Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (13): 1995-2001.doi: 10.3969/j.issn.2095-4344.1688
Previous Articles Next Articles
Effects of perfluorotributylamine/alginate/bioglass biomaterials on viability and osteogenic differentiation of adipose-derived stem cells
Luo Kai, Yang Yafeng, Ma Teng, Xia Bing, Huang Liangliang, Huang Jinghui, Luo Zhuojing
- Department of Orthopedics, Xijing Hospital, Air Force Medical University, Xi’an 710032, Shaanxi Province, China
-
Revised:2019-01-09Online:2019-05-08Published:2019-05-08 -
Contact:Luo Zhuojing, Professor, Chief physician, Department of Orthopedics, Xijing Hospital, Air Force Medical University, Xi’an 710032, Shaanxi Province, China -
About author:Luo Kai, Master candidate, Department of Orthopedics, Xijing Hospital, Air Force Medical University, Xi’an 710032, Shaanxi Province, China -
Supported by:the National Natural Science Foundation of China (General Program), No. 81672148 (to HJH)
CLC Number:
Cite this article
Luo Kai, Yang Yafeng, Ma Teng, Xia Bing, Huang Liangliang, Huang Jinghui, Luo Zhuojing. Effects of perfluorotributylamine/alginate/bioglass biomaterials on viability and osteogenic differentiation of adipose-derived stem cells[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(13): 1995-2001.
share this article
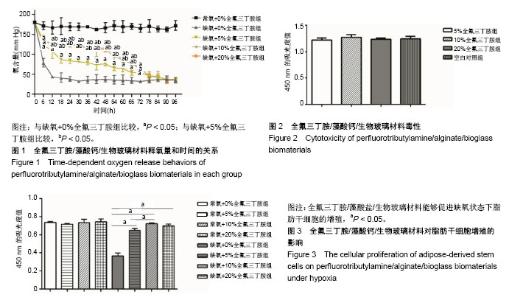
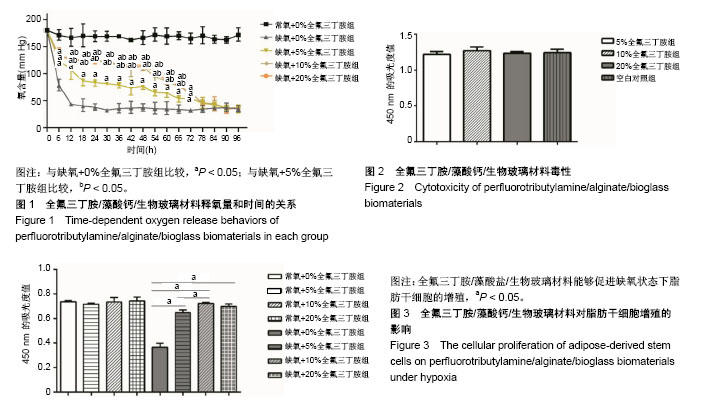
2.1 全氟三丁胺/藻酸钙/生物玻璃材料的释氧特征 将含有0%,5%,10%,20%全氟三丁胺的藻酸盐/生物玻璃材料分别置于缺氧培养箱中,每6 h抽取材料上层培养基进行血气分析。应用血气分析仪对其所取样本检测其氧含量,结果发现在72 h内各检测时间点缺氧各组培养基中氧含量显著低于常氧组,且缺氧条件下10%全氟三丁胺组与20%全氟三丁胺组在释氧能力上差异无显著性意义,见图1。 2.2 全氟三丁胺/藻酸钙/生物玻璃材料的毒性评价 各组材料浸提液毒性测定结果显示:5%全氟三丁胺组、10%全氟三丁胺组、20%全氟三丁胺组及空白对照组的CCK-8吸光度值分别为1.220±0.038,1.271±0.051,1.235±0.022,1.244±0.045。4组组间比较差异无显著性意义(P=0.401),见图2。 2.3 脂肪干细胞在全氟三丁胺/藻酸钙/生物玻璃材料上的增殖能力 CCK-8吸光度值结果显示:常氧条件下培养48 h后,5%全氟三丁胺组、10%全氟三丁胺组、20%全氟三丁胺组及空白对照组的CCK-8吸光度值为0.717±0.009,0.735± 0.035,0.755±0.031,0.737±0.009,各组间比较差异无显著性意义。缺氧条件培养48 h后,吸光度值分别为0.647±0.021,0.723±0.008,0.670±0.028,0.366± 0.031,见图3。缺氧条件下,含有全氟三丁胺各组吸光度值显著高于空白对照组。因此,全氟三丁胺/藻酸盐/生物玻璃材料能够促进缺氧状态下脂肪干细胞的增殖。在缺氧条件下,10%全氟三丁胺组与20%全氟三丁胺组的吸光度值差异无显著性意义。考虑材料应用因素,后续实验选用10%全氟三丁胺。"
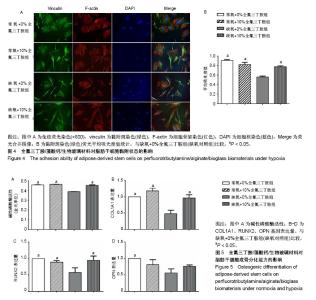
2.4 脂肪干细胞在全氟三丁胺/藻酸钙/生物玻璃材料上的黏附能力 培养48 h后,用黏附斑(绿色)、细胞骨架(红色)以及细胞核(蓝色)进行免疫荧光染色,黏附斑是位于细胞和细胞外基质之间接合的结构,可以显示脂肪干细胞的附着状态和伸展情况。荧光显微镜下可以观察到,常氧10%全氟三丁胺材料组、常氧对照组及缺氧10%全氟三丁胺材料组细胞骨架(红色)伸展良好,布满整个细胞,其黏附斑(绿色)荧光强度相近,在荧光合并图像中,可观察到黏附斑与细胞骨架连接紧密,显示出细胞的黏附状态良好,见图4A。进一步通过测定黏附斑的荧光强度来定量评价黏附状态,常氧10%全氟三丁胺材料组与常氧对照组黏附斑吸光度值为0.91±0.02及0.82±0.45,差异无显著性意义(P > 0.05)。在缺氧条件下,缺氧10%全氟三丁胺材料组黏附斑吸光度值为0.79±0.03,显著高于缺氧对照组(0.56±0.02,P < 0.05),见图4B。 2.5 全氟三丁胺/藻酸钙/生物玻璃材料对脂肪干细胞成骨分化能力的影响 应用碱性磷酸酶试剂盒检测各组脂肪干细胞的碱性磷酸酶活性。培养7 d,常氧条件下,常氧10%全氟三丁胺组与常氧对照组碱性磷酸酶活性比较差异无显著性意义,在缺氧条件下,缺氧10%全氟三丁胺组细胞碱性磷酸酶活性显著高于缺氧对照组(P < 0.05),见图5A。进一步通过RT-PCR检测OPN,RUNX2,COL1A1的基因转录水平。结果显示,常氧条件下,常氧10%全氟三丁胺组与常氧对照组3种基因的表达差异无显著性意义。缺氧条件下,缺氧10%全氟三丁胺组RUNX2、COL1A1基因表达量显著高于缺氧对照组(P < 0.05),两组OPN基因表达量比较差异无显著性意义,见图5B-D。"

| [1] Gajewski M, Wysieńska J, Wysieński L, et al. Tissue engineering in reconstructive surgery of bone and cartilage. Ortop Traumatol Rehabil. 2000;2(4):58-65.[2] Handschel J, Berr K, Depprich RA, Induction of osteogenic markers in differentially treated cultures of embryonic stem cells. Head Face Med. 2008;4:10.[3] Kahle M, Wiesmann HP, Berr K, et al. Embryonic stem cells induce ectopic bone formation in rats. Biomed Mater Eng. 2010;20(6): 371-380.[4] Cameron JA, Milner DJ, Lee JS, et al. Employing the biology of successful fracture repair to heal critical size bone defects. Curr Top Microbiol Immunol. 2013;367:113-132.[5] Lu C, Saless N, Wang X, et al. The role of oxygen during fracture healing. Bone. 2013;52(1):220-229.[6] King AT, Mulligan BJ, Lowe KC. Perfluorochemicals and Cell Culture. Nature Biotechnology. 1989; 7(10):1037-1042.[7] Luo Y, Wu C, Lode A, et al. Hierarchical mesoporous bioactive glass/alginate composite scaffolds fabricated by three-dimensional plotting for bone tissue engineering. Biofabrication. 2013;5(1):015005.[8] Yang Y, Ma T, Ge J, et al. Facilitated Neural Differentiation of Adipose Tissue-Derived Stem Cells by Electrical Stimulation and Nurr-1 Gene Transduction. Cell Transplant. 2016;25(6): 1177-1191.[9] 陈犹白,陈聪慧,Zhang Q,等. 脂肪干细胞分离、纯化和保存:研究进展与未来方向[J]. 中国组织工程研究, 2016, 20(10): 1508-1520.[10] Grayson WL, Zhao F, Izadpanah R, et al. Effects of hypoxia on human mesenchymal stem cell expansion and plasticity in 3D constructs. J Cell Physiol. 2006;207(2):331-339.[11] Malladi P, Xu Y, Chiou M, et al. Effect of reduced oxygen tension on chondrogenesis and osteogenesis in adipose-derived mesenchymal cells. Am J Physiol Cell Physiol. 2006;290(4): C1139-1146. [12] Sun Z, Luo B, Liu Z, et al. Effect of perfluorotributylamine- enriched alginate on nucleus pulposus cell: Implications for intervertebral disc regeneration. Biomaterials.2016;82:34-47.[13] Ma T, Zhu L, Yang Y, et al. Enhanced in vivo survival of Schwann cells by a synthetic oxygen carrier promotes sciatic nerve regeneration and functional recovery. J Tissue Eng Regen Med. 2018;12(1):e177-e189.[14] Allen AB, Zimmermann JA, Burnsed OA, et al. Environmental manipulation to promote stem cell survival in vivo: use of aggregation, oxygen carrier, and BMP-2 co-delivery strategies. Journal of Materials Chemistry B. 2016; 4(20):3594-3607.[15] Zhu S, Ge J, Wang Y, et al. A synthetic oxygen carrier-olfactory ensheathing cell composition system for the promotion of sciatic nerve regeneration. Biomaterials. 2014;35(5): 1450-1461.[16] Wang Y, Qi F, Zhu S, et al. A synthetic oxygen carrier in fibrin matrices promotes sciatic nerve regeneration in rats. Acta Biomater. 2013;9(7):7248-7263.[17] Ma T, Wang Y, Qi F, et al. The effect of synthetic oxygen carrier-enriched fibrin hydrogel on Schwann cells under hypoxia condition in vitro. Biomaterials. 2013;34(38): 10016-10027.[18] Benjamin S, Sheyn D, Ben-David S, et al. Oxygenated environment enhances both stem cell survival and osteogenic differentiation. Tissue Eng Part A. 2013;19(5-6):748-758.[19] Kimelman-Bleich N, Pelled G, Sheyn D, et al. The use of a synthetic oxygen carrier-enriched hydrogel to enhance mesenchymal stem cell-based bone formation in vivo. Biomaterials. 2009;30(27): 4639-4648.[20] Nomi M, Atala A, Coppi PD, et al. Principals of neovascularization for tissue engineering. Mol Aspects Med. 2002;23(6):463-483.[21] Mikos AG, Sarakinos G, Lyman MD, et al. Prevascularization of porous biodegradable polymers. Biotechnol Bioeng. 1993; 42(6):716-723.[22] Li H, Wijekoon A, Leipzig ND. Encapsulated neural stem cell neuronal differentiation in fluorinated methacrylamide chitosan hydrogels. Ann Biomed Eng. 2014;42(7):1456-1469.[23] Srinivasan S, Jayasree R, Chennazhi KP, et al. Biocompatible alginate/nano bioactive glass ceramic composite scaffolds for periodontal tissue regeneration. Carbohydrate Polymers. 2012; 87(1):274-283.[24] Bosetti M, Cannas M. The effect of bioactive glasses on bone marrow stromal cells differentiation. Biomaterials. 2005;26(18): 3873-3879.[25] Day RM, Boccaccini AR, Shurey S, et al. Assessment of polyglycolic acid mesh and bioactive glass for soft-tissue engineering scaffolds. Biomaterials. 2004;25(27):5857-5866.[26] Seidenstuecker M, Kerr L, Bernstein A, et al. 3D Powder Printed Bioglass and β-Tricalcium Phosphate Bone Scaffolds. Materials (Basel). 2017;11(1): E13.[27] Boulila S, Oudadesse H, Kallel R, et al. The performance of a scaffold bioglass–chitosan in the treatment of bone defect. Polymer Bulletin. 2018;75(12):5567-5586.[28] Jiang G, Evans ME, Jones IA, et al. Preparation of poly(epsilon-caprolactone)/continuous bioglass fibre composite using monomer transfer moulding for bone implant. Biomaterials. 2005;26(15):2281-2288.[29] Maquet V, Boccaccini AR, Pravata L, et al. Porous poly(alpha-hydroxyacid)/Bioglass composite scaffolds for bone tissue engineering. I: Preparation and in vitro characterisation. Biomaterials. 2004;25(18):4185-4194.[30] Boccaccini AR, Blaker JJ, Maquet V, et al. Preparation and characterisation of poly(lactide-co-glycolide) (PLGA) and PLGA/Bioglass®; composite tubular foam scaffolds for tissue engineering applications. Materials Science and Engineering: C. 2005;25(1):23-31.[31] Yu H, Peng J, Xu Y, et al. Bioglass Activated Skin Tissue Engineering Constructs for Wound Healing. ACS Appl Mater Interfaces. 2016;8(1):703-715.[32] Mancuso E, Bretcanu OA, Marshall M, et al. Novel bioglasses for bone tissue repair and regeneration: Effect of glass design on sintering ability, ion release and biocompatibility. Mater Des. 2017;129:239-248.[33] Vrouwenvelder WC, Groot CG, de Groot K. Histological and biochemical evaluation of osteoblasts cultured on bioactive glass, hydroxylapatite, titanium alloy, and stainless steel. J Biomed Mater Res. 1993;27(4):465-475.[34] Virolainen P, Heikkilä J, Yli-Urpo A, et al. Histomorphometric and molecular biologic comparison of bioactive glass granules and autogenous bone grafts in augmentation of bone defect healing. J Biomed Mater Res. 1997;35(1):9-17.[35] Lovelace TB, Mellonig JT, Meffert RM, et al. Clinical evaluation of bioactive glass in the treatment of periodontal osseous defects in humans. J Periodontol. 1998;69(9):1027-1035.[36] Zeng Q, Han Y, Li H, et al. Bioglass/alginate composite hydrogel beads as cell carriers for bone regeneration. J Biomed Mater Res B Appl Biomater. 2014;102(1):42-51.[37] Rottensteiner U, Sarker B, Heusinger D, et al. In vitro and in vivo Biocompatibility of Alginate Dialdehyde/Gelatin Hydrogels with and without Nanoscaled Bioactive Glass for Bone Tissue Engineering Applications. Materials (Basel). 2014;7(3): 1957-1974.[38] Mouriño V, Newby P, Boccaccini AR. Preparation and Characterization of Gallium Releasing 3-D Alginate Coated 45S5 Bioglass® Based Scaffolds for Bone Tissue Engineering. Advanced Engineering Materials. 2010; 12(7):B283-B291.[39] Arnett TR, Gibbons DC, Utting JC, et al. Hypoxia is a major stimulator of osteoclast formation and bone resorption. J Cell Physiol. 2003;196(1):2-8.[40] Nicolaije C, Koedam M, van Leeuwen JP. Decreased oxygen tension lowers reactive oxygen species and apoptosis and inhibits osteoblast matrix mineralization through changes in early osteoblast differentiation. J Cell Physiol. 2012;227(4): 1309-1318.[41] Potier E, Ferreira E, Andriamanalijaona R, et al. Hypoxia affects mesenchymal stromal cell osteogenic differentiation and angiogenic factor expression. Bone. 2007;40(4): 1078-1087. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [6] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [7] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [8] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [9] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [10] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [11] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [12] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [13] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [14] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [15] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||