215. |
| [1] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [2] | Chen Zhenyu, Zhang Xiaoning, Luo Yuxin, Liang Jianwei, Yan Chi. Evaluation of silk fibroin/curcumin composite film for promoting wound healing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2554-2561. |
| [3] | Xu Changkui, Pu Xiaobing, Lu Yao, Chen Jiarong, Pan Lei. Safety and antibacterial properties of gentamicin-loaded silk fibroin in meniscus repair [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1545-1549. |
| [4] | Liu Xiaoyin, , Zhong Lin, Zheng Bo, Wei Pan, Dai Chen, Hu Liangcong, Wang Tiantian, Liang Xiaolong, Zhang Sai, Wang Xiaoli. Diffusion tensor imaging predicting locomotor function recovery with 3D printing scaffold after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4547-4554. |
| [5] | Zhao Delu, Tie Chaorong, Yang Sisi, Sun Zhen, Wang Xin, Zhu Huaian, Yin Miao. Preparation and characterization of sodium methacrylate modified photocrosslinked alginate hydrogel scaffold [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(22): 3445-3451. |
| [6] | Zhang Yifan, Zhang Jiaying, Xu Mingen, Wang Ling, Zhang He. Optimization of the coaxial cell printing performance of bioink and printing of tissue-engineered scaffolds with vascular-like structure [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(22): 3553-3558. |
| [7] | Zhang Xiaoyun, Chen Yueping, Song Shilei, Zhang Chi, Zhuo Yinghong, Yang Nan, Zhan Huasong, Xu Canhong. Good cell compatibility and permeability of silk fibroin/chitosan composite scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(16): 2544-2550. |
| [8] | She Rongfeng, Zhang Yi, Chen Long, Wang Yuanzheng, Zhang Bin, Huang Qixiang. Biocompatibility of tissue engineered cartilage constructed in vivo by silk fibroin-chitosan scaffold carrying bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(1): 27-32. |
| [9] | Cheng Yang, Liu Min, Zhu Zhongyan, Gao Shasha. Comparative analysis of hydroxyapatite, β-tricalcium phosphate, and carbonated hydroxyapatite alginate dental pulp replacement materials [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(30): 4804-4810. |
| [10] | Chen Lei, Ge Weiming, Lü Linwei, Lei Ming, Teresa Zielińska, Xing Enhong. Mechanical properties of artificial cartilage scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(30): 4763-4768. |
| [11] | Sun Kai, Li Ruixin, Li Hao, Li Dong, Li Hui. Finite element analysis and demonstration of scaffold material stacking [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(30): 4787-4792. |
| [12] | Li Xuanze, Shu Liping, Chen Jiao, Sun Yu, Zou Qiang, Wang Weiyu, Liu Jun, Yang Long, Ma Minxian, Ye Chuan. Preparation of sodium alginate-hydroxyapatite-polyethylene glycol composite hydrogel scaffold and its biocompatibility [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(26): 4230-4236. |
| [13] | Zhou Sirui, Xiao Hongli, Huang Wenliang, Deng Jiang, Ye Peng. Supercritical bone defect repaired by composite bioscaffolds loaded with recombinant human bone morphogenetic protein 2/poly(lactic acid) [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(18): 2806-2811. |
| [14] | Zhao Delu, Tie Chaorong, Wang Xin, Zhu Huaian, Yang Sisi, Li Xiaobao, Yin Miao. Effects of adding strontium ions into photocrosslinked alginate hydrogels on its mechanical and biological properties [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(18): 2880-2887. |
| [15] |
Niu Yu-xiang, Xiong Dang-sheng.
Preparation and in vitro properties of degradable antibacterial composite hydrogel for intestinal fistulas
[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(30): 4860-4866.
|
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||