Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (30): 4860-4866.doi: 10.3969/j.issn.2095-4344.2017.30.018
Previous Articles Next Articles
Preparation and in vitro properties of degradable antibacterial composite hydrogel for intestinal fistulas
- College of Materials Science and Engineering, Nanjing University of Science and Technology, Nanjing 210094, Jiangsu Province, China
-
Received:2017-05-07Online:2017-10-28Published:2017-11-07 -
Contact:Xiong Dang-sheng, Master, Professor, College of Materials Science and Engineering, Nanjing University of Science and Technology, Nanjing 210094, Jiangsu Province, China -
About author:Niu Yu-xiang, Master, College of Materials Science and Engineering, Nanjing University of Science and Technology, Nanjing 210094, Jiangsu Province, China -
Supported by:the National Natural Science Foundation of China, No. 51575278; the Science and Technology Plan Project of Jiangsu Province, No. BY2016004-08, BA2015054
CLC Number:
Cite this article
Niu Yu-xiang, Xiong Dang-sheng.
share this article
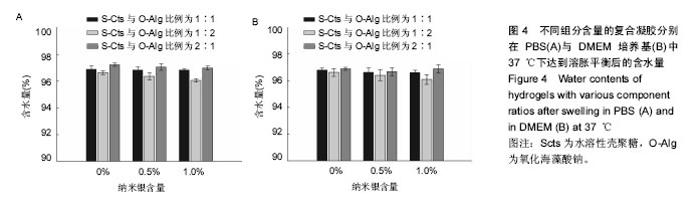
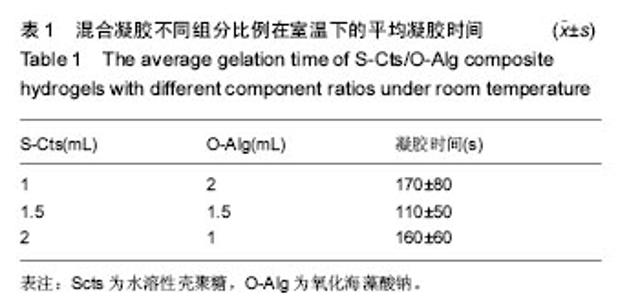
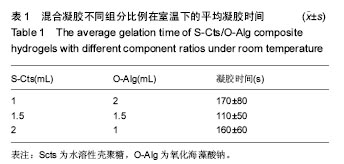
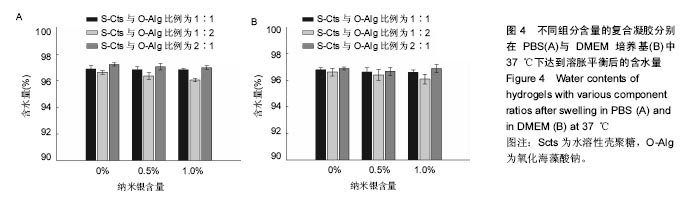
2.3 溶胀性能 图4A,B为不同纳米银含量的复合水凝胶在PBS与DMEM培养基中达到溶胀平衡之后的含水量。从图中可以发现,无论纳米银含量多少的情况下,S-Cts组分越多的凝胶其含水量就越大。这主要是由于S-Cts中接枝了-COOH,使其亲水性更优于-OH被氧化成C=O后的O-Alg,能够吸收更多水分子,而孔径大小此时并不能对含水量起到决定作用。超过90%的高含水量表明这种体系的水凝胶具有很好的仿生特点,能够在人体内维持与周围组织的体液交换。对比图4A、图4B,同样成分的水凝胶在DMEM中的含水量要稍高于在PBS中的含水量,二者并不存在明显差异。不同组分比例的样品在DMEM中的含水量差异比在PBS中更小,因为DMEM培养基中含有的氨基酸结构与壳聚糖、海藻酸钠的结构相似,外界环境对凝胶的内部改变较小。所以,种植种子细胞时直接选用DMEM培养基配置S-Cts与O-Alg溶液可以使形成的凝胶更为稳定,更利于细胞黏附固定。"
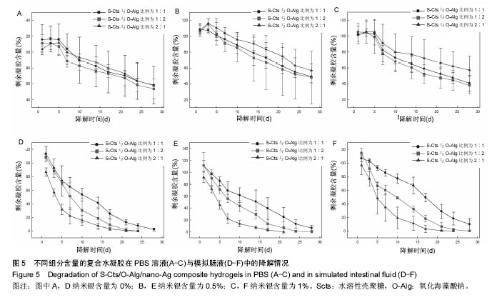
2.4 降解性能 将不同组分含量的试样置于恒温恒湿箱中,在37℃下恒温振荡,分别在PBS及模拟肠液中进行降解4周,结果如图5所示。图5A-C中可看出,在PBS环境中,S-Cts组分含量越大,降解程度越小,经过4周降解之后剩余凝胶约60%。图5D-F中可看出,模拟肠液中降解过程更为迅速,与PBS中不同的是S-Cts组分含量最多的一组降解最快,约在第3周已完全降解,其他2组在第4周结束时也只剩不到20%残余。主要是由于模拟肠液中的胰蛋白酶成分对壳聚糖这种类蛋白质多糖衍生物的分解性能更为突出,造成S-Cts含量多的一组凝胶在S-Cts与O-Alg直接形成的链结被破坏前,S-Cts自身已经被分解,导致凝胶整体迅速降解,而另外2组依然可以保持在PBS状态下的降解规律,即S-Cts组分含量大的降解速率慢。纳米银含量的改变不会影响整体水凝胶的降解规律,只是增大了不同组分变量凝胶残余量的差异。由PBS和模拟肠液中的降解变化规律可知,凝胶在模拟普通生理环境下可以保持缓慢稳定的降解特性,可以为细胞增殖分化、组织再生形成提供充足时间;但在与模拟肠液接触时降解速率显著上升,如果要实现在肠外瘘封堵的内瘘道口处保持平稳降解,需要做出针对性改善。"

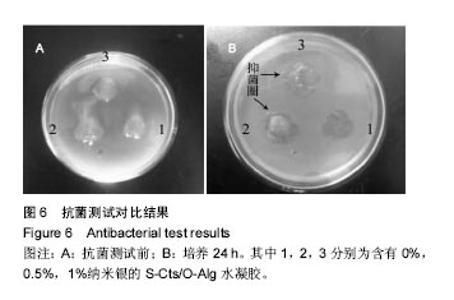
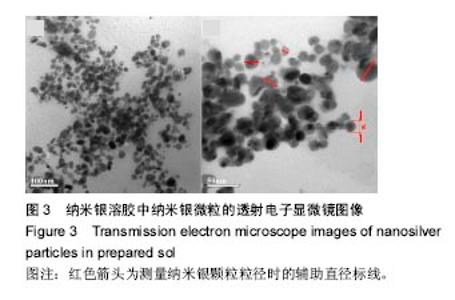
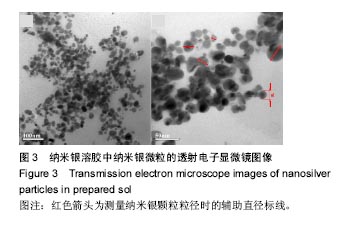
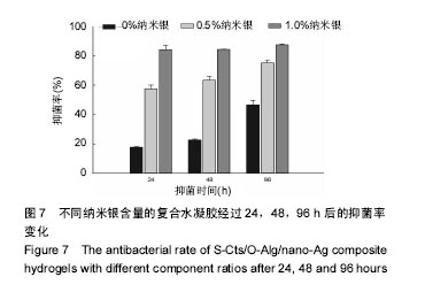
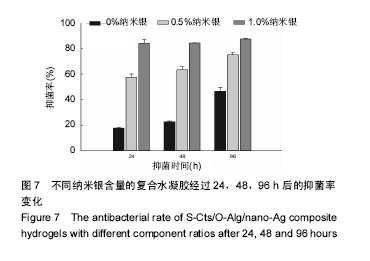
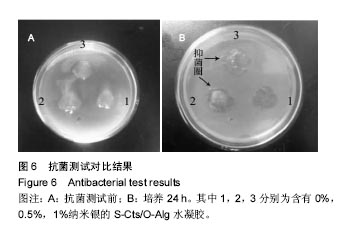
2.5 抗菌性能 采用Kirby Bauer法比较不同纳米银含量的S-Cts/O-Alg/nano-Ag复合凝胶的抑菌性能。从图6中可以直观的看出含有1%纳米银溶胶的复合水凝胶其抑菌圈略大于含有0.5%纳米银的复合水凝胶。而不含纳米银成分的原始S-Cts/O-Alg凝胶并不具备抗菌能力。银元素的抗菌机制一般认为有2种,即银离子可以直接与细菌蛋白中的巯基结合使细胞失活,或是为了抑制银离子变形从而引发DNA大分子浓缩失去复制能力。此外,纳米银微粒的粒径在20-50 nm之间,这种纳米尺寸带来了更大的表面积,对于微生物具有更强的吸附作用。 抑菌圈的观察从定性角度确定了纳米银成分具备灭菌能力,图7中的抑菌率数据进一步从定量角度得出了纳米银含量对于抗菌性能的影响。相同扩散时间下,纳米银含量越高的样品抑菌率也越高。不含纳米银的水凝胶培养 24 h抑菌率不到20%,由此可以得出复合凝胶所产生的的抗菌效果是由纳米银成分带来的。尽管不含纳米银与含有0.5%纳米银的样品,抑菌率会随着扩散时间推移而增加,但96 h后抑菌率仍远低于含有1.0%纳米银的凝胶。含1%纳米银的样品抑菌率始终保持在80%以上,且随时间变化很小。说明S-Cts/O-Alg/nano-Ag复合凝胶制备时选用纳米银含量在1%时可以达到一个稳定峰值,能够一直保持较高的抗菌能力。"
| [1]Wang X, Ren J, Zhu W, et al. Fibrin sealant prevents gastrointestinal anastomosis dehiscence in intra-abdominal sepsis. Int Surg. 2007;92(1):27-31.[2]Avalos-González J, Portilla-deBuen E, Leal-Cortés CA, et al. Reduction of the closure time of postoperative enterocutaneous fistulas with fibrin sealant. World J Gastroenterol. 2010;16(22):2793-2800.[3]Horowitz B, Busch M. Estimating the pathogen safety of manufactured human plasma products: application to fibrin sealants and to thrombin. Transfusion. 2008;48(8):1739-1753.[4]Chouhan VD, De La Cadena RA, Nagaswami C, et al. Simultaneous occurrence of human antibodies directed against fibrinogen, thrombin, and factor V following exposure to bovine thrombin: effects on blood coagulation, protein C activation and platelet function. Thromb Haemost. 1997;77(2):343-349.[5]Davoodi P, Ng WC, Yan WC, et al. Double-Walled Microparticles-Embedded Self-Cross-Linked, Injectable, and Antibacterial Hydrogel for Controlled and Sustained Release of Chemotherapeutic Agents.ACS Appl Mater Interfaces. 2016;8(35):22785-22800.[6]Hong LTA, Kim YM, Park HH, et al. An injectable hydrogel enhances tissue repair after spinal cord injury by promoting extracellular matrix remodeling. Nat Commun. 2017;8(1):533.[7]Zhuo F, Liu X, Gao Q, et al. Injectable hyaluronan- methylcellulose composite hydrogel crosslinked by polyethylene glycol for central nervous system tissue engineering.Mater Sci Eng C Mater Biol Appl. 2017;81:1-7.[8]Zhou T, Li X, Li G, et al. Injectable and thermosensitive TGF-β1-loaded PCEC hydrogel system for in vivo cartilage repair. Sci Rep. 2017;7(1):10553.[9]Gullbrand SE, Schaer TP, Agarwal P, et al. Translation of an injectable triple-interpenetrating-network hydrogel for intervertebral disc regeneration in a goat model. Acta Biomater. 2017;60:201-209.[10]Chen CH, Chen SH, Mao SH,et al. Injectable thermosensitive hydrogel containing hyaluronic acid and chitosan as a barrier for prevention of postoperative peritoneal adhesion. Carbohydr Polym. 2017;173:721-731.[11]Nguyen MK, Lee DS. Injectable biodegradable hydrogels. Macromol Biosci. 2010;10(6):563-579.[12]Bhattarai N, Gunn J, Zhang M. Chitosan-based hydrogels for controlled, localized drug delivery. Adv Drug Deliv Rev. 2010;62(1):83-99.[13]Tan H, Ramirez CM, Miljkovic N, et al. Thermosensitive injectable hyaluronic acid hydrogel for adipose tissue engineering. Biomaterials. 2009;30(36):6844-6853.[14]Gumera C, Rauck B, Wang Y. Materials for central nervous system regeneration: bioactive cues. Journal of Materials Chemistry. 2011; 21(20):7033-7051.[15]Lee KY, Mooney DJ. Hydrogels for tissue engineering. Chem Rev. 2001;101(7):1869-1879.[16]Xiao B, Lü S, Zhen C, et al. Self-reinforcing injectable hydrogel with both high water content and mechanical strength for bone repair. Chem Eng J. 2015; 288:546-556.[17]Pike DB, Cai S, Pomraning KR, et al. Heparin-regulated release of growth factors in vitro and angiogenic response in vivo to implanted hyaluronan hydrogels containing VEGF and bFGF. Biomaterials. 2006;27(30):5242-5251.[18]Liao X, Chen G, Jiang M. Hydrogels locked by molecular recognition aiming at responsiveness and functionality. Polymer Chemistry. 2012; 4(6):1733-1745.[19]Ngoenkam J, Faikrua A, Yasothornsrikul S, et al. Potential of an injectable chitosan/starch/beta-glycerol phosphate hydrogel for sustaining normal chondrocyte function. Int J Pharm. 2010;391(1-2):115-124.[20]Chiu YL, Chen SC, Su CJ, et al. pH-triggered injectable hydrogels prepared from aqueous N-palmitoyl chitosan: in vitro characteristics and in vivo biocompatibility. Biomaterials. 2009;30(28):4877-4888.[21]Zhang H, Qadeer A, Mynarcik D, et al. Delivery of rosiglitazone from an injectable triple interpenetrating network hydrogel composed of naturally derived materials. Biomaterials. 2011;32(3):890-898.[22]Marsich E, Borgogna M, Donati I, et al. Alginate/lactose-modified chitosan hydrogels: a bioactive biomaterial for chondrocyte encapsulation. J Biomed Mater Res A. 2008;84(2):364-376.[23]Leor J, Tuvia S, Guetta V, et al. Intracoronary injection of in situ forming alginate hydrogel reverses left ventricular remodeling after myocardial infarction in Swine. J Am Coll Cardiol. 2009;54(11):1014-1023.[24]Sakai S, Hirose K, Moriyama K, et al. Control of cellular adhesiveness in an alginate-based hydrogel by varying peroxidase and H(2)O(2) concentrations during gelation. Acta Biomater. 2010;6(4):1446-1452.[25]Thomas R, Soumya KR, Mathew J, et al. Inhibitory effect of silver nanoparticle fabricated urinary catheter on colonization efficiency of Coagulase Negative Staphylococci. J Photochem Photobiol B. 2015;149:68-77.[26]刘蕊,沈蕾,栗茜,等. 新型表面镀银手术器械抗菌性能研究[J]. 牙体牙髓牙周病学杂志, 2011,21(12):713-715.[27]Suliman Y AO, Ali D, Alarifi S, et al. Evaluation of cytotoxic, oxidative stress, proinflammatory and genotoxic effect of silver nanoparticles in human lung epithelial cells. Environ Toxicol. 2015;30(2):149-160.[28]邓红梅. 坤泰胶囊联合纳米银抗菌水凝胶治疗老年性阴道炎的研究[J]. 中国医学创新, 2015,12(19):94-96.[29]郑允权,郭养浩,饶强,等. 纳米氯化银/壳聚糖抗菌敷料的制备及功能评价[J]. 材料科学与工程学报, 2016, 34 (2) :188-191.[30]Yu H, Xu X, Chen X, et al. Preparation and antibacterial effects of PVA‐PVP hydrogels containing silver nanoparticles. Journal of Applied Polymer Science. 2007; 103(1): 125-133.[31]Thomas V, Yallapu MM, Sreedhar B, et al. A versatile strategy to fabricate hydrogel-silver nanocomposites and investigation of their antimicrobial activity. J Colloid Interface Sci. 2007; 315(1):389-395.[32]Sun S, Wang L, Wang A. Adsorption properties of crosslinked carboxymethyl-chitosan resin with Pb(II) as template ions. J Hazard Mater. 2006;136(3):930-937.[33]Ito T, Yeo Y, Highley CB, et al. The prevention of peritoneal adhesions by in situ cross-linking hydrogels of hyaluronic acid and cellulose derivatives. Biomaterials. 2007;28(6):975-983.[34]Ruhela D, Riviere K, Szoka FC Jr. Efficient synthesis of an aldehyde functionalized hyaluronic acid and its application in the preparation of hyaluronan-lipid conjugates. Bioconjug Chem. 2006;17(5):1360-1363.[35]Jin Y, Dong S. One-pot synthesis and characterization of novel silver-gold bimetallic nanostructures with hollow interiors and bearing nanospikes. The Journal of Physical Chemistry B. 2003; 107(47): 12902-12905.[36]Yin B, Ma H, Wang S, et al. Electrochemical synthesis of silver nanoparticles under protection of poly (N-vinylpyrrolidone). The Journal of Physical Chemistry B. 2003;107(34): 8898-8904. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [3] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [4] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [5] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [6] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [7] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [8] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [9] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [10] | Huang Chuanjun, Zou Yu, Zhou Xiaoting, Zhu Yangqing, Qian Wei, Zhang Wei, Liu Xing. Transplantation of umbilical cord mesenchymal stem cells encapsulated in RADA16-BDNF hydrogel promotes neurological recovery in an intracerebral hemorrhage rat model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 510-515. |
| [11] | Le Guoping, Zhang Ming, Xi Licheng, Luo Hanwen. Preparation and in vitro evaluation of vancomycin hydrochloride@polylactic acid-glycolic acid copolymer-chitosan-hyaluronic acid composite sustained-release microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 528-534. |
| [12] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [13] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [14] | Guan Jian, Jia Yanfei, Zhang Baoxin , Zhao Guozhong. Application of 4D bioprinting in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 446-455. |
| [15] | Huang Bo, Chen Mingxue, Peng Liqing, Luo Xujiang, Li Huo, Wang Hao, Tian Qinyu, Lu Xiaobo, Liu Shuyun, Guo Quanyi . Fabrication and biocompatibility of injectable gelatin-methacryloyl/cartilage-derived matrix particles composite hydrogel scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2600-2606. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||