Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (17): 2782-2788.doi: 10.3969/j.issn.2095-4344.1730
Dental-derived mesenchymal stem cells promote periodontal tissue regeneration: possibility and prospects
Du Shasha, Cai Zhiguo, Yang Kun, Liu Qi
- Department of Periodontology, Hospital of Stomatology, Zunyi Medical University, Zunyi 563003, Guizhou Province, China
-
Revised:2019-02-12Online:2019-06-18Published:2019-06-18 -
Contact:Liu Qi, MD, Professor, Department of Periodontology, Hospital of Stomatology, Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
About author:Du Shasha, Master candidate, Physician, Department of Periodontology, Hospital of Stomatology, Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81860196 (to LQ) and 81760199 (to YK)
CLC Number:
Cite this article
Du Shasha, Cai Zhiguo, Yang Kun, Liu Qi. Dental-derived mesenchymal stem cells promote periodontal tissue regeneration: possibility and prospects[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(17): 2782-2788.
share this article
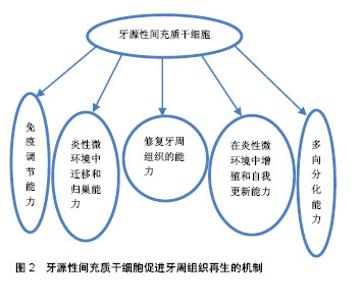
2.1 牙源性间充质干细胞 间充质干细胞是一种源自中胚层未分化的多功能干细胞,可以从胚胎早期阶段或出生后组织(成体干细胞)中分离。成人多种组织中均可获得,例如骨髓、口腔颌面部组织等。间充质干细胞能在病理条件下增殖和分化,具有修复组织缺损和自我更新的潜力[4]。研究表明,间充质干细胞可分化为成骨细胞和骨骼肌细胞等[5],且同种异体间充质干细胞具有良好的免疫耐受性,无异位组织形成。因此,间充质干细胞可作为组织工程和组织修复的理想材料[6]。 在口腔颌面部区域,牙源性间充质干细胞主要源于乳牙和恒牙的牙周膜干细胞、根尖乳头衍生干细胞、脱落乳牙干细胞、牙囊干细胞和牙髓干细胞。 2.1.1 牙源性间充质干细胞的多向分化能力 人牙周膜中含有间充质干细胞的一个亚群,可以分化为成骨细胞、成纤维细胞、成牙骨质细胞,形成牙骨质和牙周膜样组织,维持牙周组织结构和功能稳定[7]。牙髓干细胞源自颅神经嵴,位于髓腔内,具有克隆和快速增殖能力,能分化为成牙本质细胞、软骨细胞和脂肪细胞[8]。Laino等[9]在体外诱导人牙髓干细胞群分化形成骨样组织,且移植入免疫缺陷大鼠体内可形成板层骨。根尖乳头衍生干细胞和牙囊干细胞在牙萌出前均位于牙胚中,根尖乳头衍生干细胞位于牙乳头前端,牙囊干细胞位于釉质和牙乳头周围的结缔组织,两者均可分化为成牙本质细胞、成牙骨质细胞和脂肪细胞[10]。免疫缺陷大鼠体内植入根尖乳头衍生干细胞,可以在细胞载体羟磷灰石/磷酸三钙表面形成一层牙本质样组织。由于牙本质主要由成牙本质细胞样细胞分泌而成,因此这些细胞可能源于根尖牙乳头中间充质干细胞样细胞的分化。此外,根尖乳头衍生干细胞是一种独特的多能干细胞,这种细胞可以高水平表达抗细胞凋亡蛋白及人端粒末端转移酶,这两种基因可以调控细胞的增殖。根尖乳头衍生干细胞似乎比牙髓干细胞更具有组织再生的潜能,可能因为根尖乳头衍生干细胞具有更高的增殖能力[11]。牙囊干细胞可分化为牙周膜成纤维细胞、成骨细胞和成牙骨质细胞的祖细胞[12]。Guo等[13]研究发现,载有牙囊干细胞的牙本质基质支架植入小鼠3个不同部位:牙囊干细胞可以在网膜囊内促进牙本质再生;在头颅骨缺损处,牙囊干细胞能促进矿化基质形成;牙囊干细胞还可以在牙槽窝中促进牙根样组织形成牙髓牙本质复合体及似牙骨质样组织间牙周膜。脱落乳牙干细胞是从天然脱落乳牙中分离出来的,能分化为成骨细胞、成牙本质细胞、脂肪细胞和神经细胞等[14]。Yamada等[15]将狗脱落乳牙干细胞联合血小板血浆植入小型猪下颌骨缺损处,检测到缺损区域表面覆盖着结构良好的血管化新骨。牙周膜干细胞与牙髓干细胞分化受局部微环境中分子受体、生长因子、转录因子和细胞外基质蛋白等调控,体内外研究显示两者均能诱导形成矿化组织[16-17]。 2.1.2 牙源性间充质干细胞在炎性微环境中增殖和自我更新能力 任何牙周组织再生或修复的初级阶段均起始于干细胞增殖,以获得能够迁移、归巢和向特定组织分化的功能性干细胞。克隆的形成和时间依赖性倍增是干细胞自我更新的主要特征。炎性微环境能显著增强间充质干细胞的增殖能力。研究表明,源于牙周炎性组织的牙周膜干细胞比健康牙周组织的牙周膜干细胞具有更高的增殖潜能[18]。炎性条件下,CXC趋化因子8 (CXCL8)和炎性趋化因子5(CC cchcmokinc ligand 5,CCL5)升高是导致牙周膜干细胞快速增殖和克隆形成单位增加的重要原因。然而,炎性趋化因子5只能调节静息状态下牙周膜干细胞增殖能力,目前还未见炎性趋化细胞因子调节牙周膜干细胞增殖的报道[19]。白细胞介素1(1 μg/L)、肿瘤坏死因子α(10 μg/L)、γ-干扰素(100 μg/L)连续刺激7 d,牙周膜干细胞增殖和克隆形成能力明显增强[20]。同时,牙龈卟啉单胞菌来源的脂多糖刺激牙周膜干细胞48 h也能上调牙周膜干细胞的增殖能力[21]。此外,白细胞介素1β(20 μg/L)、肿瘤坏死因子α(40 μg/L)刺激120 h后,还能显著上调健康牙周膜干细胞的生存能力[22]。然而,高浓度炎性因子反而会抑制牙周膜干细胞的增殖能力,甚至诱导其凋亡。肿瘤坏死因子α联合γ-干扰素显著加速了牙周膜干细胞凋亡进程,其机制可能是高剂量肿瘤坏死因子α将γ-干扰素激活肿瘤坏死因子受体超家族成员6(Fas-6)的非凋亡信号转为半胱天冬酶3(caspase-3)和半胱天冬酶8(caspase-8)等促凋亡相关途径,降低了胞内NF-κB水平,激活细胞凋亡信号,导致牙周膜干细胞凋亡[23]。 以上研究表明,牙周膜干细胞能在轻度炎性微环境中快速增殖,同时也会在高水平炎性因子介导下凋亡。由此可以推测,牙周膜干细胞对于轻、中度牙周炎具有良好的修复能力。 2.1.3 牙源性间充质干细胞在炎性微环境中迁移和归巢能力 迁移和归巢是间充质干细胞增殖、牙周组织再生和愈合过程中的必经阶段[14]。新兴研究表明,细胞迁移和归巢是由某些特定细胞分泌的特异性细胞因子和趋化因子或损伤组织释放的特异分子共同介导的。间充质干细胞选择性迁移和归巢在一定程度上决定了炎性微环境中牙周再生组织的质与量,但其机制尚未完全阐明[24]。 已有研究表明,炎性微环境对间充质干细胞募集和归巢有着积极促进作用[25]。骨髓间充质干细胞体内趋化性和迁移率在很大程度上受关键归巢受体CXC趋化因子受体4(CXC chemokine receptor 4,CXCR4)的影响,CXCR4的表达主要由炎性微环境调控。牙周膜干细胞在白细胞介素1β(5 μg/L)、肿瘤坏死因子α(10 μg/L)中培养24 h后,CXCR4表达上调,与CXCL12特异性结合,从而增强了牙周膜干细胞迁移和归巢能力,诱导细胞迁移和归巢到特定部位[26]。但该实验中克隆形成能力尚未增加,表明牙周膜干细胞增殖和归巢两个阶段不能同时激活[27]。同时,牙周炎性组织释放的趋化因子也能显著增加牙周膜干细胞迁移能力。此外,除了前文中提到促进牙周膜干细胞增殖外,炎性趋化因子5还能调节牙周膜干细胞迁移和归巢能力。炎性趋化因子5升高显著上调了肌动蛋白细胞骨架、细胞黏附分子、F-肌动蛋白相关基因(ACTG1)的表达,促进细胞迁移信号传导[19]。 2.1.4 牙源性间充质干细胞的免疫调节能力 Sundin等[28]首次提出机体对同种异体植入的间充质干细胞仅具有弱免疫原性。间充质干细胞同种异体移植能激活细胞毒性T淋巴细胞,活化的细胞毒性T淋巴细胞将间充质干细胞作为识别靶点,产生较弱免疫排斥反应。 间充质干细胞以剂量依赖性抑制细胞因子和外源性免疫细胞(如T细胞、B细胞、NK细胞、树突状细胞)的产生,还能释放可溶性免疫抑制因子[29],如转化生长因子β、肝细胞生长因子等,抑制前细胞毒性T淋巴细胞向细胞毒性T淋巴细胞分化。因此,间充质干细胞不会被外源性细胞毒性T淋巴细胞或自然杀伤细胞裂解。此外,由于间充质干细胞缺乏Ⅱ类主要组织兼容性复合物和共刺激分子表达,其不能完全被同种异体抗原识别[30]。 健康的间充质干细胞可以抑制T细胞增殖。有学者将外周血单核细胞分别与健康间充质干细胞和炎性间充质干细胞共培养,研究发现,与炎性间充质干细胞相比,健康间充质干细胞与活化的外周血单核细胞共培养能明显抑制Th17分化及白细胞介素17分泌,而调节性T细胞分化和白细胞介素10分泌增加[31],说明炎性牙周组织中分离出的间充质干细胞对T细胞功能抑制作用明显低于健康间充质干细胞。根尖乳头衍生干细胞具有较低的免疫原性,能抑制T细胞增殖,还能以剂量依赖性的方式抑制单向混合淋巴细胞反应[32]。脱落乳牙干细胞显著抑制Th17细胞分化,促进调节性T细胞的产生。系统性红斑狼疮患者全血中输入脱落乳牙干细胞后,外周血中调节性T细胞与Th17细胞之间的比例恢复正常,相关症状减轻[33],这说明脱落乳牙干细胞具有良好的免疫调节作用。牙囊干细胞能产生转化生长因子β并抑制外周血单核细胞增殖,Toll样受体3和Toll样受体4激动剂治疗可增强牙囊干细胞的免疫抑制能力,促进转化生长因子β和白细胞介素6的分泌。研究表明,体外培养的牙髓干细胞能上调可溶性因子的表达,抑制外周血单核细胞分泌γ-干扰素,从而影响外周血单核细胞的增殖。Toll样受体通过上调转化生长因子β和白细胞介素6的表达而触发牙髓干细胞的免疫抑制[34]。因此推测牙髓干细胞可能适用于预防或治疗与器官移植相关的T细胞排斥反应。此外,牙髓干细胞还能诱导活化的T细胞凋亡,并改善结肠炎小鼠相关组织炎性损伤,但敲除FasL基因后降低了牙髓干细胞的免疫调节特性,这表明牙髓干细胞免疫调节与FasL配体的表达有关[35]。 综上所述,间充质干细胞同种异体移植后,由于具有低免疫原性,不会被免疫细胞(如T细胞、B细胞、NK细胞、树突状细胞)裂解,是牙周组织再生种子细胞的理想选择。 2.1.5 牙源性间充质干细胞的衰老 有证据表明不同年龄个体中分离的间充质干细胞都有组织再生能力,但间充质干细胞和体细胞一样,其增殖和分化潜能随着年龄的增长逐渐降低。对年轻供者(0-18岁)和老年供者(59-97岁)间充质干细胞的体外生长特性研究发现:年轻供者间充质干细胞能高速增殖,镜下观察到特征性的纺锤体形态,且碱性磷酸酶表达低;而老年供者间充质干细胞比例显著下降,生长速率低、寿命短,细胞呈现大而扁平的衰老形态,且与衰老相关的β-半乳糖苷酶活性表达升高[36]。Zheng等[37]观察到微环境对牙周膜干细胞生理学有较大影响。当年轻供者的牙周膜干细胞暴露于老化环境培养基时,其分化功能降低,成骨能力丧失。相反,改变培养环境能恢复其生物活性,逆转与年龄相关的细胞衰老。微环境也是影响间充质干细胞生理学功能的关键因素。因此,间充质干细胞微环境及其信号转导网络对间充质干细胞的影响显著,改善微环境是促进内源性和外源性间充质干细胞再生潜力的重要途径[38]。 由此可以认为:间充质干细胞“适应性”功能与年龄存在负相关关系,来自年轻供者的牙源性间充质干细胞作为牙周再生工具更具有优势,且间充质干细胞移植后,应控制局部微环境炎性反应。 2.2 牙源性间充质干细胞修复牙周组织的能力 牙源性间充质干细胞具有良好的组织修复能力,在体内成骨能力已经得到证实,可作为同种异体移植的理想载体。牙髓干细胞是成牙本质细胞和成骨细胞的祖细胞,能有效修复实验性牙周炎模型中牙周组织缺损[39]。脱落牙齿中牙髓干细胞虽不能直接分化为成骨细胞,但可以形成骨诱导模板,招募成骨细胞聚集,诱导新生牙槽骨形成[40]。 动物模型组织学也表明,牙源性间充质干细胞可以促进牙槽骨再生。牙周膜干细胞治疗大鼠牙槽骨缺损21 d后,检测到大量类似牙骨质和牙周膜样组织的充填物[41]。研究者将载有牙周膜干细胞的凝胶状支架置入绵羊晚期牙周炎模型牙槽骨缺损处,4周时观察到大量类似成骨细胞的细胞系覆盖于缺损处牙槽骨表面[42]。这与Yu等[43]在大鼠模型中得到的研究结果相同。牙囊干细胞与牙周膜干细胞同时植入免疫缺陷小鼠皮下,可见Sharpey样纤维、牙骨质及牙周膜样组织形成[44]。免疫缺陷小鼠皮下植入根尖乳头衍生干细胞后,检测到典型的牙骨质和牙周膜样组织[45]。同样,脱落乳牙干细胞也能在免疫缺陷小鼠体内形成牙髓-牙本质样复合物。另外,牙髓干细胞同种异体植入微型猪牙周炎模型中可有效修复牙周软硬组织,且无明显免疫排斥等不良反应[46],还能修复牙骨质、牙周膜和牙槽骨之间的功能性连接,促进牙周组织重建[47]。Aimetti等[48]发表了1篇56岁男性患者第三磨牙牙髓干细胞治疗牙周缺损的临床病例,术后1年从临床和放射学角度均观察到缺损处大量骨样组织填充。此外,载有同种异体牙髓干细胞的胶原支架植入人牙槽骨缺损处,形成新生牙槽骨组织的质量和数量方面均优于其他成骨技术[49]。以上研究均表明:牙源性间充质干细胞对牙周组织缺损具有良好的修复能力。 2.3 牙源性间充质干细胞促进牙周组织再生的机制 牙源性间充质干细胞促进牙周组织再生,必须具备以下3个因素:①向缺损组织前体细胞分化的能力、强增殖能力;②足量血液供应;③分子信号传导。 如前文所述,牙源性间充质干细胞可以分化为成骨细胞、成纤维细胞和成牙骨质细胞等,具有多向分化能力。此外,牙源性间充质干细胞在牙周炎早期主要表现为诱导肉芽组织形成,进展期则能促进牙周组织中大量稳定的新生血管网形成,提供丰富的血液[50]。在缺损处植入脱落乳牙干细胞后,可观察到缺损处连接受体血管的功能性血管形成[51],表明牙源性间充质干细胞可以诱导牙周组织血管重建,促进血液循环。 血液中含有丰富的营养物质,将分子信号传递到指定部位,如胰岛素样生长因子1、血管内皮生长因子、转化生长因子等通过新生血管传递到病变位置,诱导牙髓干细胞分化为成纤维细胞[52]。同时,牙髓干细胞还分泌人牙骨质蛋白和Ⅰ型胶原蛋白,合成牙周膜中的牙龈牙骨质纤维。牙周膜干细胞还能够产生多肽和蛋白质,如生物活性因子和营养因子等,抑制相邻细胞凋亡和炎症反应,减少组织炎性损伤。研究表明,生物活性分子如釉质基质衍生物和血小板衍生生长因子等能促进牙周组织再生[53]。 牙源性间充质干细胞植入动物模型后,信号分子胰岛素样生长因子1、肿瘤坏死因子α、白细胞介素6、制瘤素M和某些细胞(如Th2淋巴细胞和单核细胞)可通过新生血管网到达病变部位。胰岛素样生长因子1能诱导成纤维细胞分泌胶原纤维,并激活mTOR途径,诱导成纤维细胞分化为成骨细胞进而分泌骨钙素[54]。骨钙素可以上调骨形态发生蛋白2、Runt相关转录因子2的表达,促进成骨细胞合成细胞外基质[16]。肿瘤坏死因子α(10 μg/L)对牙髓干细胞的增殖和生长周期没有直接影响,但低浓度(1 μg/L)肿瘤坏死因子α可以激活NF-κB途径,上调矿化相关基因的表达,如骨形态发生蛋白2、碱性磷酸酶、Runt相关的转录因子2和Ⅰ型胶原蛋白,诱导细胞外基质矿化[55]。白细胞介素6与其家族细胞因子制瘤素M协同作用也能上调碱性磷酸酶表达。碱性磷酸酶是成骨细胞形成的必要因子,能促进细胞外基质的矿化,并激活JAK3/STAT3信号通路,上调成骨相关基因表达,进而诱导牙髓干细胞分化为成骨细胞[56]。此外,细胞外基质金属蛋白酶及金属蛋白酶组织抑制剂在牙周再生中起关键作用。牙周膜干细胞可以抑制基质金属蛋白酶1和基质金属蛋白酶8的活性,上调金属蛋白酶组织抑制剂2和金属蛋白酶组织抑制剂4的表达,使细胞外基质在成骨分化过程中达到最佳状态[57]。全血中单核细胞和Th2淋巴细胞都能产生白细胞介素10,促进骨保护素产生。骨保护素能下调RANKL、活化T细胞核因子c1和巨噬细胞集落刺激因子活性,从而抑制破骨细胞的分化和形成[58]。 最近,Fu等[59]提出了间充质干细胞在组织再生中的新机制:①细胞外囊泡和纳米隧道管的形成可使不同细胞相互连接,并输送各种营养因子。Aliotta等[60]研究表明受损组织中的细胞通过细胞外囊泡向干细胞传递mRNA或生长因子信号,诱导干细胞重新编码表型分子,提高组织对干细胞的亲和力。细胞外囊泡还可诱导受损组织中体细胞增殖。例如,牙源性间充质干细胞的细胞外囊泡能促进牙周损伤组织中存活的体细胞重新进入细胞周期进而分泌细胞外基质,促进牙周组织再生[28];②间充质干细胞具有抗细胞凋亡能力,当间充质干细胞与心肌细胞共培养时,可以抑制缺血心肌细胞凋亡,可能是间充质干细胞中线粒体通过纳米隧道管转入心肌细胞内,恢复了缺血心肌中线粒体的功能[61]。 以上研究表明,牙周再生机制十分复杂,众说纷纭,目前的研究尚未完全明了,但在实验性研究已经取得了一定的成果,见图2。"
| [1] Corlan Pu?cu D, Ciuluvic? RC, Anghel A, et al. Periodontal disease in diabetic patients - clinical and histopathological aspects. Rom J Morphol Embryol. 2016;57(4):1323-1329.[2] Chalisserry EP, Nam SY, Park SH, et al.Therapeutic potential of dental stem cells. J Tissue Eng. 2017; 8(1):1-17.[3] Collart-Dutilleul PY, Chaubron F, De Vos J, et al. Allogenic banking of dental pulp stem cells for innovative therapeutics. World J Stem Cells. 2015;7(7):1010-1021.[4] Liao J, Yu X, Hu X, et al. lncRNA H19 mediates BMP9-induced osteogenic differentiation of mesenchymal stem cells (MSCs) through Notch signaling. Oncotarget. 2017; 8(32):53581-53601.[5] Kim MJ, Kim ZH, Kim SM, et al. Conditioned medium derived from umbilical cord mesenchymal stem cells regenerates atrophied muscles. Tissue Cell. 2016;48(5):533-543.[6] Rohban R, Pieber TR. Mesenchymal Stem and Progenitor Cells in Regeneration: Tissue Specificity and Regenerative Potential. Stem Cells Int. 2017;2017:5173732.[7] Seo BM, Miura M, Gronthos S, et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet. 2004;364(9429):149-155.[8] Yang X, van der Kraan PM, Bian Z, et al. Mineralized tissue formation by BMP2-transfected pulp stem cells. J Dent Res. 2009;88(11):1020-1025.[9] Laino G, d'Aquino R, Graziano A, et al. A new population of human adult dental pulp stem cells: a useful source of living autologous fibrous bone tissue (LAB). J Bone Miner Res. 2005;20(8):1394-1402.[10] Sonoyama W, Liu Y, Fang D, et al. Mesenchymal stem cell-mediated functional tooth regeneration in swine. PLoS One. 2006;1:e79.[11] Sonoyama W, Liu Y, Yamaza T, et al. Characterization of the apical papilla and its residing stem cells from human immature permanent teeth: a pilot study. J Endod. 2008;34(2):166-171.[12] Chen FM, Sun HH, Lu H, et al. Stem cell-delivery therapeutics for periodontal tissue regeneration. Biomaterials.2012;33(27): 6320-6344.[13] Guo W, Gong K, Shi H, et al. Dental follicle cells and treated dentin matrix scaffold for tissue engineering the tooth root. Biomaterials. 2012;33(5):1291-1302.[14] Du ZH, Li SL, Ge XY, et al. Comparison of the secretory related molecules expression in stem cells from the pulp of human exfoliated deciduous teeth and dental pulp stem cells. Zhonghua Kou Qiang Yi Xue Za Zhi. 2018;53(11):741-747.[15] Yamada Y, Ito K, Nakamura S, et al. Promising cell-based therapy for bone regeneration using stem cells from deciduous teeth, dental pulp, and bone marrow. Cell Transplant. 2011;20(7):1003-1013.[16] Ching HS, Luddin N, Rahman IA, et al. Expression of Odontogenic and Osteogenic Markers in DPSCs and SHED: A Review. Curr Stem Cell Res Ther. 2017;12(1):71-79.[17] Bright R, Hynes K, Gronthos S, et al. Periodontal ligament-derived cells for periodontal regeneration in animal models: a systematic review. J Periodontal Res. 2015;50(2): 160-172.[18] Yang H, Gao LN, An Y, et al. Comparison of mesenchymal stem cells derived from gingival tissue and periodontal ligament in different incubation conditions. Biomaterials. 2013; 34(29):7033-7047.[19] Lee JS, Lee JB, Cha JK, et al. Chemokine in inflamed periodontal tissues activates healthy periodontal-ligament stem cell migration. J Clin Periodontol. 2017;44(5):530-539.[20] Zhang F, Si M, Wang H, et al. IL-1/TNF-α Inflammatory and Anti-Inflammatory Synchronization Affects Gingival Stem/Progenitor Cells' Regenerative Attributes. Stem Cells Int. 2017;2017:1349481.[21] Zhou L, Dörfer CE, Chen L, et al. Porphyromonas gingivalis lipopolysaccharides affect gingival stem/progenitor cells attributes through NF-κB, but not Wnt/β-catenin, pathway. J Clin Periodontol. 2017;44(11):1112-1122.[22] Tomasello L, Mauceri R, Coppola A, et al. Mesenchymal stem cells derived from inflamed dental pulpal and gingival tissue: a potential application for bone formation. Stem Cell Res Ther. 2017;8(1):179.[23] Liu Y, Wang L, Kikuiri T, et al. Mesenchymal stem cell-based tissue regeneration is governed by recipient T lymphocytes via IFN-γ and TNF-α. Nat Med. 2011;17(12):1594-1601.[24] Sordi V. Mesenchymal stem cell homing capacity. Transplantation. 2009;87(9 Suppl):S42-45. [25] Yu Y, Wu RX, Gao LN, et al. Stromal cell-derived factor-1-directed bone marrow mesenchymal stem cell migration in response to inflammatory and/or hypoxic stimuli. Cell Adh Migr. 2016;10(4):342-359.[26] Ziaei R, Ayatollahi M, Yaghobi R, et al. Involvement of TNF-α in differential gene expression pattern of CXCR4 on human marrow-derived mesenchymal stem cells. Mol Biol Rep. 2014; 41(2):1059-1066.[27] Yu Y, Bi CS, Wu RX, et al. Effects of short-term inflammatory and/or hypoxic pretreatments on periodontal ligament stem cells: in vitro and in vivo studies. Cell Tissue Res. 2016;366(2): 311-328.[28] Sundin M, Barrett AJ, Ringdén O, et al. HSCT recipients have specific tolerance to MSC but not to the MSC donor. J Immunother. 2009;32(7):755-764.[29] Rasmusson I, Ringdén O, Sundberg B, et al. Mesenchymal stem cells inhibit the formation of cytotoxic T lymphocytes, but not activated cytotoxic T lymphocytes or natural killer cells. Transplantation. 2003;76(8):1208-1213.[30] Uccelli A, Moretta L, Pistoia V. Mesenchymal stem cells in health and disease. Nat Rev Immunol. 2008;8(9):726-736.[31] Liu D, Xu J, Liu O, et al. Mesenchymal stem cells derived from inflamed periodontal ligaments exhibit impaired immunomodulation. J Clin Periodontol. 2012;39(12): 1174-1182.[32] Ding G, Liu Y, An Y, et al. Suppression of T cell proliferation by root apical papilla stem cells in vitro. Cells Tissues Organs. 2010;191(5):357-364.[33] Yamaza T, Kentaro A, Chen C, et al. Immunomodulatory properties of stem cells from human exfoliated deciduous teeth. Stem Cell Res Ther. 2010;1(1):5.[34] Morsczeck C, Völlner F, Saugspier M, et al. Comparison of human dental follicle cells (DFCs) and stem cells from human exfoliated deciduous teeth (SHED) after neural differentiation in vitro. Clin Oral Investig. 2010;14(4):433-440.[35] Zhao Y, Wang L, Jin Y, et al. Fas ligand regulates the immunomodulatory properties of dental pulp stem cells. J Dent Res. 2012;91(10):948-954.[36] Kawamura H, Nakatsuka R, Matsuoka Y, et al. TGF-β Signaling Accelerates Senescence of Human Bone-Derived CD271 and SSEA-4 Double-Positive Mesenchymal Stromal Cells. Stem Cell Reports. 2018;10(3):920-932.[37] Zheng W, Wang S, Ma D, et al. Loss of proliferation and differentiation capacity of aged human periodontal ligament stem cells and rejuvenation by exposure to the young extrinsic environment. Tissue Eng Part A. 2009;15(9): 2363-2371.[38] Li Y, Pan E, Wang Y, et al. Flk-1?Sca-1? mesenchymal stem cells: functional characteristics in vitro and regenerative capacity in vivo. Int J Clin Exp Pathol. 2015;8(9):9875-9888.[39] Aksel H, Huang GT. Human and Swine Dental Pulp Stem Cells Form a Vascularlike Network after Angiogenic Differentiation in Comparison with Endothelial Cells: A Quantitative Analysis. J Endod. 2017;43(4):588-595.[40] Miura M, Gronthos S, Zhao M, et al. SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci U S A. 2003;100(10):5807-5812.[41] Han J, Menicanin D, Marino V, et al. Assessment of the regenerative potential of allogeneic periodontal ligament stem cells in a rodent periodontal defect model. J Periodontal Res. 2014;49(3):333-345.[42] Mrozik KM, Wada N, Marino V, et al. Regeneration of periodontal tissues using allogeneic periodontal ligament stem cells in an ovine model. Regen Med. 2013;8(6): 711-723.[43] Yu N, Oortgiesen DA, Bronckers AL, et al. Enhanced periodontal tissue regeneration by periodontal cell implantation. J Clin Periodontol. 2013;40(7):698-706.[44] Shi S, Bartold PM, Miura M, et al. The efficacy of mesenchymal stem cells to regenerate and repair dental structures. Orthod Craniofac Res. 2005;8(3):191-199.[45] Han C, Yang Z, Zhou W, et al. Periapical follicle stem cell: a promising candidate for cementum/periodontal ligament regeneration and bio-root engineering. Stem Cells Dev. 2010; 19(9):1405-1415.[46] Fu X, Jin L, Ma P, et al. Allogeneic stem cells from deciduous teeth in treatment for periodontitis in miniature swine. J Periodontol. 2014;85(6):845-851.[47] Khorsand A, Eslaminejad MB, Arabsolghar M, et al. Autologous dental pulp stem cells in regeneration of defect created in canine periodontal tissue. J Oral Implantol. 2013; 39(4):433-443.[48] Aimetti M, Ferrarotti F, Cricenti L, et al. Autologous dental pulp stem cells in periodontal regeneration: a case report. Int J Periodontics Restorative Dent. 2014;34 Suppl 3:s27-33.[49] D'Aquino R, De Rosa A, Lanza V, et al. Human mandible bone defect repair by the grafting of dental pulp stem/progenitor cells and collagen sponge biocomplexes. Eur Cell Mater. 2009;18:75-83.[50] Bakopoulou A, About I. Stem Cells of Dental Origin: Current Research Trends and Key Milestones towards Clinical Application. Stem Cells Int. 2016;2016:4209891.[51] Sakai VT, Zhang Z, Dong Z, et al. SHED differentiate into functional odontoblasts and endothelium. J Dent Res. 2010; 89(8):791-796.[52] Kawai T, Katagiri W, Osugi M, et al. Secretomes from bone marrow-derived mesenchymal stromal cells enhance periodontal tissue regeneration. Cytotherapy. 2015;17(4): 369-381.[53] Liu AQ, Hu CH, Jin F, et al. Contributions of Bioactive Molecules in Stem Cell-Based Periodontal Regeneration. Int J Mol Sci. 2018;19(4): E1016.[54] Ochiai H, Okada S, Saito A, et al. Inhibition of insulin-like growth factor-1 (IGF-1) expression by prolonged transforming growth factor-β1 (TGF-β1) administration suppresses osteoblast differentiation. J Biol Chem. 2012;287(27): 22654-22661.[55] Qin Z, Fang Z, Zhao L, et al. High dose of TNF-α suppressed osteogenic differentiation of human dental pulp stem cells by activating the Wnt/β-catenin signaling. J Mol Histol. 2015; 46(4-5):409-420.[56] Feng X, Shen S, Cao P, et al. The role of oncostatin M regulates osteoblastic differentiation of dental pulp stem cells through STAT3 pathway. Cytotechnology. 2016;68(6): 2699-2709.[57] Page-McCaw A, Ewald AJ, Werb Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nat Rev Mol Cell Biol. 2007;8(3):221-233.[58] Rifas L, Arackal S, Weitzmann MN. Inflammatory T cells rapidly induce differentiation of human bone marrow stromal cells into mature osteoblasts. J Cell Biochem. 2003;88(4): 650-659.[59] Fu Y, Karbaat L, Wu L, et al. Trophic Effects of Mesenchymal Stem Cells in Tissue Regeneration. Tissue Eng Part B Rev. 2017;23(6):515-528.[60] Aliotta JM, Sanchez-Guijo FM, Dooner GJ, et al. Alteration of marrow cell gene expression, protein production, and engraftment into lung by lung-derived microvesicles: a novel mechanism for phenotype modulation. Stem Cells. 2007; 25(9): 2245-2256.[61] Han H, Hu J, Yan Q, et al. Bone marrow-derived mesenchymal stem cells rescue injured H9c2 cells via transferring intact mitochondria through tunneling nanotubes in an in vitro simulated ischemia/reperfusion model. Mol Med Rep. 2016;13(2):1517-1524. |
| [1] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [2] | Zhang Mi, Wu Saixuan, Dong Ming, Lu Ying, Niu Weidong. Expression of interleukin-24 in a mouse model of periapical periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 679-684. |
| [3] | Tang Xiaokai, Li Weiming. Role and mechanism of Nel-like molecule-1 in promoting bone fusion after spinal fusion [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3914-3920. |
| [4] | Yang Caihui, Liu Qicheng, Dong Ming, Wang Lina, Zuo Meina, Lu Ying, Niu Weidong. Serine/threonine protein kinases can promote bone destruction in mouse models of chronic periapical periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3654-3659. |
| [5] | Zhao Chuntao, Qing Mingsong, Yu Langbo, Peng Jiachen . Meta-analysis of total knee arthroplasty guided by kinematic alignment and mechanical alignment [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(9): 1435-1442. |
| [6] |
Zhang Cong, Zhao Yan, Du Xiaoyu, Du Xinrui, Pang Tingjuan, Fu Yining, Zhang Hao, Zhang Buzhou, Li Xiaohe, Wang Lidong.
Biomechanical analysis of the lumbar spine and pelvis in adolescent
idiopathic scoliosis with lumbar major curve |
| [7] | Xu Nuo, Cao Zhen, Li Xiaojie, Shi Chun. MicroRNA-21 regulates proliferation and differentiation of osteoclasts in periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1225-1230. |
| [8] |
Cen Yanhui, Xia Meng, Jia Wei, Luo Weisheng, Lin Jiang, Chen Songlin, Chen Wei, Liu Peng, Li Mingxing, Li Jingyun, Li Manli, Ai Dingding, Jiang Yunxia.
Baicalein inhibits the biological behavior of hepatocellular
carcinoma stem cells by downregulation of Decoy receptor 3 expression |
| [9] | He Yujie, Wang Haiyan, Li Zhijun, Li Xiaohe, Cai Yongqiang, Dai Lina, Xu Yangyang, Wang Yidan, Xu Xuebin. Digital measurements of the anatomical parameters of pedicle-rib unit screw fixation in thoracic vertebrae of preschoolers [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(6): 869-876. |
| [10] | Yan Shu, Lu Yan, Ouyang Zhaolian. Analysis of programs on tissue engineering funded by the National Natural Science Foundation of China between 2013 and 2018 [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(5): 731-735. |
| [11] | Lü Jianwei, Ma Jianxiong, Ma Xinlong. Melatonin promotes Schwann cell migration by activating a typical Wnt/β-catenin signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5030-5037. |
| [12] | Sun Jian, Fang Chao, Gao Fei, Wei Laifu, Qian Jun. Clinical efficacy and complications of short versus long segments of internal fixation for the treatment of degenerative scoliosis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(3): 438-445. |
| [13] | Gao Yangyang, Che Xianda, Han Pengfei, Liang Bin, Li Pengcui. Accuracy of robot-assisted and fluoroscopy-guided pedicle screw placement: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(3): 446-452. |
| [14] | Zhang Jian, Wang Xiaojian, Qin Dean, Zhao Zhongtao, Liang Qingyuan, An Qijun, Song Jiefu. Risk factors for proximal junctional kyphosis after spinal deformity surgery: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(3): 460-468. |
| [15] |
Wang Zhen, Li Xiaolan, Liu Jianguo.
Insights into immunoregulatory properties of dental-derived mesenchymal stem cells in oral diseases [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(25): 4060-4067. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||