Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (13): 2113-2120.doi: 10.3969/j.issn.2095-4344.1645
Previous Articles Next Articles
Heterogeneity of periodontal ligament stem cells: current status and prospects
Feng Qihao, Shen Mengjie, Yang Kun, Liu Qi
- Affiliated Stomatological Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
Online:2019-05-08Published:2019-05-08 -
Contact:Liu Qi, MD, Professor, Affiliated Stomatological Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Feng Qihao, Master candidate, Affiliated Stomatological Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81760199 (to YK) and 81860196 (to LQ)
CLC Number:
Cite this article
Feng Qihao, Shen Mengjie, Yang Kun, Liu Qi. Heterogeneity of periodontal ligament stem cells: current status and prospects[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(13): 2113-2120.
share this article

2.1 牙周膜干细胞异质性概述 干细胞是未分化的细胞,其特征在于在单细胞水平上能够自我更新和分化以产生成熟的子代细胞[7],牙周膜干细胞是具有成体干细胞特征的细胞群,可自我再生,并具有产生一系列不同细胞类型(成骨细胞、脂肪细胞和软骨细胞)的能力[8]。2004年Seo等首次从人牙周膜中分离出与骨髓间充质干细胞相似特征的多能干细胞,称其为牙周膜衍生的间充质干细胞,其表达与骨髓间充质干细胞相似的表型标记物,主要特征是多能性、克隆形成能力、高增殖性以及表达干细胞标记如Stro-1和血管周细胞标记CD146,但与骨髓间充质干细胞相比,牙周膜干细胞还表达肌腱特有的转录因子Scleraxis[8-9]。此外,牙周膜干细胞具有与骨髓间充质干细胞相当的免疫调节能力,由于缺乏HLAⅡDR或T细胞共刺激分子(CD80和CD86),牙周膜干细胞具有低免疫原性[10]。牙周膜干细胞可能由干细胞和定向祖细胞的异质混合物组成,具有分化成定向细胞并再生功能组织的能力,在动物模型中,植入的牙周膜衍生间充质干细胞已显示出产生类似于天然牙周复合体——牙骨质/牙周膜样结构的能力[11]。牙周膜干细胞的亚群表达胚胎干细胞(Nanog,Oct4,Klf4和Sox2 )和神经嵴(Sox10,Slug,p75和Nestin)标记,这表明它们有可能分化为成骨、成软骨(来源于中胚层)和神经源(来自外胚层)谱系[12]。更重要的是,牙周膜干细胞可以分化成胰岛素样细胞,这意味着它们也可以分化成内胚层谱系[13]。分离和纯化牙周膜干细胞是鉴别亚群的常用方法,现行分选牙周膜干细胞亚群的主要方法是通过分选技术基于细胞表面标志物分选亚群;如Saito等[14]通过有限稀释法分离单个表达CD105阳性的牙周膜干细胞克隆,并对其在体外向成骨细胞/成牙骨质细胞表型分化的克隆簇进行鉴定,发现获得的6个CD105阳性牙周膜干细胞克隆簇,其中3个为高表达成骨细胞/成牙骨质细胞克隆簇,另外3个为不能体外产生矿化基质的克隆簇;Yoo等[15]利用免疫磁珠分选(MACS)表达SSEA-4阳性的牙周膜干细胞亚群;Alvarez等[16]运用免疫荧光活化细胞分选技术(FACS),采用CD51/CD140a、CD271和Stro-1/CD146三种表面标记物组合,从牙周膜干细胞中分离出不同的牙周膜干细胞亚群,并比较其各自的成骨、成软骨分化能力。通过ATP结合盒转运蛋白(ABC)具有外排Hoechst-33342染料的特性,使用免疫荧光活化细胞分选技术(FACS)分选侧群细胞和非侧群细胞[17]。在此基础上研究者试图通过寻找具有独特表型的标志物来甄别细胞亚群,如通过追溯干细胞来源从而选择表型标记物、鉴别细胞表面蛋白质组分、基因芯片筛选未知生物学标记,同位素示踪显示细胞体内定位情况。牙周膜细胞特异性亚群远远没有得到充分的描述,而且这些亚群细胞在牙周愈合过程中的作用也有待进一步阐明。 2.2 牙周膜干细胞亚群的不同来源 2.2.1 神经嵴来源 神经嵴来自外胚层,神经嵴细胞是一种瞬时多能干细胞群,最初通过表皮和神经外胚层之间的相互作用和来自中胚层的信号在二者的交界处出现[18。在神经系统发育过程中,神经嵴细胞从神经上皮的背侧区域分层,经历上皮-间充质转化;然后它们开始迁移到胚胎中的目的地并产生各种类型的细胞[19],体内和体外实验表明神经嵴细胞产生多种细胞类型,包括脂肪细胞、神经元、平滑肌、软骨细胞、成骨细胞、成牙本质细胞、黑色素细胞[20]。牙齿发育是一个漫长而复杂的生物过程,受上皮和神经嵴衍生间充质之间相互作用的调节[21],在胚胎第6周牙齿开始发育,根据形态变化,牙齿发育通常分为萌芽期、帽状期和钟状期,从萌芽到帽状期阶段,牙源性间充质产生两种不同的细胞系:牙乳头细胞和牙囊细胞[22]。牙囊细胞被认为在牙周组织发育过程中分化为牙周膜成纤维细胞、成骨细胞和成牙骨质细胞的前体细胞。因此,可以认为牙周膜干细胞是来源于外胚层神经嵴分化而来的细胞,其前体细胞是牙囊细胞[23-24]。一项来自动物胚胎追踪实验的证据表明,牙齿组织是颅面神经嵴迁移细胞的归巢部位之一[25]。Ramírez-García等[26]将牙周膜干细胞在含有少量胎牛血清(体积分数为0.5%或1%)或缺乏胎牛血清的培养基中培养,发现越来越多的上皮样细胞共表达神经嵴特异性标志(p75,HNK-1,SOX10)、上皮形成标志(E-钙黏蛋白)和间充质细胞标志(CD73和CD105),证实了人牙周膜干细胞包含休眠神经嵴样细胞亚群,也间接证实了口腔多能干细胞是颅面神经嵴迁移细胞的后代。 2.2.2 周细胞来源 Iwasaki等[27]发现位于牙周韧带血管壁周围的牙周膜干细胞在形态学、分化潜能、细胞表型(周细胞相关标志物CD146,NG2和CD140B的表达)和形成毛细血管样的能力与周细胞具有相似的特性。最近Bae等[28]研究发现牙周膜干细胞可能是血管周围细胞的潜在来源,除表达典型的间充质特征外还表达血管周细胞标志物,如NG2、α-平滑肌肌动蛋白、血小板衍生生长因子受体β和CD146,通过体内实验检测血管生成,证实了牙周膜干细胞的体内血管生成潜力。研究表明从多种血管周围组织(脑、肺、肝、肾、肌肉、皮肤和骨髓)获得的间充质干细胞也表现出周细胞特征[29]。因此,牙周膜干细胞表达周细胞特征可能跟驻留的牙周组织位于血管周围的解剖位置有关,Roguljic等[30]通过同位素示踪技术定位牙周干细胞来源也间接证明了这点。 2.2.3 造血细胞来源 在最新的一项研究中,Wilson等[31]构造了一种独特的转基因模型,其中所有的造血细胞都表达绿色荧光蛋白(GFP+),通过体外验证了CD45+GFP与牙周膜干细胞、牙髓干细胞、牙槽骨来源的骨髓干细胞标志物共表达,还在体内实验证明了牙周组织具有造血起源,造血干细胞可以分化成牙周膜组织、牙髓、牙槽骨中存在的驻留细胞。 2.3 牙周膜干细胞特征及其影响因素 2.3.1 牙周膜干细胞的鉴定 通常描述间充质干细胞具有以下几点特征:①在标准培养条件下保持塑性黏附;②表达CD105、CD73和CD90,缺乏CD45、CD34、CD14或CD11b、CD79a或CD19和HLA-DR的表达;③体外多系分化潜能[32-33]。牙周膜干细胞作为间充质干细胞的一种,满足间充干细胞特征,但缺乏特异性标志物,为了鉴定牙周膜干细胞通常通过以下几点确认:形态学观察、细胞免疫化学鉴定、细胞增殖能力检测、多向分化潜能检测及细胞表型分析[34]。 2.3.2 分离方法对牙周膜干细胞特征的影响 常规用于分离牙周膜干细胞的方法主要有组织块法(OG)和酶消化法(EZ),见图2。酶消化法是从原始组织中获得单细胞悬液的常用方法,该方法将组织暴露于消化酶中一段最短的时间,以保持最大的细胞活力。与酶消化法相比,组织块方法在原代培养期间相当简单和方便,只要将组织碎片直接放入培养板中,适当添加少量培养基,使细胞从组织块中长出[35-36]。Tran等[37]通过组织块法和酶解离方法分别培养牙周膜干细胞,发现通过两种方法获得的牙周膜干细胞在低密度培养时能够形成贴壁的成纤维细胞克隆形成簇,而且在细胞表型和多系分化能力方面二者并无区别。研究人员在此基础上通过组织块法和酶消化法提取来源于不同供体的牙周膜干细胞,并深入比较了两种方式提取的细胞在免疫表型和多系分化能力方面的具体差别,发现典型周细胞标记物CD140b,CD146和NG2的表达在组织块法-牙周膜干细胞中显著高于酶消化法-牙周膜干细胞。实时荧光定量PCR进行基因表达分析和免疫荧光标记,结果表明组织块法-牙周膜干细胞呈现出独特的成牙骨质样性质并且在体内形成牙骨质样硬组织[38]。有趣的是通过对比来源于单个供体以两种方式提取的细胞,发现间充质干细胞标记物表达和成纤维细胞集落形成能力是相似的。在第3天和第5天,酶消化法-牙周膜干细胞的增殖能力更强,两种方法均有多系分化能力,并且酶消化法-牙周膜干细胞的矿化能力强于组织块法-牙周膜干细胞[39]。"
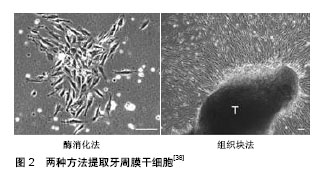
2.3.3 培养方式和条件对牙周膜干细胞特征的影响 牙周膜干细胞一般以二维单层细胞的方式在37 ℃,体积分数为5%CO2条件下培养[40]。Moritani等[41]利用标记有聚乙二醇(PEG)的微孔芯片使牙周膜干细胞形成球状细胞簇,让单层培养的牙周膜干细胞变成三维立体结构。通过对比单层细胞和球状细胞的干细胞特征,发现牙周膜干细胞球体表达与单层细胞相似的间充质干细胞标记,体内体外实验表明牙周膜干细胞球体的矿化和成骨能力显著强于单层细胞。相比于正常培养条件,缺氧似乎有助于维持牙周膜干细胞的多能性,Zhou等[42]发现牙周膜干细胞的分化潜能和多能性标志物(Oct-4、Sox-2和c-Myc)的表达在体积分数为2%氧气培养下显著增加,此外,激活p38和ERK 1/2信号通路,在缺氧(体积分数为2%氧气)条件下可以促进牙周膜干细胞的成骨潜能[43]。Choi等[44]也证实在缺氧条件下激活缺氧诱导因子1,能够增加牙周膜干细胞中牙本质蛋白1的表达和矿化以及促进体外成牙骨质发生。在最近的一项研究中Murabayashi等[45]通过设定缺氧和含氧量正常两种条件,改变培养的微环境(无血清和含血清培养基),比较牙周膜干细胞的特性。结果表明,在无血清培养基中培养不会影响牙周膜干细胞的间充质干细胞表型,并一步证明了缺氧不会改变无血清条件下牙周膜干细胞的生长;缺氧抑制牙周膜干细胞成骨和成脂分化,但无论血清含量如何,都能维持其多系分化潜能;牙周膜干细胞可能在无血清培养条件下被广泛的外源细胞毒性刺激所破坏。 2.3.4 旁分泌效应对牙周膜干细胞特征的影响 间充质干细胞不仅可以直接分化为不同的细胞类型,还可以分泌具有趋化性、有丝分裂和分化调节作用的营养因子,这些旁分泌因子被认为是间充质干细胞介导组织修复的主要机制[46]。移植的牙周膜干细胞不仅通过分化为不同类型的细胞直接参与骨组织修复,还通过分泌营养因子募集骨祖细胞,这可能是促进牙槽骨再生的主要机制[47]。研究者利用Transwell系统允许可溶性分子在分离的小室之间扩散同时阻断细胞与细胞的接触,以此模拟体内的自然环境来促进最佳的细胞表征,如Wu等[48]通过血管内皮细胞与牙周膜干细胞共培养研究血管内皮细胞介导下的旁分泌效应对牙周膜干细胞成骨分化能力的影响,发现与单独培养的牙周膜干细胞相比,共培养的牙周膜干细胞成骨分化进一步增强;Chen等[49]利用共培养系统将牙周膜干细胞与前成骨细胞MC3T3-E1或破骨细胞前体细胞RAW264.7共培养,结果表明牙周膜干细胞刺激增强了MC3T3-E1细胞成骨分化和RAW264.7细胞破骨细胞分化。另一种模拟旁分泌效应的方式是构建干细胞衍生的条件培养基,Kim等[50]将永生人牙周膜干细胞和脱落乳牙干细胞在不含胎牛血清的培养基中培养,通过收集上清液过滤浓缩的方式制备条件培养基,比较二者条件培养基的分泌蛋白,发现人牙周膜干细胞条件培养基更适合用于再生疗法的应用。Nagata等[51]研究也证实从牙周膜干细胞获取的条件培养基通过移植到手术创建的大鼠牙周缺损中,能够以浓度依赖的方式增强牙周再生作用。值得注意的是Xia等[52]通过构建来源于健康牙周膜干细胞(H-PDLSCs)和“发炎”牙周膜干细胞(I-PDLSCs)的条件培养基,并将两种牙周膜干细胞分别在两种条件培养基中培养,发现在骨诱导条件下于健康牙周膜干细胞条件培养基中孵育后,“发炎”牙周膜干细胞的成骨分化能力被部分拯救,这表明了细胞外微环境的改变对牙周膜干细胞功能的直接作用。 2.3.5 表观遗传修饰对牙周膜干细胞特征的影响 表观遗传修饰是通过DNA和相关蛋白的化学改变,以此导致染色质的重塑和基因转录的激活或失活,其主要方式是DNA甲基化、组蛋白乙酰化以及miRNA干扰等[53]。牙周膜干细胞的特征在于细胞的可塑性,然而负责这一点的潜在机制不明,Huynh等[54]发现组蛋白去乙酰化酶(HDACs)抑制人牙周膜干细胞成骨分化,提出表观遗传修饰可能是这一潜在机制。Gay等[55]比对765种miRNA,在牙周膜干细胞中发生了miRNA表达的变化,证实了牙周膜干细胞矿化组织的分化与hsa-mir-218表达的降低相关。在最近的研究中Diomede等[56]使用牙龈卟啉单胞菌脂多糖刺激牙周膜干细胞,发现在脂多糖处理组中显著抑制了牙周膜干细胞中DNMT1的表达,而DNA(胞嘧啶-5)甲基转移酶1(DNMT1)是控制DNA甲基化的关键控制因子。与此同时Sun小组[57]探讨使用Osthole(蛇床子素)是否可以通过表观遗传修饰恢复“炎性”牙周膜干细胞的成骨分化缺陷,结果表明Osthole上调“炎性”牙周膜干细胞成骨分化的关键调节因子组蛋白H3赖氨酸14(H3K14)并在牙周炎动物模型中改善了骨形成。 2.3.6 牙周膜干细胞异质性群体与亚群的特征比较 一般来说异质性的牙周膜干细胞在体内外研究中均能取得良好的效果[58],由于大多数研究是用表达多种间充质标志物的混合细胞群进行的,因此不清楚哪些细胞在牙周组织再生中起关键作用[590]。然而早期的研究证实了牙周膜干细胞中不同的细胞系具有不同的功能[60]。此后有越来越多的证据表明,相对于异质性群体,利用亚群形成特定的功能组织是更好的方法,如Fujii等[61]分离永生牙周膜干细胞获得3个克隆细胞系(第1-4,1-11和1-24细胞系),在动物实验中发现唯有1-11细胞系在移植物β-磷酸三钙表面形成牙周膜样结构;Ninomiya等[62]通过侧群细胞分选比较亚群与异质性群体在体内体外的分化能力,结果表明牙周膜干细胞的侧群细胞相对于非侧群和未分选的牙周膜干细胞具有更高的成骨潜力。然而缺乏特异性标记物识别牙周膜干性细胞群,以及无法确定标记物标识的细胞亚群的具体功能是制约亚群在体内移植应用的难点之一[63-64]。 2.3.7 牙周膜干细胞表面标志物 迄今为止间充质干细胞标记大致分为两类:单一标记和干性标记,见表1。干性标记能够识别具有成纤维细胞形成能力和多系分化潜力的间充质干细胞亚群,甚至是识别胚胎干细胞样群体,而单一标记可以从其体内环境中鉴定或纯化间充质样细胞, 基于两种不同类型标记物的性质,单一标记物通常高度表达,而干性标记物可以适度被检测[65]。Fortino等[66]通过免疫荧光检测间接发现牙周膜干细胞表达胚胎干细胞标记物Oct-4、Nanog和中胚层标记物波形蛋白,其中Nanog低表达。Li等[67]通过流式细胞术分析牙周膜干细胞中间充质干细胞标记物CD73、CD90高表达。Zhu小组[68]发现CD146(+)牙周膜干细胞与CD146(-)牙周膜干细胞相比显示出更显著的集落形成效率,更快的增殖速率以及更强的成骨潜能。源自人牙周膜干细胞的SSEA-4阳性克隆细胞具有成脂、成骨和成软骨形成潜力[69]。Alvarez等[17]也证实CD271(+)牙周膜干细胞通过强烈诱导成骨标志物如DLX5,RUNX2和BGLAP,显示出最大的成骨潜能。此外一些新的候选标记物也开始被使用,如Szepesi等[70]使用基于抗体的分选方法来富集表达ABCG2(ATP结合盒亚家族G成员2)的牙周膜干细胞亚群,表明ABCG2是一种可选择的标记物,可以增强亚群细胞的多向分化潜能,然而抗体的叠加效应无法做到单独识别具体的细胞亚群,如Torii等[71]从正常和永生牙周膜干细胞分离的SSEA3/CD44/CD105阳性细胞群具有牙骨质发生能力,而Gauthier等[72]通过比较3组STRO-1(+)/CD146(+),STRO-1(+)/CD146(+)和STRO-1(-)/CD146(-)牙周膜干细胞亚群,发现成牙骨质标记牙骨质附着蛋白的表达要比原始细胞略高。Yan等[73]将扩增后的STRO-1(+)细胞与未分选的亲代细胞比较,结果发现在扩增过程中,STRO-1(+)细胞的百分比从95.3%显著下降至2.3%。此外,经过4次体外扩增,扩增的STRO-1分选细胞和未分选的亲代细胞之间没有观察到显著的DNA含量、克隆形成差异。这说明体外培养不可避免地改变了干细胞的表面标志物表达及其生物学特性,特别是标记物的特性会随着时间的推移而减弱[74]。"
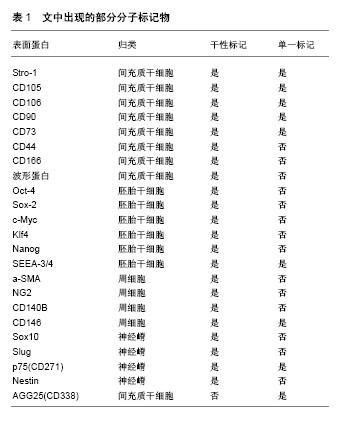
| [1] Zhai Q, Dong Z, Wang W, et al. Dental stem cell and dental tissue regeneration. Front Med. 2018 Jul 4. doi: 10.1007/s11684-018-0628-x. [Epub ahead of print][2] Gaubys A, Papeckys V, Pranskunas M. Use of Autologous Stem Cells for the Regeneration of Periodontal Defects in Animal Studies: a Systematic Review and Meta-Analysis. J Oral Maxillofac Res. 2018;9(2):e3.[3] Botelho J, Cavacas MA, Machado V, et al. Dental stem cells: recent progresses in tissue engineering and regenerative medicine. Ann Med. 2017;49(8):644-651.[4] Hart DA. Why Mesenchymal Stem/Progenitor Cell Heterogeneity in Specific Environments? —Implications for Tissue Engineering Applications Following Injury or Degeneration of Connective Tissues. J Biomedical Science and Engineering. 2014; 7: 526-532. [5] Thomas GV, Thomas NG, John S, et al. The Scope of Stem Cells in Periodontal Regeneration. J Dent Oral DisordTher. 2015;3(2): 1-9.[6] Chen FM, Gao LN, Tian BM, et al. Treatment of periodontal intrabony defects using autologous periodontal ligament stem cells: a randomized clinical trial. Stem Cell Res Ther. 2016; 7:33.[7] Bassir SH, Wisitrasameewong W, Raanan J, et al. Potential for Stem Cell-Based Periodontal Therapy. J Cell Physiol. 2016; 231(1):50-61.[8] Menicanin D, Mrozik KM, Wada N, et al. Periodontal-ligament- derived stem cells exhibit the capacity for long-term survival, self-renewal, and regeneration of multiple tissue types in vivo. Stem Cells Dev. 2014;23(9):1001-1011.[9] Seo BM, Miura M, Gronthos S, et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet. 2004;364(9429):149-155.[10] Wada N, Maeda H, Hasegawa D, et al. Semaphorin 3A induces mesenchymal-stem-like properties in human periodontal ligament cells. Stem Cells Dev. 2014;23(18): 2225-2236.[11] Zhang H, Liu S, Zhu B, et al. Composite cell sheet for periodontal regeneration: crosstalk between different types of MSCs in cell sheet facilitates complex periodontal-like tissue regeneration. Stem Cell Res Ther. 2016;7(1):168.[12] Mayo V, Sawatari Y, Huang CY, et al. Neural crest-derived dental stem cells--where we are and where we are going. J Dent. 2014; 42(9):1043-1051.[13] Lee JS, An SY, Kwon IK, et al. Transdifferentiation of human periodontal ligament stem cells into pancreatic cell lineage. Cell Biochem Funct. 2014;32(7):605-611.[14] Saito MT, Salmon CR, Amorim BR, et al. Characterization of highly osteoblast/cementoblast cell clones from a CD105-enriched periodontal ligament progenitor cell population. J Periodontol. 2014;85(6):e205-211.[15] Yoo JH, Lee SM, Bae MK, et al. Effect of orthodontic forces on the osteogenic differentiation of human periodontal ligament stem cells. J Oral Sci. 2018;60(3):438-445.[16] Alvarez R, Lee HL, Wang CY, et al. Characterization of the osteogenic potential of mesenchymal stem cells from human periodontal ligament based on cell surface markers. Int J Oral Sci. 2015;7(4):213-219.[17] Hara ES, Ono M, Eguchi T, et al. miRNA-720 controls stem cell phenotype, proliferation and differentiation of human dental pulp cells. PLoS One. 2013;8(12):e83545.[18] Tomokiyo A, Hynes K, Ng J, et al. Generation of Neural Crest-Like Cells From Human Periodontal Ligament Cell-Derived Induced Pluripotent Stem Cells. J Cell Physiol. 2017;232(2):402-416.[19] La Noce M, Mele L, Tirino V, et al. Neural crest stem cell population in craniomaxillofacial development and tissue repair. Eur Cell Mater. 2014;28:348-357.[20] Zhu Y, Zhang P, Gu RL, et al. Origin and Clinical Applications of Neural Crest-Derived Dental Stem Cells. Chin J Dent Res. 2018; 21(2):89-100.[21] Balic A, Thesleff I. Tissue Interactions Regulating Tooth Development and Renewal. Curr Top Dev Biol. 2015;115: 157-186.[22] Balic A. Biology Explaining Tooth Repair and Regeneration: A Mini-Review. Gerontology. 2018;64(4):382-388.[23] Martens W, Bronckaers A, Politis C, et al. Dental stem cells and their promising role in neural regeneration: an update. Clin Oral Investig. 2013;17(9):1969-1983.[24] Lima RL, Holanda-Afonso RC, Moura-Neto V, et al. Human dental follicle cells express embryonic, mesenchymal and neural stem cells markers. Arch Oral Biol. 2017;73:121-128.[25] Baggiolini A, Varum S, Mateos JM, et al. Premigratory and migratory neural crest cells are multipotent in vivo. Cell Stem Cell. 2015;16(3):314-322.[26] Ramirez-Garcia L, Cevallos R, Gazarian K. Unveiling and initial characterization of neural crest-like cells in mesenchymal populations from the human periodontal ligament. J Periodontal Res. 2017;52(3):609-616.[27] Iwasaki K, Komaki M, Yokoyama N, et al. Periodontal ligament stem cells possess the characteristics of pericytes. J Periodontol. 2013;84(10):1425-1433.[28] Bae YK, Kim GH, Lee JC, et al. The Significance of SDF-1α- CXCR4 Axis in in vivo Angiogenic Ability of Human Periodontal Ligament Stem Cells. Mol Cells. 2017;40(6): 386-392.[29] De Souza LE, Malta TM, Kashima Haddad S, et al. Mesenchymal Stem Cells and Pericytes: To What Extent Are They Related. Stem Cells Dev. 2016;25(24):1843-1852.[30] Roguljic H, Matthews BG, Yang W, et al. In vivo identification of periodontal progenitor cells. J Dent Res. 2013;92(8): 709-715.[31] Wilson KR, Kang IH, Baliga U, et al. Hematopoietic Stem Cells as a Novel Source of Dental Tissue Cells. Sci Rep. 2018; 8(1):8026.[32] Nakajima R, Ono M, Hara ES, et al. Mesenchymal stem/progenitor cell isolation from tooth extraction sockets. J Dent Res. 2014;93(11):1133-1140.[33] 李秋晨,肖苒. 人间充质干细胞表面标记分子的研究进展[J]. 组织工程与重建外科杂志, 2013,9(4):229-231.[34] 王冠华. 牙周膜干细胞培养及鉴定方法的研究进展[J]. 医学理论与实践, 2014,27(7):869-870.[35] Tamaki Y, Nakahara T, Ishikawa H, et al. In vitro analysis of mesenchymal stem cells derived from human teeth and bone marrow. Odontology. 2013;101(2):121-132.[36] Tominaga N, Nakahara T, Nasu M, et al. Isolation and characterization of epithelial and myogenic cells by "fishing" for the morphologically distinct cell types in rat primary periodontal ligament cultures. Differentiation. 2013;85(3): 91-100.[37] Tran Hle B, Doan VN, Le HT, et al. Various methods for isolation of multipotent human periodontal ligament cells for regenerative medicine. In Vitro Cell Dev Biol Anim. 2014; 50(7):597-602.[38] Shinagawa-Ohama R, Mochizuki M, Tamaki Y, et al. Heterogeneous Human Periodontal Ligament-Committed Progenitor and Stem Cell Populations Exhibit a Unique Cementogenic Property Under In Vitro and In Vivo Conditions. Stem Cells Dev. 2017;26(9):632-645.[39] Prateeptongkum E, Klingelhoffer C, Müller S, et al. Characterization of progenitor cells and stem cells from the periodontal ligament tissue derived from a single person. J Periodontal Res. 2016;51(2):265-272.[40] Mrozik K, Gronthos S, Shi S, et al. A Method to Isolate, Purify, and Characterize Human Periodontal Ligament Stem Cells. Methods Mol Biol. 2017;1537:413-427.[41] Moritani Y, Usui M, Sano K, et al. Spheroid culture enhances osteogenic potential of periodontal ligament mesenchymal stem cells. J Periodontal Res. 2018;53(5):870-882.[42] Zhou Y, Fan W, Xiao Y. The effect of hypoxia on the stemness and differentiation capacity of PDLC and DPC. Biomed Res Int. 2014; 2014:890675.[43] Wu Y, Yang Y, Yang P, et al. The osteogenic differentiation of PDLSCs is mediated through MEK/ERK and p38 MAPK signalling under hypoxia. Arch Oral Biol. 2013;58(10): 1357-1368.[44] Choi H, Jin H, Kim JY, et al. Hypoxia promotes CEMP1 expression and induces cementoblastic differentiation of human dental stem cells in an HIF-1-dependent manner. Tissue Eng Part A. 2014;20(1-2):410-423.[45] Murabayashi D, Mochizuki M, Tamaki Y, et al. Practical methods for handling human periodontal ligament stem cells in serum-free and serum-containing culture conditions under hypoxia: implications for regenerative medicine. Hum Cell. 2017;30(3): 169-180.[46] Liu J, Wang L, Liu W, et al. Dental follicle cells rescue the regenerative capacity of periodontal ligament stem cells in an inflammatory microenvironment. PLoS One. 2014;9(9): e108752.[47] Muhammad SA, Nordin N, Fakurazi S. Regenerative potential of secretome from dental stem cells: a systematic review of preclinical studies. Rev Neurosci. 2018;29(3):321-332.[48] Wu Y, Cao H, Yang Y, et al. Effects of vascular endothelial cells on osteogenic differentiation of noncontact co-cultured periodontal ligament stem cells under hypoxia. J Periodontal Res. 2013; 48(1):52-65.[49] Chen S, Ye X, Yu X, et al. Co-culture with periodontal ligament stem cells enhanced osteoblastic differentiation of MC3T3-E1 cells and osteoclastic differentiation of RAW264.7 cells. Int J Clin Exp Pathol. 2015;8(11):14596-14607.[50] Kim K, Jeon M, Lee HS, et al. Comparative analysis of secretory factors from permanent- and deciduous-teeth periodontal ligament cells. Arch Oral Biol. 2016;71:65-79.[51] Nagata M, Iwasaki K, Akazawa K, et al. Conditioned Medium from Periodontal Ligament Stem Cells Enhances Periodontal Regeneration. Tissue Eng Part A. 2017;23(9-10):367-377.[52] Xia Y, Tang HN, Wu RX, et al. Cell Responses to Conditioned Media Produced by Patient-Matched Stem Cells Derived From Healthy and Inflamed Periodontal Ligament Tissues. J Periodontol. 2016;87(5):e53-63.[53] Martins MD, Jiao Y, Larsson L, et al. Epigenetic Modifications of Histones in Periodontal Disease. J Dent Res. 2016;95(2): 215-222.[54] Huynh NC, Everts V, Pavasant P, et al. Inhibition of Histone Deacetylases Enhances the Osteogenic Differentiation of Human Periodontal Ligament Cells. J Cell Biochem. 2016; 117(6): 1384-1395.[55] Gay I, Cavender A, Peto D, et al. Differentiation of human dental stem cells reveals a role for microRNA-218. J Periodontal Res.2014;49(1):110-120.[56] Diomede F, Thangavelu SR, Merciaro I, et al. Porphyromonas gingivalis lipopolysaccharide stimulation in human periodontal ligament stem cells: role of epigenetic modifications to the inflammation. Eur J Histochem. 2017;61(3):2826.[57] Sun J, Dong Z, Zhang Y, et al. Osthole improves function of periodontitis periodontal ligament stem cells via epigenetic modification in cell sheets engineering. Sci Rep. 2017;7(1): 5254.[58] Zhang C, Yan B, Cui Z, et al. Bone regeneration in minipigs by intrafibrillarly-mineralized collagen loaded with autologous periodontal ligament stem cells. Sci Rep. 2017;7(1):10519.[59] Kadkhoda Z, Rafiei SC, Azizi B, et al. Assessment of Surface Markers Derived from Human Periodontal Ligament Stem Cells: An In Vitro Study. J Dent (Tehran). 2016;13(5):325-332.[60] Gay IC, Chen S, MacDougall M. Isolation and characterization of multipotent human periodontal ligament stem cells. Orthod Craniofac Res. 2007;10(3):149-160.[61] Fujii S, Maeda H, Wada N, et al. Investigating a clonal human periodontal ligament progenitor/stem cell line in vitro and in vivo. J Cell Physiol. 2008;215(3):743-749.[62] Ninomiya T, Hiraga T, Hosoya A, et al. Enhanced bone-forming activity of side population cells in the periodontal ligament. Cell Transplant. 2014;23(6):691-701.[63] Avinash K, Malaippan S, Dooraiswamy JN. Methods of Isolation and Characterization of Stem Cells from Different Regions of Oral Cavity Using Markers: A Systematic Review. Int J Stem Cells. 2017;10(1):12-20.[64] Yuan X, Pei X, Zhao Y, et al. A Wnt-Responsive PDL Population Effectuates Extraction Socket Healing. J Dent Res. 2018;97(7): 803-809.[65] Lv FJ, Tuan RS, Cheung KM, et al. Concise review: the surface markers and identity of human mesenchymal stem cells. Stem Cells. 2014;32(6):1408-1419.[66] Fortino VR, Chen RS, Pelaez D, et al. Neurogenesis of neural crest-derived periodontal ligament stem cells by EGF and bFGF. J Cell Physiol. 2014;229(4):479-488.[67] Li B, Zhang Y, Wang Q, et al. Periodontal ligament stem cells modulate root resorption of human primary teeth via Runx2 regulating RANKL/OPG system. Stem Cells Dev. 2014;23(20): 2524-2534.[68] Zhu W, Tan Y, Qiu Q, et al. Comparison of the properties of human CD146+ and CD146- periodontal ligament cells in response to stimulation with tumour necrosis factor a. Arch Oral Biol. 2013; 58(12):1791-1803.[69] Nagata M, Iwasaki K, Akazawa K, et al. Conditioned Medium from Periodontal Ligament Stem Cells Enhances Periodontal Regeneration. Tissue Eng Part A. 2017;23(9-10):367-377.[70] Szepesia, Matula Z, Szigeti A, et al. ABCG2 is a selectable marker for enhanced multilineage differentiation potential in periodontal ligament stem cells. Stem Cells Dev. 2015;24(2): 244-252.[71] Torii D, Konishi K, Watanabe N, et al. Cementogenic potential of multipotential mesenchymal stem cells purified from the human periodontal ligament. Odontology. 2015;103(1):27-35.[72] Gauthier P, Yu Z, Tran QT, et al. Cementogenic genes in human periodontal ligament stem cells are downregulated in response to osteogenic stimulation while upregulated by vitamin C treatment. Cell Tissue Res. 2017;368(1):79-92.[73] Yan XZ, Both SK, Yang PS, et al. Human periodontal ligament derived progenitor cells: effect of STRO-1 cell sorting and Wnt3a treatment on cell behavior. Biomed Res Int. 2014; 2014:145423.[74] Yasui T, Mabuchi Y, Toriumi H, et al. Purified Human Dental Pulp Stem Cells Promote Osteogenic Regeneration. J Dent Res. 2016;95(2):206-214.[75] Xiong J, Menicanin D, Zilm PS, et al. Investigation of the Cell Surface Proteome of Human Periodontal Ligament Stem Cells. Stem Cells Int. 2016;2016:1947157.[76] Taraslia V, Lymperi S, Pantazopoulou V, et al. A High-Resolution Proteomic Landscaping of Primary Human Dental Stem Cells: Identification of SHED- and PDLSC-Specific Biomarkers. Int J Mol Sci. 2018;19(1): E158.[77] Lee HS, Lee J, Kim SO, et al. Comparative gene-expression analysis of the dental follicle and periodontal ligament in humans. PLoS One. 2013;8(12):e84201.[78] Gong AX, Zhang JH, Li J, et al. Comparison of gene expression profiles between dental pulp and periodontal ligament tissues in humans. Int J Mol Med. 2017;40(3): 647-660.[79] Yamada S, Ozaki N, Tsushima K, et al. Transcriptome Reveals Cathepsin K in Periodontal Ligament Differentiation. J Dent Res. 2016;95(9):1026-1033. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [6] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [7] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [8] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [9] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [10] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [11] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [12] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [13] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [14] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [15] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||