Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (5): 815-820.doi: 10.3969/j.issn.2095-4344.1565
Mechanism and clinical application of stem cell-derived exosomes in promoting fracture healing
Zhu Wanbo, Lu Jinsen, Yang Jiazhao, Fang Shiyuan
- Anhui Provincial Hospital, Anhui Medical University, Hefei 230001, Anhui Province, China
-
Revised:2018-11-06Online:2019-02-18Published:2019-02-18 -
Contact:Fang Shiyuan, Chief physician, Anhui Provincial Hospital, Anhui Medical University, Hefei 230001, Anhui Province, China -
About author:Zhu Wanbo, Master candidate, Anhui Provincial Hospital, Anhui Medical University, Hefei 230001, Anhui Province, China
CLC Number:
Cite this article
Zhu Wanbo, Lu Jinsen, Yang Jiazhao, Fang Shiyuan. Mechanism and clinical application of stem cell-derived exosomes in promoting fracture healing[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(5): 815-820.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
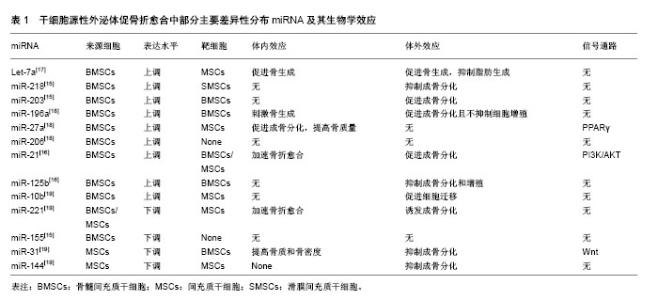
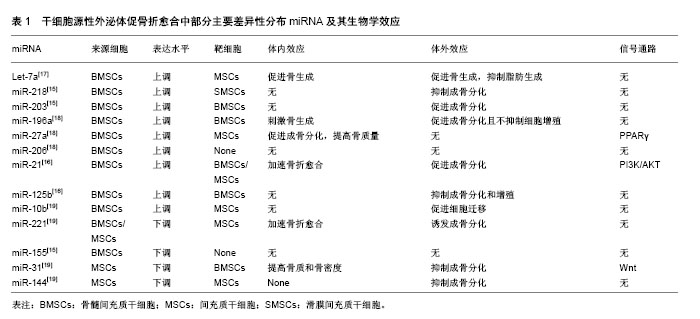
2.1 干细胞源性外泌体促骨折愈合的生物学基础 骨折愈合是一个复杂的生理过程。在骨折微环境中,骨母细胞和干细胞向成骨细胞增殖分化并移行至骨缺损和骨吸收区域,未成熟的成骨细胞产生含钙盐和磷酸盐的骨基质,并促使其矿化形成矿化骨是骨折愈合的主要机制[10]。调控干细胞和骨祖细胞的成骨分化是促骨折愈合的基础之一。骨折微环境中的新生血管能为骨折愈合带来必须的营养和无机盐,丰富的血管加快局部废物代谢的同时带来大量迁移的多能干细胞,提高成骨分化和骨重建的效率[11-12]。前期大量研究发现,干细胞源性外泌体可通过调控多能干细胞成骨分化,促进新生血管生成,提高无机盐沉积和基质矿化等途径,促进骨折愈合。这一过程是由干细胞源性外泌体选择性携带的生物活性物质所决定[9]。 2.2 外泌体携带的遗传物质调控促骨折愈合过程 干细胞源性外泌体能携带遗传物质如微小RNA(miRNA)和mRNA,在骨细胞间和干细胞间传递遗传信息,是调控干细胞源性外泌体促骨折愈合的重要组成部分[13]。miRNA被认为是成骨细胞相关骨生成和骨重建中重要的转录后调控因子,能开启一系列骨折修复相关反应[14]。在脂质膜的包裹下miRNA能稳定存在于外泌体中免于免疫系统的分解,并最终随外泌体以膜融合或直接胞吞的方式进入靶细胞,发挥效应。 2.2.1 干细胞源性外泌体miRNA具有激活靶细胞成骨分化和血管新生能力,促进骨生成的作用 Xu等[15]于2014年首次发现骨髓间充质干细胞源性外泌体(BMSC-Exo)miRNA在促骨髓间充质干细胞成骨分化过程中的分布具有差异性,其中一系列前期实验发现提高干细胞成骨分化能力的miRNA如let-7a等显著上调。还有一些体内外对比实验也相继发现一些特定的干细胞源性外泌体miRNA具有促成骨分化能力。Furuta等[16]利用外泌体沉默表达的CD9-/-小鼠做动物模型,从间充质干细胞培养基分离得到的骨髓间充质干细胞源性外泌体较对照组能加速骨折愈合,体外分析外泌体内容物发现miR-21、miR-4332等成骨分化相关miRNA与其他细胞来源外泌体相比高表达。有趣的是,被认为具有募集干细胞、促进成骨分化和血管生成功能的单核细胞趋化蛋白(MCP-1、MCP-3)和基质细胞衍生因子(SDF-1)在骨髓间充质干细胞源性外泌体内含量较对照组低,研究者推测成骨分化和血管生成相关miRNA在干细胞源性外泌体内的差异性分布,提示这些具有特殊功能的miRNA才是促骨折愈合的主要机制。表1列举了一部分研究中主要的差异性分布的miRNA及其生物学效应[15-19]。 2.2.2 不同分化阶段干细胞外泌体促成骨分化能力具有差异性 体外实验证实分化晚期人间充质干细胞源性外泌体(hMSCs-Exo)具有最强的促成骨分化能力,这可能是由于不同分化阶段干细胞选择性携带的miRNA含量不同所决定[15,19]。miR-31,miR-221和miR-144等具有抑制成骨分化作用的miRNA在分化晚期干细胞源性外泌体中含量显著降低,而miR-21,miR-10b等促成骨分化相关miRNA的含量显著上调,使干细胞源性外泌体对靶细胞成骨分化具有促调控作用[19]。 2.2.3 干细胞源性外泌体miRNA调控破骨细胞分化,促进骨吸收 骨折愈合是成骨-破骨分化的动态平衡过程,骨折微环境中骨吸收是启动一系列骨折愈合机制的开始[13]。骨髓间充质干细胞源性外泌体中携带的miR-503等miRNA具有促进干细胞破骨分化和骨吸收的作用[20]。这些miRNA通过调控RANK-RANKL信号通路调节成骨-破骨的动态平衡,促进骨折愈合[13]。 2.2.4 一些不同干细胞源性外泌体mRNA也具备激发骨折愈合的能力 Tooi等[21]发现人胚胎间充质干细胞来源外泌体(hPMSC-Exo)与骨形态发生蛋白2共培养时,细胞外基质标记物碱性磷酸酶和成骨细胞特异性转录因子OSX的mRNA表达显著上调,成纤维细胞成骨分化加速,骨矿物沉积增加。体外实验发现人脂肪来源干细胞源性外泌体(ASCs-Exo)能通过上调成骨相关基因RUNX2、ALP和COL1A1的mRNA表达,提高靶细胞成骨能力,促进骨生成[22]。 干细胞源性外泌体miRNA和mRNA是遗传信息的载体,其促进骨折愈合作用需要信号通路、蛋白质和细胞因子的共同参与。 2.3 信号通路与转录因子介导促骨折愈合过程 干细胞源性外泌体选择性携带的遗传物质和功能性蛋白作用于靶细胞后,通过借助一系列信号通路和转录因子介导促骨折愈合过程。 RUNX2是调控成骨分化的转录因子,其表达可指导多能干细胞向成骨细胞系分化并抑制成骨细胞成熟,维持未成熟成骨细胞持续的成骨能力,在干细胞修复骨损伤中扮演重要角色[23]。Qi等[24]通过体内实验发现人诱导性多能间充质干细胞源性外泌体(hiPS-MSC-Exo)在上调RUNX2等转录因子表达后,激发成骨分化,促进新生血管生成,提高动物骨折愈合率。干细胞源性外泌体miRNA在RUNX2的上调中至关重要[25]。分化晚期间充质干细胞源性外泌体中含量大幅降低的miR-31影响RUNX2下游基因Osterix和SATB2表达下调,是潜在的RUNX2作用位点[26]。体外实验证实降低或抑制miR-31表达可促进成骨分化,提高骨含量和骨密度[27]。"
| [1] Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013;200(4):373-383. [2] Bjørge IM, Kim SY, Mano JF, et al. Extracellular vesicles, exosomes and shedding vesicles in regenerative medicine - a new paradigm for tissue repair. Biomater Sci. 2017;6(1):60-78.[3] Hsu C, Morohashi Y, Yoshimura S, et al. Regulation of exosome secretion by Rab35 and its GTPase-activating proteins TBC1D10A-C. J Cell Biol. 2010;189(2):223-232.[4] Simons M, Raposo G. Exosomes--vesicular carriers for intercellular communication. Curr Opin Cell Biol. 2009;21(4): 575-581.[5] Keshtkar S, Azarpira N, Ghahremani MH. Mesenchymal stem cell-derived extracellular vesicles: novel frontiers in regenerative medicine. Stem Cell Res Ther. 2018;9(1):63.[6] Golchin A, Hosseinzadeh S, Ardeshirylajimi A. The exosomes released from different cell types and their effects in wound healing. J Cell Biochem. 2018;119(7):5043-5052.[7] Ni J, Sun Y, Liu Z. The Potential of Stem Cells and Stem Cell-Derived Exosomes in Treating Cardiovascular Diseases. J Cardiovasc Transl Res. 2018 Mar 10. doi: 10.1007/s12265-018-9799-8. [Epub ahead of print][8] Huang W, Zhang K, Zhu Y, et al. Genetic polymorphisms of NOS2 and predisposition to fracture non-union: A case control study based on Han Chinese population. PLoS One. 2018; 13(3):e0193673.[9] Hao ZC, Lu J, Wang SZ, et al. Stem cell-derived exosomes: A promising strategy for fracture healing. Cell Prolif. 2017; 50(5): e12359.[10] Komori T. Regulation of osteoblast differentiation by transcription factors. J Cell Biochem. 2006;99(5):1233-1239.[11] Stegen S, van Gastel N, Carmeliet G. Bringing new life to damaged bone: the importance of angiogenesis in bone repair and regeneration. Bone. 2015;70:19-27.[12] Ando Y, Matsubara K, Ishikawa J, et al. Stem cell-conditioned medium accelerates distraction osteogenesis through multiple regenerative mechanisms. Bone. 2014;61:82-90.[13] Yin P, Lv H, Li Y, et al. Exosome-Mediated Genetic Information Transfer, a Missing Piece of Osteoblast- Osteoclast Communication Puzzle. Front Endocrinol (Lausanne). 2017;8:336.[14] Inose H, Ochi H, Kimura A, et al. A microRNA regulatory mechanism of osteoblast differentiation. Proc Natl Acad Sci U S A. 2009;106(49):20794-20799.[15] Xu JF, Yang GH, Pan XH, et al. Altered microRNA expression profile in exosomes during osteogenic differentiation of human bone marrow-derived mesenchymal stem cells. PLoS One. 2014;9(12):e114627.[16] Furuta T, Miyaki S, Ishitobi H, et al. Mesenchymal Stem Cell-Derived Exosomes Promote Fracture Healing in a Mouse Model. Stem Cells Transl Med. 2016;5(12):1620-1630.[17] Wei J, Li H, Wang S, et al. Let-7 enhances osteogenesis and bone formation while repressing adipogenesis of human stromal/mesenchymal stem cells by regulating HMGA2. Stem Cells Dev. 2014;23(13):1452-1463.[18] Qin Y, Wang L, Gao Z, et al. Bone marrow stromal/stem cell-derived extracellular vesicles regulate osteoblast activity and differentiation in vitro and promote bone regeneration in vivo. Sci Rep. 2016;6:21961.[19] Wang X, Omar O, Vazirisani F, et al. Mesenchymal stem cell-derived exosomes have altered microRNA profiles and induce osteogenic differentiation depending on the stage of differentiation. PLoS One. 2018;13(2):e0193059.[20] Chen C, Cheng P, Xie H, et al. MiR-503 regulates osteoclastogenesis via targeting RANK. J Bone Miner Res. 2014;29(2):338-347.[21] Tooi M, Komaki M, Morioka C, et al. Placenta Mesenchymal Stem Cell Derived Exosomes Confer Plasticity on Fibroblasts. J Cell Biochem. 2016;117(7):1658-1670.[22] Li W, Liu Y, Zhang P, et al. Tissue-Engineered Bone Immobilized with Human Adipose Stem Cells-Derived Exosomes Promotes Bone Regeneration. ACS Appl Mater Interfaces. 2018;10(6):5240-5254.[23] Wang KX, Xu LL, Rui YF, et al. The effects of secretion factors from umbilical cord derived mesenchymal stem cells on osteogenic differentiation of mesenchymal stem cells. PLoS One. 2015;10(3):e0120593.[24] Qi X, Zhang J, Yuan H, et al. Exosomes Secreted by Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells Repair Critical-Sized Bone Defects through Enhanced Angiogenesis and Osteogenesis in Osteoporotic Rats. Int J Biol Sci. 2016;12(7):836-849.[25] Zhang Y, Xie RL, Gordon J, et al. Control of mesenchymal lineage progression by microRNAs targeting skeletal gene regulators Trps1 and Runx2. J Biol Chem. 2012;287(26): 21926-21935.[26] Deng Y, Zhou H, Zou D, et al. The role of miR-31-modified adipose tissue-derived stem cells in repairing rat critical-sized calvarial defects. Biomaterials. 2013;34(28):6717-6728.[27] Baglìo SR, Devescovi V, Granchi D, et al. MicroRNA expression profiling of human bone marrow mesenchymal stem cells during osteogenic differentiation reveals Osterix regulation by miR-31. Gene. 2013;527(1):321-331.[28] Ghosh-Choudhury N, Abboud SL, Nishimura R, et al. Requirement of BMP-2-induced phosphatidylinositol 3-kinase and Akt serine/threonine kinase in osteoblast differentiation and Smad-dependent BMP-2 gene transcription. J Biol Chem. 2002;277(36):33361-33368.[29] Suzuki E, Ochiai-Shino H, Aoki H, et al. Akt activation is required for TGF-β1-induced osteoblast differentiation of MC3T3-E1 pre-osteoblasts. PLoS One. 2014;9(12):e112566.[30] Zhang J, Liu X, Li H, et al. Exosomes/tricalcium phosphate combination scaffolds can enhance bone regeneration by activating the PI3K/Akt signaling pathway. Stem Cell Res Ther. 2016;7(1):136.[31] Trohatou O, Zagoura D, Bitsika V, et al. Sox2 suppression by miR-21 governs human mesenchymal stem cell properties. Stem Cells Transl Med. 2014;3(1):54-68.[32] Meng YB, Li X, Li ZY, et al. microRNA-21 promotes osteogenic differentiation of mesenchymal stem cells by the PI3K/β-catenin pathway. J Orthop Res. 2015;33(7):957-964.[33] Liu X, Li Q, Niu X, et al. Exosomes Secreted from Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells Prevent Osteonecrosis of the Femoral Head by Promoting Angiogenesis. Int J Biol Sci. 2017;13(2):232-244.[34] Leucht P, Lee S, Yim N. Wnt signaling and bone regeneration: Can't have one without the other. Biomaterials. 2018 Mar 15. doi: 10.1016/j.biomaterials.2018.03.029. [Epub ahead of print][35] McBride JD, Rodriguez-Menocal L, Guzman W, et al. Bone Marrow Mesenchymal Stem Cell-Derived CD63+ Exosomes Transport Wnt3a Exteriorly and Enhance Dermal Fibroblast Proliferation, Migration, and Angiogenesis In Vitro. Stem Cells Dev. 2017;26(19):1384-1398.[36] Martins M, Ribeiro D, Martins A, et al. Extracellular Vesicles Derived from Osteogenically Induced Human Bone Marrow Mesenchymal Stem Cells Can Modulate Lineage Commitment. Stem Cell Reports. 2016;6(3):284-291.[37] Narayanan R, Huang CC, Ravindran S. Hijacking the Cellular Mail: Exosome Mediated Differentiation of Mesenchymal Stem Cells. Stem Cells Int. 2016;2016:3808674.[38] Lamplot JD, Qin J, Nan G, et al. BMP9 signaling in stem cell differentiation and osteogenesis. Am J Stem Cells. 2013;2(1): 1-21.[39] Al Rifai O, Chow J, Lacombe J, et al. Proprotein convertase furin regulates osteocalcin and bone endocrine function. J Clin Invest. 2017;127(11):4104-4117.[40] Feng JQ, Ward LM, Liu S, et al. Loss of DMP1 causes rickets and osteomalacia and identifies a role for osteocytes in mineral metabolism. Nat Genet. 2006;38(11):1310-1315.[41] Zhang B, Yeo RWY, Lai RC, et al. Mesenchymal stromal cell exosome-enhanced regulatory T-cell production through an antigen-presenting cell-mediated pathway. Cytotherapy. 2018;20(5):687-696.[42] Peinado H, Ale?kovi? M, Lavotshkin S, et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat Med. 2012;18(6): 883-891.[43] Suárez H, Gámez-Valero A, Reyes R, et al. A bead-assisted flow cytometry method for the semi-quantitative analysis of Extracellular Vesicles. Sci Rep. 2017;7(1):11271.[44] Du T, Ju G, Wu S, et al. Microvesicles derived from human Wharton's jelly mesenchymal stem cells promote human renal cancer cell growth and aggressiveness through induction of hepatocyte growth factor. PLoS One. 2014;9(5): e96836.[45] Peak TC, Praharaj PP, Panigrahi GK, et al. Exosomes secreted by placental stem cells selectively inhibit growth of aggressive prostate cancer cells. Biochem Biophys Res Commun. 2018;499(4):1004-1010.[46] Qi J, Zhou Y, Jiao Z, et al. Exosomes Derived from Human Bone Marrow Mesenchymal Stem Cells Promote Tumor Growth Through Hedgehog Signaling Pathway. Cell Physiol Biochem. 2017;42(6):2242-2254.[47] Roccaro AM, Sacco A, Maiso P, et al. BM mesenchymal stromal cell-derived exosomes facilitate multiple myeloma progression. J Clin Invest. 2013;123(4):1542-1555.[48] Umezu T, Imanishi S, Azuma K, et al. Replenishing exosomes from older bone marrow stromal cells with miR-340 inhibits myeloma-related angiogenesis. Blood Adv. 2017;1(13): 812-823.[49] Karlsson T, Sundar R, Widmark A, et al. Osteoblast-derived factors promote metastatic potential in human prostate cancer cells, in part via non-canonical transforming growth factor β (TGFβ) signaling. Prostate. 2018;78(6):446-456. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [6] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [7] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [8] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [9] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [10] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [11] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [12] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [13] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [14] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [15] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||