Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (14): 2254-2260.doi: 10.3969/j.issn.2095-4344.1651
Previous Articles Next Articles
Antimicrobial modification for orthopedic metal implants: merits and demerits
Luo Jin1, 2, Lan Hai1, 2
- 1Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 2Affiliated Hospital of Chengdu University, Chengdu 610000, Sichuan Province, China
-
Received:2018-12-12 -
Contact:Lan Hai, Professor, Zunyi Medical University, Zunyi 563000, Guizhou Province, China; Affiliated Hospital of Chengdu University, Chengdu 610000, Sichuan Province, China -
About author:Luo Jin, Master candidate, Physician, Zunyi Medical University, Zunyi 563000, Guizhou Province, China; Affiliated Hospital of Chengdu University, Chengdu 610000, Sichuan Province, China -
Supported by:the Funded Project of Sichuan Provincial Scientific and Technological Bureau, No. 18YYJC0063 (to LH)
CLC Number:
Cite this article
Luo Jin, Lan Hai. Antimicrobial modification for orthopedic metal implants: merits and demerits[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(14): 2254-2260.
share this article

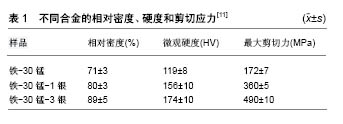
2.1 植入物自身的抗菌改性处理 目前主要通过不同金属相互混合的方法,在植入物中掺入具有抗菌活性的金属元素,起到抗菌作用。具有抗菌性能的新合金开发是目前的研究热点,已开发出具有抗菌性能的钛合金、不锈钢、镁合金、钴铬合金、锆基合金等众多体系。大量研究表明,铜离子及银离子具有良好的抗菌活性。这些合金的主要抗菌机制是通过添加具有抗菌活性的铜离子、银离子等元素,达到抗菌作用。此方法制备的抗菌合金具有抗菌持久、抗菌谱广、不容易耐药等优点,但不耐高温的抗菌活性物质无法添加,这些抗菌金属离子在体内的溶解释放速率及安全性仍有待进一步研究。Zhang等[9]采用粉末冶金法将铜元素加入纯钛中,制备了一种新型抗菌钛合金(Ti-Cu合金),经检测其对金黄色葡萄球菌及大肠埃希菌都具有较好的抗菌活性。但Ti-Cu合金仅对与合金接触的细菌有抗菌作用,说明其抗菌性能可能与铜离子的释放有关。Li等[10]在高纯度石墨坩埚中于六氟化硫SF6(体积分数1%)和CO2混合气体保护下对Mg和Cu进行熔化和混合,成功制造了Mg-Cu合金,抗菌性和组织相容性体内外评价结果表明,含质量分数0.25%Cu的Mg0.25Cu合金有治疗慢性骨感染的潜力。Sotoudehbagha等[11]采用机械合金化方法合成了新型Fe-30Mn-(1-3)Ag合金,见图2;并对其组织、力学性能、腐蚀速率、抗菌活性和细胞毒性进行了评价,结果表明,银质量分数为3%的合金具有最高的相对密度、抗剪强度、显微硬度和腐蚀速率,银质量分数为1%的合金具有较好的细胞毒性和抗菌活性,见表1。Zhang等[12]通过熔铸法在钴铬钼合金中添加铜元素,开发了具有抗菌性能的钴铬钼铜合金,发现与铜元素的加入使钴铬钼合金的腐蚀系数略有提高,对力学性能的影响有限,但对耐磨性有一定的影响,并且含铜合金对金黄色葡萄球菌有较强的抗菌能力。据推断,微量铜有助于增强抗菌活性,具有较强的抗菌性能和良好的耐蚀性,表明含铜钴合金作为一种植入物材料具有潜在的应用前景。 2.2 植入物表面抗菌改性处理 2.2.1 电化学改性 电化学改性是以溶液做电解液,沉积时主要发生化学反应。在含有H2PO4-和Ca2+的溶液中,以石墨棒做阳极,金属基体为阴极,适当调节 pH 值并控制好电极电位,通过溶液中电化学反应,正负电荷相互吸引的原理,将具有抗菌活性的物质(铜、银、锌、金等金属离子及壳聚糖等有机分子)沉积到植入体表面,从而起到抗菌作用。此方法成本较低易于实验室操作,涂层比例及浓度可调控,能够在不规则的外形表面沉积涂层,不需要经高温加热,是一种较常用的改性研究技术[13]。这些具有抗菌活性的金属离子与载体一同沉积在植入体表面,在短时间内具有良好的抗菌效果,抗菌谱广,不容易引起细菌耐药,但抗菌时间不够持久,涂层相对疏松,抗菌物质释放速率不够稳定。此方法制备的抗菌涂层,与基体的结合强度与抗菌载体关系密切,没有合金本身强度大。Hadidi等[14]通过电化学方法制备了羟基磷灰石-铜纳米复合材料,作为生物医学应用的抗菌涂层。Rasha等[15]以镍钛合金为基体制备了金纳米颗粒和壳聚糖复合涂层,以提高镍钛合金的生物相容性、生物稳定性、表面耐腐蚀性和抗菌效果,结果发现纳米尺度的金颗粒嵌入复合材料中,形成致密、厚实、光滑的涂层,电化学腐蚀测试结果表明,金纳米颗粒/壳聚糖复合镀层在不同浸泡时间和不同pH值下均能有效提高耐蚀性,说明涂层镍钛合金具有应用于骨科的潜力;此外,此生物复合涂层对细菌生长的抑制效果良好。Huang等[16]在工业纯钛(CP-Ti)上制备了锶/铜取代羟基磷灰石涂层,添加铜元素是为了提高羟基磷灰石的抗菌性能,添加锶元素是为了提高羟基磷灰石的生物相容性;锶/铜取代羟基磷灰石涂层在最初的几天内对大肠杆菌有一定程度的杀灭作用,这可能是由于涂层中的Cu取代;体外细胞实验可观察到成骨细胞黏附、增殖和碱性磷酸酶活性的增强,将有助于骨科材料的运用。 2.2.2 微弧氧化 微弧氧化法也是电化学改性的一种有效方法。Weng等[17]采用微弧氧化法制备了结构良好、附着力好的Al2O3涂层,提高了Al合金的耐磨性和耐腐蚀性,抑制了Al合金基体中有害Al离子的释放。分别将Ca、Fe、Zn掺杂到Al2O3涂层中,以提高其生物性能,发现掺杂Zn的Al2O3涂层具有优异的成骨性能,对大肠杆菌和金黄色葡萄球菌的抗菌率分别为(99.5±0.56)%和(98.77±0.52)%。Yao等[18]以含铜纳米粒子的电解质为载体,通过微弧氧化钛制备了掺杂Cu的二氧化钛抗菌涂层,可见铜纳米粒子嵌入到涂层中,并且该涂层对大肠杆菌和金黄色葡萄球菌均有良好的抗菌活性。 2.2.3 磁控溅射法 磁控溅射法利用2个直流电极,使通过电极间的气体电离而形成热等离子体,将所需涂层材料粉末送入等离子体中加热熔融,将该熔融物高速喷射在金属基体上,快速凝固而形成涂层[19]。此方法制备工艺简单,但制备的涂层材料需要能够耐高温,否则难以实现抗菌生物活性。Bai等[20]采用脉冲直流磁控溅射法制备了不同Ag含量(1.2%-1.6%)的纳米Ti-Ag涂层,随着银浓度的增加,涂层从密集的柱状晶体变为稀疏的柱状体,最终没有了柱状结构;最初Ti-Ag涂层能有效杀灭金黄色葡萄球菌,浸泡75 d后保持适度抗菌;通过调节Ti-Ag涂层中Ag的含量,可同时获得良好的抗菌活性和骨整合能力。Stranak等[21]通过等离子体辅助磁控溅射、双磁控溅射、双大功率脉冲磁控溅射3种方法制备了钛铜薄膜,采用原子吸收光谱法测定了金属钛铜薄膜中铜的含量,发现金属钛铜薄膜具有良好的杀菌效果,但对可产生生物膜细菌的杀灭作用不明显。 2.2.4 喷涂法 喷涂技术分为传统的热喷涂技术和冷喷涂2种,热喷涂分为爆炸喷涂、等离子喷涂、超速火焰喷涂等技术;冷喷涂是利用气体加速实现喷涂,其温度相对较低,可运用于不耐高温的材料。Joanna团队利用等离子热喷涂技术将羟基磷灰石涂层热喷涂在Ti6Al4V合金基体上,通过改变涂层中硝酸银浓度的浸渍和热处理获得了良好的抗菌性能,且对人体成骨细胞无细胞毒性作用[22]。Li等[23]于室温下在钛基板进行真空冷喷涂,合成了羟基磷灰石-庆大霉素硫酸盐复合粉体,该涂层具有良好的抗菌性,具有较长期的抗菌能力。Sivaraj等[24]利用一种新型的复合涂层喷涂方法成功制作了Mg取代羟基磷灰石/多壁碳纳米管复合涂层,认为Mg涂层可抑制植入物表面最初的细菌黏附,避免感染,进而促进骨整合。 2.2.5 化学转化技术 化学转化法制备涂层仿形能力强、成本低廉、工艺能耗少、容易操作,同时化学转化膜可提高涂层的结合强度,无需高温处理[25]。化学转化技术通过在植入体表面制备抗菌涂层,即添加抗菌物质而达到抗感染的目的。Hu等[26]通过化学转化处理在Mg-2Zn-1Mn-0.5Ca合金表面成功制备出了以Ca10(PO4)6(OH)2为主、少量Ag/Ag2CO3、CaHPO4•2H2O、CaSiO3的连续转化涂层,转化膜的形成过程表明,MgF2薄膜和阴极Ca2mg6Zn3相促进了Ag/羟基磷灰石的沉积,抗菌实验表明Ag/羟基磷灰石复合涂层具有良好的抗菌性能。Guo等[27]采用磷酸盐化学转化技术在纯钛上制备了Zn-Ca-PCC,抗菌研究结果表明Zn-Ca-PCC涂层对革兰阳性菌(金黄色葡萄球菌)和革兰阴性菌(铜绿假单胞菌)的生长均有明显抑制作用。 2.2.6 激光表面修饰 激光表面修饰又分为多种方法,例如飞秒激光表面处理、激光熔融、激光原位沉积等,这几种方法各自的抗菌机制完全不同。飞秒激光表面处理是通过改变植入物表面结构纹理,降低细菌的黏附和细菌生物膜的形成,达到抗菌目的。激光熔融与激光原位沉积是通过在植入物表面掺杂抗菌物质达到抗菌目的。Cunha等[28]采用中心波长为1 030 nm、脉冲持续时间为500 fs的Yb:KYW脉冲再生放大系统对植入物表面进行激光处理,制备了由激光诱导的表面结构和纳米粒子2种表面纹理,结果表明激光处理显著降低了植入物表面细菌的黏附及生物膜的形成。Chan等[29]采用近红外波长为1 064 nm的连续波光纤激光器,对商业纯Ti(2级)、Ti6Al4V(5级)和CoCrMo合金植入材料在含氮环境下进行表面激光处理,发现激光处理后的商业纯Ti和Ti6Al4V合金表面细菌黏附明显减少,这种特性是由于疏水性降低、氧化膜较厚且稳定,以及激光诱导纳米特性的存在而产生的;激光处理后的CoCrMo中没有发现类似的抗菌作用,其具体抗菌机制还有待进一步研究。激光熔融是用激光束将所要添加的粉体融化,同时将金属基体表面薄层熔化,2种物质熔融混合,从而构成冶金结合的一种表面处理技术[30]。Boutinguiza等[31]利用激光熔融技术一步法合成了纳米银粒子,并将其沉积在Ti基板上,发现含银Ti底物对唾液乳杆菌有较好的抗菌活性。Hidalgo-Robatto等[32]以脉冲激光沉积方法在金属上成功制备了富集氟的磷酸钙涂层,该涂层提高了细胞活力和抗菌性能。Vaithilingam等[33]提出了一种利用选择性激光熔融在 Ti6Al4V结构表面吸附膦酸自组装单分子膜固定环丙沙星(抗菌药物)的方法,涂层抗菌药物敏感性实验表明该药物释放后具有治疗活性。此方法展示了使用自组装单分子层修饰选择性激光熔融表面的治疗潜力。 2.2.7 等离子体注入技术 等离子体注入技术是指运用等离子体离子注入装置将不同的元素加速,再高速撞击植入物表面,在植入物表面发生一系列物理化学反应,最终通过该元素的释放或其合成的新化合物起到抗菌作用。此技术的优点是可较容易地控制掺杂物的浓度与植入深度[34]。 氧等离子体浸没注入:Yeung等[35]采用氧等离子体浸没离子注入对医用级钛合金进行抗菌改性,发现氧等离子体浸没离子注入产生的纳米TiO2层可显著抑制细菌黏附,同时保留原有的细胞相容性。纳米TiO2层在预防骨科种植体植入相关细菌感染方面具有广阔的应用前景。 氮等离子体浸没注入:Huang等[36]应用氮等离子体浸润离子注入技术在Ti6Al4V合金表面制备了氮化钛,并证明了氮化钛的存在可增强人骨髓间充质干细胞的黏附、增殖、矿化作用,且防止细菌的黏附。 其他等离子体浸没注入:Yu等[37]运用等离子体浸没离子注入沉积技术在Ti上建立了稳定的Mn离子释放平台,与蚀刻钛相比增加了抗革兰阴性菌的生存能力,增强了在含锰样品上间充质干细胞上的成骨相关基因表达。Tian等[38]利用等离子体浸没离子注入技术在氧化钛涂层表面和表面区域模拟了零价铁纳米颗粒和氧化铁纳米颗粒,这种新型体系在体外表现出铁释放、巨大的电子存储能力和优异的细胞相容性,该体系具有选择性的抗细菌生存能力,在涂层的下表面区域边界处,零价铁纳米颗粒可捕获电子,在黑暗中对金黄色葡萄球菌细胞造成氧化损伤,杀死金黄色葡萄球菌;由于大肠杆菌与底物之间缺乏电子传递能力,对大肠杆菌没有明显的杀灭作用。Echeverrigaray等[39]采用活性伏安式镀银设备通过低能量(4 keV)离子注入,对不锈钢表面银原子的抗菌性能进行了评价,结果发现其具有减少细菌黏附和生物膜形成、抗细菌定植表面的作用。Jin等[40]采用等离子体浸没离子注入技术将锌离子注入草酸腐蚀钛中,发现锌离子注入钛中锌的深度分布呈高斯分布,表面锌以ZnO的形式存在,内部锌以金属的形式存在,锌离子注入的钛能显著促进成骨细胞MC3T3-E1细胞的增殖,促进大鼠间充质干细胞的初始黏附、扩增活性、碱性磷酸酶活性、胶原分泌和细胞外基质矿化,对大肠杆菌和金黄色葡萄球菌均有一定的抗菌作用。 2.2.8 纳米球光刻技术 纳米球光刻技术是利用紫外光采用光刻手段在植入体上制作纳米级图形,通过改变植入体表面的湿润性达到抗细菌黏附的作用,由于其简单、有效、廉价等优点,在纳米制造中得到了广泛的应用。Jaggessar等[41]介绍了纳米球光刻技术在Ti6Al4V表面上的应用,利用直径1 µm和750 nm的纳米球测试了纳米球直径对纳米纹理的影响,在不同基体上成功地制备出了一致和均匀的阵列,Ti6Al4V基板表面可见纳米金字塔,但这些纳米金字塔不像玻璃那样均匀,需要进行更多的研究来搞清楚纳米球和基体材料之间的相互作用。通过进一步的调查和测试,认为纳米球光刻技术可能在产生抗菌表面、减少细菌在骨科植入物表面的黏附方面具有应用价值。 2.2.9 溶胶、凝胶法 溶胶、凝胶法是通过溶剂的水解反应生成溶胶或者凝胶,再用提拉法将植入物涂上该溶胶或者凝胶,最后再经过干燥和烧结后得到所需涂层,此法具有制备工艺简单、成本低、涂层浓度易于调节、容易操作等优点。此方法可通过在涂层中添加抗菌物质达到抗菌效果,但凝胶加热后容易产生裂痕,生成的抗菌涂层较疏松,并且凝胶与基体植入物结合强度较低等缺点。Palla-Rubio等[42]介绍了溶胶-凝胶法制备混合硅胶-壳聚糖涂料的方法,水解降解和硅释放研究表明,有效的硅释放具有促进骨骼形成的可能性;引入不同数量的壳聚糖和正硅酸四乙酯,可调节硅的释放;体外培养细胞表明,该复合涂层不具有细胞毒性,并能促进其表面的细胞增殖,其中含有5%-10%壳聚糖的涂料具有良好的抗菌性能。Keynoosh等[43]采用溶胶-凝胶法制备了含银、氟、羟基磷灰石(Ag-FHA)的复合涂层,分别用磷酸三乙酯、水合硝酸钙、氟化铵和硝酸银作为磷、钙、氟和银的前驱体,其中钙磷比为1.67、银的质量分数为0.3%,模拟体液电位动力学极化测量证实了含银、氟、羟基磷灰石涂层的腐蚀保护作用;大肠杆菌抗菌活性实验表明,随着氟化物含量的增加,含银、氟、羟基磷灰石纳米复合材料的抗菌性能显著提高,这些涂层可防止植入物的细菌感染。 2.2.10 水热法 水热法是在自然界中模拟矿化过程中晶体的生长,于一定温度和压力下,在封闭系统中使用水作为溶剂完成反应[44]。在水热条件下,水的性质,如蒸气压、密度、表面张力、黏度和离子产物将显著改变。水热法可明显降低体系的反应温度,制备高度结晶产品,晶粒小,纯度高。此外,水热法还存在设备要求高、反应时间长、能耗高等一系列缺点。此方法也是通过在涂层中添加无机抗菌物质而达到抗菌作用。但水热法对反应设备和条件要求较高,技术难度大,在实践中较难推广。Xiang等[45]采用水热法在钛金属植入物表面成功制备了聚(乳糖-乙醇酸)/银/氧化锌纳米棒涂层,并对聚(乳糖-乙醇酸)和纳米银混合物进行了自旋涂层,实验证明聚(乳酸-钴-乙醇酸)/银/氧化锌纳米棒涂层对金黄色葡萄球菌和大肠杆菌均有良好的抑菌效果。Mohandas等[46]通过一步热化学方法将纳米银加入到表面改性钛中,通过后续实验证明了此涂层具有抗菌活性和细胞相容性双重特性。 2.2.11 逐层组装 逐层组装膜主要是用带相反电荷基团的聚电解质之间层与层组装而构筑起来的膜[47],是一种构筑有机薄膜的有效方法,其抗菌活性也主要是通过在涂层中添加有机或者无机抗菌物质而实现。此方法可添加不耐受高温的抗菌物质,适用于有机物及无机离子,同时还可将多种抗菌物质共同添加进植入物涂层中,此法制备的涂层具有结合强度高、致密等优点。Zeng等[48]通过逐层组装方法在AZ31镁合金上合成了硫酸庆大霉素/聚(4-苯乙烯磺酸钠)复合涂层,所合成的复合涂层在镁合金基体上具有良好的耐蚀性和抗菌性能,使其作为医用植入物的镀层具有一定的吸引力。Tao等[49]采用逐层组装技术制备了一种由壳聚糖-1-(羟甲基)-5,5-二甲基氢氧根和明胶(凝胶)组成的生物活性涂层,通过对大肠杆菌(E. coli)和金黄色葡萄球菌(S. aureus)的抗菌实验发现,该复合涂层有效抑制了细菌的黏附和生长;同时体外细胞实验证实,该复合涂层对成骨细胞无明显毒性。此研究为抗菌钛基质的制备提供了一种很有前途的方法,可用于骨科应用。Zhao等[50]将银纳米颗粒和聚甲基三甲氧基硅烷通过逐层组装和硅氧烷自缩合反应引入AZ31镁合金中,用平板计数法测定了该薄膜对金黄色葡萄球菌的抗菌活性,发现复合镀层具有抗菌性能,可延长组装银离子的释放时间;由于聚硅氧烷的物理阻隔性和自愈性,这种表面形貌平滑且形貌单一的复合膜,能够提高镁合金的耐蚀性。 2.2.12 多种方法联合处理 抗菌修饰方法众多,各有优势,运用多种方法联合处理可相互补足,达到更好的效果。Hatada等[51]将等离子体离子注入和磁控溅射相结合,在不锈钢管内表面制备了低浓度Ag的类金刚石薄膜,在管内辅助电极的帮助下,用C2H4等离子体沉积了类金刚石膜,然后用该电极作为直流溅射靶,用气体Ar和C2H4的混合物溅射银,通过改变气流量比和工艺时间可改变薄膜的Ag含量,证明了含Ag类金刚石膜对金黄色葡萄球菌具有较好的抗菌活性。"

| [1] Berríos-Torres SI,Umscheid CA,Bratzler DW,et al. Centers for Disease Control and Prevention Guideline for the Prevention of Surgical Site Infection, 2017.JAMA Surg. 2017;152(8): 784-791.[2] Maradit Kremers H,Larson DR,Crowson CS,et al. Prevalence of Total Hip and Knee Replacement in the United States.J Bone Joint Surg Am.2015;97(17):1386-1397.[3] Antoci V,Chen AF, Parvizi J.7.9 Orthopedic Implant Use and Infection.Comprehensive Biomaterials II.2017;7:133-151.[4] Darouiche RO.Treatment of infections associated with surgical implants.N Engl J Med. 2004;350(14):1422-1429.[5] Trampuz A,Zimmerli W.Diagnosis and treatment of infections associated with fracture-fixation devices.Injury.2006;37 Suppl 2:S59-66.[6] Johnson CT,Wroe JA,Agarwal R,et al. Hydrogel delivery of lysostaphin eliminates orthopedic implant infection by Staphylococcus aureus and supports fracture healing.Proc Natl Acad Sci U S A. 2018;115(22):E4960-E4969.[7] Raphel J,Holodniy M,Goodman SB,et al.Multifunctional coatings to simultaneously promote osseointegration and prevent infection of orthopaedic implants.Biomaterials. 2016; 84:301-314.[8] Zhang R,Liu X,Xiong Z,et al.Novel Micro/nanostructured TiO2/ZnO Coating with Antibacterial Capacity and Cytocompatibility. Ceram Int.2018;44:9711-9719.[9] Zhang E,Li F,Wang H,et al.A new antibacterial titanium-copper sintered alloy: preparation and antibacterial property. Mater Sci Eng C Mater Biol Appl. 2013;33(7): 4280-4287.[10] Li Y,Liu L,Wan P,et al.Biodegradable Mg-Cu alloy implants with antibacterial activity for the treatment of osteomyelitis: In vitro and in vivo evaluations. Biomaterials.2016;106:250-263.[11] Sotoudehbagha P,Sheibani S,Khakbiz M,et al.Novel antibacterial biodegradable Fe-Mn-Ag alloys produced by mechanical alloying. Mater Sci Eng C Mater Biol Appl. 2018; 88:88-94.[12] Zhang E,Liu C.A new antibacterial Co-Cr-Mo-Cu alloy: Preparation, biocorrosion, mechanical and antibacterial property. Mater Sci Eng C Mater Biol Appl.2016;69:134-143.[13] 翁文剑,庄均珺,林素雅,等.电化学沉积生物功能涂层的研究进展[J].硅酸盐学报,2017,45(11):1539-1547.[14] Hadidi M,Bigham A,Saebnoori E,et al. Electrophoretic- deposited hydroxyapatite-copper nanocomposite as an antibacterial coating for biomedical applications. Surf Coat Technol.2017;321:171-179.[15] Rasha AA,Sahar AF,Nader E,et al.Improvement of corrosion resistance and antibacterial effect of NiTi orthopedic materials by chitosan and gold nanoparticles.Appl Surf Sci. 2014;292: 390-399.[16] Huang Y,Hao M,Nian X,et al.Strontium and copper co-substituted hydroxyapatite-based coatings with improved antibacterial activity and cytocompatibility fabricated by electrodeposition. Ceram Int.2016;42(10):11876-11888.[17] Weng Y,Liu H,Ji S,et al. A promising orthopedic implant material with enhanced osteogenic and antibacterial activity: Al2O3-coated aluminum alloy.App Surf Sci. 2018;457: 1025-1103.[18] Yao X, Zhang X, Wu H, et al. Microstructure and antibacterial properties of Cu-doped TiO 2, coating on titanium by micro-arc oxidation.Appl Surf Sci.2014;292(1):944-947.[19] 刘亚强,陈青清,李朋.磁控溅射法制备铝掺杂氧化锌薄膜研究进展[J].信阳师范学院学报(自然科学版),2017, 30(4):667-671.[20] Bai L,Hang R,Gao A, et al. Nanostructured titanium–silver coatings with good antibacterial activity and cytocompatibility fabricated by one-step magnetron sputtering.Appl Surf Sci. 2015;355:32-44.[21] Stranak V,Wulff H,Rebl H,et al.Deposition of thin titanium–copper films with antimicrobial effect by advanced magnetron sputtering methods.Mater Sci Eng C. 2011;31(7): 1512-1519.[22] Podporska-Carroll J,Quilty B,Devery R.Non-cytotoxic and antimicrobial plasma sprayed coatings for orthopaedic application. Mater Lett.2013;112(12):54-57.[23] Li D,Gong Y,Chen X,et al.Room-temperature deposition of hydroxyapatite/antibiotic composite coatings by vacuum cold spraying for antibacterial applications.Surf Coat Technol. 2017;330:87-91.[24] Sivaraj D,Vijayalakshmi K.Substantial effect of magnesium incorporation on hydroxyapatite/carbon nanotubes coatings on metallic implant surfaces for better anticorrosive protection and antibacterial ability.J Anal Appl Pyrol.2018;135:15-21.[25] 周游,姚颖悟,吴坚扎西,等.镁合金化学转化膜的研究进展[J].电镀与精饰,2013,35(5):15-18.[26] Hu G,Zeng L,Du H,et al.The formation mechanism and bio-corrosion properties of Ag/HAcomposite conversion coating on the extruded Mg-2Zn-1Mn-0.5Ca alloy for bone implant application.Surf Coat Technol. 2017;325:127-135.[27] Guo YY,Liu B,Hu BB,et al.Antibacterial activity and increased osteoblast cell functions of zinc calcium phosphate chemical conversion on titanium.Surf Coat Technol.2016;294:131-138.[28] Cunha A,Elie AM,Plawinski L,et al.Femtosecond laser surface texturing of titanium as a method to reduce the adhesion of Staphylococcus aureus and biofilm formation.Appl Surf Sci. 2016;360:485-493.[29] Chan CW,Carson L,Smith GC,et al.Enhancing the antibacterial performance of orthopaedic implant materials by fibre laser surface engineering.Appl Surf Sci.2017;404:67-81.[30] Weng F,Chen C,Yu H.Research statusof laser cladding on titanium and it alloys:A review.Mater Design.2014(58): 412-425.[31] Boutinguiza M,Fernández-Arias M,del Val J,et al.Synthesis and deposition of silver nanoparticles on cp Ti by laser ablation in open air for antibacterial effect in dental implants. Mater Lett. 2018;231:126-129.[32] Hidalgo-Robatto BM,Aguilera-Correa JJ,López-Álvarez M,et al. Fluor carbonated hydroxyapatite coatings by pulsed laser deposition to promote cell viability and antibacterial properties. Surf Coat Technol.2018;349:736-744.[33] Vaithilingam J,Kilsby S,Goodridge RD,et al.Immobilisation of an antibacterial drug to Ti6Al4V components fabricated using selective laser melting.Appl Surf Sci.2014;314(24):642-654.[34] Gao A,Hang R,Chu PK.Recent advances in anti-infection surfaces fabricated on biomedical implants by plasma-based technology.Surf Coat Technol.2017;312:2-6.[35] Yeung K,Wu S,Zhao Y, et al. Antimicrobial effects of oxygen plasma modified medical grade Tie6Ale4V alloy.Vacuum. 2013;89(1):271-279.[36] Huang HH,Shiau DK,Chen CS,et al.Nitrogen plasma immersion ion implantation treatment to enhance corrosion resistance, bone cell growth, and antibacterial adhesion of Ti-6Al-4V alloy in dental applications.Surf Coat Technol.2018. DOI: 10.1016/j.surfcoat.2018.06.023[37] Yu L,Tian Y,Qiao Y,et al.Mn-containing titanium surface with favorable osteogenic and antimicrobial functions synthesized by PIII&D. Colloids Surf B Biointerfaces.2017;152:376-384.[38] Tian Y,Cao H,Qiao Y,et al.Antibacterial activity and cytocompatibility of titanium oxide coating modified by iron ion implantation.Acta Biomaterialia.2014;10(10):4505-4517.[39] Echeverrigaray FG,Echeverrigaray S,Delamare APL.Antibacterial properties obtained by low-energy silver implantation in stainless steel surfaces.Surf Coat Technol. 2016;307:345-351.[40] Jin G,Cao H,Qiao Y,et al.Osteogenic activity and antibacterial effect of zinc ion implanted titanium. Colloids Surf B Biointerfaces.2014;117:158-165.[41] Jaggessar A,Yarlagadda P,Qiu TF,et al.Fabrication of Nano Pyramid Texture on Ti-6Al-4V Using Nanosphere Lithography. Mater Today: Proceedings. 2018;5(5): 11593-11600.[42] Palla-Rubio B,Araújo-Gomes N,Fernández-Gutiérrez M,et al.Synthesis and characterization of silica-chitosan hybrid materials as antibacterial coatings for titanium implants. Carbohydr Polym. 2019;203:331-341.[43] Keynoosh B,Abbasi KB,Abdollah A.Characterization of sol-gel derived silver/fluor-hydroxyapatite composite coatings on titanium substrate.Surf Coat Technol.2018;352:522-528.[44] Meng LY,Wang B,Ma MG,et al.The progress of microwave-assisted hydrothermal method in the synthesis of functional nanomaterials.Mater Today Chem.2016;1-2:63-83.[45] Xiang Y,Li J,Liu X,et al.Construction of poly(lactic-co-glycolic acid)/ZnO nanorods/Ag nanoparticles hybrid coating on Ti implants for enhanced antibacterial activity and biocompatibility. Mater Sci Eng C Mater Biol Appl. 2017;79:629-637.[46] Mohandas A,Krishnan AG,Biswas R,et al.Antibacterial and cytocompatible nanotextured Ti surface incorporating silver via single step hydrothermal processing.Mater Sci Eng C Mater Biol Appl. 2017;75:115-124.[47] 王海侨,文芳岱,李效玉.分子自组装技术及其在发光器件制备上的应用[J].高分子通报,2006,19(4):35-41.[48] Zeng RC,Liu LJ,Luo KJ,et al.In vitro corrosion and antibacterial properties of layer-by-layer assembled GS/PSS coating on AZ31 magnesium alloys.Trans Nonferrous Met Soc China. 2015; 25(12):4028-4039.[49] Tao B,Shen X,Yuan Z,et al.N-halamine-based multilayers on titanium substrates for antibacterial application. Colloids Surf B Biointerfaces. 2018;170:382-392.[50] Zhao Y,Shi L,Ji X,et al.Corrosion resistance and antibacterial properties of polysiloxane modified layer-by-layer assembled self-healing coating on magnesium alloy.J Colloid Interface Sci. 2018;526:43-50.[51] Hatada R,Flege S,Bobrich A,et al.Preparation of Ag-containing diamond-like carbon films on the interior surface of tubes by a combined method of plasma source ion implantation and DC sputtering. Appl Surf Sci. 2014;310(9): 257-261.[52] 刘振,张腾,尚剑.载银氧化锌涂层抗菌机制的研究进展[J].中华实验外科杂志,2018,35(7):1383-1387. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||